Abstract
Biotinidase deficiency (BD) may cause neurological symptoms and developmental problems. However, newborn screening of BD and early biotin treatment prevent the manifestation of the majority of symptoms. This study intended to examine the developmental and behavioral outcomes as well as maternal anxiety and depressive symptoms of preschool-aged children with BD and to compare these with the outcomes of healthy preschool-aged children. In total, 49 children with BD and 23 healthy children are included. All children were screened for developmental and behavioral problems. Moreover anxiety and depressive symptomatology of their mothers were evaluated. Despite the high percentage of developmental delay in BD group, the numbers of children screened positive for a developmental delay were statistically similar in children with BD and healthy children. Among patients with BD, children with risk of developmental delay had more unfavorable socio-demographic features compared to typically developing ones. Behavioral problem scores, maternal anxiety, and depressive symptoms scores of children with BD were not higher than the healthy children.
Conclusion: Children with BD were not different from their healthy peers in terms of developmental and behavioral outcomes. Developmental problems of children with BD may be related to the unfavorable socio-demographic features, not the BD itself.
What is known: • Biotinidase deficiency (BD) may result in neurological symptoms and developmental problems. • Newborn screening and early biotin supplementation prevent the manifestation of the majority of symptoms. | |
What is new: • Preschool-aged children with BD identified by newborn screening are not different from their healthy peers in terms of developmental and behavioral outcomes. • Maternal anxiety and depressive symptoms scores of children with BD are similar to scores of healthy children. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biotinidase is essential for the recycling of biotin in the body by releasing it from the biotin-dependent carboxylases (acetyl-CoA carboxylase, propionyl-CoA carboxylase, 3-methylcrotonyl-CoA carboxylase, pyruvate carboxylase). Biotinidase deficiency (BD) (OMIM# 253260), an autosomal recessive metabolic disorder, results in low activities of these enzymes [1]. While children with less than 10% of mean normal serum biotinidase activity are diagnosed with profound BD, children having 10%–30% of mean normal serum biotinidase are diagnosed with partial BD [2]. Untreated children with BD may exhibit various neurological symptoms including lethargy, hypotonia, seizures, and ataxia; cutaneous abnormalities (alopecia, skin rash, candidiasis, and dermatitis); respiratory problems (hyperventilation, laryngeal stridor, and apnea); eye abnormalities; and hearing loss [3, 4]. Moreover, varying degrees of developmental delay, learning disabilities, and autism spectrum disorders were reported in patients with BD [3, 5, 6].
Symptoms of children with BD mostly improve after biotin treatment. However, children who are treated after the onset of symptoms may exhibit irreversible problems such as hearing problems and developmental delays. While newborn screening of BD and early treatment with biotin supplementation prevent manifestation of the majority of symptoms, developmental problems have been documented in children with BD identified by newborn screening [7].
There are only a few reports on developmental and behavioral outcomes of children with partial and profound BD identified by newborn screening [7,8,9]. Therefore, the current study aimed to present the developmental and behavioral outcomes of preschool-aged children diagnosed with BD by neonatal screening and to compare the outcomes of these children with their healthy peers in a country with a high BD incidence [10,11,12]. Although inherited metabolic diseases were reported to have negative impacts on the lives and psychological well-being of parents [13, 14], the literature lacks reports presenting the effects of BD diagnosis of children on parental anxiety and depression. Therefore, this study intended to evaluate maternal anxiety and depressive symptom levels of children with BD and compare them with levels of healthy children.
Method
Study population
This study conducted in the Department of Pediatric Metabolism and Department of Developmental Pediatrics at Hacettepe University Faculty of Medicine was approved by the Ethics Committee of Hacettepe University Faculty of Medicine. Informed consent was obtained from all parents enrolled in the study.
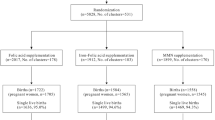
Preschool-aged children with the diagnosis of partial or profound BD, who were admitted for a follow-up visit to the Department of Pediatric Metabolism between September 1, 2019, and February 1, 2020, were invited to the study. Preschool-aged children without medical conditions or developmental-behavioral problems were recruited from well-child/sick child visits as a control group. Seven of 56 eligible mothers of a child with BD and 7 of 30 mothers of a healthy child who were admitted to the clinic during the specified period refused to take part in the study. Finally, 49 preschool-aged children with BD (45 partial BD, 4 profound BD) and 23 preschool-aged healthy children are included in the current study. Among patients with BD, while 33 had molecular analysis for BD, molecular testing was pending for the rest of 16 patients.
In what follows, management of patients with BD in the Department of Pediatric Metabolism of Hacettepe University is summarized. Every child born in Turkey has undergone mandated newborn screening for BD since 2008 [15]. Children screened positive for BD are tested by using both Guthrie test and quantitative enzyme activity method (biotin 4-aminobenzoic acid as the substrate) to confirm the diagnosis. The cutoff level for biotinidase enzyme activity is accepted as 3.5–13.8 U/L. Upon the confirmation, biotin supplementation is started and molecular analysis for the common mutations of the BTD gene is planned. While patients with profound BD are treated with 10-mg/day dose of biotin, patients with partial BD are treated with 5-mg/day dose of biotin. Patients with BD are examined at 6 months of age and 1 year of age, and then annually. All patients are referred to audiological and ophthalmological evaluations. While annual evaluation is recommended for patients with profound BD, biannual evaluations are planned for those with partial BD.
Procedures
All participants were interviewed by a semi-structured questionnaire. The socio-demographic characteristics of the participating children and their families such as parental age and educational level were questioned. The socioeconomic status (SES) was measured by utilizing the Hollingshead-Redlich index [16]. Regarding the Hollingshead-Redlich index, participants were classified based on parental education attainment (university, high school, primary school, etc.) and occupational prestige (major business, unskilled laborers, etc.). Researchers also questioned all the mothers about screen time, shared book reading, and joint play with the child to assess the quality of stimulation in the home environment. Moreover, all participating mothers were questioned for their concerns about developmental-behavioral problems and problems of vision and hearing. In this study, the information on compliance of biotin treatment was gathered from the parents of patients with BD.
After the interview, all children (n = 72) were subjected to a play-based observational evaluation, which was performed by a child development specialist and developmental pediatricians for the assessment of developmental problems including autism spectrum disorder. Moreover, screening of developmental and behavioral problems and evaluations for maternal anxiety and depressive symptomatology were made by the following instruments.
The Ages and Stages Questionnaire
ASQ, which is increasingly being recommended for the evaluation of development in clinical practice, has 19 age-specific sub-questionnaires assessing the development of children in terms of communication, gross motor skills, fine motor skills, problem solving, and personal-social skills. While children who score lower than cutoffs in at least one domain are accepted as screened positive for developmental delay, ones who score above cutoffs are accepted typically developing [17]. Although originally the Ages and Stages Questionnaire is a parent completed questionnaire, in this study, ASQ was administered via parent interviews in conjunction with the literature [18]. In the current study, the Ages and Stages Questionnaire for Turkish children (ASQ-TR) [19] was used for developmental screening. The sensitivity and specificity of ASQ-TR are 0.94 and 0.85, respectively [19].
Child behavior checklist for ages 1.5 to 5 years (CBCL/1.5–5)
This instrument is designed to be completed by parents to score their own child’s behaviors [20,21,22]. CBCL/1.5–5 has seven syndrome scores as follows: (i) emotionally reactive, (ii) anxious/depressed, (iii) somatic complaints, (iv) withdrawn, (v) sleep problems, (vi) attention problems, and (vii) aggressive. Moreover, the combination of emotionally reactive, anxious/depressed, somatic complaints, and withdrawn scores constitute the Internalizing Problems score, and the combination of attention problems and aggressive scores constitute the Externalizing Problems score. These scores, together with the seven syndrome scores and the one item added by the parents, constitute the Total Problems score. The CBCL/1.5–5 was completed by parents to evaluate the behavioral problems of the participating children in this study.
Beck depression inventory (BDI)
BDI, which is a commonly used self-rating scale for measuring depressive symptomatology, consists of 21 items with scores ranging from 0 to 3 and higher scores indicate greater depressive symptoms. In this study, the validated Turkish Beck Depression Inventory [23] was used to screen maternal depressive symptoms.
State-trait anxiety inventory-trait (STAI-T)
STAI-T is a self-report questionnaire, which evaluates relatively stable aspects of anxiety including general states of calmness, confidence, and security independent of the current conditions [24]. It consists of 20 items with scores ranging from 1 to 4 with total scores of 20 to 80, with a higher score indicating greater anxiety. The adapted Turkish STAI-T [25] was utilized to evaluate maternal anxiety in the current study.
Statistical analysis
Statistical analysis was performed with the IBM SPSS for Windows Version 23.0. Numerical variables were summarized as mean ± standard deviation or median (25th–75th percentile). Categorical variables were given as frequencies and percentages. Categorical variables were compared by Chi-square test or Fisher’s exact test. Normality of the continuous variables was evaluated by the Kolmogorov Smirnov test. Homogeneity of variances was tested by Levene’s test. Differences between the groups according to continuous variables were determined by independent samples t test or Mann Whitney U test as appropriate. Factors affecting developmental delay were determined by multiple logistic regression analysis. A p value less than 0.05 was considered as significant.
Results
In the current study, 49 preschool-aged children with BD and 23 healthy children were included. All children with BD were diagnosed via neonatal screening. In Turkey, newborn screening for biotinidase deficiency is usually completed within 2 weeks of life. However, delays may occur due to difficulties in reaching families and their referral to metabolism clinics. In the current study, while the median age at diagnosis was 22.0 (10–79) days in patients with partial BD, it was 22.5 (19–27) days in patients with profound BD. The median serum biotinidase activity was 20.5% (10–29) and 5.25% (3–7) in patients with partial and profound BD, respectively. All patients were diagnosed by newborn screening in the presymptomatic period. Therefore, additional biochemical tests, lactate, and urine organic acid analyses were not performed. All patients with BD underwent molecular analysis for BTD gene mutations and sequencing analyses were completed in 33 patients. While 10 of the 33 patients had homozygous BTD gene mutations, 23 had compound heterozygous mutations. The p.D444H (37/66), p.Q456H (6/66), p.R157H (5/66), and complex mutation p.D444H/A171T were the most common alleles in the study sample.
The compliance of biotin supplementation was very well in the majority (n = 48) of patients; yet, only one parent of a patient with partial BD reported missing the biotin supplement occasionally. Among children with BD, none of the children had dermatologic problems related to the disease or a history of seizure except two patients with a history of febrile convulsion. Physical and neurological examinations and visual and hearing assessments of all participants were normal. None of the patients had cutaneous lesions. None of the children with BD had a history of hospitalization for metabolic acidosis, lactic acidosis, or coma, regardless of the severity of enzyme deficiency.
Regarding four patients with profound BD, all of them were diagnosed before becoming symptomatic. Biotinidase enzyme activities of these patients were 3%, 5%, 6%, and 7% of the laboratory standard, respectively. The molecular analysis for BTD mutations of the patient with the enzyme activity of 3% is still pending. The other three patients had the following mutations: homozygous frameshift (p.F131Lfs*27), homozygous p.Q456H, and compound heterozygous p.D444H/A171T. Similar to the patients with partial BD, all four patients with profound BD had normal physical, neurological, and dermatological examinations.
None of the parents of children with and without BD had concerns about hearing and vision problems. None of the children were born preterm and small for gestational age. Based on play-based observational evaluations, all participants with and without BD were interested in playing and actively participated to free-play with their mothers and the researchers. All of them were able to initiate social interactions. None of the children had limited social interaction. None of the participants exhibited symptoms of autism spectrum disorders.
Features of children with BD and healthy controls are given in Table 1. There were statistically significant differences between the two groups (i.e., BD and healthy controls) in terms of child age, shared book reading, number of children, education level of the mother, and SES. Parental concerns about developmental and behavioral problems were statistically similar between the two groups.
The number of children screened positive for developmental delay by ASQ is presented in Table 2. All patients with developmental delay were accepted to a developmental follow-up program; parents of them were guided to have an enriched home environment and referred to early intervention services based on their special needs. In terms of five developmental domains, the number of children with risk of developmental delay was not statistically significantly different between biotinidase deficient and healthy groups. The number of children screened positive for a developmental delay in one domain of ASQ was statistically similar in both groups. However, the percentages of children who were screened positive for developmental delay in one domain of ASQ were 36.7% and 17.4% in children with BD and healthy children, respectively. Regarding four children with profound BD, three were typically developing and one had delayed fine motor skills.
In terms of behavior problems screened via CBCL, internalizing, externalizing, and total problem scores of patients with BD and healthy children were not significantly different. Moreover, the seven syndrome scores of CBCL were statistically similar between the two groups. Scores of CBCL are presented in Table 3. Then we analyzed the maternal BDI and STAI-T scores of the participants (see Table 3) and compared the scores of the mothers of children with BD with the mothers of healthy children. Maternal anxiety and depressive symptoms scores of children with and without BD were not significantly different.
Due to the high rates of developmental delay among children with BD, the factors that can be associated with developmental delay were analyzed. Age, gender, and CBCL scores of children, parental concern of developmental-behavioral problems, maternal anxiety, and maternal depressive symptoms scores were not found to be statistically different between typically developing children and children who were screened positive for developmental delay. It was shown that favorable socio-demographic factors (parental education, SES, shared book reading) were more common in typically developing children compared with children with risk of developmental delay (Table 4).
Discussion
To the best of the authors’ knowledge, this is the first study comparing the developmental-behavioral outcomes, as well as the maternal psychological well-being of preschool-aged children with BD and healthy controls.
In accordance with the literature, where a majority of children with BD diagnosed by neonatal screening were reported to demonstrate a typical development [9, 26, 27], this study revealed that the frequencies of developmental problems of children with and without BD deficiency were statistically similar. However, it is worth noting that more than one-third of children with BD were screened positive for developmental delay in one domain of ASQ. Although this percentage was higher compared with the general population, identification of developmental delay was reported to be more common in children who participated in a developmental screening program [28]. Moreover, based on the findings of this study, we suggest that developmental delay could be linked to unfavorable socio-demographic factors such as low SES, parental education, and poor quality of home environment rather than BD itself.
Behavioral problems screened by CBCL were not found to be more common in children with BD compared with healthy controls. This finding, which was not reported previously in the literature, suggested that newborn screening and early biotin supplementation prevented behavioral problems in children with BD. However, the similarity of reported behavior problems in children with and without BD could be linked to the fact that it is more difficult to evaluate behavior problems of younger children and problems of younger children are more likely to be accepted as normal by parents.
On the other hand, maternal anxiety and depressive symptoms scores of children with and without BD were similar. Although having a child with an inherited metabolic disease may have negative impacts on a parents’ psychological well-being, BD did not have adverse effects on anxiety and depressive symptoms of mothers. Since BD is known as a good example of the early identification and successful treatment of a genetic disease [29], diagnosis and management of BD was not seemed to be a burden on parents of patients.
While interpreting the findings of the current study, limitations should be also taken into consideration. The first limitation is the small sample size of BD and control groups due to the short recruitment period of the study. Secondly, since ASQ is a screening tool, this limited us to make a definite conclusion about the developmental status of children. The younger mean age of the children with BD than the healthy children is another limitation. Finally, due to the paucity of patients with profound BD, we could not compare the outcomes of children with partial and profound BD. One may expect an increased risk of developmental problems in patients with profound BD compared with the ones with partial BD. However, among children with profound BD, the percentage of developmental delay in one domain (25%) was not higher than the percentage of whole patients with BD (36.7%) in this study.
Despite these limitations, the current study adds several contributions to the literature. The risk of developmental and behavioral problems of children with BD ascertained by newborn screening was not higher than healthy children. However, based on the frequency of developmental problems in the BD group, we recommend regular developmental assessments in the follow-up of children with BD. On the other hand, anxiety and depressive symptom levels of mothers having a child with BD were similar to mothers of healthy children. The findings of the current study are in line with the statement of Wolf “if you have to have an inherited metabolic disease, BD is the one to have” [29].
References
Wolf B, Grier RE, Secor McVoy JR, Heard GS (1985) Biotinidase deficiency: a novel vitamin recycling defect. J Inherit Metab Dis 8(Suppl 1):53–58
Cowan TM, Blitzer MG, Wolf B (2010) Technical standards and guidelines for the diagnosis of biotinidase deficiency. Genet Med 12:464–470
Wolf B (2011) The neurology of biotinidase deficiency. Mol Genet Metab 104:27–34
Sivri HS, Genç GA, Tokatli A, Dursun A, Coşkun T, Aydin HI, Sennaroğlu L, Belgin E, Jensen K, Wolf B (2007) Hearing loss in biotinidase deficiency: genotype-phenotype correlation. J Pediatr 150:439–442
Ogundele MO (2011) Question 2. What is the incidence of biotin deficiency in preschool children with global developmental delay? Arch Dis Child 96:895–897
Zaffanello M, Zamboni G, Fontana E, Zoccante L, Tatò L (2003) A case of partial biotinidase deficiency associated with autism. Child Neuropsychol 9:184–188
Jay AM, Conway RL, Feldman GL, Nahhas F, Spencer L, Wolf B (2015) Outcomes of individuals with profound and partial biotinidase deficiency ascertained by newborn screening in Michigan over 25 years. Genet Med 17:205–209
Wolf B (2017) Successful outcomes of older adolescents and adults with profound biotinidase deficiency identified by newborn screening. Genet Med 19:396–402
Weber P, Scholl S, Baumgartner ER (2004) Outcome in patients with profound biotinidase deficiency: relevance of newborn screening. Dev Med Child Neurol 46:481–484
Pomponio RJ, Coskun T, Demirkol M, Tokatli A, Ozalp I, Hüner G, Baykal T, Wolf B (2000) Novel mutations cause biotinidase deficiency in Turkish children. J Inherit Metab Dis 23:120–128
Baykal T, Hüner G, Sarbat G, Demirkol M (1998) Incidence of biotinidase deficiency in Turkish newborns. Acta Paediatr 87:1102–1103
Karaca M, Özgül RK, Ünal Ö, Yücel-Yılmaz D, Kılıç M, Hişmi B, Tokatlı A, Coşkun T, Dursun A, Sivri HS (2015) Detection of biotinidase gene mutations in Turkish patients ascertained by newborn and family screening. Eur J Pediatr 174:1077–1084
Brown A, Crowe L, Boneh A, Anderson V (2017) Parent coping and the behavioural and social outcomes of children diagnosed with inherited metabolic disorders. JIMD Rep 31:29–36
Siddiq S, Wilson BJ, Graham ID, Lamoureux M, Khangura SD, Tingley K, Tessier L et al (2016) Experiences of caregivers of children with inherited metabolic diseases: a qualitative study. Orphanet J Rare Dis 11:168
Tezel B, Dilli D, Bolat H, Sahman H, Ozbaş S, Acıcan D, Ertek M, Köse MR, Dilmen U (2014) The development and organization of newborn screening programs in Turkey. J Clin Lab Anal 28:63–69
Hollingshead AB, Redlich FC (1958) Social class and mental illness: Community study
Squires JPL, Bricker D (1999) The ASQ User's guide for the ages & stages questionnaires: a parent-completed, child-monitoring System, 2nd edn. Paul H. Brookes publishing co, Baltimore
Small JW, Hix-Small H, Vargas-Baron E, Marks KP (2019) Comparative use of the Ages and Stages Questionnaires in low- and middle-income countries. Dev Med Child Neurol 61:431–443
Kapci EG, Kucuker S, Uslu RI (2010) How applicable are ages and stages questionnaires for use with Turkish children? Top Early Child Spec Educ 30:176–188
Achenbach TM (1992) Manual for the child behavior checklist/2–3 and 1992 profile: profile for boys and girls. Department of Psychiatry, University of Vermont, Auswertungsbögen
Achenbach TM, Rescorla LA (2000) Manual for the ASEBA preschool forms and profiles. University of Vermont, Research center for children, youth, & families, Burlington
Erol N, Simsek Z, Oner O, Munir K (2005) Behavioral and emotional problems among Turkish children at ages 2 to 3 years. J Am Acad Child Psy 44:80–87
Hisli N (1989) Reliability and validity of the Beck Depression Inventory for university students. Psikoloji Dergisi 7:3–13 (Turkish)
Spielberger CD, Gorsuch RL, Lushene R, Vagg PR, Jacobs GA (1983) Manual for the state-trait anxiety inventory Palo Alto, CA, Consulting Psychologists Press Inc.
Oner N, LeCompte A (1983) Durumluluk surekli kaygi envanteri el kitabi (the handbook of the state-trait anxiety inventory). Bogazici Publications, Istanbul
Möslinger D, Mühl A, Suormala T, Baumgartner R, Stöckler-Ipsiroglu S (2003) Molecular characterisation and neuropsychological outcome of 21 patients with profound biotinidase deficiency detected by newborn screening and family studies. Eur J Pediatr 162(Suppl 1):S46–S49
Warner-Rogers J, Waisbren SE, Levy HL (1995) Cognitive function in early treated biotinidase deficiency: follow-up of children detected by newborn screening. Screening 4:125–130
Guevara JP, Gerdes M, Localio R, Huang YV, Pinto-Martin J, Minkovitz CS, Hsu D, Kyriakou L, Baglivo S, Kavanagh J, Pati S (2013) Effectiveness of developmental screening in an urban setting. Pediatrics 131:30–37
Wolf B (2012) Biotinidase deficiency: “if you have to have an inherited metabolic disease, this is the one to have”. Genet Med 14:565–575
Author information
Authors and Affiliations
Contributions
Pinar Zengin Akkus: Conceptualization, Methodology, Investigation Writing–Original Draft
Kismet Ciki: Conceptualization, investigation, review and editing.
Ayse Mete Yesil: Investigation, review and editing.
Evin Ilter Bahadur: Investigation, review and editing.
Sevilay Karahan: Analysis and interpretation of data, review and editing.
Elif Nursel Ozmert: Conceptualization, methodology, review and editing, supervision.
Serap Sivri: Methodology, review and editing, supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
This study was approved by the Ethics Committee of Hacettepe University Faculty of Medicine.
Statement of informed consent
Informed consent was obtained from all parents enrolled in the study.
Additional information
Communicated by Peter de Winter
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zengin Akkus, P., Ciki, K., Mete Yesil, A. et al. Developmental and behavioral outcomes of preschool-aged children with biotinidase deficiency identified by newborn screening. Eur J Pediatr 180, 217–224 (2021). https://doi.org/10.1007/s00431-020-03740-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-020-03740-2