Abstract
Introduction
Chronic oxygen dependency (COD) is a common adverse outcome of very premature birth. It is, therefore, important to develop an accurate and simple predictive test to facilitate targeting of interventions to prevent COD. Our aim was to determine if a simple score based on respiratory support requirements predicted COD development.
Methods
A retrospective study of 136 infants, median gestation age (GA) 28 weeks (range: 23–33 weeks) and a prospective study of 75 infants, median GA 30 weeks (range: 23–32 weeks), were performed. The score was calculated by multiplying the inspired oxygen concentration by the level of respiratory support (mechanical ventilation: 2.5; continuous positive airway pressure: 1.5; nasal cannula or head box oxygen or air: 1.0). Scores were calculated on data from days 2 and 7, and their predictive ability compared to that of the maximum inspired oxygen concentration at those ages and (retrospective study) the results of lung volume measurement.
Results
Infants that were oxygen dependent at 28 days and 36 weeks post-menstrual age (PMA) had higher scores on days 2 (p<0.0001, p<0.0001, respectively) and 7 (p<0.0001, p<0.0001, respectively) than the non-oxygen dependent infants in both the retrospective and prospective cohorts. Construction of receiver operator characteristic curves demonstrated the score performed better than the inspired oxygen level and lung volume measurement results. A score on day 7 >0.323 had 95% specificity and 78% sensitivity in predicting COD at 28 days, and 80% specificity and 73% sensitivity in predicting COD at 36 weeks PMA.
Conclusion
Chronic oxygen dependency can be predicted using a simple scoring system.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chronic oxygen dependency (COD) is a common adverse outcome of very premature birth [14]. Affected babies are likely to have frequent readmissions to hospital during the first 2 years after birth [7], suffer troublesome respiratory symptoms even at school age and have lung function abnormalities in adolescence. To date, only systemically administered corticosteroids have been demonstrated in large randomised trials to reduce the incidence of COD [8], but this treatment has significant side-effects [6, 27]. It is, therefore, important – if systemic corticosteroids are to be used – to restrict this treatment to those infants who are at highest risk of developing COD. Essential to achieving such an aim is the development of an accurate and simple-to-use predictive test; such a tool would also be useful for facilitating targeting of other preventative strategies. Not surprisingly, then, there has been considerable interest in producing a scoring system which could determine the risk of COD by assigning scores to readily available clinical data. A number of such scoring systems have been developed [4, 11, 19, 22–24, 26, 28], but many are complex and thus unlikely to be used by the busy practitioner. Recently, a simple pulmonary score was reported [20] that required assigning scores only to the inspired oxygen concentration, the level of respiratory support and medications received. The score was developed to retrospectively assess the severity of chronic lung disease in infants who had been entered into a multicentre trial of supplemental oxygen for pre-threshold retinopathy of prematurity. The score was shown to be associated with pulmonary morbidity through to a corrected age of 3 months [20]. We, therefore, hypothesised that a modified version of the scoring system, if used in the first week after birth, might predict COD development.
In the first week after birth, infants who develop COD have abnormal lung function, a raised resistance [5], low compliance [3] and low lung volume [16]. As a consequence, there have been studies investigating whether the results of early lung function measurements might predict COD development. We have previously reported that a low functional residual capacity (FRC) on day 2, that is after surfactant treatment had been completed, was a better predictor of COD development than the results of compliance measurements or readily available clinical data [16]. A subsidiary aim of this study, therefore, was to determine whether the modified score performed better than FRC results.
Methods
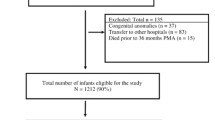
The score was initially assessed on a retrospective data set (retrospective cohort) and then further assessed on a prospective cohort (prospective cohort). The retrospective cohort included premature infants who had participated in a randomised fluid trial [17]. Very low birthweight (VLBW) infants less than 33 weeks of gestational age, ventilated within the first 6 h of birth, had been recruited into the trial, which assessed the impact of fluid balance on the development of COD [17]. Informed written parental consent had been obtained, and the trial was approved by the King’s Healthcare National Health Service (NHS) Trust’s Research Ethics Committee. Informed consent was obtained from all but three parent(s) of consecutively eligible infants. As part of the trial, data were collected regarding the infants gestational age and birthweight and their respiratory support requirements in the first week after birth. In addition, whether the infants had been oxygen dependent at 28 days and/or 36 weeks post-menstrual age (PMA) had been recorded. In a subset of infants entered into the fluid trial [17], lung volume measurements were made on day two. Lung volume had been assessed by measurement of FRC using a helium gas dilution technique and a specially designed infant circuit with a volume of 95 ml. The technique used has been described previously in detail [16]. FRC was estimated twice in each infant, with an interval of 10 min between measurements. An individual’s FRC was expressed as the mean of the paired measurements and related to body weight. The coefficient of repeatability of FRC measurements was 4.38 ml/kg in ventilated and 3.40 ml/kg in non-ventilated infants. For the purposes of this study, data from all infants surviving until discharge home were analysed from the whole of the retrospective cohort and from the subset who had FRC measurements.
Data was then collected prospectively from infants with a gestational age of less than 33 weeks, who were born during the period January 2004–May 2005 and ventilated on day 1 of life (prospective cohort). Parents gave informed written consent for their infants to take part in this study, and the study was approved by the King’s College Hospital Research Ethics Committee. Information was collected regarding the infants’ gestational ages, birthweight, respiratory support requirements during the first week and oxygen dependency status at 28 days and 36 weeks PMA. Data from infants who survived until discharge home were analysed.
The score of Madan et al. [20] was modified such that only the product of the fraction of inspired oxygen (FiO2) and level of ventilatory support was calculated to give the score. Ventilated infants received a score of 2.5, those on nasal or endotracheal continuous positive airway pressure (CPAP) a score of 1.5 and those in nasal cannula or head box supplementary oxygen or no supplementary oxygen a score of 1.0. The relative weight for each item had been decided by a consensus of three neonatologists who developed the score reported by Madan [20]. No scores were given for medications the infants were receiving. Scores were calculated on days 2 and 7, using the maximum inspired concentration and level of respiratory support on each of those days. The scores ranged from 0.21 for a baby self-ventilating in air to 2.5 for a baby who was ventilated in 100% oxygen.
As there was a marked difference in the use of postnatal corticosteroids in our two cohorts, we were interested to see if this influenced the predictive ability of the score. To do this we constructed further ROC curves using data only from infants who did not receive postnatal corticosteroids.
Patients
The retrospective cohort comprised 136 infants with a median gestational age of 28 weeks (range: 23–33 weeks) with a median birthweight of 979 g (range: 592–1494 g). Data were also analysed from 90 of those infants who had a median gestational age of 28 weeks (range: 24–32 weeks) and median birthweight of 1018 g (range: 636–1500 g) and who had had FRC measurements. The prospective cohort consisted of 75 infants with a median gestational age of 30 weeks (range: 23–32 weeks) and a median birthweight of 1246 g (range: 555–2164 g).
In both cohorts, infants born below 29 weeks of gestational age were intubated on the labour ward and also mechanically if the infant was making no or poor respiratory effort. A natural surfactant was given within 2 h of birth to infants who were fully mechanically ventilated and required an inspired oxygen concentration of more than 30%. If a second dose of surfactant was required, this was given 12 h after the first dose. In the retrospective cohort, systemic steroids were administered if, after the first week, infants remained fully ventilator dependent with an inspired oxygen concentration of at least 40% with no improvement for more than 48 h, with a patent ductus arteriosus and infection having been excluded. In the prospective cohort, systemic corticosteroids were only given to infants who remained fully ventilator dependent after the first week after birth, were in high concentrations of oxygen and had shown no improvement for more than 1 week. Forty-nine infants in the retrospective cohort and five in the prospective cohort received systemic corticosteroids. Infants who were hypotensive first received a fluid bolus if appropriate and then escalating levels and number of inotropes. Only if such therapies failed was hydrocortisone given to treat hypotension; very few infants received hydrocortisone. In the retrospective study oxygen saturations after the first weeks after birth were maintained at 95%; this was 92% in the prospective cohort except if the baby had pulmonary hypertension, at which time the oxygen saturation level was maintained at 95%.
Analysis
The results were analysed according to whether or not the infants were oxygen dependent at 28 days or 36 weeks PMA. Differences between infants with and without oxygen dependency were assessed for statistical significance using either the Mann-Whitney U or chi-square test, whichever was appropriate. As the score was made up of inspired oxygen level and the level of respiratory support, we compared the performance of the score to the inspired oxygen level alone and, in the retrospective cohort, to the lung volume results. To do this, receiver operator characteristic (ROC) curves were constructed [1], and the areas under each curve were calculated [9].
Results
Retrospective cohort
Overall (Table 1) and in the subset who had FRC measurements (data not demonstrated) infants who were oxygen dependent at 28 days or 36 weeks PMA were born more prematurely, had higher supplementary oxygen requirements on days 2 and 7 and had higher scores than those who were not. The infants who had FRC measurements and remained COD at 28 days (p<0.0001) and 36 weeks PMA (p<0.0001) had higher scores than those who did not develop COD (Table 1). The construction of ROC curves demonstrated overall (Table 2) and in the subset who had FRC measurements (Table 2) that the score on day7 had the highest areas under the ROC curves for predicting oxygen dependency at 28 days and 36 weeks PMA.
Prospective cohort
The infants who were oxygen dependent at 28 days and 36 weeks PMA were more immature, of a lower birthweight, had a longer duration of ventilation and higher oxygen requirements and had higher scores on days 2 and 7 (Table 3). The score on day 7 had the highest area under the ROC curve with regards to predicting oxygen dependency (Table 4). A score on day 7 greater than or equal to 0.323 had a 95% specificity and 78% sensitivity in predicting COD at 28 days and an 80% specificity and 73% sensitivity in predicting COD at 36 weeks PMA. A score on day 7 greater or equal to 0.42 had a 97% specificity and 72% sensitivity in predicting COD at 28 days and an 85% specificity and 73% sensitivity in predicting COD at 36 weeks PMA. A score on day 7 greater or equal to 0.65 had a 100% specificity and 53% sensitivity in predicting COD at 28 days and a 90% specificity and 55% sensitivity in predicting COD at 36 weeks PMA.
Discussion
We have demonstrated that a simple scoring system can predict COD at both 28 days and 36 weeks PMA. The score was first assessed on a retrospective cohort, and we found it to perform better than FRC results. In the prospective cohort, the score generated even higher areas under the ROC curves. The only major difference in the management of the two cohorts was a reduced use of dexamethasone in the prospective cohort, as between the two study periods the longer term complications of dexamethasone had become appreciated [6, 27]. Our results demonstrate, not surprisingly, that the use of postnatal corticosteroids does influence the predictive ability of data collected during the first week after birth. In the retrospective cohort, in which approximately one third of the infants received corticosteroids, construction of the ROC curves demonstrated higher areas under the curves when data only from infants without corticosteroid use were included. There were some differences in the levels of oxygen saturation used in the two cohorts, yet the scoring system performed well in both, thereby emphasising its robustness.
A number of other scoring systems have been developed. Romagnoli et al. [23] generated a scoring system using logistic regression. Although their system was more complex than ours, as it included scores for birthweight, gestational age, the level of inspired oxygen and the occurrence of complications, it generated an area under the ROC curve for the prediction of oxygen dependency at 28 days of 0.964, which was very similar to that generated by our score in the prospective cohort. These researchers were unable to evaluate the predictive ability of their scoring system for oxygen dependency at 36 weeks PMA because all of their infants who were oxygen dependent at 28 days received dexamethasone. Several studies have compared the predictive ability of various scores. In one study [10] of 188 VLBW infants, two mortality scores (the CRIB and the Berlin admission score) and a morbidity score developed to predict chronic lung disease (Sinkin score) were compared with regard to their ability to predict COD. The Sinkin and Berlin scores had areas under the ROC curves of 0.86 and 0.89, respectively, to predict COD beyond 28 days, and the areas under the curves to predict COD beyond 36 weeks PMA were 0.88 for the Sinkin, 0.81 for the Berlin and 0.77 for the CRIB score. Yoder et al. [28] compared the Sinkin and Ryan regression model scores with regard to predicting oxygen dependency at 36 weeks PMA and a respiratory failure score they generated from the inspired oxygen concentration, the ventilator rate, peak inspiratory pressure and mean airway pressure. Amongst infants less than 30 weeks and having a birthweight of less than 1000 gm, the areas under the ROC curves were 0.81 for the Sinkin score, 0.84 for the Ryan score and 0.90 for the respiratory failure score to predict either COD at 36 weeks PMA or death after 7 days because of severe pulmonary disease. Our score generated similar, if not better areas under the ROC curves to all of the above scores, yet was much simpler to calculate.
In both cohorts, the maximum inspired oxygen concentration on days 2 and 7 differed significantly between the infants who were and were not oxygen dependent at 28 days and 36 weeks PMA. Construction of the ROC curves, however, demonstrated that the “score” had higher areas under the curve, thus multiplying the inspired oxygen level by the level of respiratory support gave better prediction of COD.
Our scoring system was tested on two populations, but both were from the same neonatal intensive care unit and exposed to similar policies regarding respiratory support. We included in both cohorts only infants ventilated in the first 24 h after birth, as we felt such infants were at high risk of developing COD. In other institutions, nasal continuous positive airway pressure is the preferred initial mode or respiratory support rather than intubation and ventilation. Comparisons with data from other centres [2] or to historical controls [15] suggest that such a strategy rather than early intubation and ventilation may be associated with a lower incidence of COD. Yet infants who have minimal or even no respiratory distress in the first few days after birth can develop bronchopulmonary dysplasia. Such infants often have increasing respiratory support requirements by the end of the first week. We therefore hope that our results may encourage centres that operate the so-called “mini touch” technique [12] with regard to respiratory support will assess the predictive ability of our scoring system when used on day 7.
A cut-off of 0.323 had a high sensitivity and specificity in predicting COD at both 28 days and 36 weeks PMA and emphasises that if an infant is either CPAP or ventilator dependent at 7 days they have a very high chance of developing COD. We also report the specificity and sensitivity of two higher cut-offs, thereby highlighting the very high specificity of the score, which is important when considering using such a criteria to implement a change in management. A follow-up study would be helpful to further define the best cut-off.
Bronchopulmonary dysplasia has been variously defined. Initially oxygen dependency beyond 28 days was used [21], then oxygen dependency at 36 weeks PMA was reported to be a more accurate predictor of on-going morbidity [25]. That study, however, was conducted before the routine use of antenatal steroids and postnatal surfactants, and subsequent studies have demonstrated that the 28-day definition was the more accurate predictor [18, 22]. At a recent NIH workshop, the consensus was to diagnose BPD if the infant was oxygen dependent at 28 days and then to diagnose the severity of the illness at a later date chosen according to the gestational age of the infant [13]. Nevertheless, both the “28 day” and “36 week PMA” definitions are still both used. As a consequence it was important for us to assess if our scoring system could predict both outcomes, which it did, particularly when used on day 7. We conclude that a simple score assessing respiratory support requirements can predict COD and highlight infants at high risk of chronic respiratory morbidity.
Abbreviations
- BPD:
-
Bronchopulmonary dysplasia
- COD:
-
Chronic oxygen dependency
- CPAP:
-
Continuous positive airway pressure
- Fi02 :
-
Functional residual capacity
- FRC:
-
Functional residual capacity
- PMA:
-
Post-menstrual age
- ROC:
-
Receiver operator characteristic
- VLBW:
-
Very low birthweight
References
Altman DG, Bland JM (1994) Diagnostic tests 3: receiver operator characteristic plots. Br Med J [Clin Res] 309:188
Avery ME, Tooley WH, Keller JB, Hurd SS, Bryan MH, Cotton RB, Epstein MF, Fitzhardinge PM, Hansen CB, Hansen TN (1987) Is chronic lung disease in low birth weight infants preventable? A survey of eight centres. Pediatrics 79:26–30
Bhutani VK, Abbasi S (1992) Relative likelihood of bronchopulmonary dysplasia based on pulmonary mechanics measured in preterm neonates during the first week of life. J Pediatr 120:605–613
Escobar GJ, Shaheen SM, Breed EM, Botas C, Greene JD, Yoshida CK, Zupancic J, Newman TB (2004) Richardson score predicts short-term adverse respiratory outcomes in newborns >34 weeks gestation. J Pediatr 145:754–760
Goldman SL, Gerhardt T, Sonni R, Feller R, Hehre D, Tapia JL, Bancalari E (1983) Early prediction of chronic lung disease by pulmonary function testing. J Pediatr 102:613–617
Greenough A (1998) Gains and losses from dexamethasone for neonatal chronic lung disease. Lancet 352:835–836
Greenough A, Alexander J, Burgess S, Chetcuti PA, Cox S, Lenney W, Turnball F, Shaw NJ, Woods A, Boorman J, Coles S, Turner J (2002) Home oxygen status on rehospitalisation and primary care requirements of chronic lung disease infants. Arch Dis Child 86:40–43
Halliday HL, Ehrenkranz RA, Doyle LW (2003) Moderately early (7–14 days) postnatal corticosteroids for preventing chronic lung disease in preterm infants. Cochrane Database Syst Rev 1:CD001145
Hanley JA, McNeil BJ (1982) The meaning and use of the area under a receiver operator characteristic (ROC) curve. Radiology 143:29–36
Hentschel J, Friedel C, Maier RF, Bassir C, Obladen M (1998) Predicting chronic lung disease in very low birthweight infants; comparison of three scores. J Perinat Med 26:378–383
Hernandez-Ronquillo L, Tellez-Zenteno JF, Weder-Cisneros N, Salinas-Ramirez V, Zapata-Pallagi JA, da Silva O (2004) Risk factors for the development of bronchopulmonary dysplasia: a case control study. Arch Med Res 35:549–553
Jacobsen T, Gronvall J, Petersen S, Andersen GE (1993) “Minitouch” treatment of very low-birth-weight infants. Acta Pediatr 82:934–938
Jobe AH, Bancalair E (2001) Bronchopulmonary dysplasia. NICHD-NHLBI-ORD Workshop. Am J Respir Crit Care Med 163:1723–1729
Johnson AH, Peacock JL, Greenough A, Marlow N, Limb ES, Marston L, Calvert SA, for the United Kingdom Oscillation Study Group (2002) High frequency oscillatory ventilation for the prevention of chronic lung disease of prematurity. New Engl J Med 347:633–642
Kamper J, Wulff K, Larsen C, Lindequist S (1993) Early treatment with nasal continuous positive pressure in very low birthweight infants. Acta Paediatr 82:193–197
Kavvadia V, Greenough A, Dimitriou G (2000a) Early prediction of chronic oxygen dependency by lung function test results. Pediatr Pulmonol 29:19–26
Kavvadia V, Greenough A, Dimitriou G, Hooper R (2000b) Randomised trial of fluid restriction in ventilated very low birthweight infants. Arch Dis Child Fetal Neonatal Ed 83:F91–F96
Kinali M, Greenough A, Dimitriou G, Yuksel B, Hooper R (1999) Chronic respiratory morbidity following premature delivery-prediction by prolonged respiratory support requirement? Eur J Pediatr 158:493–496
Kim YD, Kim EA, Kim KS, Pi SY, Kang W (2005) Scoring method for early prediction of neonatal chronic lung disease using modified respiratory parameters. J Korean Med Sci 20:397–401
Madan A, Brozanski BS, Cole CH, Oden NL, Cohen G, Phelps DL (2005) A pulmonary score for assessing the severity of neonatal chronic lung disease. Pediatrics 115:450–457
Northway WH Jr, Moss RB, Carlisle KB, Parker BR, Popp RL, Pitlick PT, Eichler I, Lamm RL, Brown BW Jr (1990) Late pulmonary sequalae of bronchopulmonary dysplasia. N Engl J Med 323:1793–1799
Palta M, Sadek M, Barnet JH, Evans M, Weinstein MR, McGuinness G, Peters ME, Gabbert D, Fryback D, Farrel P for the Newborn Lung Project (1998) Evaluation of criteria for chronic lung disease in surviving very low birthweight infants. J Pediatr 132:57–63
Romagnoli C, Zecca E, Tortorolo L, Vento G, Tortorolo G (1998) A scoring system to predict the evolution of respiratory distress syndrome into chronic lung disease in preterm infants. Intensive Care Med 24:476–480
Ryan SW, Nycyk J, Shaw BNJ (1996) Prediction of chronic neonatal lung disease on day 4 of life. Eur J Pediatr 155:668–671
Shennan AT, Dunn MS, Ohlsson A, Lennox K, Hoskins EM (1988) Abnormal pulmonary outcomes in premature infants: prediction fro oxygen requirement in the neonatal period. Pediatrics 82:527–552
Sinkin RA, Cox C, Phelps DL (1990) Predicting risk for bronchopulmonary dysplasia: selection criteria for clinical trials. Pediatrics 85:728–736
Yeh TF, Lin YJ, Huang CC, Chen YJ, Lin CH, Lin HC, Hsieh WS, Lien YJ (1998) Early dexamethasone therapy in preterm infants: a follow up study. Pediatrics 101:e7
Yoder BA, Anwar MU, Clark RH (1999) Early prediction of neonatal chronic lung disease: a comparison of three scoring methods. Pediatr Pulmonol 27:388–394
Acknowledgements
Dr. Caroline May is supported by the Charles Wolfson Foundation and Dr. Valia Kavvadia and Dr. Gabriel Dimitriou were supported by the South Thames Regional Health Authority’s Research and Development Directorate and by the Children Nationwide/Nestle Research Fellowship, respectively. We thank Mrs. Deirdre Gibbons for secretarial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
May, C., Kavvadia, V., Dimitriou, G. et al. A scoring system to predict chronic oxygen dependency. Eur J Pediatr 166, 235–240 (2007). https://doi.org/10.1007/s00431-006-0235-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-006-0235-8