Abstract
The aim of this study was to give a microscopic description of the organization, the innervation and the slow or fast type of the striated fibers of the external urethral sphincter in the female rat. Conventional methods for photonic microscopy and immunochemistry were applied to cross and longitudinal sections of snap-frozen urethra. With hematoxylin-eosin stained cross sections, striated fibers are of small diameter and attached directly to the surrounding connective tissue. They are innervated by cholinergic endplates as shown by acetylcholinesterase techniques and alpha-bungarotoxin binding. The histological aspects of the cross sections as well as the distribution of endplates along the length of the sphincter suggest an organization of the fibers in four bundles, possibly acting as a photographic diaphragm does. Like striated skeletal muscle fibers, the fibers bind monoclonal antibodies against dystrophin with subsarcolemmal distribution and against desmin which visualizes striations. All the fibers express fast myosin heavy chains and very few co-express slow myosin heavy chains as determined by immunocytochemistry. We are taking advantage of the diaphragmatic organization of the striated sphincter to develop a longitudinal section as a model of chronic incontinence to test the efficiency of grafted myoblasts provided by fast striated skeletal muscle.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Wilson (1809) was the first to describe striated muscle surrounding the urethra in humans, but later the existence of this individual muscle was denied by most of the authors and it was considered to be a part of the whole pelvic musculature. The discovery that the cross sectional area of striated fibers of the muscle surrounding the urethra was significantly smaller than those of the other pelvic muscles (Von Hayek 1960; Oelrich 1983) gave support for the existence of an individual muscle. From this time on, morphological descriptions were mainly dedicated to the whole muscle aspect and its innervation in human beings. Most of these descriptions were done in males with the view to preserving urinary continence during prostate surgery (Myers 1991, 2001; Narayan et al. 1995). In females striated fibers were described external to the smooth fiber layers of the urethra and the striated sphincter was seen as a U-shape at the proximal and distal parts of the urethra (Oelrich 1983; Gosling 1985; Tichy 1989; Kokoua et al. 1993; Ludwikowski 2001). In males and females the pudendal nerve and the pelvic plexus are thought to innervate the striated sphincter (Snooks and Swash 1984; Snooks et al. 1984). Motor neurons located in the anterior horn of the sacral medulla (S2–S4) give rise to axons running in these two nerves (review: Burnett and Wesselmann 1999) to establish cholinergic synapses with the striated fibers (Junemann et al. 1987). The striated fibers of the human external sphincter were initially described as being of the slow type by histochemical evaluation (Gosling et al. 1981), but some fibers of the fast type were probably also present (Schroder and Reske-Nielsen 1983). Although the contribution of the striated sphincter to the urethral closure function has been established (Thind 1995), the role of these different types of fibers during the striated sphincter contraction is controversial (review: Elbadawy 1996).
To the best of our knowledge only one study of the striated sphincter was performed in rats (Russell et al. 1996). The aim of this study was to give more information on the organization, the innervation and the immunochemical properties of the urethral striated sphincter fibers in adult female rats.
Material and methods
Animals
Adult female wistar white rats (250–300 g) were purchased from the Centre d'Elevage Depré (France) and housed in plastic cages in our own animal facility with water and cube diet (UAR A03) ad libitum and natural night and day cycle. All procedures were conducted according to the "Principles of laboratory animal care" (National Institute of Health, publication # 86–23, revised 1985). All surgical procedures were performed under pentobarbital anesthesia (30 mg/kg body weight). Anesthetized animals were sacrificed by cervical dislocation.
Sample excision and treatment
The bladder was exposed by a median ventral incision. The dome of the bladder was gripped with hemostatic forceps and pulled caudally to expose the urethra. The pubis was sectioned with Maillot scissors and the urethra was removed from the meatus (urethral orifice) to the bladder with the anterior wall of the vagina. The samples were not put in buffer or fixed but were immediately placed on a slice of cork with a drop of tragacanth gum in a position suitable for cross or longitudinal section and frozen in isopentane chilled with liquid nitrogen (see Dubovitz 1985). Some of the urethras were dissected from the vagina after removing and were opened by a single longitudinal incision up to the lumen. These flattened urethras were suspended on a frame before freezing to provide frontal sections to study the distribution of the end plates. All the samples were stored at −80°C until sectioning and staining. Sectioning was performed with a cryostat (Jung Frigocut 1800 N manufactured by Leica) and sections were stained with one of the following techniques:
-
1.
10 µm-thick cross sections were stained with hematoxilin-eosin (H&E) to visualize nuclei, membranes, cytoplasm and connective tissue.
-
2.
50 µm-thick sections were subject either to acetylcholinesterase visualization by the method of Koëlle and Friedenwald (1949) using acetylthiocholine iodide as the substrate or to combined staining of acetylcholinesterase and axonal endings according to Pestronk and Drachman (1978). These two staining methods identify neuromuscular junctions at the outer surface of the myofibers. On some 50 µm-thick sections, the acetylcholine receptors were localized with rhodamin-conjugated α-bungarotoxin (α-Bgt) (1/1000: Molecular Probes, a kind gift from Daniel Hantaï, Paris).
-
3.
Immunofluorescent procedures were performed on 7 µm thick serial cross sections to visualize desmin, dystrophin, fast and slow myosin heavy chains (MHCf and MHCs). Sections were transferred to 3-aminopropyl-trietoxysilane-coated (A-3648 Sigma) glass coverslips. After blocking with mouse serum for 30 min, IgG1 monoclonal antibodies (MAbs) were incubated in a humid atmosphere for 1 h at room temperature, i.e. RPN 1101 Amersham (1/5), DYS2 Novocastra (1/50), WB-MHCf Novocastra (1/40) and WB-MHCs Novocastra (1/80). After 3 rinses with PBS (10 min each) the immunolocalization of these primary antibodies was accomplished by the use of a FITC-conjugated goat-anti-mouse secondary antibody with IgG1 specificity (1/500: Southern Biotechnology Associates) applied for 1 h.
Localization of neuromuscular junctions
One sphincter was entirely cut in 50 µm thick serial cross sections and stained for acetylcholinesterase activity. Under the microscope (100× magnification) each section was divided into four quadrants by two virtual lines, one antero-posterior (12 o'clock–6 o'clock) and the other perpendicular to the first (9 o'clock–3 o'clock). Then endplates were counted by eye in each quadrant by a single examiner. When all the sections were examined, the repartition of the endplates was calculated by two ways:
-
1.
The sum of endplates of all the sections in each quadrant gave a comparison of the density of endplates in the four quadrants
-
2.
The sections were grouped into six successive segments of identical length (with the same number of sections). The sum of the endplates of all the quadrants was calculated in each segment to give their longitudinal distribution.
Results
Macroscopic aspect
In the female rat, the urethra and its sphincter are easy to dissect from the adjacent structures owing to the presence of fatty tissue surrounding the bladder neck and urethra. The ventral wall of the vagina remains in contact with the urethra by means of connective tissue. Following dissection the whole sphincter measures between 4.5 and 5.0 mm in length (n=6). Its cranial extremity begins 0.5 mm below the bladder neck and the caudal extremity is located 17±1 mm from the meatus.
Morphology
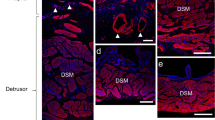
In the central part of the urethra, H&E stained cross sections revealed different layers from the lumen to the vagina wall: an epithelium layer, a connective tissue layer, two smooth muscle layers (an extensive layer of longitudinal smooth layer and a thin circular layer), another connective tissue layer and an external striated muscle layer (Fig. 1). In cross sections near the bladder neck, the dorsal wall of the sphincter was thicker than the lateral walls and the anterior wall was not detectable (not shown). By contrast, in cross sections of the distal part of the sphincter, the dorsal wall became thinner than the ventral one and distally disappeared. As shown in Fig. 1, a strip of connective tissue was visible in the lateral sides of the sphincter running from the internal smooth muscle layer to the external connective tissue and embedding the sphincter. This aspect can be observed along the whole length of the striated sphincter. In the central zone, the striated fibers of the dorsal and ventral walls were more numerous than in the lateral walls resulting in an anterior and posterior thickening of the sphincter.
Transverse section of the central part of the urethra in female rat. This 10 µm cryostat section shows different circular layers from the lumen to the periphery: the epithelium, the smooth muscle and the striated muscle. Note the thickness of the anterior and posterior part of the external striated sphincter (stars) and the presence of a lateral strip of connective tissue which divides the striated fibers in bundles (arrow). H&E, magnification ×40
Innervation
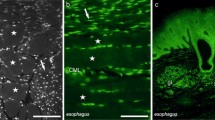
Endplates were detected by binding rhodamin-conjugated α-Bgt to the acetylcholine receptors (Fig. 2A). At high magnification (×630) these endplates showed an aspect similar to those of striated skeletal muscles: although their size was slightly smaller, "bunch of grapes" aspects can be observed (Fig. 2B) and the synaptic gutter is detectable in cross sections (Fig. 2C). The distribution of these endplates in a cross section suggests four groups, two antero-lateral and two postero-lateral with no endplates in the anterior and the posterior segments. These endplates can also be visualized by the acetylcholinesterase reaction as demonstrated by simultaneous staining of endplates and axons. A single axon innervates one endplate as is the rule in striated skeletal muscles but the motor unit organization seems to be different since one motor neuron innervates adjacent fibers (Fig. 3A) in contrast to the scattered distribution observed in skeletal muscles. Moreover endplates appeared longitudinally disposed in line on flattened longitudinal sections (Fig. 3B). To determine the repartition of the endplates in the sphincter circumference, endplates were counted into four equivalent quadrants of serial cross sections of one sphincter stained for acetylcholinesterase reaction. The total number of endplates was almost equal in each quadrant over the whole length of the urethra (n=372, 394, 371 and 452, respectively). Then, we separated the urethra in six identical segments to examine the distribution of the endplates in the whole length of the urethra. The number of endplates increased from the meatus to the bladder (segment 1: 228, segment 2: 243, segment 3: 259, segment 4: 341, segment 5: 411). A sharp decrease occurred when the sphincter became thin before its replacement by connective tissue near segment 6 (n=107).
Distribution and aspect of endplates visualized by α-bungaratoxin rhodamin-conjugated binding on a cross section of the striated external sphincter. A Endplates are distributed in four groups (assembly of six images at magnification ×160). The endplate aspect is similar to that encountered on striated skeletal muscle looking to bunch of grapes B with a synaptic gutter C. B and C magnification ×630
A Innervation of the fibers of the external striated sphincter. One motor axon innervates several endplates located on contiguous myofibers (acetylcholinesterase technique and silver impregnation, magnification ×400). B Distribution of endplates in a longitudinal section of the striated external sphincter. Endplates show an alignment along the length of the urethra from the top to the bottom of the picture (acetylcholinesterase technique, magnification ×100)
Immunohistochemistry
On cross sections, the striated fibers of the urethral sphincter expressed desmin and dystrophin (Fig. 4A,B), two markers encountered in striated skeletal muscle. Desmin binds to specific MAbs both in the fibers of striated and smooth muscles of the sphincter. In striated fibers, this immunostaining delineates the transverse striation of the fibers (Fig. 4A). Smooth muscle fibers of the sphincter do not express dystrophin. In striated fibers the immunolocalization of dystrophin is subsarcolemmal (Fig. 4B). Expression of slow and fast myosin heavy chain was also investigated using specific MAbs. Most of the fibers were positive to the fast MHC (Fig. 5A,C) and only a few fibers were positive to the slow MHC (Fig. 5B,D). The examination of several serial cross sections showed that fibers expressing slow MHC usually co-express fast MHC.
Fast and slow myosin heavy chain (MHCf, MHCs) expression in serial cross sections of the external striated sphincter. A, C Most of the fibers expressed MHCf. B and D Only a few of them expressed both MHCs and MHCf on serial sections. Arrows indicate the same fibers seen in serial cross sections (A with B, C with D). A, B, C, and D magnification ×400
Discussion
To the best of our knowledge only a single microscopic study of the urethral sphincter of the rats was done previously. In 1996 Russell et al. (1996) described the smooth muscle as an inner longitudinal layer and the striated muscle as an outer circular annulus surrounding the urethra lumen. Concerning the smooth muscle, they observed two layers as described for the female human and mouse urethra (Colleselli et al. 1998; Phillips and Davies 1980). In addition, we observed in the middle part of the urethra that the inner longitudinal layer is thick and separated from the lumen epithelium by a dense connective tissue, whereas the outer circular layer is very thin. This smooth muscle was present along the whole length of the urethra and, near the bladder neck the outer layer became thicker and replaced the striated sphincter.
The most striking feature of the striated sphincter is the small diameter of its fibers which is less than 3–5 times the diameter measured in those of striated skeletal muscle from the pelvic floor which is true for humans and rodents (rat and mouse) (Von Hayek 1960; Yiou et al. 2001). In the rat this thinness results in the absence of peripheral localization of the nuclei as is the rule in striated skeletal muscle fibers. The nuclei appear to be the same size as the diameter of the fibers and they seem to be centrally located. Another major feature of this striated muscle is the absence of visible anchorage points: the ends of the fibers are in direct contact with the surrounding connective tissue and at this point the striated urethral sphincter is distinct from the striated skeletal muscles which show myotendinous junctions. Beside these differences, the urethral striated fibers present high similarities with the skeletal muscle fibers.Firstly, the MAb against desmin delineates a striated organization of this filamentous protein identical in the external sphincter and the skeletal muscle fibers. The smooth muscle layers showed fibers uniformly stained using the same MAb. Secondly, dystrophin, a subsarcolemmal protein binding F-actin to the dystrophin-associated protein complex is detectable in these fibers by immunohistochemistry as in skeletal muscle striated fibers (Ibraghimov-Beskrovnaya et al. 1992). This protein is known to be present in normal striated skeletal muscles but not in smooth muscle (Byers et al. 1991). Thirdly, α-bungarotoxin is known as a specific blocker of the α-subunits of the acetylcholine nicotinic receptor in the endplates of the skeletal muscle fibers (Chiappinelli 1985). This blocker binds to the neuromuscular junction of the striated urethral sphincter indicating that these endplates are also nicotinic. Moreover their appearance as a "bunch of grapes" is very similar. However, the motor units are distinctly organized in the skeletal muscle where fibers innervated by a single axon are scattered since the striated urethral fibers innervated by a single axon appear contiguous.
The connective tissue surrounding the striated fibers as seen in H&E cross sections delineates four bundles of fibers. The two anterior bundles overlap in the medial anterior part of the urethra and the posterior bundles overlap in the medial posterior part. This is not the case in the lateral side where the bundles are separated by a strip of connective tissue. This disposition of the striated fibers in four bundles is supported by the visualization of their endplates using α-bungarotoxin. These endplates are divided into four groups corresponding to the four quadrants of the sphincter disposed as are the bundles of fibers. In each of the quadrants the endplates are disposed longitudinally as a column and counting of the endplates in each column showed a similar number of innervated striated fibers. Then each bundle seems to contain the same number of fibers since each of them showed a single endplate in the medial part. The striated urethral sphincter seems to be composed of four devices acting as a photographic diaphragm to close the urethral lumen via the smooth muscle layer.
It seems possible to classify the striated fibers of the urethral sphincter in fast or type I fibers and slow or type II fibers by means of specific antibodies as it is regularly done in skeletal muscle (Pette and Staron 1990). But the fiber type constitution of the urethral striated sphincter is controversial and probably depends on the species and the method used for the determination of the fast and slow types. For instance, electrophoretic studies of myosins in the rabbit determined a fast type for all the fibers of the striated sphincter (Tokunaka et al. 1984, 1986). In contrast, the same method detected a mixture of slow and fast fibers in humans with slow fibers being predominent (Tokunaka et al. 1990). A previous study in humans using histochemical methods showed slow fibers in the striated sphincter (Gosling et al.1981). In the rat it was shown that slow fibers exist, but the bias of the study was an exclusive use of slow myosin antibody for histochemistry without evaluation of concomitant fast fibers by the same method (Russell 1996). In our study, the expression profile of MHCs and MHCf showed a great predominance of MHCf-positive fibers in the external sphincter. The role of these fast fibers in the state of continence is questionable. In a recent study intravenous injection of α-Bgt reduced voiding in paraplegic rats probably by suppressing high frequency phasic sphincter activity (Yoshiyama et al. 2000). Then, it seems that the external striated sphincter in which fast fibers are prominent could play a role in the mechanism of continence though phasic contractions. The putative variability of the MHC profile between humans and rats could be due to a difference in the function of the striated sphincter between quadrupeds and bipeds to insure urethral closure.
In summary, histology of the urethra in the female rat allows us to describe small striated fibers organized in four semi-circular bundles with direct connective tissue anchorage. These cholinergic innervated fibers are mainly of the fast type giving consistency to a phasic activity of the striated sphincter in continence. We are taking advantage of the diaphragmatic organization of the striated sphincter to develop a longitudinal section as a model of chronic incontinence to test the efficiency of grafted myoblasts provided by fast striated skeletal muscles.
References
Burnett AL, Wesselmann U (1999) History of the neurobiology of the pelvis. Urology 53:1082–1089
Byers TJ, Kunkel LM, WatkinsC (1991) The subcellular distribution of dystrophin in mouse skeletal, cardiac, and smooth muscle. J Cell Biol 115:411–421
Chiappinelli VA (1985) Actions of snake venom toxins on neuronal nicotinic receptors and other neuronal receptors. Pharmacol Ther 31:1–32
Colleselli K, Stenzl A, Eder R, Strasser H, Poisel S, Bartsch G (1998) The female urethral sphincter: a morphological and topographical study. J Urol 160:49–54
Dubovitz V (1985) Muscle biopsy: a practical approach, 2nd edn. Baillière Tindall, London
Elbadawy A (1996) Functional anatomy of the organs of micturition. Urol Clin North Am 23:177–210
Gosling JA (1985) The structure of the female lower urinary tract and pelvic floor. Urol Clin North Am 12:207–214
Gosling JA, Dixon JS, Critchley HO, Thompson SA (1981) A comparative study of the human external sphincter and periurethral levator ani muscles. Br J Urol 53:35–41
Ibraghimov-Beskrovnaya O, Ervasti JM, Leveille CJ, Slaughter CA, Sernett SW, Campbell KP (1992) Primary structure of dystrophin-associated glycoproteins linking dystrophin to the extracellular matrix. Nature 355:696–702
Junemann KP, Schmidt RA, Melchior H, Tanagho EA (1987) Neuroanatomy and clinical significance of the external urethral sphincter. Urol Int 42:132–136
Koëlle GB, Friedenwald JS (1949) A histochemical method for localizing cholinesterase activity. Proc Soc Exp Biol Med 70:617–622
Kokoua A, Homsy Y, Lavigne JF, Williot P, Corcos J, Laberge I, Michaud J (1993) Maturation of the external urinary sphincter: a comparative histotopographic study in humans. J Urol 150:617–622
Ludwikowski B, Oesch Hayward I, Brenner E, Fritsh H (2001) The development of the external sphincter in humans. BJU Int 87:565–568
Myers RP (1991) Male urethral sphincteric anatomy and radical prostatectomy. Urol Clin North Am 18:211–227
Myers RP (2001) Practical surgical anatomy for radical prostatectomy. Urol Clin North Am 28:473–490
Narayan P, Konety B, Aslam K, Aboseif S, Blumenfeld W, Tanagho E (1995) Neuroanatomy of the external urethral sphincter: implications for urinary continence preservation during radical prostate surgery. J Urol 153:337–341
Oelrich TM (1983) The striated urogenital sphincter muscle in the female. Anat Rec 205:223–232
Pestronk A, Drachman DB (1978) A new stain for quantitative measurement of sprouting at neuromuscular junctions. Muscle Nerve 1:70–74
Pette D, Staron RS (1990) Cellular and molecular diversities of mammalian skeletal muscle fibers. Rev Physiol Biochem Pharmacol 116:1–76
Phillips JI, Davies I (1980) The comparative morphology of the bladder and urethra in young and old female C57BL/Icrfat mice. Exp Gerontol 15:551–562
Russell B, Baumann M, Heidkamp MC, Svanborg A (1996) Morphometry of the aging female rat urethra. Int Urogynecol J Pelvic Floor Dysfunct 7:30–36
Schroder HD, Reske-Nielsen E (1983) Fiber types in the striated urethral and anal sphincters. Acta Neuropathol (Berl) 60:278–282
Snooks SJ, Swash M (1984) Perineal nerve and transcutaneous spinal stimulation: new methods for investigation of the urethral striated sphincter musculature. Br J Urol 56:406–409
Snooks SJ, Setchell M, Swash M, Henry MM (1984) Injury to innervation of pelvic floor sphincter musculature in childbirth. Lancet 8402:546–550
Thind P (1995) The significance of smooth and striated muscles in the sphincter function of the urethra in healthy women. Neurourol Urodyn 14:585–618
Tichy M (1989) The morphogenesis of human sphincter urethrae muscle. Anat Embryol 180:577–582
Tokunaka S, Murakami U, Ohashi K, Okamura K, Yachiku S (1984) Electrophoretic and ultrastructural analysis of the rabbit's striated external urethral sphincter. J Urol 132:1040–1043
Tokunaka S, Murakami U, Okamura K, Miyata M, Yachiku S (1986) The fiber type of the rabbits' striated external urethral sphincter: electrophoretic analysis of myosin. J Urol 135:427–430
Tokunaka S, Okamura K, Fujii H, Yachiku S (1990) The proportions of fiber types in human external urethral sphincter: electrophoretic analysis of myosin. Urol Res 18:341–344
Von Hayek H (1960) Das Faserkaliber in den MM. transversus perinei und sphincter urethrae. Z Anat Entwicklungsgesch 121:455–458
Wilson D (1809) A description of two muscles surrounding the membranous part of the urethra. Med Chirurg Soc Lond 1:175–180
Yiou R, Delmas V, Carmeliet P, Gherardi RK, Barlovatz-Meimon G, Chopin D, Abbou CC, Lefaucheur JP (2001) The physiopathology of pelvic floor disorders: evidence from a histomorphologic study of the perineum and a mouse model of rectoprolapse. J Anat 199:599–607
Yoshiyama M, DeGroat WC, Fraser MO (2000) Influences of external urethral sphincter relaxation induced by alpha-bungarotoxin, a neuromuscular junction blocking agent, on voiding dysfunction in the rat with spinal cord injury. Urology 55:956–960
Acknowledgements
This study was supported by the Fondation de l'Avenir (grants ETO-0 and ETO-1 for AS). CP was granted by the Association Française contre les myopathies. The authors thank Nicolle Delphinefor technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Praud, C., Sebe, P., Mondet, F. et al. The striated urethral sphincter in female rats. Anat Embryol 207, 169–175 (2003). https://doi.org/10.1007/s00429-003-0340-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-003-0340-7