Abstract
The aim of the present study was to analyze if the left hemisphere preferentially controls flexion responses toward positive stimuli, while the right hemisphere is specialized toward extensor responses to negative pictures. To this end, right-handed subjects had to pull or push a joystick subsequent to seeing a positive or a negative stimulus in their left or right hemifield. Flexion responses were faster for positive stimuli, while negative stimuli were associated with faster extensions responses. Overall, performance was fastest when emotional stimuli were presented to the left visual hemifield. This right hemisphere superiority was especially clear for negative stimuli, while reaction times toward positive pictures showed no hemispheric difference. We did not find any interaction between hemifield and response type. Neither was there a triple interaction between valence, hemifield and response type. We suppose that response dichotomies in humans are not as tightly linked to a hemisphere- and valence-bound reaction type as previously assumed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Emotions are often seen as events that only describe subjective feelings of an individual. Yet, there are various evidences indicating that emotions may be traced back to primordial reaction types such as approach-appetitive, consumption-oriented or withdrawal-avoidant, directed at minimizing unfavorable or maximizing favorable input (Lang et al. 1990). Given this perspective, positive emotions are associated with movements toward a desirable stimulus. A bodily response that is closely associated with approach is the contraction of the arm flexor, which is involved in pulling an object closer to the self. On the contrary, negative emotions likely relate to a movement away from an aversive source and seem to be coupled with the contraction of the arm extensor to push an object away from the self. Several studies have indeed shown that responses using flexor or extensor muscles are associated with a response bias toward pleasant or unpleasant emotional stimuli, respectively (Chen & Bargh, 1999; Coombes, Cauraugh, & Janelle, 2007; Duckworth, Bargh, Garcia, & Chaiken, 2002; Solarz, 1960; Rotteveel & Phaf, 2004; Wentura, Rothermund, & Bak, 2000).
Another frequently investigated issue in neuropsychology and affective neuroscience is whether, and how, emotional processes are functionally lateralized across the hemispheres (Canli, 1999; Demaree, Everhart, Youngstrom, & Harrison, 2005). Two main theories have been proposed. The first one is the “right hemisphere hypothesis”. It posits that emotional processing of both positive and negative emotions is lateralized toward the right hemisphere (Borod, 1992; Borod, Bloom, Brickman, Nakhutina, & Curko, 2002). Numerous studies have supported this hypothesis (Borod, Kent, Koff, Martin, & Alpert, 1988; Christman & Hackworth, 1993; Hugdahl, Iversen, & Johnsen, 1993; McLaren & Bryson, 1987; Moreno, Borod, Welkowitz, & Alpert, 1990; Tamietto, Latini Corazzini, de Gelder, & Geminiani, 2006). The second theory is the “valence hypothesis”, which states that hemispheres differ according to emotional valence with the right brain dominant for the processing of negative emotions and the left brain dominant for the processing of positive emotions (Davidson, 1995; Silberman & Weingartner, 1986). Evidence for this theory has also been reported by several studies (Ahern & Schwartz, 1985; Canli, Desmond, Zhao, Glover, & Gabrieli, 1998; Davidson, Ekman, Saron, Senulis, & Friesen, 1990; Jansari, Tranel, & Adolphs, 2000; Jones & Fox, 1992; Pauli, Wiedemann, & Nickola, 1999; Reuter-Lorenz & Davidson, 1981; Wiedemann et al. 1999).
Some recent studies with brain-damaged patients have supported the right hemisphere hypothesis: patients with lesions to the right hemisphere have greater impairment in the perception of emotional faces, regardless of the valence of the expressed emotion (Adolphs, Damasio, Tranel, Cooper, & Damasio, 2000; Borod et al., 1998; Bowers, Bauer, Coslett, & Heilman, 1985). Nevertheless, other studies have also provided support for the valence hypothesis: unilateral brain damage to the right hemisphere impaired the perception of negative emotions, while the perception of positive ones was preserved (Adolphs, Damasio, Tranel, & Damasio, 1996; Adolphs, Jansari, & Tranel, 2001).
The assumption of strong relations between specific emotions (positive/negative) and specific motor behavior (approach/avoidance), and the concept of specific emotions being connected to specific cerebral hemispheres, leads to the question whether there is a relationship between motor behavior and hemispheres. In a recent study, Maxwell and Davidson (2007) have shown that flexion and extension responses are asymmetrically represented across the cerebral hemispheres, with flexion responses being associated with the left hemisphere (LH) and extension responses with the right hemisphere (RH). In this reaction time experiment, a participant was required to point a finger in response to the direction of a flashed arrow appearing either in the left or the right visual field and pointing away (extension) or toward (flexion) the subject. Facilitation of flexor responses relative to extensor responses was greater in the LH than in the RH, while facilitation of extensor responses relative to flexor responses was greater in the RH than in the LH. This pattern of hemispheric specialization was observed to a greater degree with participants who evidence a higher self-reported level of daily positive affect (according to PANAS; Watson, Clark, & Tellegen, 1988) and a lower self-reported level of dispositional anxiety (according to STAI_X2; Spielberger & Diaz-Guerrero, 1983).
Based on the above-mentioned studies, we aimed to answer the question whether flexion and extension responses toward positive or negative emotional pictures would bias motor processing toward the LH and RH in a similar manner as shown by Maxwell & Davidson (2007). Contrary to Maxwell and Davidson (2007), who induced flexion and extension responses by simple finger responses, we designed an experiment where flexion and extension responses had to be executed via arm movements with a joystick to use a more naturalistic response of approach and avoidance. In addition, we used emotional stimuli to possibly reveal a triple interaction between valence, hemifield and response type. Driven by previous evidence, we predicted faster flexion responses to positive stimuli and faster extensions to negative stimuli. We also expected a hemispheric asymmetry for this mapping between valence and motor behavior, so that faster flexions to positive stimuli would be mediated by the left hemisphere, while the right hemisphere would dominate faster extensions to negative stimuli.
Methods
Participants
Thirty neurologically healthy subjects (15 women, 15 men, aged 19–39 years) participated in this study. The mean age of the women was 25.07 years (SD = 5.35, range: 19–39 years) and that of the men was 27.87 years (SD = 4.16, range: 20–37 years). All participants were right handed, as determined by the Edinburgh Handedness Inventory (Oldfield, 1971). The laterality-index (LQ) provided by this test is calculated by LQ = [(R − L)/(R + L)] × 100, resulting in values between −100 and +100. Positive values indicate a preference for the right hand, while negative values indicate left handedness. Women had a mean LQ of 92.48 (SD = 12.35, range: 63.63–100), while the mean LQ for men was 93.59 (SD = 10.26, range: 68.42–100). All subjects were students recruited from the University of Bochum, Germany. They all gave written consent for participating in the experiment. Each subject in the experiment received a certificate stating the hours they had spent as test subjects (as required for their studies). The study was approved by the ethics committee of the Medical Faculty at the University of Bochum.
Affective stimuli
The emotional pictures were taken from the International Affective Picture System (IAPS; Lang, Bradley, & Cuthbert, 2008). Of these, we chose 40 pleasant (positive) and 40 unpleasant (negative) scenes. Positive pictures included erotic couples and happy families; negative pictures included mutilated bodies and scenes of attack and threat.Footnote 1 The pictures were selected on the basis of their mean normative ratings of valence and arousal (Lang et al., 2008) with positive and negative pictures differing in valence (positive: 7.3, negative: 2.5) but not in arousal (positive: 6.3, negative: 5.6).
Picture presentation
The participants were asked to place their head on a chin rest, at a distance of approximately 57 cm from a monitor, so that 1 cm represents 1° visual angle. The inner edge of the photographs was placed 4° from the central cross. To insure that lateralized stimuli were presented more than 2° visual angle to the left or right of a central fixation cross, we instructed our participants to keep their head and body still and to fixate that cross during the whole experiment. All stimuli were presented in a frame of 11.5 cm in width and 11.5 cm in height. Each picture was presented on a 16-in. computer screen with a frame refresh rate of 75 Hz. The stimuli were presented with Presentation 12.2 software.
Motor task and procedure
The experimental task consisted of two conditions. In one condition positive pictures were responded to by joystick flexion (i.e., self-directed movements) and negative pictures by joystick extension (i.e., movements directed away from the self); in the second condition these relations were inversed, negative pictures were responded to by joystick flexion (i.e., self-directed movements) and positive pictures by joystick extension (i.e., movements directed away from the self). Participants had to respond as quickly and as correctly as possible.
Each participant completed both task conditions with 800 trials each. Within each condition, the responding hand was changed after 400 trials. Task conditions were separated by a 15-min break and started with 40 practice trials. Within each task condition, each picture was presented ten times in a randomized order. The order of task condition and hand use was balanced using a randomized block design.
Each trial began with the presentation of a central fixation cross presented for 1,000 ms. Then the lateralized picture appeared randomly either in the left or the right visual hemifield for 180 ms. The participants were instructed to respond as fast and as accurately as possible. After their response, the subjects had to move back the joystick into start position; this immediately started the next trial.
Data analysis
Reaction time was recorded with Presentation 12.2 software. For statistical analysis of the behavioral data, SPSS Statistics 17 for Windows (SPSS Inc., Chicago, IL) was used. For both task conditions, we measured the median reaction time of all responses for each subject differentiated for the experimental conditions. Median reaction times were analyzed with a mixed 2 × 2 × 2 ANOVA with response type (flexion, extension), valence (positive, negative) and hemifield (RVH, LVH) as within-subjects factors. The assumption of normality for each condition was tested using the Kolmogorov–Smirnov test. The results of these tests showed that the data for all conditions were normally distributed (all p’s > 0.05). Thus, no significant deviation from normality was observed.
Results
Reactions were faster when stimuli were presented to the left (mean = 477.50 ms; SD = 11.06 ms) than to the right hemifield (mean = 482.17 ms; SD = 11.89).
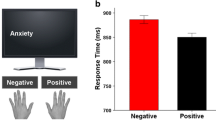
We found a significant interaction of hemifield and valence [F (1, 29) = 9.890, p = 0.004] with participants responding faster to negative stimuli presented in the left visual hemifield (LVH) than in the RVH (paired t test, p = 0.001). No such hemispheric differences in reaction times were observed for positive stimuli (paired t test, p = 0.936). For neither the RVH (paired t test, p = 0.074) nor for the LVH (paired t test, p = 0.709) did response latencies to negative and positive stimuli differ (see Fig. 1).
As shown by a significant interaction of valence and response type, [F (1, 29) = 6.335, p = 0.018] (see Fig. 2) responses to positive stimuli were faster in the flexion condition than in the extension condition (paired t test, p = 0.006), whereas responses to negative stimuli were faster in the extension condition than in the flexion condition (paired t test, p = 0.001).
There was no significant interaction of response type and hemifield (F < 1) and no triple interaction of response type, hemifield and valence (F < 1).
Discussion
We investigated if motor response patterns, i.e., arm flexion versus extension, were associated with a lateralized organization of emotions. Overall, reactions were faster for emotional stimuli presented in the left visual hemifield. Corroborating previous studies (e.g., Chen & Bargh, 1999; Duckworth et al., 2002), we also observed that positive and negative stimuli were associated with faster flexion and extension responses, respectively. This effect was further modulated by the valence, with only negative photographs presented in the LVH eliciting faster responses. No triple interaction between valence, hemifield and response type was found. We will discuss these points, one by one.
First, our results in line with previous studies (Chen & Bargh, 1999; Coombes et al., 2007; Duckworth et al., 2002; Solarz, 1960; Rotteveel & Phaf, 2004) showed that flexion and extension responses are facilitated positive and negative stimuli, respectively. The application of various stimulus types has hitherto shown this association between the direction of a motor response and a bidirectional stimulus valence. This has been revealed for valenced words (Chen & Bargh, 1999), abstract images (Duckworth et al., 2002), facial expressions (Marsh, Kleck, & Ambady, 2005), object pictures (Lavender & Hommel, 2007) and phobic objects (Rinck & Becker, 2007). The vast majority of these experiments utilized arm movements, although also finger movements (Maxwell & Davidson, 2007) and button presses (Wentura et al., 2000) were employed. Thus, our data meet with plenty of support from the literature, making it likely that the approach to a desired target and the avoidance of agonistic stimuli are evolutionarily coded by prefixed movement patterns.
Second, our results showed that reaction times for emotional stimuli presented in the LVH were overall faster than for stimuli presented in the right hemifield. This result is consistent with the right hemisphere hypothesis, which posits that this brain half is characterized by an overall superiority for emotional cues, independent of their valence (Alves, Aznar-Casanova, & Fukusima, 2009; Christman & Hackworth, 1993; Dimberg & Petterson, 2000; Hugdahl et al., 1993; McLaren & Bryson, 1987; Moreno et al., 1990; Narumoto, Okada, Sadato, Fukui, & Yonekura, 2001; Rodway & Schepman, 2007; Sato, Kochiyama, Yoshikawa, Naito, & Matsumura, 2004; Tamietto et al., 2006). However, if these results are differentiated according to valence and visual hemifield, the overall pattern changes. Now responses to negative emotional cues were faster in the LVH, with no difference to positive cues between hemifields. Thus, our results support a mixed pattern of valence and right hemisphere hypotheses. This resembles a large number of studies that show a right hemispheric specialization for negative affect and a bihemispheric processing of positive valence issues (Adolphs et al., 2001; Asthana & Mandal, 2001; Kensinger & Choi, 2009; Killgore & Yurgelun-Todd, 2007; Kimura, Yoshino, Takahashi, & Nomura, 2004; Mandal, Tandon, & Asthana, 1991; Natale, Gur, & Gur, 1983; Noesselt, Driver, Heinze, & Dolan, 2005; Sato & Aoki, 2006; Simon-Thomas, Role, & Knight, 2005; Smith & Bulman-Fleming, 2004). Thus, neither do we seem to have a simple dominance pattern for an overall processing of emotions, nor are negative and positive affects simply mapped in a complementary way onto the two hemispheres.
Third, although we revealed that fast flexion responses were related to positive and fast extensions to negative emotional stimuli, this mapping was independent of hemispheres. To some extent, this contradicts a couple of previous studies. Maxwell and Davidson (2007) revealed a differential lateralization of finger flexion and extension actions, which were associated with left hemispheric flexion and right hemispheric extension responses, respectively. Cretenet and Dru (2004) and Dru and Cretenet (2005) showed that right-arm flexion together with left-arm extension shifted evaluations toward positive values, while the opposite arm movements resulted in negative judgments. However, our study supports the previous report of Sobotka, Davidson and Senulis (1992), who also could not reveal a triple interaction between valence, movement direction and hemisphere in the alpha power of the frontal EEG.
It is always difficult to interpret nonsignificant findings, and we are therefore cautious with our following statements on the absence of a triple interaction. While doing so, we would like to emphasize that our approach differs to some extent from previous ones. First, different from some of the cited studies (Sobotka et al., 1992; Maxwell & Davidson, 2007), we included an overt emotional manipulation and additionally required our subjects to perform full arm and not mere finger movements. Second, different from Cretenet and Dru (2004) and Dru and Cretenet (2005), we did not analyze alterations in stimulus judgments, but reaction times after emotional stimulus presentations. Thus, we devised our experiment as being relatively close to a possible evolutionary scenario in which appetitive or aversive stimuli are to be responded to by means of movements that either drag them toward the subject or push them away. The fact that our triple interaction did not even approach a level of significance could imply that response dichotomies in humans are not as tightly linked to one hemisphere- and valence-bound reaction type as previously assumed.
Notes
The numbers of IAPS pictures used were as follows. Positive: 2040, 2050, 2057, 2058, 2070, 2071, 2080, 2260, 2660, 1710, 1440, 1441, 2150, 1920, 2091, 2332, 2311, 2345, 2344, 2153, 4607, 4608, 4643, 4652, 4656, 4658, 4659, 4660, 4680, 4670, 4676, 4677, 4681, 4683, 4687, 4689, 4694, 4695, 4800 and 4810; negative: 6555, 6200, 6244, 6213, 6250, 6550, 6300, 6570.1, 6510, 6230, 6212, 6540, 2811, 6560, 6313, 6350, 1930, 1300, 1301, 2120, 3000, 3010, 9435, 6021, 6022, 3016, 3062, 9433, 2800, 3069, 3005.1, 3015, 3063, 1113, 1120, 1201, 1052, 1050, 1111 and 2095.
References
Adolphs, R., Damasio, H., Tranel, D., Cooper, G., & Damasio, A. R. (2000). A role for somatosensory cortices in the visual recognition of emotion as revealed by three-dimensional lesion mapping. The Journal of Neuroscience, 20, 2683–2690.
Adolphs, R., Damasio, H., Tranel, D., & Damasio, A. R. (1996). Cortical systems for the recognition of emotion in facial expressions. The Journal of Neuroscience, 16, 7678–7687.
Adolphs, R., Jansari, A., & Tranel, D. (2001). Hemispheric perception of emotional valence from facial expressions. Neuropsychology, 15, 516–524.
Ahern, G. L., & Schwartz, G. E. (1985). Differential lateralization for positive and negative emotion in the human brain: EEG spectral analysis. Neuropsychologia, 23, 745–755.
Alves, N. T., Aznar-Casanova, J. A., & Fukusima, S. S. (2009). Patterns of brain asymmetry in the perception of positive and negative facial expressions. Laterity, 14, 256–272.
Asthana, H. S., & Mandal, M. K. (2001). Visual-field bias in the judgment of facial expression of emotion. The Journal of general psychology, 128, 21–29.
Borod, J. C. (1992). Interhemispheric and intrahemispheric control of emotion: a focus on unilateral brain damage. Journal of Consulting and Clinical Psychology, 60, 339–348.
Borod, J. C., Bloom, R. L., Brickman, A. M., Nakhutina, L., & Curko, E. A. (2002). Emotional processing deficits in individuals with unilateral brain damage. Applied Neuropsychology, 9, 23–36.
Borod, J. C., Cicero, B. A., Obler, L. K., Welkowitz, J., Erhan, H. M., Santschi, C., et al. (1998). Right hemisphere emotional perception: evidence across multiple channels. Neuropsychology, 12, 446–458.
Borod, J. C., Kent, J., Koff, E., Martin, C., & Alpert, M. (1988). Facial asymmetry while posing positive and negative emotions: support for the right hemisphere hypothesis. Neuropsychologia, 26, 759–764.
Bowers, D., Bauer, R. M., Coslett, H. B., & Heilman, K. M. (1985). Processing of face by patients with unilateral hemisphere lesions: dissociations between judgements of facial affect and facial identity. Brain and Cognition, 4, 258–272.
Canli, T. (1999). Hemispheric asymmetry in the experience of emotion: a perspective from functional imaging. The Neuroscientist, 5, 201–207.
Canli, T., Desmond, J. E., Zhao, Z., Glover, G., & Gabrieli, J. D. E. (1998). Hemispheric asymmetry for emotional stimuli detected with fMRI. Neuroreport, 9, 3233–3239.
Chen, M., & Bargh, J. A. (1999). Consequences of automatic evaluation: Immediate behavioral predispositions to approach or avoid the stimulus. Personality and Social Psychology Bulletin, 25, 215–224.
Christman, S. D., & Hackworth, M. D. (1993). Equivalent perceptual asymmetries for free viewing of positive and negative emotional expressions in chimeric faces. Neuropsychologia, 31, 621–624.
Coombes, S. A., Cauraugh, J. H., & Janelle, C. M. (2007). Emotional state and initiating cue alter central and peripheral motor processes. Emotion, 7, 275–284.
Cretenet, J., & Dru, V. (2004). The influence of unilateral and bilateral arm flexion versus extension on judgments: an exploratory case of motor congruence. Emotion, 4, 282–294.
Davidson, R. J. (1995). Cerebral asymmetry, emotion, and affective style. In R. J. Davidson & K. Hughdahl (Eds.), Brain Asymmetry (pp. 361–387). Massachusetts: MIT Press.
Davidson, R. J., Ekman, P., Saron, C. D., Senulis, J. A., & Friesen, W. V. (1990). Approach-withdrawal and cerebral asymmetry: emotional expression and brain physiology. Journal of Personality and Social Psychology, 58, 330–341.
Demaree, H. A., Everhart, D. E., Youngstrom, E. A., & Harrison, D. W. (2005). Brain lateralization of emotional processing: historical roots and a future incorporating “dominance”. Behavioral and Cognitive Neuroscience Reviews, 4, 3–20.
Dimberg, U., & Petterson, M. (2000). Facial reactions to happy and angry facial expressions: evidence for right hemisphere dominance. Psychophysiology, 37, 693–696.
Dru, V., & Cretenet, J. (2005). Influence of bilateral motor action on judgments: additional evidence for the motor congruence hypothesis. Laterality, 10, 389–398.
Duckworth, K. L., Bargh, J. A., Garcia, M., & Chaiken, S. (2002). The automatic evaluation of novel stimuli. Psychological Science, 13, 513–519.
Hugdahl, K., Iversen, P. M., & Johnsen, B. H. (1993). Laterality for facial expressions: does the sex of the subject interact with the sex of the stimulus face? Cortex, 29, 325–331.
Jansari, A., Tranel, D., & Adolphs, R. (2000). A valence-specific lateral bias for discriminating emotional facial expressions in free field. Cognition and Emotion, 14, 341–353.
Jones, N. A., & Fox, N. (1992). Electroencephalogram asymmetry during emotionally evocative films and its relation to positive and negative affectivity. Brain and Cognition, 20, 280–299.
Kensinger, E. A., & Choi, E. S. (2009). When side matters: hemispheric processing and the visual specificity of emotional memories. Journal of Experimental Psychology. Learning, Memory, and Cognition, 35, 247–253.
Killgore, W. D., & Yurgelun-Todd, D. A. (2007). The right-hemisphere and valence hypotheses: could they both be right (and sometimes left)? Social Cognitive and Affective Neuroscience, 2, 240–250.
Kimura, Y., Yoshino, A., Takahashi, Y., & Nomura, S. (2004). Interhemispheric difference in emotional response without awareness. Psychology & Behavior, 82, 727–731.
Lang, P. J., Bradley, M. M., & Cuthbert, B. N. (1990). Emotion, attention, and the startle reflex. Psychological Review, 97, 377–395.
Lang, P. J., Bradley, M. M., & Cuthbert, B. N. (2008). International affective picture system (IAPS): affective ratings of pictures and instruction manual. Technical Report A-8. Gainesville: University of Florida.
Lavender, T., & Hommel, B. (2007). Affect and action: toward an event-coding account. Emotion and Cognition, 21, 1270–1296.
Mandal, M. K., Tandon, S. C., & Asthana, H. S. (1991). Right brain damage impairs recognition of negative emotions. Cortex, 27, 247–253.
Marsh, A. A., Kleck, R. E., & Ambady, N. (2005). The effects of fear and anger facial expressions on approach- and avoidance-related behaviors. Emotion, 5, 119–124.
Maxwell, J. S., & Davidson, R. J. (2007). Emotion as motion: asymmetries in approach and avoidant actions. Psychological Science, 18, 1113–1119.
McLaren, J., & Bryson, S. E. (1987). Hemispheric asymmetries in the perception of emotional and neutral faces. Cortex, 23, 645–654.
Moreno, C. R., Borod, J. C., Welkowitz, J., & Alpert, M. (1990). Lateralization for the expression and perception of facial emotion as a function of age. Neuropsychologia, 28, 199–209.
Narumoto, J., Okada, T., Sadato, N., Fukui, K., & Yonekura, Y. (2001). Attention to emotion modulates fMRI activity in human right superior temporal sulcus. Cognitive Brain Research, 12, 225–241.
Natale, M., Gur, R. E., & Gur, R. C. (1983). Hemispheric asymmetries in processing emotional expressions. Neuropsychologia, 21, 555–565.
Noesselt, T., Driver, J., Heinze, H. J., & Dolan, R. (2005). Asymmetrical activation in the human brain during processing of fearful faces. Current Biology, 15, 424–429.
Oldfield, R. C. (1971). The assessment and the analysis of handedness: the Edinburgh inventory. Neuropsychologia, 9, 97–113.
Pauli, P., Wiedemann, G., & Nickola, M. (1999). Pain sensitivity, cerebral laterality, and negative affect. Pain, 80, 359–364.
Reuter-Lorenz, P., & Davidson, R. J. (1981). Differential contributions of the two cerebral hemispheres to the perception of happy and sad faces. Neuropsychologia, 19, 609–613.
Rinck, M., & Becker, E. S. (2007). Approach and avoidance in fear of spiders. Journal of Behavior Therapy and Experimental Psychiatry, 38, 105–120.
Rodway, P., & Schepman, A. (2007). Valence specific laterality effects in prosody: expectancy account and the effects of morphed prosody and stimulus lead. Brain and Cognition, 63, 31–41.
Rotteveel, M., & Phaf, R. H. (2004). Automatic affective evaluation does not automatically predispose for arm flexion and extension. Emotion, 4, 156–172.
Sato, W., & Aoki, S. (2006). Right hemispheric dominance in processing of unconscious negative emotion. Brain and Cognition, 62, 261–266.
Sato, W., Kochiyama, T., Yoshikawa, S., Naito, E., & Matsumura, M. (2004). Enhanced neural activity in response to dynamic facial expressions of emotion: an fMRI study. Cognitive Brain Research, 20, 81–91.
Silberman, E. K., & Weingartner, H. (1986). Hemispheric lateralization of functions related to emotion. Brain and Cognition, 5, 322–353.
Simon-Thomas, E. R., Role, K. O., & Knight, R. T. (2005). Behavioral and electrophysiological evidence of a right hemisphere bias for the influence of negative emotion on higher cognition. Journal of Cognitive Neuroscience, 17, 518–529.
Smith, S. D., & Bulman-Fleming, M. B. (2004). A hemispheric asymmetry for the unconscious perception of emotion. Brain and Cognition, 55, 452–457.
Sobotka, S. S., Davidson, R. J., & Senulis, J. A. (1992). Anterior brain electrical asymmetries in response to reward and punishment. Electroencephalography and Clinical Neurophysiology, 83, 236–247.
Solarz, A. K. (1960). Latency of instrumental responses as a function of compatibility with the meaning of eliciting verbal signs. Journal of Experimental Psychology, 59, 239–245.
Spielberger, C. D., & Diaz-Guerrero, R. (1983). Cross-cultural anxiety. Series in Clinical and Community Psychology: Stress and Anxiety, 2, 218.
Tamietto, M., Latini Corazzini, L., de Gelder, B., & Geminiani, G. (2006). Functional asymmetry and interhemispheric cooperation in the perception of emotions from facial expressions. Experimental Brain Research, 171, 389–404.
Watson, D., Clark, L. A., & Tellegen, A. (1988). Development and validation of brief measures of positive and negative affect: the PANAS scales. Journal of Personality and Social Psychology, 54, 1063–1070.
Wentura, D., Rothermund, K., & Bak, P. (2000). Automatic vigilance: the attention-grabbing power of approach- and avoidance-related social information. Journal of Personality and Social Psychology, 78, 1024–1037.
Wiedemann, G., Pauli, P., Dengler, W., Lutzenberger, W., Birbaumer, N., & Buchkremer, G. (1999). Frontal brain asymmetry as a biological substrate of emotions in patients with panic disorders. Archives of General Psychiatry, 56, 78–84.
Acknowledgments
C. Ö. -H. was supported by the Deutsche Forschungsgemeinschaft (DFG) through its Research Training Group “Emotions” (GRK 1253/1) and by the Deutscher Akademischer Austausch Dienst -STIBET. O. G. was supported by the DFG (Gu 227/11-1). P. P. is supported by the DFG through the Research Group “Emotion and Behaviour (FOR 605)”. We gratefully acknowledge the support and the comments provided by Prof. Dr. Joseph Classen.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Önal-Hartmann, C., Pauli, P., Ocklenburg, S. et al. The motor side of emotions: investigating the relationship between hemispheres, motor reactions and emotional stimuli. Psychological Research 76, 311–316 (2012). https://doi.org/10.1007/s00426-011-0337-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00426-011-0337-4