Abstract
This study employs labels for cell proliferation and cell death, as well as classical histology to examine the fates of all eight neural stem cells (neuroblasts) whose progeny generate the central complex of the grasshopper brain during embryogenesis. These neuroblasts delaminate from the neuroectoderm between 25 and 30 % of embryogenesis and form a linear array running from ventral (neuroblasts Z, Y, X, and W) to dorsal (neuroblasts 1-2, 1-3, 1-4, and 1-5) along the medial border of each protocerebral hemisphere. Their stereotypic location within the array, characteristic size, and nuclear morphologies, identify these neuroblasts up to about 70 % of embryogenesis after which cell shrinkage and shape changes render progressively more cells histologically unrecognizable. Molecular labels show all neuroblasts in the array are proliferative up to 70 % of embryogenesis, but subsequently first the more ventral cells (72–75 %), and then the dorsal ones (77–80 %), cease proliferation. By contrast, neuroblasts elsewhere in the brain and optic lobe remain proliferative. Apoptosis markers label the more ventral neuroblasts first (70–72 %), then the dorsal cells (77 %), and the absence of any labeling thereafter confirms that central complex neuroblasts have exited the cell cycle via programmed cell death. Our data reveal appearance, proliferation, and cell death proceeding as successive waves from ventral to dorsal along the array of neuroblasts. The resulting timelines offer a temporal blueprint for building the neuroarchitecture of the various modules of the central complex.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
The insect central complex is a brain-specific structure possessing a modular neuroarchitecture whose basic elements—the protocerebral bridge (PB), central body (CB), ellipsoid body (EB), noduli, and various accessory lobes—are remarkably conserved across insect species (see Strausfeld 2012 for an overview) and may even be equivalent to the mammalian basal ganglia in evolutionary terms (Strausfeld and Hirth 2013). It is involved in multi-sensory information processing and in coordinating motor behavior (Homberg 1994; Mappes and Homberg 2004; Liu et al. 2006; Heinze and Homberg 2007; Neuser et al. 2008; Weinrich et al. 2008; Weir and Dickinson 2012), and mutant analyses confirm that the neural substrates for complex behaviors appear to be mapped into its neuroarchitecture (Strauss et al. 1992; Strauss and Heisenberg 1993; Strauss 2002).
The neurons of the insect brain, like those in the ventral nerve cord, derive from bilaterally symmetrical pairs of neural stem cells (so-called neuroblasts (NBs)) which delaminate from the neuroectoderm of each hemisphere at stereotypic locations during development (Malzacher 1968; Bate 1976; Doe and Goodman 1985a, 1985b; Zacharias et al. 1993; Goodman and Doe 1994; Younossi-Hartenstein et al. 1996, 1997; Boyan and Williams 1997; Urbach and Technau 2003; Urbach et al. 2003a, 2003b; Williams et al. 2005). Each NB acquires an intrinsic capacity for neuronal proliferation and generates a specific lineage of progeny via molecular mechanisms which are now largely understood (Broadus et al. 1995; Doe and Skeath 1996; Younossi-Hartenstein et al. 1996, 1997; Pearson and Doe 2003; Urbach and Technau 2003; Urbach et al. 2003a, 2003b; Doe 2008; Reichert 2011) and which are at least partially conserved across species (Broadus and Doe 1995; Boyan and Reichert 2011; Reichert 2011). Nevertheless, once delaminated, NBs can undergo different fates according to the lifestyle of their host and their location in its nervous system.
In holometabolous insects (e.g., Drosophila) brain development is stochastic in that embryonic NBs may exit the cell cycle at a specific time, remain quiescent for a period, and then reenter the cell cycle again later (e.g., postembryonically) to generate second-order progeny (Truman and Bate 1998; Prokop and Technau 1991; Ito et al. 1997; Pereanu and Hartenstein 2006; Ito and Awasaki 2008; Lai et al. 2008; Izergina et al. 2009; Reichert 2011). Accordingly, proliferating postembryonic NBs of the brain are found associated with integrative centers such as the central complex, mushroom bodies, and optic lobes which arise during late larval stages and are thus first functional in the adult (Nordlander and Edwards 1969; Meinertzhagen 1973; Ito et al. 1997; Hartenstein et al. 1998, 2008; Renn et al. 1999; Pereanu and Hartenstein 2006; Ito and Awasaki 2008; Izergina et al. 2009; Young and Armstrong 2010a, 2010b; Boyan and Reichert 2011; Reichert 2011; Riebli et al. 2013; Viktorin et al. 2013). However, in Drosophila such NBs exit the cell cycle by the end of metamorphosis so that there are no identifiable NBs in the adult brain (Ito and Hotta 1992).
Development of the nervous system in hemimetabolous insects, by contrast, is continuous in that delaminated NBs remain proliferative during their lifespan and are not known to exit and then re-enter the cell cycle at different times (Bate 1976; Doe and Goodman 1985a, 1985b; Doe et al. 1985; Reichert and Boyan 1997; Boyan and Reichert 2011). Regional differences in NB fates exist nevertheless. All the NBs of the grasshopper ventral nerve cord, for example, were found to disappear by the end of embryogenesis (Shepherd and Bate 1990). This implies that no further central neurons are added to embryonic thoracic cellular networks and is consistent with the finding that central motor pathways can be functional before hatching even though the associated external structures (such as wings) have not completely developed (Stevenson and Kutsch 1986). In the brain, the large numbers of neurons and glia comprising the mushroom bodies and optic lobes are generated by proliferative clusters which survive embryogenesis and are active at postembryonic and even adult stages in various orthopteroid insects (Panov 1960; Vanhems 1985, 1995; Cayre et al. 1994, 1996, 2007; Farris and Sinakevitch 2003). The central complex, by contrast, develops differently.
Of the 96 NBs in each protocerebral neuromere of the grasshopper brain, eight located in the so-called pars intercerebralis region have been identified as contributing progeny to the modules of the central complex (Boyan and Williams 1997; Williams et al. 2005). These NBs are no longer present postembryonically (Boyan et al. 2010a, c) which means that the central complex must develop sufficiently during embryogenesis in order to mediate fundamental behaviors such as orientation, locomotion and feeding at the time of hatching (see Chapman 1982). This temporal restriction necessitates the presence of additional so-called intermediate progenitors within such lineages to generate the required number of progeny in time (Boyan at al. 2010c; Boyan and Reichert 2011). Significantly, the study of Shepherd and Bate (1990) on the ventral nerve cord remains the only one known to us in which the complete timelines of NBs in the grasshopper have been documented. How long central complex NBs are proliferative, and exactly how they exit the cell cycle (final terminal division vs. programmed cell death), have not been determined. The cellular and molecular bases of downstream central complex development, such as the establishment of its fine neuroarchitecture (Boyan et al. 2008) or the sequential expression of neuromodulators (Boyan et al. 2010b; Herbert et al. 2010; Boyan and Williams 2011), therefore remain unclear.
In this study, we employ molecular labels in combination with histology to document the timelines of the eight NBs of each protocerebral hemisphere that generate the central complex of the grasshopper Schistocerca gregaria during embryogenesis. Four of these NBs (Z, Y, X, and W) generate progeny whose axons project to the PB and then the columnar system of the CB and the EB via the so-called z, y, x, and w tracts, while the progeny from four others (1-2, 1-3, 1-4, and 1-5) direct axons initially into other central complex modules such as the noduli and various accessory lobes (Boyan et al. 2010b; Boyan and Niederleitner 2011; Boyan and Williams 2011). We document the time period of proliferation in all these NBs, show when and how they exit the cell cycle—via programmed cell death rather than a final terminal division. Significantly, proliferation and death proceed in temporally ordered waves along the array of NBs. The resulting timelines offer a temporal blueprint for building the neuroarchitecture of the various modules of the central complex.
Materials and methods
Animals
Animals were held in crowded cages maintained on a 12-h/12-h light/dark cycle with air humidity at a constant 35 %. An internal lamp in each cage ensured a constant day temperature of around 30 °C; night temperatures hovered around 20 °C. S. gregaria eggs from our own culture were incubated in moist aerated containers at 30 °C. Embryos were staged at time intervals equal to percentage of embryogenesis according to Bentley et al. (1979). All experiments were performed strictly according to the guidelines for animal welfare as laid down by the German Research Council (DFG).
Dissection and preincubation
Staged embryos were dissected out of the egg into ice-cold 0.1 M phosphate-buffered saline (PBS) and freed from embryonic membranes. Heads were detached and immersion fixed overnight at 4 °C in 3.7 % paraformaldehyde in 0.1 M PBS for anti-1C10, anti-8B7, anti-HRP, anti-PH3 labeling. Labeling for PCNA, terminal deoxynucleotidyl transferase deoxyuridine triphosphate nucleotides (dUTP) nick end labeling (TUNEL), BrdU, and 4,6-diamidino-2-phenylindole (DAPI) proceeded as described in the appropriate sections below. Preparations were then washed repeatedly in PBS prior to embedding in 5 % Agarose/PBS at 55–60 °C, the solution allowed to cool, and the resulting block serially sectioned on a Vibratome (Leica VT 1000S) at 50 μm thickness. Sections were collected and washed overnight in 0.1 M PBS at 4 °C. Sections were then freed from agarose, positioned onto Superfrost® Plus (Menzel-Gläser) microscope slides, and covered with preincubation medium comprising 1 % normal goat serum (NGS), 0.1 % bovine serum albumin (BSA), PBT (0.05 % Triton X-100 in 0.1 M PBS, pH 7.4) for 1 h at room temperature to block unspecific binding sites. Immunolabeling was performed directly on this sectioned material. Brain slices were exposed to primary antibodies for 48 h at 4 °C in the dark unless otherwise stated.
Cell identification marker
Anti-1C10
The 1C10 (Lachesin) antibody recognizes a cell surface molecule belonging to the Ig superfamily (Karlstrom et al. 1993). In grasshopper, expression of the molecule occurs initially on all differentiating epithelial cells, but only cells involved in neurogenesis such as NBs continue to express the molecule. Lachesin has additionally been found to play roles in neuronal differentiation and axogenesis. Mutant analyses in Drosophila show that the molecule is required for the blood–brain barrier (Strigini et al. 2006) and morphogenesis of the tracheal system (Llimargas et al. 2003). The 1C10 antibody (mouse, gift of M. Bastiani) was diluted 1:5,000 in preincubating medium.
Cell proliferation markers
Anti-phospho-Histone H3 (Ser10)
H3, one of the four core histones wrapped inside genomic DNA and forming the core nucleosome complex, is involved in the packing of DNA into chromatin (see Takizawa and Meshorer 2008). The PH3 antibody recognizes and binds the phosphorylated form of the amine terminal of Histone 3. This binding is only possible when the chromatin lies dissociated from the nucleosome complex, as occurs during mitotic chromosome condensation. Chromosomes in late meta- and early anaphase are labeled best because phosphorylation is then a maximum (see Hendzel et al. 1997; Adams et al. 2001 for details). The PH3 antibody (rabbit, Millipore) was diluted 1:250 in preincubating medium.
Anti-PCNA
Proliferating cell nuclear antigen, also known as cyclin (Bravo and Celis 1980) is an evolutionarily highly conserved 36 kD acidic nuclear protein associated with the cell cycle (Celis et al. 1987; Hall et al. 1990; Zudaire et al. 2004). This protein functions as a co-factor for DNA polymerase δ, therefore being an essential requirement for DNA repair synthesis (Celis et al. 1987; Fairman 1990) as well as chromosome replication (Bravo and Macdonald-Bravo 1987; Shreeram and Blow 2003) occurring immediately prior to mitosis. Immunocytochemical detection of PCNA has been shown to be a useful and straightforward tool to assess the cell proliferation status of mammalian (Celis and Celis 1985) and insect (Ng et al. 1990; Yamaguchi et al. 1991, 1995; Zudaire et al. 2004) cells. Prior to PCNA antibody incubation, the preparations were placed in HIER buffer (19.0 ml 011 M citric acid, 81.0 ml 0.1 M sodium citrate, pH 6.0) and heat-shocked at 80 °C for 20 min. The PCNA primary antibody (mouse, Dako Cytomation) was diluted 1:500 in a preincubating medium comprising 5 % BSA, 0.5 % Triton X-100, and 0.1 % saponin in 0.1 M PBS (pH 7.4).
5-Bromodeoxyuridine
5-bromodeoxyuridine (BrdU) incorporation occurs during DNA replication in the S-phase of the cell cycle, is therefore an indicator of mitotically active cells. For 5-bromodeoxyuridine (BrdU) incorporation in whole embryo culture, eggs were sterilized in 70 % ethanol for 60 s, blotted dry on filter paper, and then transferred into filter-sterilized Mitsuhashi and Maramorosch insect medium (Sigma) supplemented with 10 % NGS or FCS, 5 μl/ml penicillin-streptomycin and 150 μl/l 20-hydroxyecdysone. This medium also served for dissecting the staged embryos out of the egg. The embryos were then incubated in a cell culture dish containing the above medium together with BrdU (final concentration 10−2 M). The incubation took place at 30 °C with gentle agitation over pulses of 15–16 h (this represents approximately 3 % of development for each stage examined). Afterwards the embryos were restaged and fixed in Pipes-FA (100 mM Pipes, 2 mM EGTA, 1 mM MgSO4, and 3.7 % paraformaledhyde; pH 6.8–7.4) for 1 h. Following fixation, the embryos were washed thoroughly in PBS (3 × 15 min), their DNA denatured in a solution of 2 N HCL in PBS for 20 min, and then washed again.
For BrdU immunolabeling, the embryos were incubated at room temperature in a blocking solution of 0.4 % PBT (PBS plus 0.4 % Triton X-100), 5 % NGS, and 0.2 % BSA for 45 min. The primary antibody (mouse anti-BrdU, Sigma) was diluted 1:200 in the blocking solution. Incubation was at 4 °C with gentle agitation for 24 h. Embryos were then washed in 0.025 % PBT (PBS plus 0.025 % Triton X-100), and incubated in the secondary antibody (Dako EnVision™, GAM-PO, dilution 1:3 in PBS) at room temperature under gentle agitation for 15–16 h. Embryos were then washed in PBS, stained with diaminobenzidine (DAB) containing 0.02 M NiCl2 for signal intensification (DAB fast tablets, Sigma). The reaction was stopped through several rinses with PBS. Preparations were embedded in soft Epon, sectioned at 20 μm, covered in 90 % glycerol in PBS and coverslipped for viewing.
Cell death markers
Acridine orange
Acridine orange (AO) is an acknowledged fluorescent dye for labeling apoptotic cells in insect tissues (e.g., Spreij 1971; Abrams et al. 1993; Jiang et al. 1997; Stacey et al. 2010; Liu and Boyan 2013). Wholemount brains were exposed to AO solution (5 μg/ml in PBS) for 30 min at room temperature, and this was followed by four washing cycles each of 20 min duration in 0.1 M PBS. The brains were then embedded in agarose medium and sectioned with a Vibratome at 55 μm thinkness. TUNEL technology and DAPI counterstaining was then directly applied to the brain slices as described elsewhere in the “Materials and methods” in order to reveal the pattern of cell death in NBs of the embryonic central complex.
TUNEL
TUNEL is a method for detecting DNA fragmentation in cells undergoing apoptosis (Gavrieli et al. 1992; Bello et al. 2003). So-called nicks or strand breaks in DNA occurring during fragmentation can be identified by the presence there of accessible 3′-hydroxyl (3′-OH) groups. The assay detects dUTPs, which are added to the 3′-OH end of the DNA strand breaks by the enzyme terminal deoxynucleotidyl transferase, and have been previously labeled with an appropriate fluorescent marker.
After dissection, brains were fixed overnight in 3.7 % PFA at 4 °C, embedded in Agarose as above and sectioned with a Vibratome at 60 μm. After washing in PBS at 4 °C, brain slices were bathed in Citrate buffer and heated with a microwave oven (5 min, 360 W) and washed again overnight in PBS at 4 °C. We then incubated the brain slices in preincubating medium (TrisHCl, 0.1 M; pH 7.5 with 20 % NGS and 3 % BSA) for 30 min at room temperature. Samples were then washed 3 times (each time for 30 min) with PBS. The TUNEL (In Situ Cell Death Detection Kit, TMR red; Roche) reaction was performed according to the instructions accompanying the product. Brain slices were then washed in PBS for 2 h at room temperature, counterstained with DAPI (see below), covered in Vectashield® (Vector laboratories) and coverslipped for microscopy.
Secondary antibodies
After exposure to the primary antibody, brain slices were washed thoroughly in 0.1 M PBS and then placed in preincubation medium (see above) to which the relevant secondary antibody was added for 24 h. at 4 °C as follows: for anti-PH3 (GAR-Alexa 488, Invitrogen, 1:250 dilution); for anti-1C10 (GAM-Cy3, Dianova, 1:150 dilution); for PCNA (GAM-Cy3, Sigma, 1:150 dilution). For double stainings appropriate secondary antibodies were selected in each case so as to prevent spectral confusion. Sections were then washed overnight in 0.1 M PBS at 4 °C in the dark, covered with Vectashield® (Vector laboratories) and coverslipped for microscopy. Specificity of each secondary antibody was confirmed by its application in the absence of the primary antibody (in no case was a staining pattern observed).
Nuclear marker
DAPI
DAPI (Sigma) is a cell permeable fluorescent probe which binds to the minor groove of double-stranded DNA (Naimski et al. 1980), and has additionally been previously proven to label proliferative cells especially at metaphase and anaphase in Xenopus (Sumara et al. 2000) and in grasshopper as confirmed by comparing the staining patterns for DAPI and the mitosis marker anti-phospho-Histone 3 (Liu and Boyan 2013). DAPI was diluted 1:100 in 0.1 M PBS. Brain slices were exposed to DAPI for 30 min at room temperature, and this was followed by 6 washing cycles each of 20 min duration in 0.1 M PBS.
Histology
Osmium tetroxide-ethyl gallate
Staged embryos were dissected out of the egg into PBS, freed from membranes, then fixed in Carnoy fixative (60 % ethanol, 30 % chloroform, and 10 % glacial acetic acid) for one hour and brought to distilled water through a descending ethanol series. Postfixation of the brain tissue in 1 % osmium tetroxide for 17 to 48 h was followed by thorough washing in distilled water. For signal intensification, the embryos were maintained in a saturated aqueous solution of ethyl gallate (several changes over 48 h at room temperature). The tissue was then washed in distilled water, dehydrated through an ascending ethanol series to propylene oxide, embedded in soft Epon, sectioned at between 10 and 14 μm thickness, and mounted on a glass slide in Permount.
Imaging
Optical sections of preparations were acquired with a Leica TCS SP5 confocal laser scanning microscope equipped with ×20 and ×63 oil immersion objectives. Fluorochromes were visualized on the confocal microscope using an argon laser with excitation wavelengths of 488 nm for Alexa® 488, 561 nm for Cy3. Z-stacks of confocal images were collated using public domain software (Image J) and only the contrast, resolution and color were subsequently adjusted. Histological sections of the brain were viewed under a Zeiss Axioskop 2 microscope using differential interference contrast or bright field optics. Images were captured with a 1.3 MP color CCD camera (Scion Corp.) using Scion Visicapture™ software and processed with Canvas™ X.
Terminology
Neuroblasts
NBs were mapped according to Boyan and Williams (1997) and Williams et al. (2005). The equivalences between the nomenclatures in the two studies are: W (NB 1-1), X (NB 2-1), Y (NB 3-1), and Z (NB 4-1); NBs 1-2, 1-3, and 1-4 are common to both.
Axes
Unless otherwise stated, the axes used for anatomical analysis in this study are neuraxes, not body axes. The top of the brain (in the head) is neurally anterior, the front of the brain is neurally ventral, the back of the brain is neurally dorsal, and the base of the brain is neurally posterior (see Boyan et al. 1993). Planes of section are also defined with respect to the neuraxis (a horizontal section is parallel to, a transverse section is orthogonal to, the neuraxis).
Results
Temporal appearance and organization of central complex NBs
In this study we examine the temporal fates of neural stem cells (NB) whose progeny generate the central complex in the protocerebral neuromere of the grasshopper brain during embryogenesis. Previous studies have identified eight NBs (Z, Y, X, W, 1-2, 1-3, 1-4, and 1-5) of the total complement of 96 in each hemisphere as contributing progeny to the central complex (Boyan and Williams 1997; Williams et al. 2005). In order to visualize them early in development we employed 5-bromodeoxyuridine (BrdU) incorporation (Fig. 1). This occurs during DNA replication in the S-phase of the cell cycle, so that BrdU labels those cells en route to the mitotic phase of the cell cycle. The time window we selected for BrdU incorporation (25–34 % of embryogenesis) covers the period over which NBs first appear in the ventral nerve cord (Bate 1976; Doe and Goodman 1985a, b; Shepherd and Bate 1990) and when pioneer neurons are first establishing the tract system from the PB to the CB in the brain (Williams et al. 2005; Boyan and Williams 2011).
Temporal appearance and organization of central complex neuroblasts. a–c Photomicrographs of sections of the protocerebral midbrain following 5-bromodeoxyuridine (BrdU) incorporation and subsequent anti-BrdU immunolabeling reveal organization of central complex neuroblasts (NBs) during early embryogenesis. Ventral according to the neuraxis is to the top throughout. a At 25 % of embryogenesis NBs of the protocerebrum (PC) are still in the process of delaminating from the neuroectoderm. Central complex NBs Z, Y, X, W, and 1-2 of each brain hemisphere are already occupying their stereotypic locations in the array. More dorsally, NB 1-4 on the left has appeared as has 1-3 on the right. Spaces (circled) are for NBs which have yet to incorporate BrdU. Dashed line indicates brain midline. b Between 29 and 30 % of embryogenesis, the linear array of NBs from Z through to 1-3 is complete (NB 1-3 on the left is out of picture). Note the bilaterally symmetrical organization. Not all central complex NBs have synchronously incorporated BrdU. c At 34 % of embryogenesis, the compliment of protocerebral NBs including those not associated with the central complex is largely complete and NBs Y, W have generated their first progeny (black stars). Cells of the median domain (MD) are also proliferative. d The stereotypic location of NBs established early in embryogenesis is subsequently maintained. Photomicrograph of a section through the brain at low magnification following osmium ethyl gallate (OsEG) histology at 47 % of embryogenesis and visualized via DIC optics reveals the complete linear array of protocerebral NBs which contribute to the central complex: Z, Y, X, W, 1-2, 1-3, 1-4, and 1-5, along with some of their lineages (z, y, x, and w). Note the bilateral symmetry. Borders of each protocerebral hemisphere are dashed white. e Image from (d) but at higher magnification reveals stereotypic differences in NB size and nuclear morphology in representative NBs (Y and X). Note bilateral symmetry. f Photomicrograph of the bilateral NBs (X) from a different preparation to d, e above shows consistent morphological features which allow this type to be distinguished from other NBs in the array. g Photomicrographs of NB 1-2 from three different preparations (i–iii) confirm consistency of nuclear morphology (white stars) characterizing this NB type. h Stereotypic locations of NBs allow a reference map to be generated for the protocerebrum. NBs whose progeny contribute to the central complex are colored black. Panel modified with permission of Elsevier Press from Boyan and Williams (2011). Other abbreviations: mOc median ocellus, MP midline precursor, OL optic lobe. Scale bar represents 65 μm in (a), 40 μm in (b) and (c); 50 μm in (d), 20 μm in (e)–(g), and 115 μm in (h)
Brain slices of the protocerebral midbrain following BrdU incorporation and subsequent anti-BrdU immunolabeling at 25 % of embryogenesis reveal NBs of the protocerebrum (PC) still in the process of delaminating from the neuroectoderm (Fig. 1a). Central complex NBs appear to be among the earliest to delaminate in the PC, and of these the more ventral ones (Z, Y, X, and W) appear and incorporate BrdU first. Of the more dorsal subset, NBs 1-2 of each brain hemisphere already occupy their future stereotypic locations in the array (c.f. Fig. 1h), while NB 1-4 on the left and 1-3 on the right have appeared. Spaces are evident where other NBs will delaminate and then incorporate BrdU, consistent with this occurring at predetermined locations in the neuroectoderm. Between 29 and 30 % of embryogenesis, the linear array of central complex NBs in each protocerebral hemisphere is complete and their organization is remarkably bilaterally symmetrical (Fig. 1b). At 34 % of embryogenesis, the remaining compliment of protocerebral NBs—such as those not associated with the central complex—is largely complete and NBs Y and W have generated progeny (Fig. 1c).
The locations of the eight central complex NBs early in embryogenesis suggest a topological organization. To see if this is maintained, and to examine the cellular morphology of individual NBs which cannot be revealed using BrdU incorporation, we stained NBs with osmium ethyl gallate. Sectioning at the appropriate depth in the brain at mid-embryogenesis (47 %) for example, allows the complete linear array of central complex NBs (Z, Y, X, W, 1-2, 1-3, 1-4, and 1-5) to be visualized within a single brain slice (Fig. 1d). The bilateral symmetry in NB arrays from both hemispheres is clear, the locations at which individual NBs are found within the array are like those seen earlier in embryogenesis (c.f. Fig. 1c), consistent with a stereotypical topology. At higher magnification (Fig. 1e), NBs can been seen to have a cell-specific size, as well as a characteristic nuclear morphology, which together allow them to be individually identifiable within the array. These features are consistent in the homologs from both brain hemispheres (Fig. 1e, f), and repeatable for a given NB from preparation to preparation (Fig. 1e–g(i–iii)). The resulting consistency in location and appearance allow a reference map to be created in which the central complex NBs are seen organized into a linear array stretching from ventral to dorsal symmetrically along the median edge of each protocerebral hemisphere (Fig. 1h). The array can be subdivided functionally. Progeny of the more ventral NBs (Z, Y, X, and W) are known to generate so-called small-field neurons whose axons project via the so-called w, x, y, and z tract system through the PB and then to the CB where they form a stereotypic columnar system (Williams 1975; Boyan and Williams 1997, 2011; Heinze and Homberg 2008). The more dorsal NBs (1-2, 1-3, 1-4, and 1-5) generate so-called wide-field progeny which direct axons initially into other central complex modules such as the noduli and various accessory lobes (Heinze and Homberg 2008; Boyan et al. 2010b; Boyan and Niederleitner 2011; Boyan and Reichert 2011). In contrast to the ventral nerve cord (Shepherd and Bate 1990), the fates of these brain NBs have remained unresolved to date, but are documented below.
Visualization of all central complex NBs through embryogenesis
Application of the map derived from osmium ethyl gallate histology allows all eight central complex NBs to be identified up to 70 % of embryogenesis (Fig. 2). As is obvious from the compilation, individual NBs maintain their morphological features (cell size, nuclear shape) over a broad time spectrum. However, with progressive age (beyond 67 %), shrinkage in size by up to one third occurs, and particularly ventral NBs such as Z, Y, and X also become flattened in shape. As of 72 %, ventral NBs—first Z and Y and then X and W—can no longer be uniquely identified by histological methods alone, although they may still be present. The more dorsal NBs (1-2, 1-3, 1-4, and 1-5) by contrast, are still clearly identifiable even at 77 %, and can even be proliferative at this age (see Figs. 3 and 4 below). No central complex NBs were identifiable after 80 % using osmium-based histological techniques alone.
Photomicrographs of all individual, identified, central complex NBs following staining with osmium ethyl gallate (OsEG) and visualized via DIC optics between 37 and 77 % of embryogenesis. Note that with progressive age (67 %) NBs such as Z, Y, and X become flattened in shape, while all clearly shrink in size. As of 72 %, ventral NBs—first Z, Y and then X, W—become histologically not recognizable (nr) at their stereotypic locations although they may still be present. More dorsal NBs (1-2, 1-3, 1-4, and 1-5) by contrast, are still clearly identifiable. After 77 %, no NBs were identifiable using OsEG alone. Scale bar represents 15 μm for all NBs up to and including 67 % and 10 μm at 72 and 77 %
Timeline of proliferation in central complex NBs during embryogenesis. Panels show brain slices imaged confocally at the level of the protocerebral bridge (PB) in the right protocerebral hemisphere (PC; approximate border shown white; m brain midline) following immunolabeling against the proliferation marker PCNA (red) and counterstaining with the DNA label DAPI (blue). At 50 (a), 55 (b), 60 (c), 65 (d), 68 (e), and 70 % (f), all central complex NBs are proliferative; however, PCNA immunoreactivity progressively weakens in all NBs with age. Absence of the PCNA label indicates that ventral NBs (Z, Y, X, and W) cease proliferation earlier than dorsal ones (1-2, 1-3, and 1-4), so that at 75 % (g) only the most dorsal two, NBs 1-4 and 1-5 (not visible in section), are still proliferative. NB locations at 70 %, 75 % are circled white for clarity. Scale bar represents 40 μm in (a)–(g)
Timeline of mitosis in central complex NBs during embryogenesis. a–h Confocal images of a representative ventral NB (Z) and dorsal NB (1-3) at successive ages during embryogenesis: 50 (a, b), 60 (c, d), 68 (e, f), and 72 % (g, h) following double immunolabeling with the mitosis marker phospho-histone 3 (PH3; green) and the cell marker 1C10 (red). Approximate dimensions of each NB are indicated dashed white. Both NBs are mitotically active up to 68 %; at 72 %, only NB 1-3 is PH3-immunoreactive (h, white arrows) and therefore proliferative. Note change in shape of the Z NB at 72 % (g). Scale bar represents 15 μm in (a)–(h)
Timeline of proliferation in central complex NBs during embryogenesis
Mitotic activity was followed first in the complete array of central complex NBs via immunolabeling with the proliferation marker PCNA (see “Materials and methods”) in conjunction with the nuclear stain DAPI. We found the patterns of proliferation (and of programmed cell death, see below) among central complex NBs to be identical in both brain hemispheres throughout development so that we reference only one hemisphere in our data set below.
Brain slices imaged confocally at the level of the PB reveal all central complex NBs of a given protocerebral hemisphere to be uniformly proliferative between 50 and 70 % of embryogenesis (Fig. 3). However, there is also a noticeable progressive weakening of PCNA labeling in these NBs with age (Fig. 3a–f), and, there is also a discernible pattern according to which the labeling within the array ceases. Absence of PCNA labeling consistent with a cessation of proliferation occurs first in ventral NBs (Z, Y, X, and W) and then spreads dorsally, so that at 75 % (Fig. 3g) only NBs from the dorsal group (e.g., 1-3, 1-4, and 1-5) are still proliferative. Examination of brain slices at 80 % of embryogenesis and beyond failed to detect proliferative cells in locations previously occupied by central complex NBs (see also Legl 2009).
In order to confirm the timeline of proliferation suggested by the PCNA labeling above, we repeated the analysis using the true mitosis marker PH3 which labels cells in the meta- to anaphase, rather than just those within the S-phase as is the case for PCNA (see “Materials and methods”). We chose one representative NB (Z) from the ventral group and one from the dorsal group (1-3) for comparison over a time span of 50–72 % of embryogenesis (Fig. 4). Our data show that both NBs are mitotically active up to 68 % of embryogenesis, but subsequently only NBs 1-3 retains mitotic activity while the Z NB has not only changed shape, it is barely discernible with the 1C10 cell marker—consistent with it degenerating (c.f. Fig. 2 and see below). In keeping with our PCNA data, PH3 labeling failed to detect mitotically active stem cells of the central complex after about 77 % of embryogenesis.
Mitotic activity in non-central complex NBs
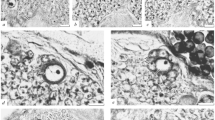
In order to ascertain how representative the timeline for mitotic activity in central complex NBs is for the PC in general, we examined NBs in three other regions of the brain at 80 % of embryogenesis—the lateral PC (Fig. 5a), around the calyces of the mushroom bodies (Fig. 5b), and in the lobula of the visual system (Fig. 5c). In all three regions, osmium staining revealed actively dividing NBs as evidenced by chromatin condensed into chromosomes (e.g., Fig. 5a(ii), b(i–iv), c), and the presence of mitotic spindles (e.g., Fig. 5b (i, ii)). Surrounding these neural stem cells were freshly generated ganglion mother cells (Fig. 5c) which in one case had just divided symmetrically into sibling progeny (Fig. 5a). The data are consistent with NBs in other brain regions maintaining proliferative activity for longer than central complex NBs.
NBs in the brain not associated with the central complex are proliferative even at 80 % of embryogenesis. Photomicrographs (DIC optics) following osmium staining of brain slices at 80 % of embryogenesis reveal chromatin condensed into chromosomes in NBs of the LPC (a i, ii, white arrowhead), of the MB (b i–iv), and of the lobula (c, black star), as well as mitotic spindles in MB neuroblasts (b i, ii, white arrowheads). Furthermore, new sibling progeny generated by the symmetrical division of a ganglion mother cell are visible in the LPC (a i, black stars) while a further ganglion mother cell (GMC) is visible in the lobula (c). Scale bar represents 20 μm in (a)–(c)
Timeline of apoptosis in individual central complex NBs
Our analyses above suggest that: (a) central complex NBs change morphologically with age (Fig. 2); (b) the ventral subset (Z, Y, X, and W) are no longer morphologically recognizable at 77 %, the dorsal subset at 80 % (Fig. 2); (c) the pattern of proliferative activity ceases in these respective NB subsets over these same ages (Figs. 3 and 4). It is conceivable that programmed cell death is the means for exiting the cell cycle and is involved in these changes.
To investigate this possibility, we first examined representative NBs Z and Y along with their lineages at three different ages during embryogenesis using the expression pattern of the apoptosis marker AO along with the nuclear marker DAPI (Fig. 6). Confocal images at 65 % of embryogenesis reveal apoptotic cells present in both Z (Fig. 6a) and Y (Fig. 6b) lineages, but the NBs themselves are AO negative. By 72 % of embryogenesis, both the Z NB (Fig. 6c) and the Y NB (Fig. 6d) express AO as do other progeny in the lineages. At 77 % of embryogenesis, progeny in the respective lineages are still apoptotic but neither the Z NB (Fig. 6e) nor the Y NB (Fig. 6f) can be detected via AO or DAPI, indicating that they have disappeared.
Timeline of apoptosis in central complex NBs. Confocal images reveal expression of the apoptosis marker acridine orange (AO; yellow) in the lineages of the Z and Y neuroblasts at three different ages during embryogenesis. 3D lineages were reconstructed from series of optical sections following counterstaining with the DNA label DAPI (cyan). At 65 % of embryogenesis, apoptotic cells are present in both Z (a) and Y (b) lineages, but the NBs themselves (white stars) are AO negative. At 72 % of embryogenesis, the Z neuroblast (c, white arrowhead) and the Y neuroblast (d, white arrowhead) express AO as do other progeny in the lineages. At 77 % of embryogenesis, neither the Z neuroblast (e) nor the Y neuroblast (f) can be detected by DAPI indicating that they have died (dashed circles mark their previous locations). Other cells in both lineages are still undergoing apoptosis (yellow). Scale bar represents 20 μm in (a)–(f)
In order to confirm that the disappearance of the NBs was consistent with programmed cell death, we then examined all ventral NBs (Z, Y, X, and W) in the array, as well as representative dorsal NBs (1-2 and 1-3) over these critical ages, but at greater temporal resolution, using the acknowledged apoptosis-detecting system TUNEL (see “Materials and methods”; Bello et al. 2003 for Drosophila). We additionally controlled that AO and TUNEL label the same apoptotic cells in a central complex lineage of the embryonic (80 %) grasshopper brain (Supplementary Fig. 1).
Confocal images of single NBs Z, Y, X, W, 1-2, and 1-3 at different ages during embryogenesis (65, 68, 70, 75, and 77 %) reveal that the first NBs to be labeled via TUNEL are Z and W at 70 % of embryogenesis (Fig. 7). At this age, DAPI staining reveals condensed chromatin in the form of chromosomes in NB 1-3 indicating the cell is still mitotically active. We have previously shown that DAPI staining and anti-PH3 labeling co-label chromosomes in proliferative NBs of the grasshopper brain (Liu and Boyan 2013). At 75 % of embryogenesis, ventral NBs Z, Y, X, and W are labeled via TUNEL, but not the more dorsal NBs 1-2 and 1-3. At 77 %, the ventral group of NBs comprising Z, Y, and W can no longer be labeled via TUNEL or DAPI, suggesting they have disappeared, while the dorsal group of 1-2 and 1-3, then 1-4 and 1-5 (1-5 data not shown) are now labeled via TUNEL and are therefore undergoing programmed cell death. After 80 % no central complex NBs can be labeled via death markers, whereas cells undergoing apoptosis are still prevalent within their lineages.
Timeline of apoptosis in individual central complex NBs. Confocal images of single NBs Z, Y, X, W, 1-2, and 1-3 at different ages during embryogenesis (65, 68 70, 75, and 77 %) as revealed by double labeling with TUNEL (red) which reveals DNA fragmentation in cells undergoing apoptosis, and the DNA stain DAPI (blue). The first NBs to be labeled via TUNEL (white arrowheads) are Z and W at 70 % of embryogenesis. At this same age, chromosomes (white star) are visible in NB 1-3 indicating it is still mitotically active. At 75 % of embryogenesis, neuroblasts Z, Y, X, and W are labeled via TUNEL but not the more dorsal NBs 1-2 and 1-3. At 77 %, the ventral group of NBs comprising Z, Y, and W can no longer be labeled via TUNEL or DAPI, suggesting they have disappeared, while the dorsal group of 1-2 and 1-3 are now labeled via TUNEL (white arrowheads). Progeny (white stars) of NB 1-2 are labeled via TUNEL indicating they are also undergoing programmed cell death. Scale bar represents 10 μm in all panels
We conclude that the eight central complex NBs exit the cell cycle via programmed cell death rather than a final terminal division.
Patterns of proliferation and apoptosis among central complex NBs during embryogenesis.
Our analyses based on cell appearance, proliferation, and death, reveal consistent patterns in the lifespans of central complex NBs as summarized schematically in Fig. 8. Histology and BrdU incorporation show that NBs appear in a progressive wave beginning with the ventral subset (Z, Y, X, and W) shortly before 25 % of embryogenesis, followed by the more dorsal cells (1-2, 1-3, 1-4, and 1-5), so that the array is complete at 34 % (Fig. 8a–f). The array remains stable in appearance up to 70 % after which ventral NBs (72 %) and then dorsal NBs (>77 %) become progressively more unrecognizable morphologically suggesting they may be degenerating. No central complex NBs are unequivocally recognizable after 80 % of embyrogenesis.
Schematic summarizes the major trends in proliferation and apoptosis among central complex neuroblasts during embryogenesis. Neuroblasts are individually identified by color; dashed open circle indicates morphologically unrecognizable and/or absence of a molecular label. The eight NBs involved are organized in a linear array beginning with Z (most ventral) and ending with 1-5 (most dorsal). According to histological data (a–f), NBs first are recognizable at or shortly before 25 % of embryogenesis (a) and appear in a progressive wave from ventral to dorsal along the array. The array remains stable in appearance up to 70 % of embryogenesis (a–c), after which ventral NBs (Z, Y, X, and W) become progressively unrecognizable first (72–75 %, d) suggesting they may degenerate before the more dorsal ones (1-2, 1-3, 1-4, and 1-5). At 77 % (e), only the most dorsal NBs remain recognizable, but after 80 % (f), none are recognizable. Proliferation markers such as BrdU incorporation, PCNA, PH3 show that all NBs are proliferative from 34 to 70 % (g–i), after which the ventral group cease proliferating between (72–75 %, j), followed by the dorsal NBs (77–80 %, k, l). Proliferation markers fail to label central complex NBs after 80 %. Apoptosis (AO, TUNEL) is first evident in ventral NBs (70 %, o), includes the complete ventral group at 72–75 % (p), and spreads to dorsal NBs at 77 % (q). No death labels are expressed by central complex neuroblasts after 80 % of embryogenesis (r). The data are consistent with cell proliferation and death proceeding as waves from ventral to dorsal in the array with a transition phase occurring between 70 and 72 % of embryogenesis
Proliferation markers such as 5-BrdU incorporation, PCNA, PH3 (Fig. 8g–l) show that all NBs are proliferative from 34 to 70 % (Fig. 8g–i), after which the ventral group cease proliferating between 72 and 75 % (Fig. 8j), followed by the dorsal NBs (77–80 %, Fig. 8 k, l). Proliferation markers fail to label central complex NBs after 80 %.
Apoptosis labels (AO and TUNEL) are first evident in ventral NBs at 70 % (Fig. 8o), include the complete ventral group at 72–75 % (Fig. 8p), and spread to dorsal NBs at 77 % (Fig. 8q). No death labels are expressed by central complex NBs after 80 % of embryogenesis (Fig. 8r).
Our data are consistent with cell proliferation and death proceeding as waves from ventral to dorsal in the array, with a transition from proliferation to cell cycle exit and death occurring between 70 and 72 % of embryogenesis.
Discussion
During insect nervous system development, neural stem cells (NBs) delaminate from the neuroectoderm at stereotypic locations so that maps reveal very similar NB distributions for the ventral nerve cord (Bate 1976; Doe and Goodman 1985a, 1985b; Doe et al. 1985; Broadus and Doe 1995; Broadus et al. 1995) and brain (Malzacher 1968; Zacharias et al. 1993; Younossi-Hartenstein et al. 1996, 1997; Boyan and Williams 1997; Urbach and Technau 2003; Urbach et al. 2003a, 2003b; Williams et al. 2005) in various insect species, and may even include crustacea (Sintoni et al. 2012). Each NB generates a lineage of neural progeny which is nearly invariant and unique (Doe and Skeath 1996; Younossi-Hartenstein et al. 1996, 1997; Pearson and Doe 2003; Doe 2008; Reichert 2011), and in the grasshopper, this stereotypy has allowed lineages to be profiled temporally, biochemically and physiologically (VNC, Goodman and Spitzer 1979; Goodman et al. 1979, 1980; brain, Boyan et al. 2010b; Herbert et al. 2010; Boyan and Niederleitner 2011).
The mechanisms regulating cell identity and fates in early NBs may also be conserved. Molecular markers, such as seven-up, prospero, engrailed, fushi-tarazu/Dax label homologous early-forming NBs in the ventral nerve cord of both the grasshopper and Drosophila (Broadus and Doe 1995). Conserved mechanisms have been shown to regulate the formation of the initial axon scaffold, and subsequent central complex circuitry, in the brain of grasshopper and Drosophila (Reichert and Boyan 1997; Boyan et al. 2010c; Young and Armstrong 2010a, b; Boyan and Reichert 2011; Boyan and Williams 2011; Riebli et al. 2013), and may also regulate the times and manner in which proliferative NBs exit the cell cycle despite the divergent lifestyles of holometabolous and hemimetabolous insects (Reichert 2011).
Proliferation can be terminated by cell cycle exit either through a terminal symmetrical division generating sibling progeny or via programmed cell death, and the molecular mechanisms regulating these different fates in Drosophila have been largely identified (Maurange and Gould 2005; Doe 2008; Maurange et al. 2008; Reichert 2011; Jiang and Reichert 2012, 2013). In holometabolous insects such as Manduca and Drosophila, programmed cell death is prevalent in ventral nerve cord lineages in embryonic (Abrams et al. 1993; White et al. 1994; Karcavich and Doe 2005), postembryonic (Booker and Truman 1987), postmetamorphic (Kimura and Truman 1990) and even adult (Truman 1983) stages, as well as among the postembryonic progeny of central complex NBs (Kumar et al. 2009). Pulses of Hox gene proteins at specific times during postembryonic development have been shown to terminate NB proliferation through programmed cell death in the ventral nerve cord (Abd-A, Bello et al. 2003; Cenci and Gould 2005) and brain (Labial: Kuert et al. 2012). In the grasshopper, the mode of finally exiting the cell cycle has up to now not been clear. NBs of the ventral nerve cord disappear during embryogenesis (Shepherd and Bate 1990)—most probably via programmed cell death, although cell cycle exit through a terminal symmetric division could not be excluded in that study. Whatever the mechanism, in ventral nerve cord ganglia NB fates do not obviously follow a pattern that correlates with cell location across the 30 cell matrix (Shepherd and Bate 1990). In the brain, by contrast, both NB proliferation and death do follow well-defined temporal patterns according to location (Fig. 8). The contingent of NBs which builds the central complex is established early in embryogenesis and remains unchanged until 70–72 % of embryogenesis. Subsequently, these NBs take on different fates according to whether they are located more ventrally or more dorsally within the array. NBs located more ventrally along the linear array (Z, Y, X, and W) cease proliferation earlier (Figs. 3 and 4), and undergo programmed cell death earlier (Figs. 6, 7, and 8) than their more dorsal counterparts (1-2, 1-3, 1-4, and 1-5). Furthermore, these NBs do not undergo cell cycle exit via a final terminal division, they actively undergo programmed cell death (see Reichert 2011 for Drosophila).
Waves of apoptosis also traverse central complex lineages (Boyan et al. 2010a) even, as we now know (Figs. 6 and 7), after the NBs themselves have died, suggesting a cell autonomous regulation of this process. Apoptotic activity in the Z and Y lineages, for example, reaches a maximum just after that for mitotic activity, and is still high when neurogenesis has all but ceased (Boyan et al. 2010a; Boyan and Williams 2011). Apoptosis ceases altogether, however, in such lineages at 90–100 % of embryogenesis and has not been observed postembryonically (Legl 2009). Programmed cell death therefore appears timed to prune excessive cell populations from the these lineages as they approach their maximum size, and so shape them for a postembryonic future. With the death of the NBs, and in the absence of further proliferation (Figs. 3, 4, and 8), we surmise that the complement of neurons generating central complex neuroarchitecture in the grasshopper is complete before hatching.
It is conceivable that the gradients in cell cycle exit and death along the array of central complex NBs result from ventral and dorsal cells being differentially exposed to Hox gene proteins like those described for Drosophila (Bello et al. 2003; Cenci and Gould 2005; Kuert et al. 2012). Different timelines in NB proliferation and death may then translate into a temporal topology according to which the central complex neuroarchitecture is established during embryogenesis. The more ventral NBs (Z, Y, X, and W) commence proliferation earlier, and their progeny include small-field neurons which project axons via the associated z, y, x, and w tracts to the protocerebral bridge, then to the CB where they form part of its characteristic columnar system and from there to the EB where they terminate (Boyan and Williams 1997; Williams et al. 2005; Heinze and Homberg 2008). A subset of these neurons has been shown to form the polarization-sensitive system subserving grasshopper visual navigation (Heinze and Homberg 2007). Pioneer neurons from these same Z, Y, X, and W lineages have been shown to establish this system of tracts at around 33–34 % of embryogenesis (Williams et al. 2005; Williams and Boyan 2008) well before axon pathways from progeny of the 1-2, 1-3, 1-4, and 1-5 system appear and target their central complex modules. We speculate that in forming earlier, the neuroarchitecture generated by the early projections from progeny of ventral Z, Y, X, W and NBs may act as a scaffold onto which later developing subsystems of neurons can direct their processes. Our proposal for the grasshopper mirrors central complex development in Drosophila where early-born neurons from the four DM NBs equivalent to the Z, Y, X, and W system of the grasshopper (see Boyan and Reichert 2011) first generate the fan-shaped body (CB) primordium during larval development and subsequently a layer-specific innervation of the mature structure (Riebli et al. 2013).
Our data further suggest that the dorsal group of NBs displays a greater degree of heterogeneity in timelines than the ventral group (Fig. 8). We speculate that this may relate to the respective central complex modules to which the lineages of these two NB subgroups contribute. Current evidence suggests that whereas the ventral group (Z, Y, X, and W) acts more uniformly in generating lineages for the z, y, x, and w tracts and the columnar system of the CB (Williams et al. 2005), progeny from the dorsal group (1-2, 1-3, 1-4, and 1-5) contribute to a neuroarchitecturally diverse range of modules including the lateral and medial accessory lobes, the ellipsoid body and the noduli (Boyan and Williams 2011). This diversity may necessitate greater differences in cellular organization between the various dorsal lineages than between ventral lineages, thus requiring divergent programs of proliferation and death as reflected in their NB timelines (Fig. 8).
An ordered temporal component to the development of central complex neuroarchitecture in the grasshopper as we report here is consistent with previous studies demonstrating a temporal topology in neuromodulator expression patterns in various central complex modules (Boyan et al. 2010b; Herbert et al. 2010), as well as with the incremental addition of central complex modules over successive instars in Tenebrio (Wegerhoff and Breidbach 1992). Highly organized neuropils elsewhere in the brain also appear to be generated along such temporal lines. An age gradient has been reported among Kenyon cells in the cockroach Periplaneta americana where cells of different ages project axons into different layers of the calyx and pedunculus of the developing mushroom body (Farris and Strausfeld 2001; Farris and Sinakevitch 2003), and age gradients are present in the optic anlagen in Drosophila (Meinertzhagen 1973; Viktorin et al. 2013), where finely tuned waves of cell production, cell differentiation and cell death contribute to the modular organization of the medulla and lobula (Panov 1960; Nordlander and Edwards 1969).
While the complement of neurons of the grasshopper central complex may be complete by the end of embryogenesis, maturation processes at the biochemical level continue into postembryonic stages (Herbert et al. 2010). This suggests that epigenic factors then act on the embryonic set of progeny to adapt the existing central complex neural circuitry to that required for behavior patterns involving adult structures such functional wings and a reproductive apparatus (see Chapman 1982).
References
Abrams JM, White K, Fessler LI, Steller H (1993) Programmed cell death during Drosophila embryogenesis. Development 117:29–43
Adams RR, Maiato H, Earnshaw WC, Carmena M (2001) Essential roles of Drosophila inner centromere protein (INCENP) and Aurora B in histone H3 phosphorylation, metaphase chromosome alignment, kinetochore disjunction, and chromosome segregation. J Cell Biol 153:865–879
Bate CM (1976) Embryogenesis of an insect nervous system I. A map of the thoracic and abdominal neuroblasts in Locusta migratoria. J Embryol Exp Morph 35:107–123
Bello BC, Hirth F, Gould AP (2003) A pulse of the Drosophila Hox protein abdominal-A schedules the end of neural proliferation via neuroblast apoptosis. Neuron 37:209–219
Bentley D, Keshishian H, Shankland M, Toroian-Raymond A (1979) Quantitative staging of embryonic development of the grasshopper, Schistocerca nitens. J Embryol Exp Morph 54:47–74
Booker R, Truman JW (1987) Postembryonic neurogenesis in the CNS of the tobacco hornworm. Manduca sexta. I. neuroblast arrays and the fate of their progeny during metamorphosis. J Comp Neurol 255:548–559
Boyan G, Niederleitner B (2011) Patterns of dye coupling involving serotonergic neurons provide insights into the cellular organization of a central complex lineage of the embryonic grasshopper Schistocerca gregaria. Dev Genes Evol 220:297–313
Boyan GS, Reichert H (2011) Mechanisms for complexity in the brain: generating the insect central complex. Trends Neurosci 34:247–257
Boyan GS, Williams JLD (1997) Embryonic development of the pars intercerebralis/central complex of the grasshopper. Dev Genes Evol 207:317–329
Boyan G, Williams L (2011) Embryonic development of the insect central complex: insights from lineages in the grasshopper and Drosophila. Arthr Struct Devel 40:334–348
Boyan G, Williams L, Meier T (1993) Organization of the commissural fibers in the adult brain of the locust. J Comp Neurol 332:358–377
Boyan GS, Williams JL, Herbert Z (2008) Fascicle switching generates a chiasmal neuroarchitecture in the embryonic central body of the grasshopper Schistocerca gregaria. Arthr Struct Dev 37:539–544
Boyan G, Herbert Z, Williams L (2010a) Cell death shapes embryonic lineages of the central complex in the grasshopper Schistocerca gregaria. J Morphol 271:949–959
Boyan G, Williams JL, Herbert Z (2010b) Multipotent neuroblasts generate a biochemical neuroarchitecture in the central complex of the grasshopper Schistocerca gregaria. Cell Tissue Res 340:13–28
Boyan GS, Williams L, Legl A, Herbert Z (2010c) Proliferative cell types in embryonic lineages of the central complex of the grasshopper Schistocerca gregaria. Cell Tissue Res 341:259–277
Bravo R, Celis JE (1980) A search for differential polypeptide synthesis throughout the cell cycle of Hela cells. J Cell Biol 84:795–802
Bravo R, Macdonald-Bravo H (1987) Existence of two populations of cyclin/proliferating cell nuclear antigen during the cell cycle: association with DNA replication sites. J Cell Biol 105:1549–1554
Broadus J, Doe CQ (1995) Evolution of neuroblast identity: seven-up and prospero expression reveal homologous and divergent neuroblast fates in Drosophila and Schistocerca. Development 121:3989–3996
Broadus J, Skeath JB, Spana EP, Bossing T, Technau G, Doe CQ (1995) New neuroblast markers and the origin of the aCC/pCC neurons in the Drosophila central nervous system. Mech Dev 53:393–402
Cayre M, Strambi C, Strambi A (1994) Neurogenesis in an adult insect brain and its hormonal control. Nature 368:57–59
Cayre M, Strambi C, Charpin P, Augier R, Meyer MR, Edwards JS, Strambi A (1996) Neurogenesis in adult insect mushroom bodies. J Comp Neurol 371:300–310
Cayre M, Scotto-Lomassese S, Malaterre J, Strambi C, Strambi A (2007) Understanding the regulation and function of adult neurogenesis: contribution from an insect model, the house cricket. Chem Senses 32:385–395
Celis JE, Celis A (1985) Cell cycle-dependent variations in the distribution of the nuclear protein cyclin proliferating cell nuclear antigen in cultured cells: subdivision of S phase. Proc Natl Acad Sci U S A 82:3262–3266
Celis JE, Madsen P, Celis A, Nielsen HV, Gesser B (1987) Cyclin (PCNA, auxiliary protein of DNA polymerase delta) is a central component of the pathway(s) leading to DNA replication and cell division. FEBS Lett 220:1–7
Cenci C, Gould AP (2005) Drosophila grainyhead specifies late programmes of neural proliferation by regulating the mitotic activity and Hox-dependent apoptosis of neuroblasts. Development 132:3835–3845
Chapman RF (1982) The insects: structure and function. Hodder and Stoughton, London
Doe CQ (2008) Neural stem cells: balancing self-renewal with differentiation. Development 135:1575–1587
Doe CQ, Goodman CS (1985a) Early events in insect neurogenesis. I. Development and segmental differences in the pattern of neuronal precursor cells. Dev Biol 111:193–205
Doe CQ, Goodman CS (1985b) Early events in insect neurogenesis. II. The role of cell interactions and cell lineage in the determination of neuronal precursor cells. Dev Biol 111:206–219
Doe CQ, Skeath JB (1996) Neurogenesis in the insect central nervous system. Curr Opin Neurobiol 6:18–24
Doe CQ, Kuwada JY, Goodman CS (1985) From epithelium to neuroblasts to neurones: the role of cell interactions and cell lineage during insect neurogenesis. Philos Trans R Soc Lond B 312:67–81
Fairman MP (1990) DNA polymerase delta/PCNA: actions and interactions. J Cell Sci 95:1–4
Farris SM, Sinakevitch I (2003) Development and evolution of the insect mushroom bodies: towards the understanding of conserved developmental mechanisms in a higher brain center. Arthr Struct Dev 32:79–101
Farris SM, Strausfeld NJ (2001) Development of laminar organization in the mushroom bodies of the cockroach: Kenyon cell proliferation, outgrowth, and maturation. J Comp Neurol 439:331–351
Gavrieli Y, Sherman Y, Ben-Sasson SA (1992) Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 119:493–501
Goodman CS, Doe CQ (1994) Embryonic development of the Drosophila central nervous system. In: Bate M, Martinez-Arias A (eds) The development of Drosophila, vol 1. Cold Spring Harbor Press, New York, pp 1131–1206
Goodman CS, Spitzer NC (1979) Embryonic development of identified neurones: differentiation from neuroblast to neurone. Nature 280:208–214
Goodman CS, O'Shea M, Caman R, Spitzer NC (1979) Embryonic development of identified neurons: temporal pattern of morphological and biochemical differentiation. Science 204:1219–1222
Goodman CS, Pearson KG, Spitzer NC (1980) Electrical excitability: a spectrum of properties in the progeny of a single embryonic neuroblast. Proc Natl Acad Sci U S A 77:1676–1680
Hall PA, Levison DA, Woods AL, Yu CC-W, Kellock DB, Watkins JA, Barnes DM, Gillett CE, Camplejohn R, Dover R, Waseem NH, Lane DP (1990) Proliferating cell nuclear antigen (PCNA) immunolocalization in paraffin sections: an index of cell proliferation with evidence of deregulated expression in some neoplasms. J Pathol 162:285–294
Hartenstein V, Nassif C, Lekven A (1998) Embryonic development of the Drosophila brain. II. Pattern of glia cells. J Comp Neurol 402:32–47
Hartenstein V, Spindler S, Pereanu W, Fung S (2008) The development of the Drosophila larval brain. Adv Exp Med Biol 628:1–31
Heinze S, Homberg U (2007) Maplike representation of celestial e-vector orientations in the brain of an insect. Science 315:995–997
Heinze S, Homberg U (2008) Neuroarchitecture of the central complex of the desert locust: intrinsic and columnar neurons. J Comp Neurol 511:454–478
Hendzel MJ, Wei Y, Mancini MA, Hooser AV, Ranalli T, Brinkley BR, Bazett-Jones DP, Allis CD (1997) Mitosis-specific phosphorylation of histone H3 initiates primarily within pericentromeric heterochromatin during G2 and spreads in an ordered fashion coincident with mitotic chromosome condensation. Chromosoma 106:348–360
Herbert Z, Rauser S, Williams L, Kapan N, Güntner M, Walch A, Boyan G (2010) Developmental expression of neuromodulators in the central complex of the grasshopper Schistocerca gregaria. J Morphol 271:1509–1526
Homberg U (1994) Flight-correlated activity changes in neurons of the lateral accessory lobes in the brain of the locust Schistocerca gregaria. J Comp Physiol A 175:597–610
Ito K, Awasaki T (2008) Clonal unit architecture of the adult fly brain. In: Technau GM (ed) Brain development in Drosophila melanogaster. Springer, New York, pp 137–158
Ito K, Hotta Y (1992) Proliferation pattern of postembryonic neuroblasts in the brain of Drosophila melanogaster. Dev Biol 149:134–148
Ito K, Awano W, Suzuki K, Hiromi Y, Yamamoto D (1997) The Drosophila mushroom body is a quadruple structure of clonal units each of which contains a virtually identical set of neurones and glial cells. Development 124:761–771
Izergina N, Balmer J, Bello B, Reichert H (2009) Postembryonic development of transit amplifying neuroblast lineages in the Drosophila brain. Neural Dev 4:44
Jiang Y, Reichert H (2012) Programmed cell death in type II neuroblast lineages is required for central complex development in the Drosophila brain. Neural Dev 7:3
Jiang Y, Reichert H (2013) Analysis of neural stem cell self-renewal and differentiation by transgenic RNAi in Drosophila. Arch Biochem Biophys 534:38–43
Jiang C, Baehrecke EH, Thummel CS (1997) Steroid regulated programmed cell death during Drosophila metamorphosis. Development 124:4673–4683
Karcavich R, Doe CQ (2005) Drosophila neuroblast 7-3 cell lineage: a model system for studying programmed cell death, Notch/Numb signaling, and sequential specification of ganglion mother cell identity. J Comp Neurol 481:240–251
Karlstrom RO, Wilder LP, Bastiani MJ (1993) Lachesin: an immunoglobulin superfamily protein whose expression correlates with neurogenesis in grasshopper embryos. Development 118:509–522
Kimura KI, Truman JW (1990) Postmetamorphic cell death in the nervous and muscular systems of Drosophila melanogaster. J Neurosci 10:403–411
Kuert PA, Bello BC, Reichert H (2012) The labial gene is required to terminate proliferation of identified neuroblasts in postembryonic development of the Drosophila brain. Biology Open 1:1006–1015
Kumar A, Bello B, Reichert H (2009) Lineage-specific programmed cell death in postembryonic brain development of Drosophila. Development 136:3433–3442
Lai SL, Awasaki T, Ito K, Lee T (2008) Clonal analysis of Drosophila antennal lobe neurons: diverse neuronal architectures in the lateral neuroblast lineage. Development 135:2883–2893
Legl A (2009) Dynamik mitotischer und absterbender Zellen in den W-, X-, Y- und Z-Lineages des Zentralkomplexes bei Schistocerca gregaria [dissertation]. Ludwig-Maximilians-Universität München, Munich, Germany, p 108p
Liu Y, Boyan G (2013) Glia associated with central complex lineages in the embryonic brain of the grasshopper Schistocerca gregaria. Dev Genes Evol 223:213–223
Liu G, Seiler H, Wen A, Zars T, Ito K, Wolf M, Heisenberg M, Liu L (2006) Distinct memory traces for two visual features in the Drosophila brain. Nature 439:551–556
Llimargas M, Strigini M, Katidou M, Karagogeos D, Casanova J (2003) Lachesin is a component of a separate junction-based mechanism that controls tube size and epithelial integrity in the Drosophila tracheal system. Development 131:181–190
Malzacher P (1968) Die Embryogenese des Gehirns paurometaboler Insekten. Untersuchungen an Carausius morosus und Periplaneta americana. Zool Morphol Tiere 62:103–161
Mappes M, Homberg U (2004) Behavioural analysis of polarisation vision in tethered flying locusts. J Comp Physiol A 190:61–68
Maurange C, Gould AP (2005) Brainy but not too brainy: starting and stopping neuroblast divisions in Drosophila. Trends Neurosci 28:30–36
Maurange C, Chen L, Gould AP (2008) Temporal transcription factors and their targets schedule the end of neural proliferation in Drosophila. Cell 133:891–902
Meinertzhagen IA (1973) Compound eye and optic lobe development. In: Young D (ed) Developmental neurobiology of arthropods. Cambridge University Press, Cambridge, pp 51–104
Naimski P, Bierzyimageski A, Fikus M (1980) Quantitative fluorescent analysis of different conformational forms of DNA bound to the dye 4′,6-diamidine-2-phenylindole, and separated by gel electrophoresis. Anal Biochem 106:471–475
Neuser K, Triphan T, Mronz M, Poeck B, Strauss R (2008) Analysis of a spatial orientation memory in Drosophila. Nature 453:1244–1247
Ng L, Prelich G, Anderson CW, Stillman B, Fisher PA (1990) Drosophila proliferating cell nuclear antigen. J Biol Chem 266:11948–11954
Nordlander RH, Edwards JS (1969) Postembryonic brain development in the monarch butterfly, Danaus plexippus plexippus, L. I. Cellular events during brain morphogenesis. Wilh Roux'Archiv 162:197–217
Panov AA (1960) The structure of the insect brain during successive stages of postembryonic development. III. Optic lobes. Entomol Rev 39:55–68
Pearson BJ, Doe CQ (2003) Regulation of neuroblast competence in Drosophila. Nature 425:624–628
Pereanu W, Hartenstein V (2006) Neural lineages of the Drosophila brain: a three-dimensional digital atlas of the pattern of lineage location and projection at the late larval stage. J Neurosci 26:5534–5553
Prokop A, Technau GM (1991) The origin of postembryonic neuroblasts in the ventral nerve cord of Drosophila melanogaster. Development 111:79–88
Reichert H (2011) Drosophila neural stem cells: cell cycle control of self-renewal, differentiation, and termination in brain development. In: Kubiak JZ (ed) Cell cycle in development, results and problems in cell differentiation. Springer, Berlin, pp 529–546
Reichert H, Boyan GS (1997) Building a brain: insights from insects. Trends Neurosci 20:258–264
Renn SCN, Armstrong JD, Yang M, Wang Z, An X, Kaiser K, Taghert PH (1999) Genetic analysis of the Drosophila ellipsoid body neuropil: organization and development of the central complex. J Neurobiol 41:189–207
Riebli N, Viktorin G, Reichert H (2013) Early-born neurons in type II neuroblast lineages establish a larval primordium and integrate into adult circuitry during central complex development in Drosophila. Neural Dev 8:6
Shepherd D, Bate CM (1990) Spatial and temporal patterns of neurogenesis in the embryo of the locust Schistocerca gregaria. Development 108:83–96
Shreeram S, Blow JJ (2003) The role of the replication licensing system in cell proliferation and cancer. Prog Cell Cycle Res 5:287–293
Sintoni S, Benton JL, Beltz BS, Hansson BS, Harzsch S (2012) Neurogenesis in the central olfactory pathway of adult decapod crustaceans: development of the neurogenic niche in the brains of procambarid crayfish. Neural Dev 7:1
Spreij THE (1971) Cell death during the development of the imaginal disks of Calliphora erythrocephala. Netherl J Zool 21:221–264
Stacey SM, Muraro NI, Peco E, Labbe A, Thomas GB, Baines RA, van Meyel DJ (2010) Drosophila glial glutamate transporter Eaat1 is regulated by fringe-mediated Notch signaling and is essential for larval locomotion. J Neurobiol 30:14446–14457
Stevenson PA, Kutsch W (1986) Basic circuitry of an adult-specific motor program completed with embryogenesis. Naturwissenschaften 73:741–743
Strausfeld NJ (2012) Arthropod brains. Harvard University Press, Cambridge
Strausfeld NJ, Hirth F (2013) Deep homology of arthropod central complex and vertebrate basal ganglia. Science 340:157–161
Strauss R (2002) The central complex and the genetic dissection of locomotor behaviour. Curr Opin Neurobiol 12:633–638
Strauss R, Heisenberg M (1993) A higher control center of locomotor behavior in the Drosophila brain. J Neurosci 13:1852–1861
Strauss R, Hanesch U, Kinkelin M, Wolf R, Heisenberg M (1992) No-bridge of Drosophila melanogaster: portrait of a structural brain mutant of the central complex. J Neurogenet 8:125–155
Strigini M, Cantera R, Morin X, Bastiani MJ, Bate M, Karagogeos D (2006) The IgLON protein Lachesin is required for the blood–brain barrier in Drosophila. Mol Cell Neurosci 32:91–101
Sumara I, Vorlaufer E, Gieffers C, Peters BH, Peters J-M (2000) Characterization of vertebrate cohesin complexes and their regulation in prophase. J Cell Biol 151:749–761
Takizawa T, Meshorer E (2008) Chromatin and nuclear architecture in the nervous system. Trends Neurosci 31:343–352
Truman JW (1983) Programmed cell death in the nervous system of an adult insect. J Comp Neurol 216:445–452
Truman JW, Bate M (1998) Spatial and temporal patterns of neurogenesis in the central nervous system of Drosophila melanogaster. Dev Biol 125:145–157
Urbach R, Technau GM (2003) Early steps in building the insect brain: neuroblast formation and segmental patterning in the developing brain of different insect species. Arthr Struct Dev 32:103–123
Urbach R, Schnabel R, Technau GM (2003a) The pattern of neuroblast formation, mitotic domains, and proneural gene expression during early brain development in Drosophila. Development 130:3589–3606
Urbach R, Technau GM, Breidbach O (2003b) Spatial and temporal pattern of neuroblasts, proliferation, and Engrailed expression during early brain development in Tenebrio molitor L. (Coleoptera). Arthr Struct Dev 32:125–140
Vanhems E (1985) An in vitro autoradiographic study of gliogenesis in the embryonic locust brain. Dev Brain Res 23:269–275
Vanhems E (1995) Insect glial cells and their relationships with neurons. In: Vernadakis A, Roots B (eds) Neuron-glia interrelationships during phylogeny: plasticity and regeneration. Humana Press Inc, Totowa, NJ, pp 49–77
Viktorin G, Riebli N, Reichert H (2013) A multipotent transit-amplifying neuroblast lineage in the central brain gives rise to optic lobe glial cells in Drosophila. Dev Biol 379:182–194
Wegerhoff R, Breidbach O (1992) Structure and development of the larval central complex in a holometabolous insect, the beetle Tenebrio molitor. Cell Tissue Res 268:341–358
Weinrich A, Kunst M, Wirmer A, Holstein GR, Heinrich R (2008) Suppression of grasshopper sound production by nitric oxide-releasing neurons of the central complex. J Comp Physiol A 194:763–776
Weir PT, Dickinson MH (2012) Flying Drosophila orient to sky polarization. Curr Biol 22:21–27
White K, Grether ME, Abrams JM, Young L, Farrell K, Steller H (1994) Genetic control of programmed cell death in Drosophila. Science 264:677–683
Williams JLD (1975) Anatomical studies of the insect central nervous system: a ground-plan of the midbrain and an introduction to the central complex in the locust, Schistocerca gregaria (Orthoptera). J Zool (Lond) 176:67–86
Williams JLD, Boyan GS (2008) Building the central complex of the grasshopper Schistocerca gregaria: axons pioneering the w, x, y, z tracts project onto the primary commissural fascicle of the brain. Arthr Struct Dev 37:129–140
Williams JLD, Guentner M, Boyan GS (2005) Building the central complex of the grasshopper Schistocerca gregaria: temporal topology organizes the neuroarchitecture of the w, x, y, z tracts. Arthr Struct Dev 34:97–110
Yamaguchi M, Date T, Matsukage A (1991) Distribution of PCNA in Drosophila embryo during nuclear division cycles. J Cell Sci 100:729–733
Yamaguchi M, Nishimoto Y, Hirose F, Matsukage A (1995) Distribution of PCNA during postblastoderm cell division cycles in the Drosophila melanogaster embryo: effect of a string-mutation. Cell Struc Funct 20:47–57
Young JM, Armstrong JD (2010a) Structure of the adult central complex in Drosophila: organization of distinct neuronal subsets. J Comp Neurol 518:1500–1524
Young JM, Armstrong JD (2010b) Building the central complex in Drosophila: the generation and development of distinct subsets. J Comp Neurol 518:1525–1541
Younossi-Hartenstein A, Nassif C, Hartenstein V (1996) Early neurogenesis in the Drosophila brain. J Comp Neurol 370:313–329
Younossi-Hartenstein A, Green P, Liaw GJ, Rudolph K, Lengyel J, Hartenstein V (1997) Control of early neurogenesis of the Drosophila brain by the head gap genes tll, otd, ems and btd. Dev Biol 182:270–283
Zacharias D, Williams JLD, Meier T, Reichert H (1993) Neurogenesis in the insect brain: cellular identification and molecular organization of the brain neuroblasts in the grasshopper embryo. Development 118:941–955
Zudaire E, Simpson SJ, Illa I, Montuenga LM (2004) Dietary influences over proliferating cell nuclear antigen expression in the locust midgut. J Exp Biol 207:2255–2265
Acknowledgments
We thank Dr. J.L.D. Williams for assistance with NB identifications and Karin Fischer for excellent technical assistance. This study is supported by the Graduate School of Systemic Neuroscience, University of Munich (to G.B.).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Siegfried Roth
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Confirmation that acridine orange (AO) and TUNEL label the same apoptotic cells in a central complex lineage of the embryonic (80 %) grasshopper brain. a DAPI staining reveals three putative degenerated cells (white arrowheads) in the Z lineage. AO (b, green) and TUNEL (c, red) label three cells independently demonstrating that these are undergoing programmed cell death. Merging the AO and TUNEL labeling (d, yellow) confirms these are the same cells labeled by DAPI within the Z lineage (a). Scale bar represents 5 μm. (JPEG 12 kb)
Rights and permissions
About this article
Cite this article
Boyan, G., Liu, Y. Timelines in the insect brain: fates of identified neural stem cells generating the central complex in the grasshopper Schistocerca gregaria . Dev Genes Evol 224, 37–51 (2014). https://doi.org/10.1007/s00427-013-0462-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00427-013-0462-8