Abstract
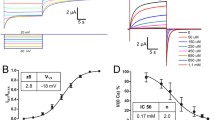
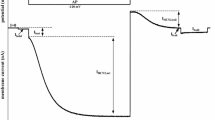
Hemichannels of rat connexin 46 (rCx46) were expressed in Xenopus laevis oocytes and analysed by two-electrode voltage-clamp experiments. It is established that rCx46 hemichannels can be activated at low external Ca2+ and positive membrane potentials. Upon larger depolarizations, the hemichannels of oocytes activate in a time-dependent manner, occasionally followed by a spontaneous inactivation. We found that, in the absence of inactivation, treatment of oocytes with 1-oleoyl-2-acetyl-sn-glycerol (OAG), an activator of protein kinase C (PKC), reversibly reduced the amplitude of the rCx46-mediated current and, after an incubation time of about 30 min, induced inactivation of the voltage-dependent current. After wash-out of OAG the corresponding membrane conductance increased and the inactivation behaviour disappeared. The OAG-induced inactivation, as well as the spontaneous inactivation, could be removed by application of the specific PKC inhibitor calphostin C and also by phloretin. The data provide evidence that the activation and inhibition of PKC affect the rCx46-mediated membrane conductance as well as the voltage-dependent current inactivation in an inverse manner.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 13 March 1998 / Received after revision and accepted: 18 June 1998
Rights and permissions
About this article
Cite this article
Ngezahayo, A., Zeilinger, C., Todt, I. et al. Inactivation of expressed and conducting rCx46 hemichannels by phosphorylation. Pflügers Arch 436, 627–629 (1998). https://doi.org/10.1007/s004240050681
Issue Date:
DOI: https://doi.org/10.1007/s004240050681