Abstract
The SLC34 family of sodium-driven phosphate cotransporters comprises three members: NaPi-IIa (SLC34A1), NaPi-IIb (SLC34A2), and NaPi-IIc (SLC34A3). These transporters mediate the translocation of divalent inorganic phosphate (HPO4 2−) together with two (NaPi-IIc) or three sodium ions (NaPi-IIa and NaPi-IIb), respectively. Consequently, phosphate transport by NaPi-IIa and NaPi-IIb is electrogenic. NaPi-IIa and NaPi-IIc are predominantly expressed in the brush border membrane of the proximal tubule, whereas NaPi-IIb is found in many more organs including the small intestine, lung, liver, and testis. The abundance and activity of these transporters are mostly regulated by changes in their expression at the cell surface and are determined by interactions with proteins involved in scaffolding, trafficking, or intracellular signaling. All three transporters are highly regulated by factors including dietary phosphate status, hormones like parathyroid hormone, 1,25-OH2 vitamin D3 or FGF23, electrolyte, and acid–base status. The physiological relevance of the three members of the SLC34 family is underlined by rare Mendelian disorders causing phosphaturia, hypophosphatemia, or ectopic organ calcifications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Phosphate homeostasis
Essential role of phosphate
Phosphate is the third most abundant anion in the body and required for a variety of fundamental biologic processes. Inorganic phosphate is essential for bioenergetics (ATP, GTP), metabolic regulation (e.g., in glycolysis or oxidative phosphorylation), intracellular signaling pathways, cell proliferation (as part of the DNA and RNA), and for structures such as bones and membranes [25, 26]. Phosphate contributes also to acid–base balance as buffer in blood and in urine. Approximately 85 % of total body phosphate is accumulated in bone and teeth, about 14 % is in soft tissues such as skeletal muscle and erythrocytes, and only 1 % circulates as free phosphate in extracellular fluids. The concentration of intracellular inorganic phosphate may be in the range of 0.7–2.5 mM as determined by 31P-NMR and chemical analyses [51]. Extracellular concentrations of inorganic phosphate vary between 0.8 and 1.2 mM in humans. In plasma, phosphate exists in both the monovalent and the divalent form. Based on the pK value of 6.8, at blood pH of 7.4, 72 % of plasma phosphate is present in the divalent (HPO4 2−) and 28 % is present in the monovalent (H2PO4 −) form.
The extracellular concentration of phosphate depends to a large extent on mechanisms that control renal excretion of phosphate. Renal handling of phosphate, and to a lesser extent gastrointestinal absorption of phosphate, is controlled by complex regulatory networks that involve several organs and several endocrine factors [20, 38, 88, 96, 98].
Deviations from normal serum phosphate concentrations cause severe clinical disorders. Even slight chronic elevations have been associated with increased rates of death due to cardiovascular complications that are common among patients with chronic kidney disease [32, 115, 131, 132]. On the other hand, prolonged hypophosphatemia, caused by, e.g., malabsorption, renal phosphate losses, or inherited disorders such as X-linked hypophosphatemia [18], results in symptoms such as osteomalacia, hypercalciuria, and bone demineralization [29].
Intestinal phosphate absorption
Organic phosphate ingested in foods is hydrolyzed in the gastrointestinal tract releasing inorganic phosphate. Phosphate absorption along the gastrointestinal tract is mediated by two pathways, a transcellular absorptive component, which involves SLC34 and SLC20 sodium-dependent phosphate cotransporters [54, 96, 118], and a concentration- or load-dependent absorptive component that may permeate the paracellular route, which, however, is poorly characterized. Together, these two processes results in an overall fractional absorption of phosphate that ranges between 65 and 70 % of the amount ingested. Notably, segmental distribution of absorption of phosphate varies among different species. In humans and rats, most of the ingested phosphate is absorbed in duodenum and jejunum, whereas in mice, most of the phosphate absorption occurs in the ileum [96, 97, 114]. The role of the colon is uncertain.
Intestinal absorption of phosphate is regulated and adapts to dietary phosphate intake as well as to acid–base status and various hormones such as 1,25-OH2 vitamin D3 or glucocorticoids (see below, regulation of NaPi-IIb).
Renal phosphate handling
Phosphate is almost freely filtered by the glomerulus and subsequently reabsorbed along the nephron. The extent of reabsorption depends on dietary intake and a variety of other factors (see below). For the average diet, fractional excretion of phosphate in adults is in the range of 10–30 % of the filtered load. There is no evidence for secretion of phosphate. The main site of phosphate reabsorption is the proximal tubule, whether and to which extent more distal segments contribute to phosphate reabsorption has not been fully clarified [21, 71, 72]. In rodents, the early proximal segments have higher phosphate transport rates and juxtamedullary nephrons are more active than superficial nephrons [21, 103, 104].
Renal phosphate handling is highly regulated by many factors including dietary phosphate intake, acid–base homeostasis, electrolyte status (e.g., hypokalemia), and a variety of hormones. The renal excretion of phosphate is increased by high dietary intake of phosphate, during acidosis, hypokalemia, by parathyroid hormone (PTH), 1,25-OH2-vitamin D3, fibroblast growth factor 23 (FGF23), dopamine, or glucocorticoids. In contrast, increased renal phosphate reabsorption occurs due to low dietary phosphate intake, during alkalosis, or stimulated by insulin, insulin-like growth factor 1(IGF1), 1,25-OH2-vitamind D3, and thyroid hormone [17, 21, 27, 103, 104].
The SLC34 family of sodium-dependent phosphate transporters
NaPi-IIa (SLC34A1)
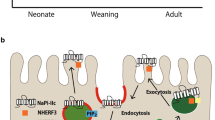
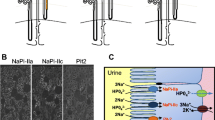
The human NaPi-IIa isoform comprises 639 amino acids. Rat and human NaPi-II cDNAs were first isolated by expression cloning [93]. NaPi-IIa is predominantly expressed in kidney and localizes to the apical brush border membrane of the proximal tubule (Fig. 1) [34]. Expression is higher in early convoluted proximal tubules (S1 segments) and in juxtamedullary nephrons but spreads to the late proximal tubule (S2/S3 segments) and to superficial nephrons during phosphate depletion [89]. NaPi-IIa mRNA has also been detected in bone and brain, but protein expression has not been confirmed.
Localization of renal phosphate cotransporters in rodent kidney. In kidney, at least three Na+-dependent phosphate cotransporters are involved in phosphate reabsorption: the two SLC34 family members NaPi-IIa and NaPi-IIc and the SLC20 family member Pit-2. The upper panels show localization of the three transporters in the murine kidney cortex of animals kept on a low phosphate diet. The nephron models depict localization of the three transporters under conditions of normal–high phosphate intake (red shaded parts) which is restricted to the initial S1 segment of proximal tubules of juxtamedullary nephrons. During phosphate depletion, expression of all three transporters spreads to proximal tubules of superficial nephrons and NaPi-IIa expression extends also to S2 and S3 segments of the proximal tubule (yellow shaded nephron segments)
NaPi-IIb (SLC34A2)
NaPi-IIb is a protein of 689 amino acids in humans, and its mRNA has been detected in many organs including lungs, testis, salivary gland, thyroid gland, small intestine, liver, mammary gland, and uterus [68]. In small intestine, NaPi-IIb is expressed at the luminal brush border membrane, in rats and humans in the duodenum and jejunum, whereas in mice mostly in ileum [68, 105, 114].
NaPi-IIc (SLC34A3)
Human NaPi-IIc comprises 599 amino acids and like NaPi-IIa is mostly expressed in kidney. In kidney, NaPi-IIc is found in the brush border membrane of the early proximal tubule (S1 segment) of juxtamedullary nephrons and is absent from other portions of the proximal nephron (Fig. 1). During phosphate depletion, NaPi-IIc protein is also detected in the early proximal tubule of superficial nephrons [24, 112, 121]. NaPi-IIc may also be expressed in bone, but its function there has not been elucidated [125].
Structural and functional characteristics of SLC34 transporters
Transport mechanism and kinetic characteristics of SLC34 proteins
The three SLC34 isoforms (NaPi-IIa,b,c) are selective for divalent Pi (HPO4 2−) [8, 49] and use the inwardly directed Na+ electrochemical gradient to catalyze uphill movement of Pi. Although Na+ and Pi are the preferred substrates, cation replacement studies have established that Li+ can partly substitute for Na+ to drive Pi transport, albeit at a significantly lower rate [4]. Transport by NaPi-IIb of the phosphate mimetic arsenate has been demonstrated [138], and given its intestinal localization, this may have relevance for providing a potential route for arsenate accumulation, for example in aquatic species [16]. Transport capacity of SLC34 proteins is also strongly dependent on external pH. As pH defines the monovalent/divalent Pi distribution in the extracellular compartment, it may, for example, influence transport rates for NaPi-IIa and NaPi-IIc along the proximal tubule. Protons can also directly modulate the transport kinetics by competing with Na+ binding and by modifying the apparent rate constants associated with specific kinetic steps in the transport cycle [45].
The SLC34 isoforms display similar apparent substrate affinity constants of ≤100 μM and ∼40 mM for Pi and Na+, respectively, at pH 7.4. The transporters probably function close to their maximum rates at least in the initial segment of the renal proximal tubule, given that these apparent affinities are generally well below the typical concentrations found under normal physiological conditions in the intestine and renal proximal tubule. The apparent affinity for Pi is somewhat larger for the mammalian NaPi-IIb compared with the other isoforms (∼10 μM), and this may partially compensate for the reduced availability of divalent Pi in a low-pH environment [47, 50].
The salient functional feature that distinguishes the three isoforms is whether or not net charge movement accompanies cotransport. NaPi-IIa and NaPi-IIb are electrogenic and translocate one net positive charge per transport cycle. They mediate transport with a 3:1 Na+–Pi stoichiometry [49]. Importantly, their transport rates are a strong function of membrane potential. In contrast, NaPi-IIc is electroneutral with no net charge translocation and its transport kinetics are insensitive to membrane potential. It mediates transport with a 2:1 stoichiometry [8, 122]. The difference in stoichiometries between these isoforms means that the theoretical Pi concentrating capacity is approximately 100-fold higher for NaPi-IIa/b compared with NaPi-IIc, at the cost of a 10-fold greater inward flux of Na+ ions, together with net charge movement, both of which must be compensated by the cell through the action of the Na+/K+-ATPase.
The transport process for the electrogenic isoforms can be described as a sequence of partial reactions between states that correspond to unique conformations of the protein (Fig. 2). This model is consistent with an alternating access transport mechanism, whereby substrate binding can occur from either the extra- or intra-cellular medium, but not from both simultaneously. The development of this model for the electrogenic NaPi-IIa,b and its extension to include NaPi-IIc has relied on several experimental approaches based on applying the voltage clamp technique to single Xenopus oocytes that heterologously express the specific isoform. This technique allows control of a key driving force for transport, membrane potential. Moreover, assays in which both substrate flux (Na+, Pi) and net charge translocation are measured simultaneously on the same oocyte have allowed a definitive determination of stoichiometry and specification of the preferred Pi species to be made [49]. Importantly, these assays also confirm the validity of using Pi-induced membrane current as a measure of transport activity. Using steady-state and presteady-state kinetic analysis, electrogenic partial reactions were identified and apparent substrate affinities, turnover rate, pH dependence, and the substrate binding order were determined (for review, see [47]).
Transport mechanism of SLC34 proteins. For all SLC34 proteins, transmembrane transport for an inwardly directed Na+ gradient comprises an ordered sequence of binding steps in which two Na+ ions in the extracellular medium bind sequentially, followed by divalent Pi and a third Na+ ion. Cartoons indicate two conformations of the protein. Substrate release to the cytosol occurs after reorientation of the fully loaded carrier. For electrogenic NaPi-IIa and NaPi-IIb (top), an intrinsic negative charge (hypothesized to be a conserved aspartic acid located between the third and fourth predicted transmembrane spanning regions (Fig. 3) and which senses the transmembrane field) confers voltage dependence to transport cycle. This allows binding of the first Na+ ion (1) within the transmembrane electric field and subsequent translocation together with the other substrates (2, 3, 4), giving a 3:1 Na+–Pi stoichiometry. When substrates are released to the cytosol, the intrinsic charge senses the transmembrane field, which leads to a voltage-dependent reorientation of the empty carrier, ready for the next transport cycle. The electroneutral NaPi-IIc (bottom), which conserved glycine instead of aspartic acid at the critical site, has one Na+ ion bound (1) (like NaPi-IIa/b), but cannot be translocated [53], thus resulting in a 2:1 Na+–Pi transport stoichiometry. In the absence of phosphate, NaPi-IIa/b shows a Na+-dependent leak mode, hypothesized to involve the translocation of the first Na+ ion [3]. Cartoons modified from [47]
Presteady-state charge relaxations, which result from rapid changes in membrane potential, have proven indispensable to our understanding of the transduction of membrane potential as a transport driving force. They are ubiquitous property of many electrogenic cation-driven symport systems (e.g., SGLT1 [63, 91], GAT1 [101], EAAT [58]) and provided the first indirect evidence that conformational changes occur as the protein moves from one state to another in response to changes in membrane potential. For NaPi-IIa/b, the transport voltage dependence arises intrinsically from the proteins themselves that adopt favored orientations (outward facing or inward facing, Fig. 2), together with Na+ ion movement to binding sites within the transmembrane electric field (for review, see [47]). From measurements of the total charge displacement as a function of membrane potential, estimates of an effective valence per transporter can be obtained, amounting to 0.3–0.4 for the intrinsic carrier and 0.6–0.7 for the carrier with Na+ bound.
By substituting cysteines at functionally important sites and labeling these covalently with fluorophores, it was established that two Na+ ions bind sequentially and cooperatively before Pi and a third Na+ binding transition precedes the reorientation of the fully loaded carrier (see Fig. 2) [53, 142]. This conclusion was supported by a detailed cation substitution study in which presteady-state analysis identified the Li+ interaction site as corresponding to the first Na+ ion binding site [4]. The apparent affinity for Na+ is determined largely by the first Na+ interactions, whereas the third Na+ is a strong determinant of the transport turnover rate. Presteady state kinetic analysis can also be used to estimate the functional expression from the ratio of total mobile change to effective valence. Moreover, by combining with steady-state electrophysiological measurements, the transport turnover rate can be estimated. For selected electrogenic NaPi-IIa/b, this was typically <100 s−1 and may vary among isoforms and species [50].
The electroneutral NaPi-IIc transport mechanism differs from the electrogenic scheme in that the first Na+ ion to bind to the outward facing empty carrier is not transported (Fig. 2). Thus, for NaPi-IIc, three Na+ ions bind, but only two are translocated and released to the cytosol [53]. For the electrogenic isoforms, the first Na+ ion is thought to contribute to an uncoupled leak in the absence Pi [3]. Although we propose that Li+ can also compete with Na+ for occupancy at the first cation binding site and that in the cotransport mode Li+ ions can indeed translocate, their contribution to the leak current has not been demonstrated. This “uniport” mode is not detected in NaPi-IIc, but when electrogenicity is restored through mutagenesis, the leak mode returns, as evidenced by a change in holding current induced by the inhibitor phosphonoformic acid (PFA), a SLC34 inhibitor [8]. Interestingly, naturally occurring mutations in NaPi-IIc were shown to result in a significant Na+ leak [73]. Despite considerable progress in characterizing NaPi-II proteins at the mechanistic level, two areas still deserve attention. First, we are lacking concrete insight into the cytosolic release of substrates, principally because of the experimental limitations of the intact Xenopus oocyte preparation. In one study, NaPi-IIc expressing oocytes were preloaded with tracer substrate, and by establishing an outward driving force, reverse transport of Pi was demonstrated in agreement with the kinetic scheme [53]. Recently, evidence based on presteady-state analysis of the kinetics of the electroneutral NaPi-IIc, which was modified by mutagenesis to exhibit electrogenic behavior [8], suggests that the last step in the transport cycle is indeed the cytosolic release of one Na+ ion [110]. This also agrees with earlier studies on the uncoupled Na+ leak mode [4] in which it was proposed that the leak mode is essentially an uniporter involving the empty carrier and two Na+ bound states [43]. Further insight will only be obtained using patch clamp or cut-open oocyte techniques to controlling the cytosol composition.
Second, there is a lack of specific high-affinity inhibitors for SLC34 proteins. PFA acts as a competitive inhibitor for all isoforms [135, 137] and is the compound readily available. However its apparent inhibition constant is relatively large [90] (typically 1 mM (NaPi-IIa), 0.16 mM (NaPi-IIb), and 0.9 mM (NaPi-IIc) [136]), and at high concentrations, PFA can induce nonspecific leak in the cell membrane (I. C. Forster, unpublished observations). Other commercially synthesized substances with greater efficacies show mixed competitive and non-competitive behavior (e.g., JTP-59557, Japan Tobacco [99], see also [146]). Having access to specific and high-affinity inhibitors would obviously have considerable potential for control of dietary Pi intake, for which NaPi-IIb is a clinical target.
Structure and structure–function relationships
SLC34 proteins are a unique class of membrane transport proteins and share no obvious homology with the other solute carrier families, even at the bacterial level. At present a 3-D structure of the mammalian SLC34 proteins or their bacterial homologs is lacking, and therefore, all structural information has been derived from indirect biophysical and biochemical studies on wild-type and engineered mutations (for review, see [47]). NaPi-IIa was shown to be a functional monomer [82]; however, indirect evidence indicates dimeric or tetrameric structures in the membrane [46, 56, 59]. The primary sequences of the mammalian isoforms vary from 599 amino acids (NaPi-IIc) to ∼640 amino acids (NaPi-IIa and NaPi-IIb). Sequence differences appear mainly in the intracellular C- and N-terminal regions and the large extracellular loop which contains two N-glycosylation sites and a disulfide bridge that links the two halves of the protein (Fig. 3). The proposed secondary topology of the eukaryotic isoforms comprises eight transmembrane domains (TMDs) and two opposed reentrant loop domains that most likely contain short α-helical motifs. This model incorporates predictions from biochemical and biophysical studies including epitope labeling, cysteine scanning mutagenesis, and in vitro glycosylation assays (for review, see [44, 46, 48, 139]). The C-terminal region is important for targeting, hormonal regulation, and protein–protein interactions. For example, the TRL motif in the C-terminal plays a role as a PDZ binding motif [67, 77], and a KR motif located in an intracellular linker region (Fig. 3) is critical for PTH sensitivity [76]. Of note from the transport function perspective is the signature inverted repeat motif that is conserved among SLC34 isoforms and homologs in all phyla [147]. Cysteine substitution studies (see below) confirm the functional role of these repeats in defining the transport pathway [48, 52]. Given that similar motifs are found in the 3-D structures of transporters with identified architecture, e.g. [1, 42, 120], they most likely contain substrate coordination sites for NaPi-II proteins also. Thus, all SLC34 proteins, including bacterial homologs, are expected to have similar functional core elements comprising TMDs 2–10 (Fig. 3).
Topology and structure–function features of SLC 34 proteins. Topology model for SLC34 shows the predicted transmembrane domains (numbered) and repeat regions (dark shading) for each family. The 3-D folding is currently unknown. For SLC34, evidence suggests that the reentrant regions (3, 4) and (8, 9) physically associate to form a substrate coordination site [52]. The disulfide bridge and two glycosylation sites in the large extracellular loop are indicated for the SLC34 proteins. Critical residues for targeting/regulation and electrogenicity are highlighted
Cysteine scanning mutagenesis, in which cysteines are substituted at potentially functionally important sites in the protein and the accessibility by thiol-reactive reagents (methanesulfonates) is determined (e.g., [79]), has been applied extensively to SLC34 proteins (for review, see [47, 48]) (see Fig. 3). These studies have yielded both structural information as well as mechanistic insights. Substitutions have been made at sites in the predicted re-entrant regions, and accessibility studies confirm the reentrant topology [83, 87]. Accessibility of some sites is strongly membrane potential and substrate dependent, confirming that the protein conformation is sensitive to these variables [86, 142], and cross-linking studies suggest that the two reentrant regions indeed associate [52]. Finally, new insights into the transport mechanism and structure–function relationships have been gained by covalently linking fluorophores to engineered cysteines. The change in fluorescence emission in response to change in membrane potential and external substrate is an indication of specific conformational changes affecting the microenvironment of the fluorophore. In addition to establishing the cation binding order [53, 110, 142], they also provide compelling evidence of reciprocal movements of the two halves of the protein during the transport cycle [141].
Finally, insight into the molecular determinants of functional differences between isoforms can be gained by comparing sequences and identifying critical amino acids. For example, the electroneutral NaPi-IIc [122] was used to elucidate the molecular determinants of electrogenicity. Three regions were highlighted that differ significantly in amino acid sequence between electrogenic and electroneutral isoforms. In one of these regions, three critical amino acids were found by mutagenesis that are conserved in all electrogenic isoforms. Their presence confers electrogenic Na+ interactions and transport-dependent charge translocation [8], and recently, it was shown that this “re-engineering” of electrogenicity has most likely only restored charge movement to the empty carrier and internal release of one Na+ ion [110].
Regulation of SLC34 transporters
Interacting proteins
In vivo and in vitro experiments have shown that NaPi-IIa physically interacts with several proteins in the renal brush border membrane and that some of these interactions have effects on the expression as well as the regulation of the cotransporter. Most of the known interactions are PDZ (PSD-95, Disc-large, ZO-1)-based. The C-terminal residues of NaPi-IIa (TRL) constitute a PDZ-binding domain that is engaged in association with the PDZ domains of the four members of the NHERF (NHE3 regulatory factor) family [56, 57], as well as with Shank2E [100] (Fig. 4).
Network of proteins interacting with NaPi-IIa in renal proximal tubule cells. NaPi-IIa interacts with the four members of the NHERF family as well as with Shank in the brush border membrane. These interactions take place between the C-terminal PDZ-binding motif of NaPi-IIa (TRL) and the PDZ domains of the NHERFs/Shank proteins indicated with an asterisk. Association with NHERF1/2 controls the stability of the cotransporter in the apical membrane. In addition, NHERF1/2 may play a role in the PTH-induced downregulation of NaPi-IIa by determining the intracellular signaling activated upon receptor stimulation. The physiological meaning of the association of NaPi-IIa with NHERF3/4 is less clear, and it is not discussed in this review. In addition to a PDZ-domain, Shank2E also contains ankyrin repeats as well as SH3 and proline-rich domains that are not shown in the figure. PTH-R PTH receptor, PLC phospholipase C, PDZ post-synaptic density protein (PSD95), Dlg1 Drosophila disc large tumor suppressor, ZO-1 zonula occludens, and MERM-BD merlin–ezrin–radixin–moesin-binding domain
Binding to NHERF1 seems to be critical for the stability of the cotransporter at the brush border membrane, as either preventing the association in cell culture models [66] or the absence of NHERF1 in animal models [128] results in a reduced expression of NaPi-IIa at the cell membrane. Consistently with this reduction, Nherf1 −/− mice have an increased urinary excretion of phosphate that results in hypophosphatemia [128]. In humans, mutations in NHERF1 have been reported in patients with hypophosphatemia and relatively low TmP/GFR values [78]. NHERF1 has the potential to indirectly connect with the actin cytoskeleton via its association with ezrin, suggesting that the anchoring of NaPi-IIa to the plasma membrane depends on the formation of a multiprotein complex. Supporting this hypothesis, Ezrin −/− mice have a reduced expression of NHERF1 and NaPi-IIa at the brush border membrane that leads to urinary loss of phosphate and hypophosphatemia [60]. Therefore, it is not surprising that factors that reduce the expression of NaPi-IIa at the brush border membrane (such as PTH, dopamine or FGF23) induce the dissociation of NaPi-IIa from NHERF1. Treatment of renal proximal tubular cells with PTH leads to phosphorylation of a serine residue (S77) located within the PDZ domain of NHERF1 involved in interaction with NaPi-IIa. NHERF1 phosphorylation at S77 then results in a reduced binding to NaPi-IIa, allowing the retrieval of the cotransporter from the cell surface [35, 143]. A similar mechanism has been proposed for dopamine [145] and FGF23 [144]. PTH and dopamine act at least partially via activation of PKC and/or PKA [10, 14], and pharmacological activation of either kinase also results in phosphorylation of S77 [143]. Recently, PKA was also shown to phosphorylate ezrin in vitro, and this modification reduces the interaction of ezrin with NHERF1 [150]. Phosphorylation of ezrin may provide an additional molecular mechanism for the destabilization of NaPi-IIa at the cell surface upon hormonal activation. NHERF2 has been suggested to control the intracellular signaling pathway activated upon binding of PTH to its receptor [95]. Thus, in the absence of NHERF2, treatment with PTH results in activation of PKA, whereas the presence of NHERF2 leads preferentially to activation of phospholipase C (PLCβ). This switch is based on the capability of NHER2 to bind simultaneously to the PTH1R and PLCβ, a feature also shared with NHERF1 [95]. Accordingly, mice lacking NHERF1 show impaired activation of PLC by luminal PTH receptors and reduced internalization of NaPi-IIa [30].
The PDZ-binding motif of NaPi-IIa is also responsible for binding to Shank2E [36, 100]. Shank2E also interacts with dynamin II, a GTPase involved in the pinching off and endocytosis of clathrin-coated vesicles [107]. Unlike the NHERF proteins, Shank2E is internalized together with NaPi-IIa in response to high phosphate, suggesting that Shank2E controls the intracellular trafficking of the cotransporter.
The interaction of NaPi-IIa with GABA receptor-associated protein (GABARAP) is independent of the PDZ-binding motif [56, 116]. GABARAP is known to interact with proteins involved in intracellular trafficking, including tubulin, the N-ethylmaleimide-sensitive factor, and clathrin (for review, see [102]). GABARAP−/− mice are characterized by reduced urinary excretion of phosphate associated with increased expression of NaPi-IIa; also, NHERF1 was found to be upregulated in mutant animals [116]. The increase in NaPi-IIa does not result in higher levels of phosphate in plasma, probably due to a reduction in the expression of the intestinal NaPi-IIb cotransporter (see below).
Regulation of renal NaPi-IIa and NaPi-IIc
Regulation of SLC34 phosphate transporters has been best defined in the case of NaPi-IIa. In general, regulation occurs mostly on the posttranscriptional level by altering the rate of synthesis and degradation and by changing the amount of NaPi-IIa transporter molecules in the brush border membrane. There is little evidence for a regulation of NaPi-IIa transporters by altering their activity in the brush border membrane through phosphorylation or other similar mechanisms.
The biosynthetic pathway and mechanisms governing insertion of NaPi-IIa into the brush border membrane are little known. NaPi-IIa is N-glycosylated, and experiments in Xenopus oocytes indicated that lack of glycosylation is not altering transport characteristics but decreased membrane expression [62]. Thus, glycosylation may be required for insertion into and/or stability in the plasma membrane.
Retrieval of NaPi-IIa from the brush border membrane occurs rapidly within minutes after injecting PTH into rodents or after increasing dietary phosphate intake [13, 24]. NaPi-IIa is internalized via a route involving clathrin-coated pits as well as early and late endosomes and is routed to lysosomes for degradation [13, 81, 111, 134]. There is no evidence for recycling of internalized NaPi-IIa molecules. The pathway of NaPi-IIa internalization is shared with receptor-mediated endocytosis occurring in the same cells of the proximal tubule. The integrity of the machinery involved in receptor-mediated endocytosis is required for NaPi-IIa internalization. Genetic deletion of the major endocytic receptor megalin or its chaperone receptor-associated protein (RAP) alters the structure of the subapical compartment and reduces steady-state as well as PTH-provoked endocytosis leading to accumulation of NaPi-IIa in the brush border membrane and enhanced phosphate reabsorption [9, 11].
As indicated above, internalization of NaPi-IIa is initialized by phosphorylation of NHERF1 at serine 77 and subsequent dissociation of NaPi-IIa and NHERF1. NaPi-IIa is internalized, whereas NHERF1 and other NaPi-IIa-associated proteins remain at the apical membrane [35, 143]. There is no evidence for phosphorylation of NaPi-IIa by parathyroid hormone. The exact molecular mechanisms that address NaPi-IIa to the lysosome and are required for its routing are presently unknown.
In most cases, NaPiIIa internalization appears to occur without direct modification of the NaPiIIa transporter in the plasma membrane. However, downregulation by FGF23-klotho may involve also an additional mechanism. FGF23-klotho may induce direct phosphorylation of NHERF1 and internalization of NaPi-IIa [5]. In addition, klotho alone may proteolytically degrade NaPi-IIa located in the plasma membrane through a beta-glucuronidase-like mechanism. Klotho seems to first modify glycan residues on NaPi-IIa, allowing for cleavage of NaPi-IIa inducing its lysosomal degradation [69].
Regulation of NaPi-IIc abundance and activity is much less well studied. NaPi-IIc removal from the brush border membrane in response to PTH or intake of phosphate-rich diets occurs with much slower kinetics than for NaPi-IIa and requires hours [112, 126, 127]. In contrast to NaPi-IIa, NaPi-IIc may undergo partial recycling [126].
Regulation by signaling networks and kinases
Activation of parathyroid hormone receptors downregulates NaPi-IIa abundance at the brush border membrane within minutes through at least two different signaling pathways involving either PKA or PKC. Both pathways may eventually merge in ERK1/2 and lead to the phosphorylation of NHERF1. PTH leads also to downregulation of NaPi-IIc; however, this requires hours and the signals mediating this effect are not known [112].
The signals mediating the downregulation of NaPiIIa and NaPi-IIc by FGF-23 are not fully elucidated. FGF23 signals through binding to the FGF1c and FGF4 receptors together with its co-ligand klotho to reduce NaPi-IIa and NaPi-IIc protein and mRNA abundance [75, 119, 124]. FGF1c and klotho are expressed in the proximal tubule and with much higher abundance in the distal convoluted tubule. Activation of FGF1c causes phosphorylation of FRS2a and ERK1/2 and acts downstream on Egr1 [85]. Injection of FGF23 into mice increases staining for phosphorylated ERK1/2 in the distal convoluted tubule, and it has thus been suggested that the distal convoluted tubule may generate signals acting on the proximal tubule to reduce phosphate reabsorption [37]. Alternatively, FGF23 may directly activate in the proximal tubule a signaling cascade involving sgk1, ERK1/2, and NHERF1 phosphorylation to regulate NaPi-IIa [5].
Also activation of guanylate cyclases and increased intracellular cGMP production reduces NaPi-IIa expression in the brush border membrane. Atrial natriuretic peptide and NO may reduce renal phosphate reabsorption involving cGMP-dependent retrieval of NaPi-IIa [12].
Little is known about signals involved in the upregulation of NaPi-IIa expression and stimulated renal phosphate reabsorption. The insulin-like growth factor 1 (IGF1) stimulates NaPi-IIa expression and activity and may mediate some of the age- and growth-dependent effects on renal phosphate handling. The phosphorylation and activation of tyrosine kinase may be required for the IGF1-dependent effects [74].
Various additional kinases are involved in the regulation of NaPi-IIa function as evident from gene-modified mouse models. However, it is not clear in all instances if this is by directly affecting signaling networks in the proximal tubule or more indirectly by primarily altering the regulation of hormones acting on the kidney. A role for GSK3, Akt2/protein kinase beta 2, Jak2, Osr1, Spak, and Sgk3 has been found [41, 80, 108, 109, 129].
Regulation of NaPi-IIb
The abundance of NaPi-IIb at the apical membrane of the small intestine is under the control of a number of factors. Early studies indicated that phosphate absorption across the intestinal epithelia decreases during the suckling–weaning transition [22, 23, 39]. This reduction in the phosphate absorbing capacity coincides with an increase in the plasma levels of corticosterone, a glucocorticoid involved in intestinal maturation [65]. Arima et al. found that corticosterone administration results in downregulation of NaPi-IIb at the mRNA and protein level [6], an effect that may underlay the ontogenic regulation of the cotransporter.
As for the renal cotransporters, reduction in dietary intake of phosphate results in increased expression of NaPi-IIb in the intestinal epithelia [61]. This regulation is most probably independent of 1,25-OH2 vitamin D3, as it is preserved in VDR−/− as well as 1αOHase−/− mice [31, 123]. There are some discrepancies regarding whether or not the dietary adaptation involves changes in NaPi-IIb mRNA expression, with reports in favor [114, 123] and against [31, 61] transcriptional regulation. Recently, it was shown that NaPi-IIb and NHERF1 are coexpressed in intestinal epithelia and that they interact with each other via a C-terminal PDZ-binding motif of the cotransporter [55]. Furthermore, the dietary upregulation of NaPi-IIb is blunted in NHERF1−/− mice. Improper dietary regulation of the cotransporter has also been described in mice deficient for myosin VI [64]. Myosin VI is a minus-end actin-motor protein involved in a wide range of cellular functions including clathin-mediated endocytosis. Hegan et al. reported that feeding a low-Pi diet results in high and similar levels of NaPi-IIb in wild type and myosin VI−/− mice. However, upon administration of high phosphate, the dowregulation of the cotransporter at the cell surface is smaller in myosin VI−/− than wild-type mice [64]. These findings suggest that myosin VI is required for the membrane retrieval of the cotransporter. In addition to NHERF1- and myosin VI-deficient mice, the expression of NaPi-IIb is also altered in GABARAP−/− mice. As indicated above, NaPi-IIb expression is reduced in these mice, most probably as a compensatory mechanism for the increased expression of the renal NaPi-IIa [116, 117].
Although not required for the dietary adaptation, 1,25-OH2 vitamin D3 directly upregulates the expression of NaPi-IIb [61]. Moreover, the abundance of NaPi-IIb (protein and mRNA) is reduced in the intestinal epithelia of VDR−/− mice [31, 123]. Experiments in rats indicated that the effect of 1,25-OH2 vitamin D3 on NaPi-IIb expression involves changes in mRNA transcription in young but not in adult animals [148]. This last report also shows that the activity of the human NaPi-IIb promoter increases in response to 1,25-OH2 vitamin D3. The levels of 1,25-OH2 vitamin D3 are reduced in patients chronically treated with antimicrobial (rifampicin) or antiepileptic (phenobarbital) drugs [28]. Both drugs activate the pregnane X receptor (PXR), a nuclear xenobiotic receptor and ligand-activated transcription factor responsible for the inactivation and excretion of drugs. Although the levels of 1,25-OH2 vitamin D3 seem unaffected in PXR−/− mice, these animals have low serum phosphate levels associated with reduced intestinal expression of NaPi-IIb [84]. Furthermore, activation of PXR with rifampicin was reported to induce the NaPi-IIb promoter activity.
Estrogen administration increases the Na/Pi cotransport across the intestinal epithelia by upregulating the expression of NaPi-IIb [149]. Estrogens are known to regulate the production of 1,25-OH2 vitamin D3 [7]. However, their effect on the intestinal cotransporter is, at least partially, independent of 1,25-OH2 vitamin D3 as the activity of the human NaPi-IIb promoter measured in cell culture is directly stimulated by β-estradiol [149].
Normalization of the acid–base status upon metabolic acidosis involves an increased urinary excretion of protons. Acidosis leads to the release of phosphate from bones as well as to inhibition of renal reabsorption of phosphate, with the latter contributing to the supply of titrable acids in the urine [2, 106]. In contrast, both the intestinal Na/Pi cotransport and the expression of NaPi-IIb are increased in metabolic acidosis [130], suggesting that the gut compensates for the urinary loss of phosphate.
Inherited disorders of phosphate transport in the SLC34 family
Mutations in SLC34A1 and renal phosphate handling
The role of mutations and gene variants in the SLC34A1 gene on renal phosphate handling and kidney function are still not fully understood. Prie and colleagues reported on three patients with reduced renal threshold for phosphate reabsorption (TmP/GFR < 0.7 mM), hypercalciuria, and kidney stones. In one patient, a A48F mutation was detected; in the two other related patients, a V147M mutation was found. In all three patients, only one allele was found mutated and no further data were available whether there was cosegregation of the mutation with the phenotype in relatives of the patients [113]. Subsequent functional analysis of the mutations did not reveal any differences in the functional transport properties of the mutants or their trafficking in a mammalian renal cell line [140]. A second report presented patients from a consanguineous Arab Israeli family that had rickets, hypophosphatemic Fanconi syndrome, and hypercalcuria with highly elevated 1,25-OH2 vitamin D3 levels. The patients had all a homozygous in-frame duplication (21 bp) G154-V160dup that completely abolished phosphate-induced currents in the mutant expressed in Xenopus laevis oocytes [94]. Since the inserted stretch of amino acids affects a putative transmembrane domain of NaPi-IIa, it is very likely that the overall structure of the transporter is severely altered. The occurrence of the Fanconi syndrome is not complete since plasma bicarbonate levels are unchanged and patients are able to increase levels of active 1,25-OH2 vitamin D3 [94]. Moreover, in two different mouse models of Slc34a1, deficiency either due to genetic deletion or to the spontaneous occurrence of the compound heterozygous mutations at L499V and V528M no Fanconi syndrome has been observed [15, 70]. Thus, our understanding of the importance of SLC34A1 for renal phosphate handling in man and the impact of mutations on generalized proximal tubule functions is incomplete to date.
Mutations in SL34A2: pulmonary microlithiasis
Mutations in SLC34A2 cause pulmonary microlithiasis that is in some cases associated with testicular microlithiasis [33]. Different mutations have been described including missense mutations and truncations [40]. The cellular mechanisms how mutations in SLC34A2 lead to impaired NaPi-IIb function has not been addressed to date. Pulmonary alveolar microlithiasis is a very rare lung disease characterized by calcifications within the alveoli that may eventually lead to loss of lung function [40]. The pathogenesis of the disease has not been clarified and may relate to functions of NaPi-IIb in alveolar type II cells [133]. Whether NaPi-IIb is relevant for removing phosphate from the alveolar fluid or has other functions has not been determined. Unfortunately, no information is available whether patients with SLC34A2 mutations show other systemic disturbances of phosphate homeostasis and whether intestinal phosphate absorption is altered.
Mice lacking NaPi-IIb (Slc34a2) are not viable, whereas inducible NaPi-IIb KO mice with deletion of Slc34a2 in adult mice cause pulmonary calcification and reduced intestinal phosphate absorption [118]. Loss of NaPi-IIb is associated with abolished sodium-dependent phosphate transport in ileum suggesting that NaPi-IIb is the major if not only important phosphate transporter in this segment. However, overall intestinal phosphate absorption was only mildly reduced, suggesting that other segments of the small and large intestine absorb the largest quantity of phosphate and/or that other pathways such as the paracellular route may provide for major phosphate fluxes in intestine.
Mutations in SLC34A3: hereditary hypophosphatemic rickets with hypercalcuria
Hereditary hypophosphatemic rickets with hypercalcuria (HHRH) is a rare autosomal recessive disorder caused by mutations in the SLC34A3 gene encoding for NaPi-IIc [19, 92]. Patients suffer from hypophosphatemia due to renal phosphate losses. Reduced phosphate availability impairs bone mineralization and growth leading to rickets. Compensatory increases in 1,25-OH2 vitamin D3 synthesis and levels stimulate intestinal phosphate and calcium absorption. However, excessive calcium uptake is counterbalanced by increased urinary excretion where high urinary levels of calcium and phosphate cause nephrolithiasis and nephrocalcinosis.
Patients with HHRH showed SLC34A3 deletions and truncations but also splice site mutations, homozygous missense, or compound missense mutations [18]. Missense mutations affect trafficking to the plasma membrane and/or abundance [73].
Mice lacking NaPi-IIc have been generated, and surprisingly, no major phenotype could be detected. Renal phosphate handling is similar to wild-type mice, and no hypophosphatemia or hyperphosphaturia develops. However, NaPi-IIc shows hypercalciuria and slightly increased 1,25-OH2 vitamin D3 levels [125]. Also, a kidney-specific NaPi-IIc-deficient mouse model has been developed and this mouse model has normal renal phosphate and calcium handling with no signs for altered 1,25-OH2 vitamin D3 metabolism (Myakala et al., unpublished results). Thus, while NaPi-IIc has a major role in phosphate metabolism in humans, the role in mice appears to be minor.
Summary and future perspectives
Twenty years after the molecular identification of the first member of the SLC34 family, much progress has been made in understanding the hormonal regulation of these transporters, identification of various interacting proteins, and gaining insights into the transport mechanism of these transporters. Genetic studies demonstrated that all three transporters have important physiological functions. However, major questions have remained unanswered or are only partially understood. At least three fields are certainly of major interest and not fully explored to date.
Structural models of SLC34 transporters rely on structure–functions studies mostly using mutagenesis, chemical labeling, and electrophysiology experiments. Obviously, crystal structures of mammalian SLC34 transporters or homologs would greatly enhance our understanding of the transport mechanism and would possibly facilitate development of isoform specific inhibitors.
NaPi-IIa served as a model protein to examine regulation of phosphate and other epithelial transporters. The identification of interacting proteins helped to understand the role of some of these proteins in epithelial physiology as exemplified for NHERF1. Nevertheless, our understanding of the “life cycle” of these transport proteins is incomplete. We miss insight into the processes regulating translation and trafficking of transporters to the cell membrane and mechanisms that affect the residence at the plasma membrane.
A third field that will require further attention is the regulation of these transporters by phosphate. This is also of major clinical importance as dysregulation of phosphate handling by intestine, bone, and kidney may contribute to chronic kidney and bone disease and subsequently may increase the risk for cardiovascular diseases. FGF23 has gained much attention over the last years, but the mechanisms how increases in phosphate intake or body content trigger changes in major phosphate regulating hormones such as parathyroid hormone, FGF23, or 1,25-OH2 vitamin D3 are largely unknown. The possibility that phosphate may regulate these transporters also independent from known factors remains also open. Thus, “phosphate-sensing” mechanisms and mediators of regulation are not fully explored and understood.
References
Abramson J, Wright EM (2009) Structure and function of Na+-symporters with inverted repeats. Curr Opin Struct Biol 19:425–432
Ambuhl PM, Zajicek HK, Wang H, Puttaparthi K, Levi M (1998) Regulation of renal phosphate transport by acute and chronic metabolic acidosis in the rat. Kidney Int 53:1288–1298
Andrini O, Ghezzi C, Murer H, Forster IC (2008) The leak mode of type II Na+-Pi cotransporters. Channels (Austin) 2:346–357
Andrini O, Meinild AK, Ghezzi C, Murer H, Forster IC (2012) Lithium interactions with Na+-coupled inorganic phosphate cotransporters: insights into the mechanism of sequential cation binding. Am J Physiol Cell Physiol 302:C539–C554
Andrukhova O, Zeitz U, Goetz R, Mohammadi M, Lanske B, Erben RG (2012) FGF23 acts directly on renal proximal tubules to induce phosphaturia through activation of the ERK1/2-SGK1 signaling pathway. Bone 51:621–628
Arima K, Hines ER, Kiela PR, Drees JB, Collins JF, Ghishan FK (2002) Glucocorticoid regulation and glycosylation of mouse intestinal type IIb Na-Pi cotransporter during ontogeny. Am J Physiol Gastrointest Liver Physiol 283:G426–G434
Ash SL, Goldin BR (1988) Effects of age and estrogen on renal vitamin D metabolism in the female rat. Am J Clin Nutr 47:694–699
Bacconi A, Virkki LV, Biber J, Murer H, Forster IC (2005) Renouncing electrogenicity is not free of charge: switching on electrogenicity in a Na+-coupled phosphate cotransporter. Proc Natl Acad Sci U S A 102:12606–12611
Bachmann S, Schlichting U, Geist B, Mutig K, Petsch T, Bacic D, Wagner CA, Kaissling B, Biber J, Murer H, Willnow TE (2004) Kidney-specific inactivation of the megalin gene impairs trafficking of renal inorganic sodium phosphate cotransporter (NaPi-IIa). J Am Soc Nephrol 15:892–900
Bacic D, Capuano P, Baum M, Zhang J, Stange G, Biber J, Kaissling B, Moe OW, Wagner CA, Murer H (2005) Activation of dopamine D1-like receptors induces acute internalization of the renal Na+/phosphate cotransporter NaPi-IIa in mouse kidney and OK cells. Am J Physiol Renal Physiol 288:F740–F747
Bacic D, Capuano P, Gisler SM, Pribanic S, Christensen EI, Biber J, Loffing J, Kaissling B, Wagner CA, Murer H (2003) Impaired PTH-induced endocytotic down-regulation of the renal type IIa Na+/Pi-cotransporter in RAP-deficient mice with reduced megalin expression. Pflugers Arch 446:475–484
Bacic D, Hernando N, Traebert M, Lederer E, Völkl H, Biber J, Kaissling B, Murer H (2001) Regulation of the renal type IIa Na/Pi cotransporter by cGMP. Pflugers Arch 443:306–313
Bacic D, Lehir M, Biber J, Kaissling B, Murer H, Wagner CA (2006) The renal Na+/phosphate cotransporter NaPi-IIa is internalized via the receptor-mediated endocytic route in response to parathyroid hormone. Kidney Int 69:495–503
Bacic D, Schulz N, Biber J, Kaissling B, Murer H, Wagner CA (2003) Involvement of the MAPK-kinase pathway in the PTH-mediated regulation of the proximal tubule type IIa Na+/Pi cotransporter in mouse kidney. Pflugers Arch 446:52–60
Beck L, Karaplis AC, Amizuka N, Hewson AS, Ozawa H, Tenenhouse HS (1998) Targeted inactivation of Npt2 in mice leads to severe renal phosphate wasting, hypercalciuria, and skeletal abnormalities. Proc Natl Acad Sci U S A 95:5372–5377
Beene LC, Halluer J, Yoshinaga M, Hamdi M, Liu Z (2011) Pentavalent arsenate transport by zebrafish phosphate transporter NaPi-IIb1. Zebrafish 8:125–131
Bergwitz C, Juppner H (2010) Regulation of phosphate homeostasis by PTH, vitamin D, and FGF23. Annu Rev Med 61:91–104
Bergwitz C, Juppner H (2012) FGF23 and syndromes of abnormal renal phosphate handling. Adv Exp Med Biol 728:41–64
Bergwitz C, Roslin NM, Tieder M, Loredo-Osti JC, Bastepe M, Abu-Zahra H, Frappier D, Burkett K, Carpenter TO, Anderson D, Garabedian M, Sermet I, Fujiwara TM, Morgan K, Tenenhouse HS, Juppner H (2006) SLC34A3 mutations in patients with hereditary hypophosphatemic rickets with hypercalciuria predict a key role for the sodium-phosphate cotransporter NaPi-IIc in maintaining phosphate homeostasis. Am J Hum Genet 78:179–192
Berndt T, Kumar R (2007) Phosphatonins and the regulation of phosphate homeostasis. Annu Rev Physiol 69:341–359
Biber J, Hernando N, Forster I (2013) Phosphate transporters and their function. Annu Rev Physiol 75:535–550
Borowitz SM, Granrud GS (1992) Glucocorticoids inhibit intestinal phosphate absorption in developing rabbits. J Nutr 122:1273–1279
Borowitz SM, Granrud GS (1992) Ontogeny of intestinal phosphate absorption in rabbits. Am J Physiol 262:G847–G853
Bourgeois S, Capuano P, Stange G, Muhlemann R, Murer H, Biber J, Wagner CA (2013) The phosphate transporter NaPi-IIa determines the rapid renal adaptation to dietary phosphate intake in mouse irrespective of persistently high FGF23 levels. Pflugers Arch 465:1557–1572
Brazy PC, Gullans SR, Mandel LJ, Dennis VW (1982) Metabolic requirement for inorganic phosphate by the rabbit proximal tubule. J Clin Invest 70:53–62
Brazy PC, Mandel LJ, Gullans SR, Soltoff SP (1984) Interactions between phosphate and oxidative metabolism in proximal renal tubules. Am J Physiol 247:F575–F581
Breusegem SY, Takahashi H, Giral-Arnal H, Wang X, Jiang T, Verlander JW, Wilson P, Miyazaki-Anzai S, Sutherland E, Caldas Y, Blaine JT, Segawa H, Miyamoto K, Barry NP, Levi M (2009) Differential regulation of the renal sodium-phosphate cotransporters NaPi-IIa, NaPi-IIc, and PiT-2 in dietary potassium deficiency. Am J Physiol Renal Physiol 297:F350–F361
Brodie MJ, Boobis AR, Hillyard CJ, Abeyasekera G, Stevenson JC, MacIntyre I, Park BK (1982) Effect of rifampicin and isoniazid on vitamin D metabolism. Clin Pharmacol Ther 32:525–530
Brunelli SM, Goldfarb S (2007) Hypophosphatemia: clinical consequences and management. J Am Soc Nephrol 18:1999–2003
Capuano P, Bacic D, Roos M, Gisler SM, Stange G, Biber J, Kaissling B, Weinman EJ, Shenolikar S, Wagner CA, Murer H (2007) Defective coupling of apical PTH receptors to phospholipase C prevents internalization of the Na+-phosphate cotransporter NaPi-IIa in Nherf1-deficient mice. Am J Physiol Cell Physiol 292:C927–C934
Capuano P, Radanovic T, Wagner CA, Bacic D, Kato S, Uchiyama Y, St-Arnoud R, Murer H, Biber J (2005) Intestinal and renal adaptation to a low-Pi diet of type II NaPi cotransporters in vitamin D receptor- and 1alphaOHase-deficient mice. Am J Physiol Cell Physiol 288:C429–C434
Coladonato JA (2005) Control of hyperphosphatemia among patients with ESRD. J Am Soc Nephrol 16(Suppl 2):S107–S114
Corut A, Senyigit A, Ugur SA, Altin S, Ozcelik U, Calisir H, Yildirim Z, Gocmen A, Tolun A (2006) Mutations in SLC34A2 cause pulmonary alveolar microlithiasis and are possibly associated with testicular microlithiasis. Am J Hum Genet 79:650–656
Custer M, Lötscher M, Biber J, Murer H, Kaissling B (1994) Expression of Na-Pi cotransport in rat kidney: localization by RT-PCR and immunohistochemistry. Am J Physiol 266:F767–F774
Deliot N, Hernando N, Horst-Liu Z, Gisler SM, Capuano P, Wagner CA, Bacic D, O’Brien S, Biber J, Murer H (2005) Parathyroid hormone treatment induces dissociation of type IIa Na+-P(i) cotransporter-Na+/H+ exchanger regulatory factor-1 complexes. Am J Physiol Cell Physiol 289:C159–C167
Dobrinskikh E, Lanzano L, Rachelson J, Cranston D, Moldovan R, Lei T, Gratton E, Doctor RB (2013) Shank2 contributes to the apical retention and intracellular redistribution of NaPiIIa in OK cells. Am J Physiol Cell Physiol 304:C561–C573
Farrow EG, Davis SI, Summers LJ, White KE (2009) Initial FGF23-mediated signaling occurs in the distal convoluted tubule. J Am Soc Nephrol 20:955–960
Farrow EG, White KE (2010) Recent advances in renal phosphate handling. Nat Rev Nephrol 6:207–217
Ferraro C, Ladizesky M, Cabrejas M, Montoreano R, Mautalen C (1976) Intestinal absorption of phosphate: action of protein synthesis inhibitors and glucocorticoids in the rat. J Nutr 106:1752–1756
Ferreira Francisco FA, Pereira e Silva JL, Hochhegger B, Zanetti G, Marchiori E (2013) Pulmonary alveolar microlithiasis. State-of-the-art review. Respir Med 107:1–9
Foller M, Kempe DS, Boini KM, Pathare G, Siraskar B, Capuano P, Alesutan I, Sopjani M, Stange G, Mohebbi N, Bhandaru M, Ackermann TF, Judenhofer MS, Pichler BJ, Biber J, Wagner CA, Lang F (2011) PKB/SGK-resistant GSK3 enhances phosphaturia and calciuria. J Am Soc Nephrol 22:873–880
Forrest LR, Kramer R, Ziegler C (2011) The structural basis of secondary active transport mechanisms. Biochim Biophys Acta 1807:167–188
Forster I, Hernando N, Biber J, Murer H (1998) The voltage dependence of a cloned mammalian renal type II Na+/Pi cotransporter (NaPi-2). J Gen Physiol 112:1–18
Forster I, Hernando N, Sorribas V, Werner A (2011) Phosphate transporters in renal, gastrointestinal, and other tissues. Adv Chron Kidney Dis 18:63–76
Forster IC, Biber J, Murer H (2000) Proton-sensitive transitions of renal type II Na+-coupled phosphate cotransporter kinetics. Biophys J 79:215–230
Forster IC, Hernando N, Biber J, Murer H (2006) Proximal tubular handling of phosphate: a molecular perspective. Kidney Int 70:1548–1559
Forster IC, Hernando N, Biber J, Murer H (2012) Phosphate transport kinetics and structure-function relationships of SLC34 and SLC20 proteins. Curr Top Membr 70:313–356
Forster IC, Kohler K, Biber J, Murer H (2002) Forging the link between structure and function of electrogenic cotransporters: the renal type IIa Na+/Pi cotransporter as a case study. Prog Biophys Mol Biol 80:69–108
Forster IC, Loo DD, Eskandari S (1999) Stoichiometry and Na+ binding cooperativity of rat and flounder renal type II Na+-Pi cotransporters. Am J Physiol 276:F644–F649
Forster IC, Virkki LV, Bossi E, Murer H, Biber J (2006) Electrogenic kinetics of a mammalian intestinal Na+/Pi-cotransporter. J Membr Biol 212:177–190
Freeman D, Bartlett S, Radda G, Ross B (1983) Energetics of sodium transport in the kidney. Saturation transfer 31P-NMR. Biochim Biophys Acta 762:325–336
Ghezzi C, Meinild AK, Murer H, Forster IC (2011) Voltage- and substrate-dependent interactions between sites in putative re-entrant domains of a Na+-coupled phosphate cotransporter. Pflugers Arch 461:645–663
Ghezzi C, Murer H, Forster IC (2009) Substrate interactions of the electroneutral Na+-coupled inorganic phosphate cotransporter (NaPi-IIc). J Physiol 587:4293–4307
Giral H, Caldas Y, Sutherland E, Wilson P, Breusegem S, Barry N, Blaine J, Jiang T, Wang XX, Levi M (2009) Regulation of rat intestinal Na-dependent phosphate transporters by dietary phosphate. Am J Physiol Renal Physiol 297:F1466–F1475
Giral H, Cranston D, Lanzano L, Caldas Y, Sutherland E, Rachelson J, Dobrinskikh E, Weinman EJ, Doctor RB, Gratton E, Levi M (2012) NHE3 regulatory factor 1 (NHERF1) modulates intestinal sodium-dependent phosphate transporter (NaPi-2b) expression in apical microvilli. J Biol Chem 287:35047–35056
Gisler SM, Kittanakom S, Fuster D, Wong V, Bertic M, Radanovic T, Hall RA, Murer H, Biber J, Markovich D, Moe OW, Stagljar I (2008) Monitoring protein-protein interactions between the mammalian integral membrane transporters and PDZ-interacting partners using a modified split-ubiquitin membrane yeast two-hybrid system. Mol Cell Proteomics 7:1362–1377
Gisler SM, Stagljar I, Traebert M, Bacic D, Biber J, Murer H (2001) Interaction of the type IIa Na/Pi cotransporter with PDZ proteins. J Biol Chem 276(12):9206–9213
Grewer C, Rauen T (2005) Electrogenic glutamate transporters in the CNS: molecular mechanism, pre-steady-state kinetics, and their impact on synaptic signaling. J Membr Biol 203:1–20
Haito-Sugino S, Ito M, Ohi A, Shiozaki Y, Kangawa N, Nishiyama T, Aranami F, Sasaki S, Mori A, Kido S, Tatsumi S, Segawa H, Miyamoto KI (2011) Processing and stability of type IIc sodium-dependent phosphate cotransporter mutations in patients with hereditary hypophosphatemic rickets with hypercalciuria. Am J Physiol Cell Physiol 302:C1316–C1330
Hatano R, Fujii E, Segawa H, Mukaisho K, Matsubara M, Miyamoto K, Hattori T, Sugihara H, Asano S (2013) Ezrin, a membrane cytoskeletal cross-linker, is essential for the regulation of phosphate and calcium homeostasis. Kidney Int 83:41–49
Hattenhauer O, Traebert M, Murer H, Biber J (1999) Regulation of small intestinal Na-Pi type IIb cotransporter by dietary phosphate intake. Am J Physiol 277:G756–G762
Hayes G, Busch A, Lötscher M, Waldegger S, Lang F, Verrey F, Biber J, Murer H (1994) Role of N-linked glycosylation in rat renal Na/Pi-cotransport. J Biol Chem 269:24143–24149
Hazama A, Loo DD, Wright EM (1997) Presteady-state currents of the rabbit Na+/glucose cotransporter (SGLT1). J Membr Biol 155:175–186
Hegan PS, Giral H, Levi M, Mooseker MS (2012) Myosin VI is required for maintenance of brush border structure, composition, and membrane trafficking functions in the intestinal epithelial cell. Cytoskeleton (Hoboken) 69:235–251
Henning SJ (1978) Plasma concentrations of total and free corticosterone during development in the rat. Am J Physiol 235:E451–E456
Hernando N, Deliot N, Gisler SM, Lederer E, Weinman EJ, Biber J, Murer H (2002) PDZ-domain interactions and apical expression of type IIa Na/Pi cotransporters. Proc Natl Acad Sci U S A 99:11957–11962
Hernando N, Karim-Jimenez Z, Biber J, Murer H (2001) Molecular determinants for apical expression and regulatory membrane retrieval of the type IIa Na/Pi cotransporter. Kidney Int 60:431–435
Hilfiker H, Hattenhauer O, Traebert M, Forster I, Murer H, Biber J (1998) Characterization of a murine type II sodium-phosphate cotransporter expressed in mammalian small intestine. Proc Natl Acad Sci U S A 95:14564–14569
Hu MC, Shi M, Zhang J, Pastor J, Nakatani T, Lanske B, Shawkat Razzaque M, Rosenblatt KP, Baum MG, Kuro OM, Moe OW (2010) Klotho: a novel phosphaturic substance acting as an autocrine enzyme in the renal proximal tubule. FASEB J 24:3438–3450
Iwaki T, Sandoval-Cooper MJ, Tenenhouse HS, Castellino FJ (2008) A missense mutation in the sodium phosphate co-transporter Slc34a1 impairs phosphate homeostasis. J Am Soc Nephrol 19:1753–1762
Jaeger P, Bonjour JP, Karlmark B, Stanton B, Kirk RG, Duplinsky T, Giebisch G (1983) Influence of acute potassium loading on renal phosphate transport in the rat kidney. Am J Physiol 245:F601–F605
Jaeger P, Karlmark B, Stanton B, Kirk RG, Duplinsky T, Giebisch G (1980) Micropuncture study of distal tubular activation of phosphate reabsorption in the rat. Adv Exp Med Biol 128:77–82
Jaureguiberry G, Carpenter TO, Forman S, Juppner H, Bergwitz C (2008) A novel missense mutation in SLC34A3 that causes hereditary hypophosphatemic rickets with hypercalciuria in humans identifies threonine 137 as an important determinant of sodium-phosphate cotransport in NaPi-IIc. Am J Physiol Renal Physiol 295:F371–F379
Jehle AW, Forgo J, Biber J, Lederer E, Krapf R, Murer H (1998) IGF-I and vanadate stimulate Na/Pi-cotransport in OK cells by increasing type II Na/Pi-cotransporter protein stability. Pflugers Arch 437:149–154
Juppner H (2011) Phosphate and FGF-23. Kidney Int Suppl:S24–27
Karim-Jimenez Z, Hernando N, Biber J, Murer H (2000) Requirement of a leucine residue for (apical) membrane expression of type IIb NaPi cotransporters. Proc Natl Acad Sci U S A 97:2916–2921
Karim-Jimenez Z, Hernando N, Biber J, Murer H (2001) Molecular determinants for apical expression of the renal type IIa Na+/Pi-cotransporter. Pflugers Arch 442:782–790
Karim Z, Gerard B, Bakouh N, Alili R, Leroy C, Beck L, Silve C, Planelles G, Urena-Torres P, Grandchamp B, Friedlander G, Prie D (2008) NHERF1 mutations and responsiveness of renal parathyroid hormone. N Engl J Med 359:1128–1135
Karlin A, Akabas MH (1998) Substituted-cysteine accessibility method. Methods Enzymol 293:123–145
Kempe DS, Ackermann TF, Boini KM, Klaus F, Umbach AT, Dermaku-Sopjani M, Judenhofer MS, Pichler BJ, Capuano P, Stange G, Wagner CA, Birnbaum MJ, Pearce D, Foller M, Lang F (2010) Akt2/PKBbeta-sensitive regulation of renal phosphate transport. Acta Physiol (Oxf) 200:75–85
Keusch I, Traebert M, Lötscher M, Kaissling B, Murer H, Biber J (1998) Parathyroid hormone and dietary phosphate provoke a lysosomal routing of the proximal tubular Na/Pi-cotransporter type II. Kidney Int 54:1224–1232
Kohler K, Forster IC, Lambert G, Biber J, Murer H (2000) The functional unit of the renal type IIa Na+/Pi cotransporter is a monomer. J Biol Chem 275:26113–26120
Kohler K, Forster IC, Stange G, Biber J, Murer H (2002) Identification of functionally important sites in the first intracellular loop of the NaPi-IIa cotransporter. Am J Physiol 282:F687–F696
Konno Y, Moore R, Kamiya N, Negishi M (2010) Nuclear xenobiotic receptor PXR-null mouse exhibits hypophosphatemia and represses the Na/Pi-cotransporter SLC34A2. Pharmacogenet Genomics 20:9–17
Kurosu H, Ogawa Y, Miyoshi M, Yamamoto M, Nandi A, Rosenblatt KP, Baum MG, Schiavi S, Hu MC, Moe OW, Kuro-o M (2006) Regulation of fibroblast growth factor-23 signaling by klotho. J Biol Chem 281:6120–6123
Lambert G, Forster IC, Stange G, Biber J, Murer H (1999) Properties of the mutant Ser-460-Cys implicate this site in a functionally important region of the type IIa Na+/Pi cotransporter protein. J Gen Physiol 114:637–652
Lambert G, Forster IC, Stange G, Kohler K, Biber J, Murer H (2001) Cysteine mutagenesis reveals novel structure-function features within the predicted third extracellular loop of the type IIa Na+/Pi cotransporter. J Gen Physiol 117:533–546
Lee DB, Walling MW, Brautbar N (1986) Intestinal phosphate absorption: influence of vitamin D and non-vitamin D factors. Am J Physiol 250:G369–G373
Levi M, Lötscher M, Sorribas V, Custer M, Arar M, Kaissling B, Murer H, Biber J (1994) Cellular mechanisms of acute and chronic adaptation of rat renal Pi transporter to alterations in dietary Pi. Am J Physiol 267:F900–F908
Loghman-Adham M (1996) Use of phosphonocarboxylic acids as inhibitors of sodium-phosphate cotransport. Gen Pharmacol 27:305–312
Loo DD, Hirayama BA, Cha A, Bezanilla F, Wright EM (2005) Perturbation analysis of the voltage-sensitive conformational changes of the Na+/glucose cotransporter. J Gen Physiol 125:13–36
Lorenz-Depiereux B, Benet-Pages A, Eckstein G, Tenenbaum-Rakover Y, Wagenstaller J, Tiosano D, Gershoni-Baruch R, Albers N, Lichtner P, Schnabel D, Hochberg Z, Strom TM (2006) Hereditary hypophosphatemic rickets with hypercalciuria is caused by mutations in the sodium-phosphate cotransporter gene SLC34A3. Am J Hum Genet 78:193–201
Magagnin S, Werner A, Markovich D, Sorribas V, Stange G, Biber J, Murer H (1993) Expression cloning of human and rat renal cortex Na/Pi cotransport. Proc Natl Acad Sci U S A 90(13):5979–5983
Magen D, Berger L, Coady MJ, Ilivitzki A, Militianu D, Tieder M, Selig S, Lapointe JY, Zelikovic I, Skorecki K (2010) A loss-of-function mutation in NaPi-IIa and renal Fanconi’s syndrome. N Engl J Med 362:1102–1109
Mahon MJ, Donowitz M, Yun CC, Segre GV (2002) Na+/H+ exchanger regulatory factor 2 directs parathyroid hormone 1 receptor signalling. Nature 417:858–861
Marks J, Debnam ES, Unwin RJ (2010) Phosphate homeostasis and the renal-gastrointestinal axis. Am J Physiol Renal Physiol 299:F285–F296
Marks J, Srai SK, Biber J, Murer H, Unwin RJ, Debnam ES (2006) Intestinal phosphate absorption and the effect of vitamin D: a comparison of rats with mice. Exp Physiol 91:531–537
Martin A, David V, Quarles LD (2012) Regulation and function of the FGF23/klotho endocrine pathways. Physiol Rev 92:131–155
Matsuo A, Negoro T, Seo T, Kitao Y, Shindo M, Segawa H, Miyamoto K (2005) Inhibitory effect of JTP-59557, a new triazole derivative, on intestinal phosphate transport in vitro and in vivo. Eur J Pharmacol 517:111–119
McWilliams RR, Breusegem SY, Brodsky KF, Kim E, Levi M, Doctor RB (2005) Shank2E binds NaP(i) cotransporter at the apical membrane of proximal tubule cells. Am J Physiol Cell Physiol 289:C1042–C1051
Meinild AK, Forster IC (2012) Using lithium to probe sequential cation interactions with GAT1. Am J Physiol Cell Physiol 302(11):C1661–C1675
Mohrluder J, Schwarten M, Willbold D (2009) Structure and potential function of gamma-aminobutyrate type A receptor-associated protein. FEBS J 276:4989–5005
Murer H, Hernando N, Forster I, Biber J (2000) Proximal tubular phosphate reabsorption: molecular mechanisms. Physiol Rev 80:1373–1409
Murer H, Hernando N, Forster I, Biber J (2003) Regulation of Na/Pi transporter in the proximal tubule. Annu Rev Physiol 65:531–542
Nishimura M, Naito S (2008) Tissue-specific mRNA expression profiles of human solute carrier transporter superfamilies. Drug Metab Pharmacokinet 23:22–44
Nowik M, Picard N, Stange G, Capuano P, Tenenhouse HS, Biber J, Murer H, Wagner CA (2008) Renal phosphaturia during metabolic acidosis revisited: molecular mechanisms for decreased renal phosphate reabsorption. Pflugers Arch 457:539–549
Okamoto PM, Gamby C, Wells D, Fallon J, Vallee RB (2001) Dynamin isoform-specific interaction with the shank/ProSAP scaffolding proteins of the postsynaptic density and actin cytoskeleton. J Biol Chem 276:48458–48465
Pathare G, Foller M, Daryadel A, Mutig K, Bogatikov E, Fajol A, Almilaji A, Michael D, Stange G, Voelkl J, Wagner CA, Bachmann S, Lang F (2012) OSR1-sensitive renal tubular phosphate reabsorption. Kidney Blood Press Res 36:149–161
Pathare G, Foller M, Michael D, Walker B, Hierlmeier M, Mannheim JG, Pichler BJ, Lang F (2012) Enhanced FGF23 serum concentrations and phosphaturia in gene targeted mice expressing WNK-resistant SPAK. Kidney Blood Press Res 36:355–364
Patti M, Ghezzi C, Forster IC (2013) Conferring electrogenicity to the electroneutral phosphate cotransporter NaPi-IIc (SLC34A3) reveals an internal cation release step. Pflugers Arch 465:1261–1279
Pfister MF, Ruf I, Stange G, Ziegler U, Lederer E, Biber J, Murer H (1998) Parathyroid hormone leads to the lysosomal degradation of the renal type II Na/Pi cotransporter. Proc Natl Acad Sci U S A 95:1909–1914
Picard N, Capuano P, Stange G, Mihailova M, Kaissling B, Murer H, Biber J, Wagner CA (2010) Acute parathyroid hormone differentially regulates renal brush border membrane phosphate cotransporters. Pflugers Arch 460:677–687
Prie D, Huart V, Bakouh N, Planelles G, Dellis O, Gerard B, Hulin P, Benque-Blanchet F, Silve C, Grandchamp B, Friedlander G (2002) Nephrolithiasis and osteoporosis associated with hypophosphatemia caused by mutations in the type 2a sodium-phosphate cotransporter. N Engl J Med 347:983–991
Radanovic T, Wagner CA, Murer H, Biber J (2005) Regulation of intestinal phosphate transport. I. Segmental expression and adaptation to low-P(i) diet of the type IIb Na+-Pi cotransporter in mouse small intestine. Am J Physiol Gastrointest Liver Physiol 288:G496–G500
Razzaque MS (2013) Phosphate toxicity and vascular mineralization. Contrib Nephrol 180:74–85
Reining SC, Gisler SM, Fuster D, Moe OW, O’Sullivan GA, Betz H, Biber J, Murer H, Hernando N (2009) GABARAP deficiency modulates expression of NaPi-IIa in renal brush-border membranes. Am J Physiol Renal Physiol 296:F1118–F1128
Reining SC, Liesegang A, Betz H, Biber J, Murer H, Hernando N (2010) Expression of renal and intestinal Na/Pi cotransporters in the absence of GABARAP. Pflugers Arch 460:207–217
Sabbagh Y, O’Brien SP, Song W, Boulanger JH, Stockmann A, Arbeeny C, Schiavi SC (2009) Intestinal npt2b plays a major role in phosphate absorption and homeostasis. J Am Soc Nephrol 20:2348–2358
Saito H, Kusano K, Kinosaki M, Ito H, Hirata M, Segawa H, Miyamoto K, Fukushima N (2003) Human fibroblast growth factor-23 mutants suppress Na+-dependent phosphate co-transport activity and 1alpha,25-dihydroxyvitamin D3 production. J Biol Chem 278:2206–2211
Screpanti E, Hunte C (2007) Discontinuous membrane helices in transport proteins and their correlation with function. J Struct Biol 159:261–267
Segawa H, Kaneko I, Takahashi A, Kuwahata M, Ito M, Ohkido I, Tatsumi S, Miyamoto K (2002) Growth-related renal type II Na/Pi cotransporter. J Biol Chem 277:19665–19672
Segawa H, Kaneko I, Takahashi A, Kuwahata M, Ito M, Ohkido I, Tatsumi S, Miyamoto K (2002) Growth-related renal type II Na/Pi cotransporter. J Biol Chem 277:19665–19672
Segawa H, Kaneko I, Yamanaka S, Ito M, Kuwahata M, Inoue Y, Kato S, Miyamoto K (2004) Intestinal Na-P(i) cotransporter adaptation to dietary P(i) content in vitamin D receptor null mice. Am J Physiol Renal Physiol 287:F39–F47
Segawa H, Kawakami E, Kaneko I, Kuwahata M, Ito M, Kusano K, Saito H, Fukushima N, Miyamoto K (2003) Effect of hydrolysis-resistant FGF23-R179Q on dietary phosphate regulation of the renal type-II Na/Pi transporter. Pflugers Arch 446:585–592
Segawa H, Onitsuka A, Kuwahata M, Hanabusa E, Furutani J, Kaneko I, Tomoe Y, Aranami F, Matsumoto N, Ito M, Matsumoto M, Li M, Amizuka N, Miyamoto K (2009) Type IIc sodium-dependent phosphate transporter regulates calcium metabolism. J Am Soc Nephrol 20:104–113
Segawa H, Yamanaka S, Ito M, Kuwahata M, Shono M, Yamamoto T, Miyamoto K (2005) Internalization of renal type IIc Na-Pi cotransporter in response to a high-phosphate diet. Am J Physiol Renal Physiol 288:F587–F596
Segawa H, Yamanaka S, Onitsuka A, Tomoe Y, Kuwahata M, Ito M, Taketani Y, Miyamoto K (2007) Parathyroid hormone-dependent endocytosis of renal type IIc Na-Pi cotransporter. Am J Physiol Renal Physiol 292:F395–F403
Shenolikar S, Voltz JW, Minkoff CM, Wade JB, Weinman EJ (2002) Targeted disruption of the mouse NHERF-1 gene promotes internalization of proximal tubule sodium-phosphate cotransporter type IIa and renal phosphate wasting. Proc Natl Acad Sci U S A 99:11470–11475
Shojaiefard M, Hosseinzadeh Z, Pakladok T, Bhavsar SK, Lang F (2013) Stimulation of Na+ coupled phosphate transporter NaPiIIa by janus kinase JAK2. Biochem Biophys Res Commun 431:186–191
Stauber A, Radanovic T, Stange G, Murer H, Wagner CA, Biber J (2005) Regulation of intestinal phosphate transport. II. Metabolic acidosis stimulates Na(+)-dependent phosphate absorption and expression of the Na+-Pi cotransporter NaPi-IIb in small intestine. Am J Physiol Gastrointest Liver Physiol 288:G501–G506
Tonelli M (2013) Serum phosphorus in people with chronic kidney disease: you are what you eat. Kidney Int 84:871–873
Tonelli M, Curhan G, Pfeffer M, Sacks F, Thadhani R, Melamed ML, Wiebe N, Muntner P (2009) Relation between alkaline phosphatase, serum phosphate, and all-cause or cardiovascular mortality. Circulation 120:1784–1792
Traebert M, Hattenhauer O, Murer H, Kaissling B, Biber J (1999) Expression of type II Na-Pi cotransporter in alveolar type II cells. Am J Physiol 277:L868–L873
Traebert M, Roth J, Biber J, Murer H, Kaissling B (2000) Internalization of proximal tubular type II Na-Pi cotransporter by PTH: immunogold electron microscopy. Am J Physiol Renal Physiol 278:F148–F154
Villa-Bellosta R, Bogaert YE, Levi M, Sorribas V (2007) Characterization of phosphate transport in rat vascular smooth muscle cells. Implications for vascular calcification. Arterioscler Thromb Vasc Biol 27:1030–1036
Villa-Bellosta R, Sorribas V (2008) Role of rat sodium/phosphate cotransporters in the cell membrane transport of arsenate. Toxicol Appl Pharmacol 232:125–134
Villa-Bellosta R, Sorribas V (2009) Different effects of arsenate and phosphonoformate on P(i) transport adaptation in opossum kidney cells. Am J Physiol Cell Physiol 297:C516–C525
Villa-Bellosta R, Sorribas V (2010) Arsenate transport by sodium/phosphate cotransporter type IIb. Toxicol Appl Pharmacol 247:36–40
Virkki LV, Biber J, Murer H, Forster IC (2007) Phosphate transporters: a tale of two solute carrier families. Am J Physiol Renal Physiol 293:F643–F654
Virkki LV, Forster IC, Hernando N, Biber J, Murer H (2003) Functional characterization of two naturally occurring mutations in the human sodium-phosphate cotransporter type IIa. J Bone Miner Res 18:2135–2141
Virkki LV, Murer H, Forster IC (2006) Mapping conformational changes of the type IIb Na+/Pi cotransporter by voltage clamp fluorometry. J Biol Chem 281:28837–28849
Virkki LV, Murer H, Forster IC (2006) Voltage clamp fluorometric measurements on a type II Na+-coupled Pi cotransporter: shedding light on substrate binding order. J Gen Physiol 127:539–555
Weinman EJ, Biswas RS, Peng G, Shen L, Turner CL, E X, Steplock D, Shenolikar S, Cunningham R (2007) Parathyroid hormone inhibits renal phosphate transport by phosphorylation of serine 77 of sodium-hydrogen exchanger regulatory factor-1. J Clin Invest 117:3412–3420
Weinman EJ, Steplock D, Shenolikar S, Biswas R (2011) Fibroblast growth factor-23-mediated inhibition of renal phosphate transport in mice requires sodium-hydrogen exchanger regulatory factor-1 (NHERF-1) and synergizes with parathyroid hormone. J Biol Chem 286:37216–37221
Weinman EJ, Steplock D, Zhang Y, Biswas R, Bloch RJ, Shenolikar S (2010) Cooperativity between the phosphorylation of Thr95 and Ser77 of NHERF-1 in the hormonal regulation of renal phosphate transport. J Biol Chem 285:25134–25138
Weinstock J (2004) Inhibitors of sodium-dependent phosphate transport. Expert Opin Ther Pat 14:3
Werner A, Kinne RK (2001) Evolution of the Na-Pi cotransport systems. Am J Physiol 280:R301–R312
Xu H, Bai L, Collins JF, Ghishan FK (2002) Age-dependent regulation of rat intestinal type IIb sodium-phosphate cotransporter by 1,25-(OH)2 vitamin D3. Am J Physiol Cell Physiol 282:C487–C493
Xu H, Uno JK, Inouye M, Xu L, Drees JB, Collins JF, Ghishan FK (2003) Regulation of intestinal NaPi-IIb cotransporter gene expression by estrogen. Am J Physiol Gastrointest Liver Physiol 285:G1317–G1324
Yamada F, Horie D, Nakamura A, Tanimura A, Yamamoto H, Segawa H, Ito M, Miyamoto K, Taketani Y, Takeda E (2013) Role of serine 249 of ezrin in the regulation of sodium-dependent phosphate transporter NaPi-IIa activity in renal proximal tubular cells. J Med Invest 60:27–34
Acknowledgments
The work in the laboratories of the authors has been supported by the Swiss National Science foundation with individual grants to C.A. Wagner, N. Hernando, and J. Biber, and to I. Forster. Additional support comes from the National Center for Competence in Research NCCR Kidney. CH is supported by the Swiss National Science Foundation. The authors thank also H. Murer for his on-going interest in and support for all “NaPis.”
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wagner, C.A., Hernando, N., Forster, I.C. et al. The SLC34 family of sodium-dependent phosphate transporters. Pflugers Arch - Eur J Physiol 466, 139–153 (2014). https://doi.org/10.1007/s00424-013-1418-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-013-1418-6