Abstract.
Human papillomavirus (HPV) infections are thought to be one of the causal factors in the development of head and neck squamous cell carcinomas (HNSCC), particularly in tumors arising from the Waldeyer's tonsillar ring. We screened 98 carefully stratified HNSCC and different control tissues for the presence of HPV DNA by nested polymerase chain reaction (PCR) specific for genital- and Epidermodysplasia verruciformis (EV)-associated HPVs and by HPV16-specific single step PCR. Typing was performed by direct sequencing and/or sequencing of cloned amplimers. On average HNSCC showed rather low HPV DNA prevalences; 18% of the oral cavity cancers, 8% of nasopharyngeal cancers, 25% of hypopharyngeal cancers and 7% of laryngeal cancers were HPV DNA positive. In contrast, HPV sequences could be detected in 45% of the oropharyngeal cancers, particularly tonsillar carcinomas (58%). Tonsillar carcinomas were significantly more likely to be HPV positive than tumors from any other site (P<0.001). All tonsillar cancers contained oncogenic HPV types, predominantly HPV16 (13 of 14; 93%). Unaffected tonsils were available from two of these patients, but both tested negative for HPV DNA. Furthermore, no HPV DNA could be found in tonsillar biopsy specimens from control groups. Localization and load of HPV DNA was determined in HPV16-positive tonsillar carcinomas, their metastases and in unaffected mucosa using laser-assisted microdissection and subsequent real time fluorescence PCR. We demonstrated that the HPV genome is located in the cancer cells, whereas the infection of normal mucosa is a rare event. Quantification of HPV16 DNA in samples of seven patients yielded viral loads from 6 to 153 HPV DNA copies per β-globin gene copy and the load values in both locations were roughly comparable. These loads are comparable with data shown for other HPV-associated lesions. Statistical evaluation of data related to clinicopathological parameters showed a significant correlation of the HPV positivity of tonsillar carcinomas with tumor grading (P=0.008) and alcohol consumption (P=0.029). Taken together our findings show a preferential association of HPV DNA with tonsillar carcinomas. Furthermore our results argue for HPV-positive tonsillar carcinomas representing a separate tumor entity, which is less dependent on conventional HNSCC risk factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Human papillomavirus (HPV) infections are responsible for a broad spectrum of epithelial tumors of skin and mucosa, ranging from benign warts, papillomas and dysplasias to malignant lesions of the cervix uteri [7, 25]. The causal role of HPV infection in carcinogenesis and the involved molecular mechanisms could be elucidated best in the case of cervical carcinomas. Overwhelming epidemiological and molecular evidence indicates the existence of several so-called high-risk HPV types (e.g., HPV16 or 18), which are consistently associated with genital malignancies and encode proteins that deregulate the control network of cellular proliferation/differentiation processes [32]. Furthermore, other neoplasias of men are currently being intensively examined in view of their possible etiological association with HPV infections. According to epidemiological data, the most probable candidates are the non-melanoma skin cancers and some of the head and neck squamous cell carcinomas (HNSCC) [6, 20].
HNSCC represent 6% of all new cancer cases [18]. Cigarette smoking and alcohol consumption have been shown to significantly increase the risk for developing HNSCC [28]. However, a significant proportion of HNSCC patients do not contribute to these risk groups. Despite correction for stage and localization of the tumors the prognosis is diverse [3], suggesting that HNSCC are a heterogenic group of tumors with different cell biology and etiology. Tonsillar carcinomas are the most common head and neck malignancy, surpassed only by laryngeal tumors [8]. There are several recent studies indicating that there is a correlation between HPV infection and tonsillar carcinomas.
We reported on the prevalence of HPV DNA in a series of carefully stratified HNSCC, homologous metastases and different control groups using highly sensitive PCR protocols. By laser-assisted microdissection we tested for the association of HPV DNA with either neoplastic or normal epithelial cells [13]. As shown in retrospective case-control studies high HPV16 load is associated with a strongly elevated risk of developing cervical carcinomas [11, 31]. Focusing on tonsillar carcinomas, quantification of HPV DNA was carried out using a real time quantitative PCR assay in primary and metastatic tumor. The PCR analyses of 98 HNSSC cases employing both group- and type-specific primers revealed 26% of the tested samples to be HPV positive. Depending on the anatomical location of the tumors significant differences in the HPV DNA prevalence were detected [13]. On average, HNSCC showed rather low HPV-DNA prevalences; 18% of the oral cavity cancers, 8% of nasopharyngeal cancers, 25% of hypopharyngeal cancers and 7% of laryngeal cancers were HPV DNA positive. In contrast, HPV sequences could be detected in 45% of the oropharyngeal cancers, and here especially among tumors arising from the palatine tonsils (58%). The other sites of the oropharynx were HPV negative or showed low HPV DNA prevalences (base of the tongue, 17%) [13]. Sequence analysis revealed that 84% of tested DNA samples (21 out of 25 HPV-positive biopsy samples) carried sequences of the HPV type 16, all showing 99–100% homology with the HPV16 prototype described by Seedorf et al. [24]. HPV33 was only detected once in a biopsy of a tonsillar carcinoma. Four of the analyzed tumors contained sequences of typical cutaneous papillomaviruses: HPV19 (1 hypopharyngeal and 1 tongue cancer), IA09 (accession no. AFO42003, nasopharyngeal tumor) and ADX1 (accession no. AJ001483, tonsillar carcinoma). In two tonsillar cancers double infections by HPV5/HPV16 and ADX1/HPV16 were identified. Normal mucosa controls of patients with HPV-positive tumors showed HPV DNA in 7 of 11 (64%) cases. HPV DNA was not detected in any of the tonsillar specimens from the non-tumor controls.
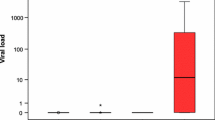
Viral load determination was performed for 8 patients with HPV16 and for 1 patient with HPV33 positive tonsillar carcinoma. Viral loads were determined in microdissected samples from neoplastic and histologically normal areas of primary tumor and lymph node metastasis and from unaffected mucosa. The mean HPV16 loads in primary tumors ranged from 6 to 153 copies per β-globin gene copy [13]. These values are comparable to the HPV16 loads we measured in other HPV-associated lesions as erythroplasia of Queyrat [29] and cervical scrapes of dysplasia patients (Weissenborn et al., submitted). Load values in the primary tumors and metastases were roughly comparable. In contrast, except for one case all the microdissected samples of histologically normal mucosa from tumor margins were HPV negative. For the tonsillar subset, a significant correlation of the HPV status and primary tumor grading, alcohol consumption and cigarette smoking was found. Poorly differentiated tumors were more likely to be HPV positive (P=0.008). Patients with HPV-positive tumors had a significantly lower alcohol intake per day than patients with HPV-negative tumors (P=0.029). Further, there was a trend for patients with HPV-positive tumors to be nonsmokers or light smokers (P=0.037). Statistical testing revealed no significant correlation of the HPV status with TMN stage or local tumor control. Furthermore, no significant correlation between HPV-positive and -negative carcinomas in regard of the median age (P=0.97, t-test) or gender (P=0.44, Chi-square test) could be seen [13].
Summarizing, using PCR methods capable of detecting more than 90% of presently known HPV-types we found papillomavirus sequences in 58% (14 of 24) of the tonsillar carcinomas. All of these contained oncogenic HPV types, predominantly HPV16 (13 of 14; 93%). In other tumor localizations oncogenic HPV types were detected in only 8% (6 out of 74). These results show that tonsillar carcinomas are significantly more likely to be HPV positive than tumors from all other sites (P<0.001). In view of the number of tested tumors (n=98), and especially because of the high sensitivity of the employed detection methods, we believe this estimate to reflect the true prevalence of HPV DNA. Our prevalence data based on the so-far largest patient collective are in line with previous reports [2, 6, 19, 27, 30].
The cumulative data of HPV DNA prevalence among HNSCC reveal carcinomas arising from the palatine tonsil as being preferentially infected with HPV16, suggesting a potential role of this virus in the tumor etiology. The central question is whether HPV-associated tonsillar carcinomas are a different tumor entity, distinct from other HNSCC in regard of risk factors, tumor biology and prognosis. Serological studies have shown that HPV-16-seropositive subjects have an extensively increased risk (odds ratio 10.2) for the development of a tonsillar carcinoma [14, 17]. Furthermore, patients with human papillomavirus (HPV)-associated anogenital cancers and husbands of women with HPV-associated neoplastic lesions of the cervix have an increased risk for tonsillar squamous cell carcinoma, suggesting HPV as a risk factor [4, 10]. Indeed, known risk factors for cervical carcinomas, such as younger age at first intercourse, high number of sex partners and a history of genital warts were also shown to increase the chance for developing HPV-positive HNSCC [23]. These findings led to the hypothesis regarding HPV-positive HNSCC as a sexually transmitted disease [4, 5].
Sexual transmission of oncogenic HPV is generally accepted for the anogenital region [7, 25], whereas the routes of infection for the oropharyngeal region are unclear. The fact that 51.7% of buccal samples from 267 healthy children aged 3–11 years were shown to be HPV16 positive suggests that vertical transmission is an event. In contrast, we did not detect HPV sequences in the tonsils and adenoids of 19 children in a similar age group, indicating transmission later in life (Klussmann et al., submitted).
There are only rare data on the prevalence of oncogenic HPV in the normal mucosa of the oropharynx in healthy adults (for overview see [16]). We did not detect HPV DNA in tonsils of the adult control group using highly sensitive PCR techniques [13]. Consequently, infection of the oropharynx with oncogenic HPV seems to be an infrequent event, in contrast to the genital region where in many cases normal cervix smears contain oncogenic HPV. HPV-associated cervical carcinomas mostly arise from the so-called transformation zone, which is more susceptible to HPV infections due to exposition of the basal layer of the epithelium. The anatomic structure of the tonsils is characterized by deep invaginations of mucosal surface (tonsillar crypts), which can probably trap infectious agents simply by lowering their mechanical clearance. These crypts are lined with a monolayer epithelium showing similarities to basal keratinocytes of mucosa. Evidence that the HPV-positive tonsillar carcinomas arise in the tonsillar crypts is also gained from histological and clinical experience. In Fig. 1 a histological section of a typical HPV-positive tonsillar carcinoma is shown. The carcinoma is located exclusively in the crypt and has not reached the tonsillar surface in any section, indicating the origin of the carcinoma from the crypt epithelium. This is in line with the clinical observation that first symptoms in these patients are often the cervical masses, indicating the lymph node metastasis, whereas the tonsils macroscopically show no abnormality [8].
Concerning the different tumor pathology of HPV-positive tonsillar carcinomas, a correlation with poor keratinization [30] and a decreased expression of the retinoblastoma tumor suppressor protein has been reported [1]. This is consistent with our data showing a significant correlation of HPV positivity with tumor grading, suggesting an influence of the viral oncogenes on tumor cell differentiation. The influence of HPV on the cell cycle in tonsillar carcinomas is also shown by preliminary data concerning the expression of the p16 tumor suppressor protein (Klussmann, manuscript in preparation). p16 is a cyclin-dependent kinase (CDK) inhibitor that decelerates the cell cycle by inactivating CDKs that phosphorylate retinoblastoma (Rb) protein [15]. The p16 expression is markedly influenced by the status of Rb expression [21], and p16 overexpression has been demonstrated in cervical cancers as a result of functional inactivation of Rb by HPV E7 protein [12, 22]. Using immunohistochemical techniques, we demonstrated a strong and diffuse expression of p16 in the HPV16-positive tonsillar carcinomas, whereas the HPV-negative tonsillar carcinomas showed no or only focal p16 expression. This may indicate the effect of functional expression of E7.
Our finding that patients with HPV-positive carcinomas had a significantly lower exposure to the known risk factors for HNSCC argues for HPV-positive tonsillar carcinomas representing a different tumor entity. Interestingly, a better prognosis for patients with HPV-positive HNSCC is reported [1, 6]. We [13] and others [9] could not confirm this, probably due to short follow-up periods. It has been suggested that the reason for better survival of the HPV-positive tumor patients might be a higher susceptibility to radiation or chemotherapy [1], but this hypothesis has not yet be substantiated. Speculations that the HPV-positive HNSCC might be associated with a younger age of the patients are refuted by our findings [13] and earlier investigations [6, 26].
Taken together, our data strongly supports the idea that HPV-positive tonsillar carcinomas represent a tumor entity separate from other HNSCC.
References
Andl T, Kahn T, Pfuhl A, Nicola T, Erber R, Conradt C, Klein W, Helbig M, Dietz A, Weidauer H, Bosch FX (1998) Etiological involvement of oncogenic human papillomavirus in tonsillar squamous cell carcinomas lacking retinoblastoma cell cycle control. Cancer Res 58:5–13
Brandsma JL, Abramson AL (1989) Association of papillomavirus with cancers of the head and neck. Arch Otolaryngol Head Neck Surg 115:621–625
DeVita VT, Helman S, Rosenberg SA (1997) Principles and practice of oncology. Lippincott Williams & Wilkins, Philadelphia
Frisch M, Biggar RJ (1999) Aetiological parallel between tonsillar and anogenital squamous-cell carcinomas. Lancet 354:1442–1443
Gillison ML, Koch WM, Shah KV (1999) Human papillomavirus in head and neck squamous cell carcinoma: are some head and neck cancers a sexually transmitted disease? Curr Opin Oncol 11:191–199
Gillison ML, Koch WM, Capone RB, Spafford M, Westra WH, Wu L, Zahurak ML, Daniel RW, Viglione M, Symer DE, Shah KV, Sidransky D (2000) Evidence for a causal association between human papillomavirus and a subset of head and neck cancers. J Natl Cancer Inst 92:709–720
Gross GE, Barasso R (1997) Human papillomavirus infection. Ullstein Mosby, Wiesbaden
Guay ME, Lavertu P (1995) Tonsillar carcinoma. Eur Arch Otorhinolaryngol 252:259–264
Haraf DJ, Nodzenski E, Brachman D, Mick R, Montag A, Graves D, Vokes EE, Weichselbaum RR (1996) Human papilloma virus and p53 in head and neck cancer: clinical correlates and survival. Clin Cancer Res 2:755–762
Hemminki K, Dong C and Frisch M (2000) Tonsillar and other upper aerodigestive tract cancers among cervical cancer patients and their husbands. Eur J Cancer Prev 9:433–437
Josefsson AM, Magnusson PK, Ylitalo N, Sorensen P, Qwarforth-Tubbin P, Andersen PK, Melbye M, Adami HO, Gyllensten UB (2000) Viral load of human papilloma virus 16 as a determinant for development of cervical carcinoma in situ: a nested case-control study. Lancet 355:2189–2193
Klaes R, Friedrich T, Spitkovsky D, Ridder R, Rudy W, Petry U, Dallenbach-Hellweg G, Schmidt D, Knebel DM von (2001) Overexpression of p16(INK4A) as a specific marker for dysplastic and neoplastic epithelial cells of the cervix uteri. Int J Cancer 92:276–284
Klussmann JP, Weissenborn S, Wieland U, Dries V, Kolligs J, Jungehuelsing M, Eckel HE, Dienes HP, Pfister H, Fuchs P (2001) Prevalence, distribution and viral load of human papillomavirus 16 DNA in tonsillar carcinomas. Cancer 92:2875–2884
Klussmann JP, Weissenborn S, Fuchs PG (2001) Human papillomavirus infection as a risk factor for squamous-cell carcinoma of the head and neck. N Engl J Med 345:376
Koh J, Enders GH, Dynlacht BD, Harlow E (1995) Tumour-derived p16 alleles encoding proteins defective in cell-cycle inhibition. Nature 375:506–510
McKaig RG, Baric RS, Olshan AF (1998) Human papillomavirus and head and neck cancer: epidemiology and molecular biology. Head Neck 20:250–265
Mork J, Lie AK, Glattre E, Hallmans G, Jellum E, Koskela P, Moller B, Pukkala E, Schiller JT, Youngman L, Lehtinen M, Dillner J (2001) Human papillomavirus infection as a risk factor for squamous-cell carcinoma of the head and neck. N Engl J Med 344:1125–1131
Parkin DM, Pisani P, Ferlay J (1999) Estimates of the worldwide incidence of 25 major cancers in 1990. Int J Cancer 80:827–841
Paz IB, Cook N, Odom-Maryon T, Xie Y, Wilczynski SP (1997) Human papillomavirus (HPV) in head and neck cancer. An association of HPV 16 with squamous cell carcinoma of Waldeyer's tonsillar ring. Cancer 79:595–604
Pfister H, Schegget J ter (1997) Role of HPV in cutaneous premalignant and malignant tumors. Clin Dermatol 15:335–347
Sakaguchi M, Fujii Y, Hirabayashi H, Yoon HE, Komoto Y, Oue T, Kusafuka T, Okada A, Matsuda H (1996) Inversely correlated expression of p16 and Rb protein in non-small cell lung cancers: an immunohistochemical study. Int J Cancer 65:442–445
Sano T, Oyama T, Kashiwabara K, Fukuda T, Nakajima T (1998) Expression status of p16 protein is associated with human papillomavirus oncogenic potential in cervical and genital lesions. Am J Pathol 153:1741–1748
Schwartz SM, Daling JR, Doody DR, Wipf GC, Carter JJ, Madeleine MM, Mao EJ, Fitzgibbons ED, Huang S, Beckmann AM, McDougall JK, Galloway DA (1998) Oral cancer risk in relation to sexual history and evidence of human papillomavirus infection. J Natl Cancer Inst 90:1626–1636
Seedorf K, Krammer G, Durst M, Suhai S, Rowekamp WG (1985) Human papillomavirus type 16 DNA sequence. Virology 145:181–185
Shah KV, Howley PM (1996) Papillomaviruses. In: Fields BN, Knipe DM, Howley PM, Channock RM, Melnick JL, Monath TP, Roizman B (eds) Fields virology. Lippincott-Raven, Philadelphia, pp 2077–2109
Sisk EA, Bradford CR, Jacob A, Yian CH, Staton KM, Tang G, Harris MO, Carey TE, Lancaster WD, Gregoire L (2000) Human papillomavirus infection in "young" versus "old" patients with squamous cell carcinoma of the head and neck. Head Neck 22:649–657
Snijders PJ, Cromme FV, Brule AJ van den, Schrijnemakers HF, Snow GB, Meijer CJ, Walboomers JM (1992) Prevalence and expression of human papillomavirus in tonsillar carcinomas, indicating a possible viral etiology. Int J Cancer 51:845–850
Vokes EE, Weichselbaum RR, Lippman SM, Hong WK (1993) Head and neck cancer. N Engl J Med 328:184–194
Wieland U, Jurk S, Weissenborn S, Krieg T, Pfister H, Ritzkowsky A (2000) Erythroplasia of queyrat: coinfection with cutaneous carcinogenic human papillomavirus type 8 and genital papillomaviruses in a carcinoma in situ. J Invest Dermatol 115:396–401
Wilczynski SP, Lin BT, Xie Y, Paz IB (1998) Detection of human papillomavirus DNA and oncoprotein overexpression are associated with distinct morphological patterns of tonsillar squamous cell carcinoma. Am J Pathol 152:145–156
Ylitalo N, Sorensen P, Josefsson AM, Magnusson PK, Andersen PK, Ponten J, Adami HO, Gyllensten UB, Melbye M (2000) Consistent high viral load of human papillomavirus 16 and risk of cervical carcinoma in situ: a nested case-control study. Lancet 355:2194–2198
Zur Hausen H (1999) Papillomaviruses in human cancers. Proc Assoc Am Physicians 111:581–587
Acknowledgements.
This work was supported by funding from the Jean Uhrmacher Foundation and from the Köln Fortune-Program of the Faculty of Medicine, University of Cologne (No. 63/1999) and a grant from the Center for Molecular Medicine of Cologne (ZMMK).
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented in part at the Joint Meeting of the Arbeitsgemeinschaft Dermatologische Infektiologie der Deutschen Dermatologischen Gesellschaft and the Arbeitsgemeinschaft Oralpathologie on "Oral Viral Infection and Oral Tumors" in Rostock, Germany, 6–7 July 2001
Rights and permissions
About this article
Cite this article
Klussmann, J.P., Weissenborn, S.J., Wieland, U. et al. Human papillomavirus-positive tonsillar carcinomas: a different tumor entity?. Med Microbiol Immunol 192, 129–132 (2003). https://doi.org/10.1007/s00430-002-0126-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-002-0126-1