Abstract
In the present study, based on injections of retro- or retro-anterograde tracers at the cortical level, we analyzed the amygdalar connections of the caudal ventrolateral prefrontal areas 45A and 45B of the macaque and compared them with those of the adjacent areas 8/FEF, 8r, 46v, and 12r. The results showed that areas 45A and 45B display reciprocal amygdalar connections, which appear to be considerably richer than those of their neighboring areas. Specifically, these two areas are a target of differentially weighted projections originating predominantly from the magnocellular and the intermediate subdivisions of the basal nucleus and are a source of projections mostly directed to the magnocellular subdivision of the basal nucleus and the dorsal part of the lateral nucleus. The present data, together with previous data on the thalamic connectivity of areas 45A and 45B (Contini et al. Eur J Neurosci 32:1337–53, 2010), suggest that direct and indirect—trans-thalamic—amygdalar connectivity is a characterizing connectional feature of these two areas. Specifically, the amygdalar connections of area 45A, for which a role in communication behavior has been proposed, could convey information on the emotional significance of communicative signals to this area, where it could play a crucial role in guiding appropriate social interactions. Furthermore, the amygdalar connections of area 45B, possibly involved in higher-order aspects of visual guidance of gaze, could convey information related to the relevance of visual stimuli, which could contribute to a representation of priority maps in this VLPF area.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The caudal part of the macaque ventrolateral prefrontal (VLPF) cortex hosts two architectonically distinct areas: a caudal one—45B—located ventrally in the prearcuate cortex and a rostral one—45A—located on the rostrally adjacent inferior frontal convexity (Petrides and Pandya 1994, 2002; Gerbella et al. 2007). These two areas display connectivity patterns clearly distinguishing them from one another and from their neighboring areas, suggesting a differential role in non-spatial information processing (Gerbella et al. 2010). Specifically, the robust extraprefrontal connectivity with higher-order auditory and multisensory areas of the superior temporal gyrus, as well as functional data showing involvement in the processing of communication stimuli (see Romanski and Averbeck 2009) suggest a role in communication behavior for area 45A. In contrast, the robust connectivity with inferotemporal ventral visual stream areas and with frontal (8/FEF, 8r, supplementary eye field) and parietal (LIP) oculomotor areas suggest a role in higher-order aspects of visually guided oculomotor behavior for area 45B.
Areas 45A and 45B, though differing in their thalamic connectivity (Contini et al. 2010), are both a target of strong projections from the magnocellular subdivision of the thalamic nucleus medialis dorsalis (MDmc), a well-known target of the amygdala (Porrino et al. 1981; Aggleton and Mishkin 1984; Russchen et al. 1987), suggesting that these two areas are the recipient of robust trans-thalamic amygdalar input. Amygdalar projections are important for affective and purposive behavior and can reach the frontal cortex directly, or indirectly, through the MDmc (e.g. Porrino et al. 1981; Aggleton and Mishkin 1984; Russchen et al. 1987; Ray and Price 1993; Ghashghaei et al. 2007). Specifically, it has been suggested that the direct and the indirect pathways would carry more detailed and less detailed, more integrated information, respectively (Russchen et al. 1987; Ray and Price 1993). Areas 45A and 45B could also have direct connections with the amygdala. Indeed, Ghashghaei et al. (2007), after tracer injections in the amygdala, found retro- and anterograde labeling in the location of areas 45A and 45B, but considered this sector as part of area 12. Furthermore, in our previous study focused on the thalamic connectivity of areas 45A and 45B (Contini et al. 2010), we have provided preliminary evidence that these two areas are directly connected to the amygdala.
In the present study, to obtain more detailed information on the involvement of areas 45A and 45B in the “triangular” amygdalo-thalamo-cortical circuits, we analyzed the direct amygdalar connections of these two areas and of their neighbors, based on tracer injections at the cortical level. As these circuits are the possible substrate for the conveyance of emotional and motivational information to the prefrontal cortex (Barbas 2007), these data could be useful to gain additional insight into the possible proposed roles of areas 45A and 45B in communication behavior and in visually guided aspects of gaze, respectively.
Methods
Subjects, surgical procedures, and selection of the injection sites
The present study is based on results from injections of neural tracers placed in the architectonic areas 45A, 45B, 8/FEF, and 8r and in the caudal part of areas 46v and 12r (Fig. 1; Table 1) in eight macaque monkeys (5 Macaca mulatta, 2 Macaca fascicularis, and 1 Macaca nemestrina). All these cases, except for the tracer injection in area 8r in Case 56l, have already been presented in previous studies focused on the cortical connectivity of these VLPF areas (Gerbella et al. 2010, 2012; Borra et al. 2011). The animal handling as well as surgical and experimental procedures complied with the European guidelines (86/609/EEC and 2003/65/EC Directives) and Italian laws in force on the care and use of laboratory animals, and were approved by the Veterinarian Animal Care and Use Committee of the University of Parma and authorized by the Italian Health Ministry.
Composite view of the location of all the injection sites mapped on a template hemisphere. Each injection site is numbered and shown as a black circle. Dashed lines mark cytoarchitectonic borders. IA inferior arcuate sulcus, SA superior arcuate sulcus, C central sulcus, L lateral fissure, P principal sulcus
Under general anesthesia and aseptic conditions, each animal was placed in a stereotaxic apparatus, and an incision was made in the scalp. The skull was trephined to remove the bone overlying the target region, and the dura was opened to expose the VLPF. The criteria for the selection of the injection sites have been described in detail in previous studies (see Table 1). After the tracer injections were placed, the dural flap was sutured, the bone was replaced, and the superficial tissues were sutured in layers. During surgery, hydration was maintained with saline, and temperature was maintained using a heating pad. Heart rate, blood pressure, respiratory depth, and body temperature were continuously monitored. Upon recovery from anesthesia, the animals were returned to their home cages and closely monitored. Dexamethasone and prophylactic broad-spectrum antibiotics were administered pre- and post-operatively. Furthermore, analgesics were administered intra- and post-operatively.
Tracer injections and histological procedures
Once the appropriate site was chosen, the mostly anterograde tracer biotinylated dextran amine [(BDA) 10,000 molecular weight (MW), 10 % 0.1 M phosphate buffer, pH 7.4; Invitrogen], the retro-anterograde tracers dextran conjugated with tetramethylrhodamine [Fluoro-Ruby (FR), 10,000 MW, 10 % 0.1 M phosphate buffer, pH 7.4; Invitrogen] or with lucifer yellow [Lucifer Yellow Dextrane (LYD), 10,000 MW, 10 % in 0.1 M phosphate buffer, pH 7.4; Invitrogen-Molecular Probes] and the retrograde tracers Fast Blue (FB, 3 % in distilled water, Dr Illing Plastics GmbH, Breuberg, Germany) and Diamidino Yellow (DY, 2 % in 0.2 M phosphate buffer at pH 7.2, Dr Illing Plastics) were slowly pressure-injected through a glass micropipette (tip diameter: 50–100 μm) attached to a 1- or 5-μl Hamilton microsyringe (Reno, NV) at about 1.2–1.5 mm below the cortical surface in the inferior frontal convexity, or at different depths in the ventral prearcuate bank. Table 1 summarizes the locations of injections, the injected tracers, and their amounts.
After appropriate survival periods following the injections (28 days for BDA, LYD, and FR, 12–14 days for FB and DY), each animal was deeply anesthetized with an overdose of sodium thiopental and perfused consecutively with saline, 3.5–4 % paraformaldehyde, and 5 % glycerol, prepared in 0.1 M phosphate buffer and pH 7.4, through the left cardiac ventricle. Each brain was then blocked coronally on a stereotaxic apparatus, removed from the skull, photographed, and placed in 10 % buffered glycerol for 3 days and 20 % buffered glycerol for 4 days. Finally, it was cut frozen into coronal sections of 60-μm thickness.
In Case 36r, one series of each fifth section was processed for the visualization of BDA (incubation 60 h), using a Vectastain ABC kit (Vector Laboratories, Burlingame, CA) and 3,3-diaminobenzidine (DAB) as a chromogen. In this same Case and in Cases 37r, 39l, 43r, 48l, and 56l in which BDA was injected in other areas, one series of each fifth section was processed to visualize FR and BDA, or LYD and BDA, using the double-labeling protocol described in detail in Gerbella et al. (2010). Briefly, the sections were first processed to visualize BDA, except for a shorter incubation period in the ABC solution (overnight), and then BDA was stained brown using DAB. Then, the sections were incubated overnight in avidin–biotin blocking reagent (Vector SP-2001), for 72 h at 4 °C in a primary antibody solution of rabbit anti-FR or rabbit anti-LY (1:3,000; Invitrogen) in 0.3 % Triton, 5 % normal goat serum in PBS, and for 1 h in biotinylated secondary antibody (1:200, Vector) in 0.3 % Triton, 5 % normal goat serum in PBS. Finally, FR or LYD labeling was visualized using the Vectastain ABC kit (Vector Laboratories) and the Vector SG peroxidase substrate kit (SK-4700, Vector) as a chromogen. With this procedure, BDA labeling was stained brown, and the FR or the LYD labeling was stained blue in the same tissue sections. In all cases in which FB and DY were injected, every fifth section was mounted, air-dried, and quickly coverslipped for fluorescence microscopy. In all cases, one series of each fifth section was stained with the Nissl method (0.1 % thionin in 0.1 M acetate buffer, pH 3.7).
Data analysis
The distribution of the retrograde and anterograde labeling in the amygdala was analyzed and plotted in sections every 300 μm, together with the outlines of the sections, the ventricles, and the blood vessels. Immunohistochemically labeled small spherical endings (0.5–2.0 μm in diameter) visible as boutons en passant or boutons terminaux were considered as anterograde labeling. The attribution of the labeling to the nuclear subdivisions of the amygdala was made by superimposing adjacent Nissl-stained sections on the plots of the labeling, with the aid of a microprojector. Differences in shrinkage between adjacent, differentially processed sections were corrected by slightly changing the magnification of the microprojector. The amygdalar complex was subdivided according to the criteria described by Price et al. (1987), Pitkänen and Amaral (1998), and Amaral et al. (2003). According to these criteria, the “deep” nuclei of the amygdala, where virtually all the labeling observed in the present study was located, include a lateral, a basal, an accessory basal, and a paralaminar nucleus. Furthermore, the basal nucleus was subdivided into a magnocellular, an intermediate, and a parvicellular subdivision.
In all cases, the relative contribution of the inputs from the various subdivisions of the amygdalar complex to the areas under study was assessed by counting, for each tracer injection, the number of labeled cells in each amygdalar nucleus and/or subdivision. The absolute number of labeled neurons was largely variable across cases, which may be accounted for by several factors (e.g., differences in amount, spread, and sensitivity of injected tracers). Thus, for each tracer injection, afferents to the injected field were expressed in terms of percent of labeled neurons found in a given amygdalar nucleus and/or subdivision, with respect to the total number of labeled cells in the amygdala.
In order to graphically describe the distribution of BDA, FR, and LYD anterograde labeling in the various amygdalar nuclei, similarly to Dancause et al. (2005) and Borra et al. (2010), a grid pattern (100 × 100 μm) was overlaid on digitized images of the sections. A marker was placed in a square of the grid when at least two labeled terminals were located within the square. After preliminary analysis, we defined three types of marked squares based on the number of labeled terminals located within: squares having 2 terminals, 3–8 terminals, and >8 terminals. The definition of these three types of marked squares appeared appropriate to graphically describe relatively sparser (e.g., en-passant varicosities), intermediate, and denser (e.g., clusters of synaptic endings) labeling in 100 × 100 × 60 μm tissue voxels.
Results
The results showed that areas 45A and 45B display afferent and efferent connections with the amygdala, involving mostly the basal and, for the efferent connections, also the lateral nucleus. Furthermore, qualitative and quantitative differences in the distribution of the labeling mostly in the various subdivisions of the basal nucleus, distinguished the amygdalar connectivity of the two areas from one another and from their neighboring areas.
Before describing in detail the results, it seems appropriate to note that the retrograde labeling observed in the amygdala after injections in areas 45A and 45B of the dextran conjugate FR was very rich and far richer than that observed after injections in the same areas of the fluorescent tracers FB and DY, which was the opposite of that observed in these same cases at the cortical level (personal observations). Interestingly, the number of FR-labeled cells was considerably higher than that of FB- and DY-labeled cells also in the thalamus in these same cases (Contini et al. 2010; personal observations). Thus, it is possible that these two types of neural tracers (FR vs. fluorescent tracers) display a differential sensitivity in retrogradely tracing cortical vs. subcortical connections. However, for the purposes of the present study, it is noteworthy that, in spite of the marked differences in their absolute number, the percentage distribution of the marked cells in the various subdivisions of the amygdala was remarkably similar when different cases of tracer injections in the same area are compared (Table 2).
Amygdalar connections of areas 45A and 45B
The distribution of the retrogradely labeled cells observed in the amygdala after FR injections in areas 45A (Case 39l) or 45B (Case 37r) is shown in Fig. 2 and Table 2. The distribution of the marked cells observed after FB injections in area 45A (Case 37l), or 45B (Case 36l) was remarkably similar (Table 2).
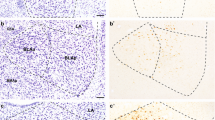
Distribution of retrogradely labeled amygdalar neurons (represented by dark gray circles) observed after FR injections in area 45A (upper part, Case 39l) and in area 45B (lower part, Case 37r). Each dot corresponds to one labeled neuron. The labeling is shown in drawings of coronal sections in rostral to caudal order (a–f, a′–f′), selected at different AP levels according to the atlas of Paxinos et al. (2000). The dashed lines mark the borders of the magnocellular, intermediate, and parvocellular subdivisions of the basal nucleus. AAA anterior amygdaloid area, AB accessory basal nucleus, Bmc magnocellular subdivision of the basal nucleus, Bi intermediate subdivision of the basal nucleus, Bpc parvocellular subdivision of the basal nucleus, L lateral nucleus, PL paralaminar nucleus
In all the cases, the retrograde labeling was virtually all located within the basal nucleus, showing, however, a clear differential distribution according to the injected area. Specifically, after tracer injections in area 45A, most of the labeled amygdalar cells (73 %) were located in the intermediate subdivision (Figs. 2, upper part; 3a, b). A relatively consistent proportion of the labeling was located in the magnocellular subdivision. In both these subdivisions, the labeled cells tended to distribute along the entire rostro-caudal and dorso-ventral extent. Weaker retrograde labeling was located in the parvocellular subdivision. Except for a few clusters of cells in the anterior amygdaloid area, no other nuclei were significantly labeled. In contrast, after injections in area 45B, the retrograde labeling was more equally distributed between the intermediate and the magnocellular subdivision of the basal nucleus, and the labeling was very weak in the parvocellular subdivision (Figs. 2, lower part; 4a, b). Furthermore, though the magnocellular subdivision was almost diffusely involved by the retrograde labeling, in the intermediate subdivision the labeled cells were by far more concentrated in its rostral part.
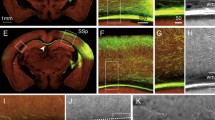
Photomicrographs of a pair of adjacent coronal sections from Case 39l FR, showing in a the distribution of retro- and anterograde labeling in the basal nucleus after tracer injections in area 45A, at approximately AP level 16.4, compared with cytoarchitectonic subdivisions, shown in b. Scale bar in a applies also to b. Higher magnification views from the photomicrograph shown in a, centered on the Bmc nucleus (c), the Bi nucleus (d), and the dorsal part of the L nucleus (e). Abbreviations as in Fig. 2
Photomicrographs of a pair of adjacent coronal sections from Case 37r FR, showing in a the distribution of retro- and anterograde labeling in the basal nucleus after tracer injections in area 45B, at approximately AP level 16.1, compared with cytoarchitectonic subdivisions, shown in b. Scale bar in a applies also to b. Higher magnification views from the photomicrograph shown in a, centered on the Bmc nucleus (c), the Bi nucleus (d), and the dorsal part of the L nucleus (e). IM intercalated masses. Other abbreviations as in Fig. 2
The distribution of the anterograde labeling observed in the amygdala after tracer injections in either area 45A (Case 39l FR), or area 45B (Cases 37r FR and 36r BDA) was quite similar and only partially matched the distribution of the retrograde labeling (Fig. 5a–c). Specifically, in all three cases labeled terminals and axons with preterminal varicosities were quite dense in almost the entire extent of the magnocellular subdivision of the basal nucleus (Figs. 3a, c; 4a, c). In contrast, the intermediate and the parvocellular subdivisions were almost devoid of labeled terminals (Figs. 3a, d; 4a, d). Furthermore, relatively dense anterograde labeling was also observed in the dorsalmost part of the lateral nucleus (Figs. 3a, e; 4a, e), in some cases marginally involving the intercalated masses.
Distribution of the anterograde labeling observed in Case 39l after FR injection in area 45A (a), in Case 37r after FR injection in area 45B (b), in Case 36r after BDA injection in area 45B (c), in Case 36r after FR injection in area 8/FEF (d), and in Case 48l after LYD injection in area 12r (e). For each case the sections are shown in rostral to caudal order. Each dot corresponds to a labeled 100 × 100 × 60 μm voxel containing 2 terminals (blue), 3–8 terminals (yellow), >8 terminals (red). Conventions and abbreviations as in Fig. 2
Amygdalar connections of areas 8/FEF, 8r, 12r, and 46v
After tracer injections in areas 8/FEF, 8r, 12r, and 46v (Fig. 1), the retro- or retro-anterograde labeling observed in the amygdala was much less rich and extensive than that observed after injections in areas 45A and 45B, suggesting preferential amygdalar connectivity, in the VLPF, of areas 45A and 45B. However, it is noteworthy that the present data, based on tracer injections in different cortical areas, do not allow us to make any conclusion on the possible quantitative differences of the amygdalar projections to the areas under study. Indeed, after tracer injections, the overall number of labeled cells and terminals can vary largely across different cases because of factors other than actual differences in the strength of the traced connections and intrinsic to this experimental approach (e.g., differences in amount, spread, and sensitivity of injected tracers).
In the Case 36r (FR injection in the ventral part of area 8/FEF), relatively dense clusters of labeled cells were mostly located in the magnocellular subdivision of the basal nucleus (Fig. 6, upper part; Table 2) and much less densely involved in the intermediate subdivision. Anterograde labeling was almost completely confined to the magnocellular subdivision of the basal nucleus and only few, sparse terminals were located in the lateral nucleus (Fig. 5d). In Case 36l DY, the distribution of the retrograde labeling was very similar, though the number of labeled cell observed in this case was considerably low (Table 2).
Distribution of retrogradely labeled amygdalar neurons (represented by dark gray circles) observed in Case 36r after FR injection in area 8/FEF (upper part) and in Case 48l after LYD injection in area 12r (lower part). The labeling is shown in drawings of coronal sections in rostral to caudal order (a–c, a′–c′). Conventions and abbreviations as in Fig. 2
After injections in the caudal part of area 12r (Cases 26l FB and 48l LYD), just rostral to area 45A, the retrograde labeling was relatively poor and showed a distribution pattern similar to that observed after injections in area 45B, involving almost equally the magnocellullar and the intermediate subdivisions of the basal nucleus (Fig. 6, lower part; Table 2). The anterograde labeling observed in Case 48l LYD was relatively sparse and limited to the magnocellular subdivision of the basal nucleus (Fig. 5e).
Finally, after injections in area 8r and in the caudal part of area 46v only very few and sparse labeled cells and terminals were observed in the magnocellular part of the basal nucleus.
Discussion
The present study shows that the two caudal VLPF areas 45A and 45B are reciprocally connected with the amygdala. Specifically, these two areas receive differentially weighted projections originating predominantly from the different subdivisions of the basal nucleus and send projections mostly to the magnocellular subdivision of the basal nucleus and to the lateral nucleus. Furthermore, the striking differences in the amount of retro- or retro-anterograde amygdalar labeling observed after tracer injections, especially of dextran conjugates, in areas 45A and 45B vs. areas 8/FEF, 8r, caudal 46v, and 12r, suggest that the amygdalar connections of areas 45A and 45B are much richer than those of their neighboring areas. Areas 45A and 45B are also a target of strong projections from the thalamic nucleus MDmc (Contini et al. 2010), a well-known thalamic relay of amygdalar projections (Porrino et al. 1981; Aggleton and Mishkin 1984; Russchen et al. 1987; Ray and Price 1993). Accordingly, relatively rich direct and indirect—trans-thalamic—connectivity with the amygdala appears to be a characterizing connectional feature of areas 45A and 45B and could provide the neural substrate for the selective conveyance, in the VLPF cortex, of emotional information to these two areas.
Amygdalar connectivity of areas 45A and 45B
The connections of the prefrontal cortex with the amygdala have been described in several studies based on tracer injections placed either in different prefrontal areas or regions (e.g., Porrino et al. 1981; Barbas and De Olmos 1990; Carmichael and Price 1995; Stefanacci and Amaral 2002; Ghashghaei and Barbas 2002), or in the amygdala (e.g., Amaral and Price 1984; Stefanacci and Amaral 2000; Ghashghaei et al. 2007). In general, these studies have substantially agreed on some main principles of the organization of the amygdalo-prefrontal projections. First, these connections involve the lateral prefrontal cortex quite weakly, but much more densely caudal orbitofrontal and caudal medial prefrontal areas. Second, there is a differential pattern of connectivity of the various amygdalar nuclei with the prefrontal cortex. Specifically, (a) the basal nucleus displays the most extensive connections, being reciprocally connected to orbital, medial, and lateral prefrontal areas (Barbas and De Olmos 1990; Carmichael and Price 1995; Ghashghaei et al. 2007); (b) the accessory basal nucleus is reciprocally connected mainly with caudal orbital and medial prefrontal areas (Carmichael and Price 1995; Ghashghaei and Barbas 2002), and (c) the lateral nucleus is reciprocally connected to caudal orbitofrontal areas and is the target of projections originating from the lateral prefrontal areas (Carmichael and Price 1995; Ghashghaei and Barbas 2002).
Our data appear in contrast with the general principle that the lateral prefrontal cortex is only weakly connected with the amygdala. However, in a recent study Ghashghaei et al. (2007) placed tracer injections in the amygdala and analyzed quantitatively the distribution of the retro- and anterograde labeling in the prefrontal cortex. They found that in a very caudal VLPF sector, considered part of area 12, the percentage of marked cells and the density of anterogradely labeled terminals was considerably higher than in the remaining of the lateral prefrontal cortex and nearly comparable to that observed in orbitofrontal areas. The present data clearly indicate that this caudal VLPF sector corresponds to areas 45A and 45B. Furthermore, the data of Ghashghaei et al. (2007) provide strong quantitative support to our observations that the amygdalar connections of areas 45A and 45B are relatively rich and much richer than those of the neighboring areas.
Functional considerations
The amygdala is an anatomically complex subcortical structure, associated to the limbic system, where sensory information originating mostly from higher-order sensory areas is endowed with emotional and motivational significance. Accordingly, this structure is an important node in emotion pathways and, through its efferent cortical and subcortical projections plays a crucial role in controlling appropriate emotional and behavioral responses to biologically relevant sensory stimuli. In this context, the reciprocal amygdalo-orbitofrontal connections, through cortico-cortical connections between orbital and lateral prefrontal areas, would mediate emotional influences on cognitive functions processed in lateral prefrontal areas, as well as the cognitive regulation of emotion (see McDonald 1998; Barbas 2007; Salzman and Fusi 2010).
Emotional information from the amygdala can reach the prefrontal cortex not only directly, but also indirectly, through the MDmc (e.g., Porrino et al. 1981; Aggleton and Mishkin 1984; Russchen et al. 1987; Ray and Price 1993; Barbas 2007; Barbas et al. 2011). It was originally proposed that the direct and the indirect pathways would carry more detailed and less detailed, more integrated information, respectively (Russchen et al. 1987; Ray and Price 1993).
The present data, suggesting that amygdalar information directly reaches areas 45A and 45B, appear helpful in providing additional insight into the possible functional role of these two areas.
Specifically, we found that amygdalar projections to areas 45A and 45B mostly originate from two subdivisions of the basal nucleus—magnocellular and intermediate—also connected to visual and auditory temporal areas (Webster et al. 1991; Stefanacci and Amaral 2002; Yukie 2002; Amaral et al. 2003), some of them in turn connected to areas 45A and 45B through cortico-cortical connections (Gerbella et al. 2010). Thus, as suggested for the posterior orbitofrontal cortex (Barbas 2007; Barbas et al. 2011), amygdalar projections to areas 45A and 45B could broadcast signals related to the emotional significance of the sensory information that is conveyed to these two areas by cortico-cortical connections. In this respect, the relatively stronger projections to area 45A from the intermediate vs. the magnocellular part of the basal nucleus could reflect the preferential linkage of auditory areas, within the basal nucleus, with the intermediate subdivision. Furthermore, we found that areas 45A and 45B send projections to the magnocellular basal nucleus subdivision and to the dorsal part of the lateral nucleus, considered as a sensory interface in the organization of the amygdalar circuitry (Carmichael and Price 1995). Thus, the present data suggest that both areas 45A and 45B could modulate at different levels visual and auditory processing in the amygdala.
Area 45A is robustly connected to higher-order auditory and multisensory areas of the superior temporal gyrus (Gerbella et al. 2010). Functional data, showing that this sector is involved in multisensory processing of communication stimuli (see Romanski and Averbeck 2009) and activates during the observation of faces (Tsao et al. 2008) or actions made by others (Nelissen et al. 2005), suggested a role in communication behavior for this area. Furthermore, the connections of area 45A to the dorsal part of area 8/FEF (Gerbella et al. 2010) could represent the neural substrate of the role in communication behavior of gaze direction, an important communicative signal in social interactions (e.g., Emery 2000; Ghazanfar et al. 2006).
The amygdala plays a well established important role in social cognition (see, e.g., Adolphs 2010). Indeed, this structure hosts neurons coding face identity and expressions (e.g., Gothard et al. 2007) or gaze directions (Tazumi et al. 2010), and neurons involved in detection of direct gaze (Tazumi et al. 2010), or activated when looking at the eyes of conspecifics (Zimmerman et al. 2012). Accordingly, the amygdala could be a source of information on the emotional significance of communicative signals, conveyed to area 45A for guiding appropriate social interactions.
Area 45B is robustly connected to higher-order ventral visual stream inferotemporal areas, rostral prefrontal and orbitofrontal areas, and to frontal (8/FEF, 8r, SEF) and parietal (LIP) oculomotor areas (Gerbella et al. 2010). Though the functional properties of this area are still poorly understood, based on these connectional data we suggested that area 45B is a ‘‘preoculomotor’’ area, where rostral prefrontal, orbitofrontal, and inferotemporal inputs guide the exploration of visual scenes for the perception of objects, actions, and faces (Gerbella et al. 2010).
One proposed general role of the amygdala is the involvement in processing selectivity based on value, saliency, and relevance of sensory stimuli (Adolphs 2010). In the visual domain, when looking at a visual scene, the selection of the objects (or of the object’s parts) on which the gaze shifts for more detailed analysis is determined by (a) their saliency, i.e., bottom–up factors related to their distinctiveness and (b) by their relevance, i.e., top–down factors related to the goals and experience of the viewers. Object saliency and relevance are then combined to provide, in the oculomotor system, a priority map that dictates the visual scanpath (see Fecteau and Munoz 2006). Recent data provided evidence for neural activity in the macaque amygdala related to saccades or fixations done over behaviorally relevant parts of visual scenes during free-viewing of natural images, suggesting an involvement in detection of priority maps (Gonzalez Andino and De Peralta Menendez 2012). Thus, it is possible that the amygdalar connections of area 45B convey information related to the relevance of visual stimuli, which could contribute to a representation of priority maps in this VLPF area. Then, area 45B could represent a node of the oculomotor cortical network where priority maps, based on physical distinctiveness and behavioral relevance of objects, guide the exploration of visual scenes, further supporting the hypothesis that this area is involved in higher-order aspect of oculomotor behavior.
References
Adolphs R (2010) What does the amygdala contribute to social cognition? Ann NY Acad Sci 1191:42–61
Aggleton JP, Mishkin M (1984) Projections of the amygdala to the thalamus in the cynomolgus monkey. J Comp Neurol 222:56–68
Amaral DG, Price JL (1984) Amygdalo-cortical projections in the monkey (Macaca fascicularis). J Comp Neurol 230:465–496
Amaral D, Behniea H, Kelly J (2003) Topographic organization of projections from the amygdala to the visual cortex in the macaque monkey. Neuroscience 118:1099–1120
Barbas H (2007) Specialized elements of orbitofrontal cortex in primates. Ann NY Acad Sci 1121:10–32
Barbas H, De Olmos J (1990) Projections from the amygdala to basoventral and mediodorsal prefrontal regions in the rhesus monkey. J Comp Neurol 300:549–571
Barbas H, Zikopoulos B, Timbie C (2011) Sensory pathways and emotional context for action in primate prefrontal cortex. Biol Psychiatry 69:1133–1139
Borra E, Belmalih A, Gerbella M, Rozzi S, Luppino G (2010) Projections of the hand field of the macaque ventral premotor area F5 to the brainstem and spinal cord. J Comp Neurol 518:2570–2591
Borra E, Gerbella M, Rozzi S, Luppino G (2011) Anatomical evidence for the involvement of the macaque ventrolateral prefrontal area 12r in controlling goal-directed actions. J Neurosci 31:12351–12363
Carmichael ST, Price JL (1995) Limbic connections of the orbital and medial prefrontal cortex in macaque monkeys. J Comp Neurol 363:615–641
Contini M, Baccarini M, Borra E, Gerbella M, Rozzi S, Luppino G (2010) Thalamic projections to the macaque caudal ventrolateral prefrontal areas 45A and 45B. Eur J Neurosci 32:1337–1353
Dancause N, Barbay S, Frost SB, Plautz EJ, Chen D, Zoubina EV, Stowe AM (2005) Extensive cortical rewiring after brain injury. J Neurosci 25:10167–10179
Emery NJ (2000) The eyes have it: the neuroethology, function and evolution of social gaze. Neurosci Biobehav Rev 24:581–604
Fecteau JH, Munoz DP (2006) Salience, relevance, and firing: a priority map for target selection. Trends Cogn Sci 10:382–390
Gerbella M, Belmalih A, Borra E, Rozzi S, Luppino G (2007) Multimodal architectonic subdivision of the caudal ventrolateral prefrontal cortex of the macaque monkey. Brain Struct Funct 212:269–301
Gerbella M, Belmalih A, Borra E, Rozzi S, Luppino G (2010) Cortical connections of the macaque caudal ventrolateral prefrontal areas 45A and 45B. Cereb Cortex 20:141–168
Gerbella M, Borra E, Tonelli S, Rozzi S, Luppino G (2012) Connectional heterogeneity of the ventral part of the Macaque Area 46. Cereb Cortex. doi:10.1093/cercor/bhs096.Online
Ghashghaei HT, Barbas H (2002) Pathways for emotion: interactions of prefrontal and anterior temporal pathways in the amygdala of the rhesus monkey. Neuroscience 115:1261–1279
Ghashghaei HT, Hilgetag CC, Barbas H (2007) Sequence of information processing for emotions based on the anatomic dialogue between prefrontal cortex and amygdala. Neuroimage 34:905–923
Ghazanfar AA, Nielsen K, Logothetis NK (2006) Eye movements of monkey observers viewing vocalizing conspecifics. Cognition 101:515–529
Gonzalez Andino SL, de Peralta Grave, Menendez R (2012) Coding of saliency by ensemble bursting in the amygdala of primates. Front Behav Neurosci 6:38
Gothard KM, Battaglia FP, Erickson CA, Spitler KM, Amaral DG (2007) Neural responses to facial expression and face identity in the monkey amygdala. J Neurophysiol 97:1671–1683
McDonald AJ (1998) Cortical pathways to the mammalian amygdala. Prog Neurobiol 55:257–332
Nelissen K, Luppino G, Vanduffel W, Rizzolatti G, Orban GA (2005) Observing others: multiple action representation in the frontal lobe. Science 310:332–336
Paxinos G, Huang XF, Toga AW (2000) The rhesus monkey brain in stereotaxic coordinates. Academic Press, San Diego
Petrides M, Pandya DN (1994) Comparative architectonic analysis of the human and the macaque frontal cortex. In: Boller F, Grafman J (eds) Handbook of neuropsychology, vol 9. Elsevier, Amsterdam, pp 17–58
Petrides M, Pandya DN (2002) Comparative cytoarchitectonic analysis of the human and the macaque ventrolateral prefrontal cortex and cortico-cortical connection patterns in the monkey. Eur J Neurosci 16:291–310
Pitkänen A, Amaral DG (1998) Organization of the intrinsic connections of the monkey amygdaloid complex: projections originating in the lateral nucleus. J Comp Neurol 398:431–458
Porrino LJ, Crane AM, Goldman-Rakic PS (1981) Direct and indirect pathways from the amygdala to the frontal lobe in rhesus monkeys. J Comp Neurol 198:121–136
Price JL, Russchen FT, Amaral DG (1987) The limbic region. II. The amygdaloid complex. In: Bjorkland A, Hokfelt T, Swanson L (eds) Handbook of chemical neuroanatomy. Elsevier, Amsterdam, pp 279–381
Ray JP, Price JL (1993) The organization of projections from the mediodorsal nucleus of the thalamus to orbital and medial prefrontal cortex in macaque monkeys. J Comp Neurol 337:1–31
Romanski LM, Averbeck BB (2009) The primate cortical auditory system and neural representation of conspecific vocalizations. Annu Rev Neurosci 32:315–346
Russchen FT, Amaral DG, Price JL (1987) The afferent input to the magnocellular division of the mediodorsal thalamic nucleus in the monkey Macaca fascicularis. J Comp Neurol 256:175–210
Salzman CD, Fusi S (2010) Emotion, cognition, and mental state representation in amygdala and prefrontal cortex. Annu Rev Neurosci 33:173–202
Stefanacci L, Amaral DG (2000) Topographic organization of cortical inputs to the lateral nucleus of the macaque monkey amygdala. J Comp Neurol 421:52–79
Stefanacci L, Amaral DG (2002) Some observations on cortical inputs to the macaque monkey amygdala: an anterograde tracing study. J Comp Neurol 451:301–323
Tazumi T, Hori E, Maior RS, Ono T, Nishijo H (2010) Neural correlates to seen gaze-direction and head orientation in the macaque monkey amygdala. Neuroscience 169:287–301
Tsao DY, Moeller S, Freiwald WA (2008) Comparing face patch systems in macaques and humans. Proc Natl Acad Sci USA 105:19514–19519
Webster MJ, Ungerleider LG, Bachevalier J (1991) Connections of inferior temporal areas TE and TEO with medial temporal-lobe structures in infant and adult monkeys. J Neurosci 11:1095–1116
Yukie M (2002) Connections between the amygdala and auditory cortical areas in the macaque monkey. Neurosci Res 42:219–229
Zimmerman PE, Mosher C, Gothard KM (2012) Looking at the eyes engages single unit activity in the primate amygdala during naturalistic social interactions. Program No. 402.02. Neuroscience Meeting Planner. New Orleans, LA: Society for Neuroscience, 2012
Acknowledgments
The work was supported by Ministero dell’Istruzione, dell’Università e della Ricerca (Grant number: PRIN 2008, no. 2008J7YFNR_002); European Commission Grant Cogsystems FP7-250013.
Author information
Authors and Affiliations
Corresponding author
Additional information
Marzio Gerbella and Matteo Baccarini equally contributed to this work.
Rights and permissions
About this article
Cite this article
Gerbella, M., Baccarini, M., Borra, E. et al. Amygdalar connections of the macaque areas 45A and 45B. Brain Struct Funct 219, 831–842 (2014). https://doi.org/10.1007/s00429-013-0538-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-013-0538-2