Abstract
Associations between specific placental histological abnormalities and obstetric outcomes are reported. However, most data are based either on high-risk cases or relate to case–control studies selected from those with abnormal placental histology findings, with the unavoidable biases that these approaches entail. This study reports the frequency of the several common, objective and predefined histological abnormalities of the placenta as identified by pathologists blinded to all clinical information. A total 1,153 women were recruited from an unselected population delivering at 34–43 weeks. Histological findings in common obstetric outcome groups were compared to those of the unselected population, and odds ratios and predictive values were calculated. Normal histological findings were present in 72.1% of pregnancies with normal outcomes and in 79.1%, 66.6%, 80%, and 74.8% of pregnancies affected by pre-eclampsia (PET), pregnancy-induced hypertension (PIH), gestational diabetes (GDM), and small for gestational age (SGA), respectively. Chronic placental underperfusion was seen more frequently in PIH (odds ratio (OR) 2) and SGA (OR 1.4), while villitis of unknown aetiology was observed more commonly in cases with PIH (OR 3.2). Fetal thrombotic vasculopathy was twice as common in cases with GDM whilst massive perivillous fibrin deposition was much more frequent in those with PET (OR 20.2) and SGA (OR 8.9). Chorangiomata were 13 times more common in pregnancies with PET. However, in all cases, positive predictive values were low, with the majority of cases with histological abnormalities being associated with normal outcome. At term, specific placental histological lesions are significantly more common in complicated pregnancies, but the clinical significance of such lesions in a specific case remains uncertain, since the majority will be identified from clinically uncomplicated normal pregnancies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Though recommendations for placental histological examination in the UK and USA exist [1, 2], the clinical significance of many histologically detectable placental lesions remains uncertain, especially in the context of an unselected low-risk population. Most studies in this area are based on retrospective review of clinical cases from categories of abnormal histological findings or abnormal obstetric outcomes; the placentas thus submitted are by definition at increased risk of being abnormal. In addition, since most studies are retrospective, reporting pathologists have had access to the clinical information available at submission when reporting the cases, introducing a source of bias. Clinical information provided can significantly affect interpretation of placental histological features [3]. Little data exist reporting the frequency and pattern of histological lesions present in the placenta in an unselected population at or near term, with histological findings identified by pathologists entirely blinded to all clinical information.
Complications such as pre-eclampsia (PET), pregnancy-induced hypertension (PIH), gestational diabetes (GDM), and small for gestational age (SGA) are associated with prematurity and increased perinatal morbidity and mortality [4–6]. Several placental lesions have been reported in association with these complications [7–9] in selected populations, and it is apparent that no specific placental lesion is characteristic of any one particular obstetric outcome category. For example, in pre-eclampsia, spiral arteries remain ‘untransformed’ [10], remaining muscular due to the incomplete invasion of trophoblast, leading to high-resistance blood flow passing through these vessels and abnormal utero-placental blood flow. Though ischaemic-type changes have been observed in the majority of severe preterm cases [8, 11], there may be subjectively normal placental histological findings even in cases of clinically severe disease at or near term [12, 13]. Features such as infarction, extensive perivillous fibrin deposition and chronic villitis are more frequent in SGA in some studies [14] but not others [15] and are well-reported in clinically normal pregnancies.
The frequency of common histological lesions present in an unselected, prospectively recruited population of placentas from deliveries at or near term with histological features reported by pathologists completely blinded to all clinical information is unknown. The aims of this study are to use this approach to determine the frequency of predefined placental histopathological abnormalities in an unselected population delivering at or near term in relation to common obstetric pregnancy outcomes and to determine the clinical significance of the interpretation of such lesions by calculating sensitivity, positive predictive values and odds ratio in relation to obstetric outcomes.
Methods
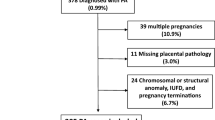
This was a prospective study performed in a large maternity unit offering care and delivery both for ‘low risk’ pregnancies and those requiring tertiary services. The study was approved by the Cambridgeshire ethical committee (ref. no. 07/Q0106/51), and written consent was obtained from the women taking part in the study. No attempt was made at selection by any maternal characteristics. Unselected women were recruited over a 13-month period (2007–2008). Of 1,159 women with singleton pregnancy initially recruited antenatally, 1,153 women were included who delivered at 34–43 weeks gestation and in whom the placenta was available for examination. This study includes unselected cases recruited specifically for the study purposes and is not a retrospective review of cases submitted as part of clinical practice to a histopathology department.
The placentas were placed in a labelled double bag within 20 min of delivery, which was kept in a dry, clean 1–2-l plastic container with an airtight lid and in a fridge with the temperature maintained at 4–6°C. All placentas were examined macroscopically by technicians trained specifically for this study under the direct supervision of a consultant paediatric pathologist in a specialist histopathology department. The cord was excised from the point of insertion into the chorionic plate; blood was drained from the placenta, and adherent blood clots removed from the maternal surface. All extraplacental membranes were trimmed from the chorionic plate, and the placenta was weighed on a fixed electronic scale. Details of the methods are entirely standard and are described in detail elsewhere [16, 17]. The placenta was serially sliced and examined macroscopically, and blocks were obtained from membranes, umbilical cord and the full thickness of the placental parenchyma according to standard diagnostic protocols [18]. Three standard samples were taken from the umbilical cord: maternal end, fetal end and from the centre. Three full-thickness central and one peripheral parenchymal block were taken, and one ‘en face’ block was taken from the maternal surface. Additional blocks were taken from any macroscopic abnormality. To avoid sampling bias, all samples that were taken were examined histologically.
Histological examination of haematoxylin and eosin-stained sections was subsequently performed by two specialist paediatric pathologists (NJS/FAJ) who classified any histological features according to previously agreed and standard criteria (Appendix 1). Pathological lesions were recorded for the purpose of this study as ‛present’ or ‛absent’. In cases where there was presence of more than one histological lesion, each lesion was recorded. The pathologists performing the histological examinations were completely blinded to all clinical information, including gestational age, the cases being identified by study number only and reported in batches purely for the purposes of the study. Cases were reported independently by two pathologists, and any discrepancy was resolved by consensus, giving priority to the possible presence of any abnormality. Specific histological subtypes predefined in this study included no significant histological abnormality, ascending genital tract infection, chronic placental underperfusion, intervillous thrombus, villitis of unknown aetiology (VUE), fetal thrombotic vasculopathy, massive perivillous fibrin deposition, chronic histiocytic intervillositis, chorioangiosis, chorangioma and retroplacental haematoma/abruption. Brief descriptions of diagnostic criteria for these abnormalities are provided in Appendix 1 but are based on widely accepted and published standard criteria from major textbooks [18] (Fig. 1).
Histological findings of placentas from cases with normal outcome were compared with those from patients with obstetric complications: pre-eclampsia (PET), pregnancy-induced hypertension (PIH), gestational diabetes (GDM) and small for gestational age (SGA). Definitions of the common obstetric outcome groups of PET, PIH, GDM and SGA are briefly described: Pre-eclampsia was defined as blood pressure ≥140/90 on two or more occasions after 24 weeks, at least 6 h apart, with significant proteinuria of more than 300 mg/l in a 24-h collection in a previously normotensive woman [19, 20]. We also included women with ≥2+ in a urine sample in the absence of a demonstrable urinary tract infection in this group. PIH was defined as blood pressure ≥140/90 in pregnancy after 20 weeks without any evidence of proteinuria. According to WHO criteria, gestational diabetes is carbohydrate intolerance resulting in hyperglycaemia of variable severity with onset or first recognition during pregnancy. The definition applies irrespective of whether or not insulin is used for treatment or the condition persists after pregnancy. We defined the gestational diabetes according to WHO criteria: fasting blood glucose level ≥7 mmol/l or 2 h post 75 mg of oral glucose load ≥7.8 mmol/l [21]. Birth weight <10th centile for gestational age defined the SGA group. To calculate these, we used the standard UK 4-in-1 charts (girls and boys), which are the growth charts designed principally for hospital use for preterm and term babies and their follow-up [22].
Descriptive statistics were performed for each obstetric outcome category, and the frequency of each common histological diagnosis was reported (Table 1). Comparison of the significance of differences in frequencies of common histological lesions between groups was performed using chi-square test (comparison of proportions). Pearson chi-square test was used to determine significance of differences in frequencies between the groups where the number of cases was more than five, with Yates's continuity correction. P ≤ 0.05 was considered significant. Odds ratios were calculated for histological categories in the obstetric outcome groups (disease present versus absent). Odds ratio, positive predictive value and sensitivity were calculated for histological features for obstetric outcome groups.
Results
Of the total 1,153 cases, placental histological findings were available in 1,122. Details of the obstetric outcome groups are provided in Table 1. Table 2 reports maternal, neonatal and placental characteristics of the study participants. The frequency of histological lesions in the clinical outcome groups is provided in Table 3. The majority of cases in all groups revealed no significant histological abnormality: 72.1%, 79.1%, 66.6%, 80% and 74.8%, respectively, in the normal, PET, PIH, GDM and SGA groups. There was no difference in the incidence of ascending genital tract infection between any of these obstetric outcome groups at or near term. Certain placental histological findings were more common in particular outcome groups, such as VUE and PIH, FTV and GDM, MPFD with PET and SGA, and chorangioma with PET. However, despite the increased odds ratios of these specific histological findings for obstetric complications, the overall sensitivity and positive predictive value for any specified histological lesions was low, with the majority of histological abnormalities being associated with normal obstetric outcome (Table 4).
Discussion
The findings of this study have demonstrated first, that in an unselected population delivering at or near term, the majority of placentas demonstrate no significant histological abnormality. Secondly, we confirm that certain histological features are more frequent in association with specific obstetric complications, but no histological feature is associated with definitive diagnosis of a clinical complication. Finally, since most pregnancies are uncomplicated, the sensitivity and positive predictive value of most histological findings for pregnancy outcome in this population is low, with the vast majority of pregnancies demonstrating any particular histological feature having a normal outcome, even when that feature is strongly associated with a complication such as PIH or pre-eclampsia. Therefore, whilst data based on highly selected groups of complicated pregnancies, in whom placentas were submitted according to indications as part of clinical care, have correctly reported a range of histological lesions in pregnancies with adverse obstetric outcomes, most—if not all—are not specific to any particular outcome group and importantly are also commonly found in normal pregnancies.
The lack of finding statistically significant differences in some placental lesions is unsurprising. For example, the incidence of histological chorioamnionitis is strongly related to the gestational age at delivery, being strongly associated with severe preterm delivery, but is not related to complications such as PIH or SGA. The frequency reported at 20–24 weeks of gestation is >80%, falling to <10% of deliveries at term [23]. We report a similar frequency of histological chorioamnionitis in this cohort of a near-term unselected population, indicating that the criteria used for classification are consistent with previous studies and therefore supporting correct interpretation. Similarly, the frequencies of clinical complications such as PET, PIH, SGA and GDM and histological findings, such as fetal thrombotic vasculopathy, are also broadly similar to published epidemiological data, indicating that both our population and interpretation of histological findings are consistent with expected frequencies. This supports the generalisibility of the findings in placentas from deliveries at or near term; however, these findings should not be applied to pregnancies with more severe complications resulting in preterm deliveries.
The sensitivity for most histological findings was low. It is recognized that diagnostic macroscopic and histological changes in the placenta from cases with PET and PIH may not be present even in clinically severe disease. Previous studies have reported increased frequency of abnormal histological features, such as placental infarcts, in placentas from pregnancies which are complicated by preterm PET/PIH. In the current study, the frequency of chronic placental underperfusion, VUE, MPFD and chorioangiomata were indeed greater in pregnancies with PET/PIH; however, since the incidence of PET and PIH in an unselected population is so low compared to the frequency of normal uncomplicated pregnancies, the positive predictive value of such histological features is very low. For example, even though this study has demonstrated that chorioangiomata are much commoner in cases with PET, most cases of PET will not show such lesions (low sensitivity), and the majority of cases with chorioangiomata present will have a normal outcome (low positive predictive value).
Previously described histological features in gestational diabetes are similar to those of pre-existing diabetes mellitus; however they are usually less pronounced and reduced in frequency [24, 25]. The pathological findings of the placenta do not predict the severity of the disease or relate to the duration of the disease, [25] and in practice, any morphological changes are usually subtle and difficult to identify on routine examination. Some specific histological lesions are more frequent, for example, placentas from diabetic pregnancies show increased incidence of vascular pathological changes such as FTV and chorangiosis which we also demonstrate [18]. In the present study, both of these histological lesions were more than twice as common in pregnancies complicated with GDM compared to the normal population, but both FTV and chorangiosis have very low sensitivities for the detection of GDM and positive predictive values of 10%; on finding FTV in an unselected placenta at term, >90% of cases will not be associated with GDM or any other obstetric complication. Nevertheless, it should be recognized that, based on previous case–control study data, FTV may be associated with fetal/neonatal complications such as visceral thrombotic lesions, regardless of whether they are GDM related or not. However, since the majority of these cases will be apparently clinically uncomplicated pregnancies, as demonstrated in the present series, according to most guidelines and the practice of the majority of centres, these placentas will not be routinely examined, and hence, the presence of such lesions will remain unrecognized!
Histological lesions found in association with SGA were, as expected, primarily those of chronic placental underperfusion, VUE, MPFD, chorangioma, fetal thrombotic vasculopathy and chronic histiocytic intervillositis. SGA by our definition encompasses not only small, growth restricted babies but also those constitutionally small. These are all expected histological features based on previous case–control study data and represent changes secondary to the maternal vascular compromise leading to abnormal placental perfusion or other specific IUGR associations. Interestingly, the frequency of histological changes of chronic placental underperfusion was only slightly greater in cases with SGA compared to normals, highlighting the fact that in pregnancies delivering with mild growth restriction at term, there may be minimal morphological changes in the placental parenchyma, the sensitivity for SGA being only 10%. Conversely, similar morphological features may be commonly seen at term in pregnancies resulting in normal outcomes, with >85% of all cases with histological changes such as infarcts being included in the normal group. Previous studies considering placentas from clinically preselected outcome groups will therefore intrinsically overestimate the importance of any identified histological finding since the vast majority of pregnancies with normal outcome do not undergo placental examination. Furthermore, selecting known ‘normal’ cases as controls is also inappropriate because the pathologist will be biassed as to the supposed clinical significance of a histological finding if the outcome is already known.
The major strengths of this study are the prospective recruitment and collection of a large number of unselected cases near term, specifically for study purposes, with uniform methodology of placental examination and outcome definitions. A unique feature to remove any interpretative bias is that all histological examinations were performed by two independent paediatric pathologists who were blinded to all clinical details, including outcome and gestational age. All histological features were recorded in an unbiased manner for the entire population, comparison of frequencies of histological lesions across subgroups in the absence of any interpretive bias. The weaknesses of the study are that, despite of prospective recruitment of over 1,000 women, many histological abnormalities have a low incidence, and only small numbers of cases were recorded in some outcome groups. Therefore, where we have low number of cases in outcome groups, the odds ratio with wide confidence interval may not be robust. Also, these findings cannot be extrapolated to preterm cases, in which the frequency of complications and pathophysiology of disease is different. The data from this study must not therefore be used to suggest that there is no value in placental histological examination, since strong data exist from high-risk case control studies regarding association between certain placental lesions and obstetric complications. However, the data do provide contemporaneous reference data regarding the frequencies of a range of placental histological features in an unselected population delivering at or near term in the UK and highlight the important finding that simply demonstrating the presence of a placental histological ‘lesion’ should not necessarily be regarded as evidence of a clinically significant obstetric complication or causation, and the clinical significance of placental pathological features should not be over interpreted.
In summary, we present the frequencies of specific placental histological features in an unselected population in the late third trimester and report that whilst certain histological features are found more commonly in association with complicated pregnancies, the sensitivity and positive predictive values for these findings are generally low. The vast majority of placentas with histological abnormalities will be seen in clinically normal pregnancies, and histological examination may be normal even in the presence of clinically severe obstetric disease.
References
Langston C, Kaplan C, Macpherson T, Manci E, Peevy K, Clark B, Murtagh C, Cox S, Glenn G (1997) Practice guideline for examination of the placenta: developed by the Placental Pathology Practice Guideline Development Task Force of the College of American Pathologists. Arch Pathol Lab Med 121:449–476
Hargitai B, Marton T, Cox PM (2004) Best practice no 178. Examination of the human placenta. J Clin Pathol 57:785–792
Khong TY, Gordijn SJ (2003) Quality of placental pathology reports. Pediatr Dev Pathol 6:54–58
Sibai B, Dekker G, Kupferminc M (2005) Pre-eclampsia. Lancet 365:785–799
Macintosh MC, Fleming KM, Bailey JA, Doyle P, Modder J, Acolet D, Golightly S, Miller A (2006) Perinatal mortality and congenital anomalies in babies of women with type 1 or type 2 diabetes in England, Wales, and Northern Ireland: population based study. BMJ 333:177
Garite TJ, Clark R, Thorp JA (2004) Intrauterine growth restriction increases morbidity and mortality among premature neonates. Am J Obstet Gynecol 191:481–487
Zhang P, Schmidt M, Cook L (2006) Maternal vasculopathy and histologic diagnosis of preeclampsia: poor correlation of histologic changes and clinical manifestation. Am J Obstet Gynecol 194:1050–1056
Roberts DJ, Post MD (2008) The placenta in pre-eclampsia and intrauterine growth restriction. J Clin Pathol 61:1254–1260
Daskalakis G, Marinopoulos S, Krielesi V, Papapanagiotou A, Papantoniou N, Mesogitis S, Antsaklis A (2008) Placental pathology in women with gestational diabetes. Acta Obstet Gynecol Scand 87:403–407
Brosens IA, Robertson WB, Dixon HG (1972) The role of the spiral arteries in the pathogenesis of preeclampsia. Obstet Gynecol Annu 1:177–191
Salafia CM, Pezzullo JC, Ghidini A, Lopez-Zeno JA, Whittington SS (1998) Clinical correlations of patterns of placental pathology in preterm pre-eclampsia. Placenta 19:67–72
Sebire NJ, Goldin RD, Regan L (2005) Term preeclampsia is associated with minimal histopathological placental features regardless of clinical severity. J Obstet Gynaecol 25:117–118
Shanklin DR, Sibai BM (1989) Ultrastructural aspects of preeclampsia. I. Placental bed and uterine boundary vessels. Am J Obstet Gynecol 161:735–741
Oliveira LH, Xavier CC, Lanaa M (2002) [Changes in placental morphology of small for gestational age newborns]. J Pediatr (Rio J) 78:397–402
Altemania M, Gonzattia R (2003) [Villitis of unknown etiology in placentas of pregnancies with hypertensive disorders and of small-for-gestational-age infants]. Rev Assoc Med Bras 49:67–71
Pathak S, Hook E, Hackett G, Murdoch E, Sebire NJ, Jessop F, Lees C (2010) Cord coiling, umbilical cord insertion and placental shape in an unselected cohort delivering at term: relationship with common obstetric outcomes. Placenta 31:963–968
Pathak S, Sebire NJ, Hook L, Hackett G, Murdoch E, Jessop F, Lees C (2011) Relationship between placental morphology and histological findings in an unselected population near term. Virchows Arch 459:11–20
Fox H, Sebire NJ (2007) Pathology of the Placenta. W.B.Saunders, London
Davey DA, Macgillivray I (1988) The classification and definition of the hypertensive disorders of pregnancy. Am J Obstet Gynecol 158:892–898
Brown MA, Lindheimer MD, De Swiet M, Van Assche A, Moutquin JM (2001) The classification and diagnosis of the hypertensive disorders of pregnancy: statement from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Hypertens Pregnancy 20:IX–XIV
WHO (2006) World Health Organisation/International Diabetes Federation, definition and diagnosis of diabetes mellitus and intermediate hyperglycemia
http://Www.Healthforallchildren.Co.Uk/. Four-in-one (decimal) growth charts (birth-20 yrs). London: Child Growth Foundation 1996/1
Sebire NJ, Goldin RD, Regan L (2001) Histological chorioamnionitis in relation to clinical presentation at 14–40 weeks of gestation. J Obstet Gynaecol 21:242–245
Fox H (1969) Pathology of the placenta in maternal diabetes mellitus. Obstet Gynecol 34:792–798
Jones CJ, Fox H (1976) Placental changes in gestational diabetes. An ultrastructural study. Obstet Gynecol 48:274–280
Funding
The study was supported by grants from Cambridge Fetal Care and Addenbrooke's Charitable Trust.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Appendix 1
Appendix 1
Rights and permissions
About this article
Cite this article
Pathak, S., Lees, C.C., Hackett, G. et al. Frequency and clinical significance of placental histological lesions in an unselected population at or near term. Virchows Arch 459, 565–572 (2011). https://doi.org/10.1007/s00428-011-1157-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-011-1157-z