Abstract
Despite multiple recommendations, intramuscular epinephrine is poorly prescribed in emergency department receiving pediatric anaphylaxis. To evaluate the role of severity symptoms on this use, we included all admissions for a diagnosis linked to possible allergy in the two pediatric emergency departments of our institution between January 2010 and December 2015. Selection and analysis were restricted to children under 18 years fulfilling Sampson’s criteria for anaphylaxis. We retrospectively ranked these admissions with the Ring and Messmer anaphylaxis severity score and compared the use of epinephrine according to this classification. Among 422,483 admissions, 204 (0.05%) fulfilled the anaphylaxis criteria (170 (83.3%) grade II anaphylaxis, and 34 (16.7%) grade III; mean age 7.9 years). Previous allergy, anaphylaxis, and asthma were found in respectively 60.8%, 36.8%, and 35.1%. Food allergy was the main suspected causal trigger. Epinephrine was used in 32.7% (n = 65/199), before admission (11.4% (n = 23/201)) or in the emergency department (22.2% (n = 45/202)). Epinephrine was more frequently prescribed in grade III than in grade II anaphylaxis (84.8% vs 22.3%, p < 0.001; OR = 19.05 [7.05–54.10]). Upon discharge, epinephrine auto-injectors prescription and allergy referral were rare (31.7% and 44.2%).
Conclusion: Pediatricians intuitively adapt their epinephrine use to the severity of the anaphylaxis and contribute to epinephrine underuse in pediatric anaphylaxis.
What is known: • Intramuscular epinephrine is the recommended treatment for pediatric anaphylaxis. However, most of the European and North-American studies show a low prescription rate of epinephrine in both prehospital and pediatric emergency department management. • Reasons for such a low prescription rate are unknown. | |
What is new: • This study confirms that intramuscular epinephrine is poorly prescribed in pediatric anaphylaxis (about one case among 10 before admission and one among 5 in pediatric emergency departments). • Despite recommendations, pediatricians intuitively adapt their prescription to the clinical severity of anaphylaxis, with a fourfold increase prescription in grade III compared to grade II anaphylaxis. This medical behavior ascertainment may be in part explained by the delay between the ED admission/management and the anaphylactic episode onset. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anaphylaxis is the most severe manifestation of immediate hypersensitivity with an incidence of 0.05 to 2% in general population [25]. Anaphylaxis concerns 4.5 to 18 admissions on 10,000 in pediatric emergency departments (EDs), with a trend to increase during the last years [14, 15]. In France, the anaphylaxis-related mortality rate in children is 0.83 per million per year [19]. A rapid clinical diagnosis, based on Sampson’s criteria [23], is required for administering epinephrine as quickly as possible in accordance with the different recommendations [16, 26]. However, many studies underline constant under-use of epinephrine in children with anaphylaxis not only in pre-hospital but also in ED management [6, 11, 21, 24, 30]. One possible explanation for this gap between recommendations and practice could be a kind of “intuitive” adaptation of epinephrine delivery according to the severity of symptoms. This study aimed to analyze pediatricians’ epinephrine prescription as a function of clinical severity during a 6-year period in children admitted for anaphylaxis in the two pediatric EDs of the University Hospitals of Marseille, France.
Methods
Patients
Design and setting
This was a retrospective study performed in the two pediatric EDs of the Assistance Publique-Hôpitaux de Marseille, France, between January 2010 and December 2015. These EDs received patients up to 18 years.
Data source and inclusion criteria
In France, for each patient admitted to an ED, an Electronic Emergency Department Abstract (EEDA), directly available from the patients’ computerized medical files, is anonymously transmitted to the French Institute of Public Health Surveillance. EEDAs report on date and hour of ED admission, age, sex, disposition after ED visit (home discharge, hospitalization, death), and final diagnosis according to the national standardized thesaurus based on the International Classification of Diseases, Tenth Revision (ICD-10), codes were collected. We also analyzed the clinical severity appreciated by the triage nurse scale upon admission (high levels of severity were levels 1–2), which was therefore not specific for anaphylaxis.
Visits related to “anaphylaxis” (“anaphylactic shock” or “Quincke edema”), “urticaria,” and “other allergic reaction” were defined using grouping of ICD-10 codes. The medical files of all patients with an ICD-code related to anaphylaxis were reviewed. Patients who met Sampson’s criteria were considered to have confirmed anaphylaxis. Patients were excluded if Sampson’s criteria were not fulfilled. The anaphylaxis severity was defined by the Ring and Messmer classification (grade I: mucocutaneous symptoms; grade II: moderate multivisceral failure; grade III: severe mono/multivisceral failure; grade IV: cardiac arrest) [20]. Medical history, anaphylaxis features, and management (biological tests, treatments, planned allergist visit, and auto-injectable epinephrine prescription) were noted for each patient.
Statistical analysis
Visits related to anaphylaxis, urticaria, and other allergic reaction were compared for age, sex, severity, and hospitalization rate. A descriptive analysis of the admissions for anaphylaxis according to Sampson’s criteria was performed. Comparison of the data according to anaphylaxis severity rank was realized. Variables were described as percent, and mean (standard deviation). Associations were tested with simple logistic regression for qualitative variables and quantified by unadjusted odd ratio (OR) and their confidence interval. Quantitative variables were compared with a Student or Fisher test depending on the number of data. Analysis was performed with SPSS version 20.0® (SPSS Inc., Chicago, IL, USA). A p value of 0.05 or less was considered as statistically significant.
Ethical considerations
This study was approved by the Ethics Committee of the French Society of Pediatrics (Société Française de Pédiatrie, No. 2016_018_2) and the National Commission of Informatics and Liberty (Commission Nationale Informatique et Libertés, No. 2016-16).
Results
Admissions with a diagnosis related to allergy
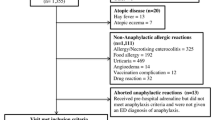
During the study period, 422,483 admissions took place (Fig. 1). The diagnosis discharge was missing for 18,029 (4.2%). Overall, 4141 visits were coded urticaria (102.13/10,000 admissions), 1358 other allergic reaction (33.49/10,000), and 235 anaphylaxis (5.76/10,000).
Compared with visits coded urticaria and to other allergic reaction, visits coded anaphylaxis concerned older children, more severe patients (according to the triage scale), and had a higher hospitalization rate (Table 1). Under 12 years, the rate of admissions for anaphylaxis was higher in boys than in girls (5.27/10,000 vs 3.25/10,000, p = 0.006). Conversely, above 12 years old, this rate was higher in girls (16.25/10,000 vs 9.25/10,000, p = 0.011).
Admissions with confirmed anaphylaxis
History and clinical presentation
Among visits related to anaphylaxis, 204 (86.8%) fulfilled Sampson’s criteria (Fig. 1 and Table 2), underlying a high predictive value of our coding for correct diagnosis. The mean age was 7.9 years (2 months to 17.7 years) with a sex ratio of 1.27 (n = 114 boys, 55.9%). A history of allergy was reported in 124 files (60.8%). When a previous episode of anaphylaxis was reported (n = 75; 36.8%), an epinephrine auto-injector was available at home. Asthma was reported in 61 files (35.1%), with a prophylactic daily treatment in 24 (39.3%). A total of 174 different children were concerned.
The current anaphylactic episode mainly occurred in private homes (n = 63/111, 56.7%) or schools (n = 32/111, 28.8%), during or after food intake (n = 122/155, 78.8%). Initial symptoms were mucocutaneous (n = 200/204, 98.5%), respiratory (n = 154/204, 75.7%), gastrointestinal (n = 47/204, 23%), cardiovascular (n = 23/204, 11.3%), or neurological (n = 21/204, 10.3%). No cardiac arrest or death was noted.
In 67.2% of admissions, patients self-presented to the ED. At ED admission, 170 cases (83.3%) were classified grade II according to the Ring and Messmer classification and 34 grade III (16.7%). A causal allergen was suspected in 129 cases (63.2%): food allergen (n = 101, 78.3%, with nuts n = 42, 32.5%; fruits n = 17, 16.8%; milk n = 15, 14.9%; egg n = 6, 5.9%; others n = 21, 16.3%), drugs (n = 11, 8.5%), insect venom (n = 6, 4.7%), and other allergens (n = 11, 8.5%). Specific IgEs were measured in 28 cases (13.7%) and tryptase in 42 cases (20.6%).
Treatment
Data concerning treatments administered before arrival at the EDs were available for 201 episodes. Intramuscular epinephrine was administered in 23 cases (11.4%). Admissions with recurrent anaphylaxis received epinephrine fourfold more often than others (16/75 vs 7/126, 21.3% vs 5.6%). Epinephrine was mainly injected by family or school staff (14/23, 60.9%). Other treatments included antihistamines (n = 93/201, 46.3%), steroids (n = 83/201, 41.3%), inhaled beta-2 agonists (n = 26/201, 12.9%), or inhaled epinephrine (n = 5/201, 2.5%).
Data on ED treatments were available for 202 episodes. Intramuscular epinephrine was administered in 45 cases (22.3%). Overall, 65/199 (32.7%) children received epinephrine before arrival or in the ED. The epinephrine injection rate was higher for grade III than for grade II anaphylaxis (84.8% vs 22.3%, p < 0.001; OR = 19.05 [7.05–54.10]). Epinephrine was also more often prescribed when the patients were classified as urgent by triage nurses (level 1 and 2), but in a less important proportion (47.8% vs 24.6%; p < 0.001; OR = 2.81 [1.51–5.21]). Other treatments included antihistamines (n = 156/202, 90.7%), steroids (n = 126/202, 73.3%), inhaled beta-2 agonists (n = 25/202, 14.5%), inhaled epinephrine (n = 10/202, 5.8%), oxygen (n = 9/202, 5.2%), and vascular filling (n = 8/202, 4.7%). The length of stay in the ED was longer (378 min versus 197 min; p < 0.001), and the hospitalization rate higher (76.2% versus 34.1%, p < 0.001) in case of epinephrine administration. To note, all hospitalized children did not received epinephrine (rate of administration 52.7%; n = 48/91). When the child was discharged at home (n = 104/204), an epinephrine auto-injector was prescribed in 33 (31.7%) cases, and an allergist visit planned in 46 (44.2%) cases, solely (Table 2).
Discussion
To our knowledge, this is the first study reporting the link between epinephrine administration and the severity of pediatric anaphylaxis. Our study also shows that epinephrine is underused during pediatric anaphylaxis management. Among the ED admissions that met Sampson’s anaphylaxis criteria, only one third received epinephrine before or during ED management.
Our study has several limitations due to its retrospective design. First, inclusion criteria (Sampson’s criteria) and Ring and Messmer classification are not, in daily practice, pre-registered in the medical record, but they have been deducted by investigators from the medical data. However, in our institution, Sampson’s criteria and Ring and Messmer classification are specified in the institutional protocol for anaphylaxis management, are well-known to pediatricians, and are well reported by physicians in the medical files. Also because of the retrospective design of the study, many data that would have been relevant to explain the underutilization of epinephrine, such as the specific delay between the onset of symptoms and ED visit, are not available. From this point of view, a qualitative approach exploring the reasons given by pediatricians to justify the absence of adrenaline prescription would also be relevant.
To minimize some bias, and in accordance with other studies [7, 12, 13, 22], we have carefully selected our cases of anaphylaxis following Sampson’s criteria. First, we have compared the visits for possible allergic reactions (urticaria and “nonspecific allergy”) to those for anaphylaxis, and we have shown that visits for anaphylaxis clearly concerned more severe patients as assessed by the clinical score value and the hospitalization rate. Second, when analyzing all the files coded as anaphylaxis, we have observed that anaphylaxis was correctly coded in 86.8% of the cases, better than the 63.1% figure reported by Walsh in the same context [29]. In another study in Australian children, ED diagnosis of anaphylaxis had a sensitivity of 43.2% and specificity of 97.9% [27].
Admissions with confirmed anaphylaxis had the same clinical characteristics as these described in the most recently published studies [10, 21, 24, 30]: age around 6–7 years, boys, mainly mucocutaneous symptoms, and food trigger. Our results also confirm the female predominance of anaphylaxis in children older than 12 years [1]. In keeping with the European Anaphylaxis Registry [11], a previous anaphylactic episode was found in about one third of current admissions for anaphylaxis. The utility of assessing tryptase and specific IgE is still a matter of debate [8], and these laboratory tests were seldom prescribed during the ED stay. Our rate of hospitalization was in agreement with the literature, between 36 and 54% [9, 15].
We have retrospectively classified anaphylaxis episodes according to the Ring and Messmer severity classification. Grade III cases (16.7% of our patients) presented well-known risk factors of severe anaphylaxis, such as asthma or allergy [3], more frequently than grade II cases. They also had higher blood tryptase levels and rate of hospitalization. Others have defined the anaphylaxis severity on the basis of anaphylaxis outcome [10, 14], estimating at 13% to 34.3% the part of severe anaphylaxis. Using this definition, severe anaphylaxis has been linked to drug allergy [10], or fewer administrations of epinephrine [14].
Current treatment recommendations for anaphylaxis highlight a prompt intramuscular epinephrine injection as the gold standard allowing to reduce morbidity, mortality, and hospitalization [16, 26]. However, the use of epinephrine is still insufficient in almost all pediatric studies. Prehospital administration of epinephrine varies in the literature from 36 to 62% [2, 6, 21, 28]. Anaphylaxis occurring at school and a one-organ system involvement would be favoring factors for use [21]. Our rate of ED administration of epinephrine is similar to that described in other European pediatric EDs that has nevertheless doubled between 2011 and 2014 [11]. In USA, EDs’ epinephrine prescription concerns about one admission among three for the first episode [24, 30], increasing to two admissions among three in case of recurrence [17]. In Asia, epinephrine is given in about 90% of the admissions for anaphylaxis [10, 14]. Such gaps between medical epinephrine prescriptions in EDs may reflect inclusion of different phenotypes of anaphylaxis and/or different prehospital management attitudes among countries.
Our results suggest that pediatricians had an intuitive approach of severity and reserved epinephrine for the most severe admitted children (grade III). This medical behavior ascertainment may be explained by the delay between the ED admission/management and the episode onset, and maybe also by a relative fear of the epinephrine side effects, even if its safety has been clearly demonstrated (21.6% of usually mild and transient side effects, such as tremors, palpitations, anxiety, more frequent among adults) [5]. Indeed, if anaphylaxis signs lowered or disappeared before ED admission, practitioners might have considered that the epinephrine administration was useless. The absence of hemodynamic or respiratory dysfunctions might also contribute to under-prescription as it has been previously shown on the management of a 2-year-old child with peanut-induced anaphylaxis where one in five interviewed pediatricians reported not using epinephrine because of this reason [18]. It also could explain why 15% of grade III anaphylaxis episodes had not received epinephrine in our study. Our data do not allow us to determine whether the behavior of our pediatricians induces more hospitalization, but a lack of epinephrine prescription in nearly half of the hospitalized patients is clearly amazing and questioning. Currently, to improve epinephrine prescription in EDs, the use of auto-injectors should be encouraged. Easy to use, they could also favor family education and encourage a too low medical prescription upon home discharge, as also found in other studies [24, 30]. However, auto-injectors have therefore some limitations such as a too high dose (0.15 mg) for the lightest children, especially < 7.5 kg [26]. In addition, auto-injectors with self-retracted needle should be clearly preferred in order to reduce the risk of lacerations and embedded needles [4]. Finally, better medical education on allergy is clearly required. Indeed, recent attendance of continuing medical education on food allergy induces significantly more epinephrine prescription [18].
Conclusion
This study shows that epinephrine was underused in EDs face to children with anaphylaxis, but that its prescription was intuitively adapted to the clinical severity. Auto-injector devices may enhance the epinephrine use in EDs, as well as family education.
References
Alonso T, Moro Moro M, García M (2015) Epidemiology of anaphylaxis. Clin Exp Allergy 45:1027–1039
Andrew E, Nehme Z, Bernard S, Smith K (2018) Pediatric anaphylaxis in the prehospital setting: incidence, characteristics, and management. Prehosp Emerg Care 19:1–7
Ben-Shoshan M, Clarke AE (2011) Anaphylaxis: past, present and future. Allergy 66:1–14
Brown JC, Tuuri RE, Akhter S, Guerra LD, Goodman IS, Myers SR, Nozicka C, Manzi S, Long K, Turner T, Conners GP, Thompson RW, Park E (2016) Lacerations and embedded needles caused by epinephrine autoinjector use in children. Ann Emerg Med 67:307–315
Cardona V, Ferré-Ybarz L, Guilarte M, Moreno-Pérez N, Gómez-Galán C, Alcoceba-Borràs E, Delavalle MB, Garriga-Baraut T, on behalf of the AdreSCAIC Research Group (2017) Safety of adrenaline use in anaphylaxis: a multicenter register. Int Arch Allergy Immunol 173:171–177
Carrillo E, Hern HG, Barger J (2016) Prehospital administration of epinephrine in pediatric anaphylaxis. Prehosp Emerg Care 20:239–244
de Silva IL, Mehr SS, Tey D, Tang ML (2008) Paediatric anaphylaxis: a 5 year retrospective review. Allergy 63:1071–1076
Farbman KS, Michelson KA (2016) Anaphylaxis in children. Curr Opin Pediatr 28:294–297
Farbman KS, Michelson KA, Neuman MI, Dribin TE, Schneider LC, Stack AM (2017) Reducing hospitalization rates for children with anaphylaxis. Pediatrics 139:e20164114
Ganapathy S, Lwin Z, Ting DH, Goh LS, Chong SL (2016) Anaphylaxis in children: experience of 485 episodes in 1,272,482 patient attendances at a tertiary paediatric emergency department from 2007 to 2014. Ann Acad Med Singap 45:542–548
Grabenhenrich LB, Dölle S, Moneret-Vautrin A, Köhli A, Lange L, Spindler T, Ruëff F, Nemat K, Maris I, Roumpedaki E, Scherer K, Ott H, Reese T, Mustakov T, Lang R, Fernandez-Rivas M, Kowalski ML, Bilò MB, Hourihane JO'B, Papadopoulos NG, Beyer K, Muraro A, Worm M (2016) Anaphylaxis in children and adolescents: the European anaphylaxis registey. J Allergy Clin Immunol 137:1128–1137
Hochstadter E, Clarke A, De Schryver S et al (2016) Increasing visits for anaphylaxis and the benefits of early epinephrine administration: a 4-year study at a pediatric emergency department in Montreal, Canada. J Allergy Clin Immunol 137:1888–1890
Huang F, Chawla K, Järvinen KM, Nowak-Wegrzyn A (2012) Anaphylaxis in a New York City pediatric emergency department: triggers, treatments, and outcomes. J Allergy Clin Immunol 129:162–168
Manuyakorn W, Benjaponpitak S, Kamchaisatian W, Vilaiyuk S, Sasisakulporn C, Jotikasthira W (2015) Pediatric anaphylaxis: triggers, clinical features, and treatment in a tertiary-care hospital. Asian Pac J Allergy Immunol 33:281–288
Michelson KA, Monuteaux MC, Neuman MI (2016) Variation and trends in anaphylaxis care in United States children’s hospitals. Acad Emerg Med 23:623–627
Muraro A, Roberts G, Worm M, Bilò MB, Brockow K, Fernández Rivas M, Santos AF, Zolkipli ZQ, Bellou A, Beyer K, Bindslev-Jensen C, Cardona V, Clark AT, Demoly P, Dubois AEJ, DunnGalvin A, Eigenmann P, Halken S, Harada L, Lack G, Jutel M, Niggemann B, Ruëff F, Timmermans F, Vlieg-Boerstra BJ, Werfel T, Dhami S, Panesar S, Akdis CA, Sheikh A, the EAACI Food Allergy and Anaphylaxis Guidelines Group (2014) Anaphylaxis: guidelines from the European academy of allergy and clinical immunology. Allergy 69:1026–1045
O’Keefe A, Clarke A, St Pierre Y et al (2017) The risk of recurrent anaphylaxis. J Pediatr 180:217–221
Pouessel G, Galand J, Beudouin E et al (2017) The gaps in anaphylaxis diagnosis and management by French physicians. Pediatr Allergy Immunol 28:295–298
Pouessel G, Tanno LK, Claverie C, Lejeune S, Labreuche J, Dorkenoo A, Renaudin JM, Eb M, Leteurtre S, Deschildre A (2018) Fatal anaphylaxis in children in France: analysis of national data. Pediatr Allergy Immunol 29:101–104
Ring J, Messmer K (1997) Incidence and severity of anaphylactoid reactions to colloid volume substitutes. Lancet 309:466–469
Robinson M, Greenhawt M, Stukus DR (2017) Factors associated with epinephrine administration for anaphylaxis in children before arrival to the emergency department. Ann Allergy Asthma Immunol 119:164–169
Russell S, Monroe K, Losek JD (2010) Anaphylaxis management in the pediatric emergency department: opportunities for improvement. Pediatr Emerg Care 26:71–76
Sampson HA, Muñoz-Furlong A, Campbell RL, Adkinson NF Jr, Bock SA, Branum A, Brown SGA, Camargo CA Jr, Cydulka R, Galli SJ, Gidudu J, Gruchalla RS, Harlor AD Jr, Hepner DL, Lewis LM, Lieberman PL, Metcalfe DD, O'Connor R, Muraro A, Rudman A, Schmitt C, Scherrer D, Simons FER, Thomas S, Wood JP, Decker WW (2006) Second symposium on the definition and management of anaphylaxis: summary report—second National Institute of allergy and infectious disease/Food Allergy and Anaphylaxis Network symposium. J Allergy Clin Immunol 117:391–397
Sidhu N, Jones S, Perry T, Thompson T, Storm E, Melguizo Castro MS, Nick TG (2016) Evaluation of anaphylaxis management in a pediatric emergency department. Pediatr Emerg Care 32:508–513
Simons FER (2010) Anaphylaxis. J Allergy Clin Immunol 125:S161–S181
Sischerer SH, Simons FER (2017) Section on allergy and immunology. Epinephrine for first-aid management of anaphylaxis. Pediatrics 139:e20164006
Thomson H, Seith R, Craig S (2017) Inaccurate diagnosis of paediatric anaphylaxis in three Australian emergency departments. J Paediatr Child Health 53:698–704
Tiyyagura GK, Arnold L, Cone DC, Langhan M (2014) Pediatric anaphylaxis management in the prehospital setting. Prehosp Emerg Care 18:46–51
Walsh KE, Cutrona SL, Foy S, Baker MA, Forrow S, Shoaibi A, Pawloski PA, Conroy M, Fine AM, Nigrovic LE, Selvam N, Selvan MS, Cooper WO, Andrade S (2013) Validation of anaphylaxis in the Food and Drug Administration’s mini-sentinel. Pharmacoepidemiol Drug Saf 22:1205–1213
Wright CD, Longjohn M, Lieberman PL, Lieberman JA (2017) An analysis of anaphylaxis cases at a single pediatric emergency department during a 1-year period. Ann Allergy Asthma Immunol 118:461–464
Author information
Authors and Affiliations
Contributions
JCD, MSL, and GN conceived the study and designed the trial. JCD, JV, MSL, PM, AB, AC, and GV supervised data collection. GN provided statistical advice on study design and analyzed the data. JCD, MSL, and JV drafted the manuscript, and all authors contributed substantially to its revision. JCD takes responsibility for the paper as a whole.
Corresponding author
Ethics declarations
This study was approved by the Ethics Committee of the French Society of Pediatrics (Société Française de Pédiatrie, No. 2016_018_2) and the National Commission of Informatics and Liberty (Commission Nationale Informatique et Libertés, No. 2016-16).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Additional information
Communicated by Nicole Ritz
Rights and permissions
About this article
Cite this article
Dubus, JC., Lê, MS., Vitte, J. et al. Use of epinephrine in emergency department depends on anaphylaxis severity in children. Eur J Pediatr 178, 69–75 (2019). https://doi.org/10.1007/s00431-018-3246-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-018-3246-3