Abstract
Although birds and mammals have evolved from primitive tetrapods and advanced divergently, both can conserve water by producing hyperosmotic urine. Unique aspects in the avian system include the presence of loopless and looped nephrons, lack of the thin ascending limb of Henle’s loop, a corticomedullary osmotic gradient primarily consisting of NaCl without contribution of urea, and significant postrenal modification of final urine. The countercurrent multiplier mechanism operates between the descending and ascending limbs of Henle via recycling of a single solute (NaCl) with no water accompaniment, forming an osmotic gradient along the medullary cone. Bird kidneys and developing rat kidneys share morphological and functional characteristics. Avian kidneys express aquaporin (AQP) 1, 2, and 4 homologues that share considerable homology with mammalian counterparts, but their distribution and function may not be the same. AQP2 expression in Japanese quail (q) evolves in the collecting duct of early metanephric kidneys and continues to increase in intensity and distribution during nephrogenesis and maturation. qAQP2 mRNA and protein are increased by arginine vasotocin (avian ADH), but vasotocin-induced enhancement of cAMP production and water permeability are less marked than in mammalian kidneys. Nephrogenesis is delayed by insufficient nutrition in avian embryos and newborns and results in fewer nephrons and an impaired water balance in adults. Diabetes insipidus quail with homozygous autosomal recessive mutation and an unaffected vasotocin system have low AQP2 expression, underdeveloped medullary cones. Comparative studies will provide important insight into integrative, cellular, and molecular mechanisms of epithelial water transport and its control by humoral, neural, and hemodynamic mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For terrestrial vertebrates, water economy is a prerequisite for survival. Reptiles, birds, and mammals share a common evolutionary origin, but the ancient evolutionary line leading to mammals (Synapsida) departed from the reptilian–avian line (Archosauria) at a very early stage of tetrapod evolution [33, 81]. Although birds and mammals represent parallel lines of divergent evolution, both are able to concentrate urine to produce hyperosmotic urine (the urine to plasma osmolality ratio is over 1.0) and thus conserve water in the body. In birds, body temperature is 40–42°C and extracellular fluid volume is generally higher than in other vertebrate animals. This is considered to be beneficial for activities that demand high energy consumption such as flight [20].
Avian kidneys have several unique morphological and functional features [9, 17, 52, 54]. First, avian kidneys have short loopless (reptilian type) cortical nephrons and longer looped (mammalian type) nephrons that are situated deeper in the medullary zone; the former comprise 70–85% of the entire nephron population. Second, looped nephrons lack the thin ascending limb; thus, the avian kidney has an outer medulla but lacks an inner medulla (Fig. 1). In general, the corticomedullary osmotic gradient and urine concentration ability are lower in avian kidneys than in mammalian kidneys [16]. Third, the corticomedullary osmotic gradient in birds is primarily composed of NaCl with no complement by a nitrogenous compound such as urea. Uric acid, the major nitrogen metabolite in birds, has low solubility and thus does not contribute to the formation of the osmotic gradient along the renal medulla.
A proposed model for urine concentration in the mammalian-type nephron of Coturnix quail, showing transport properties of NaCl and water in various nephron segments. Left NaCl is actively extruded from the thick ascending limb (TAL) and enters the descending limbs (DLs) by simple diffusion without the osmotic accompaniment of water; the NaCl is recirculated for maximum operation of a single-effect countercurrent multiplier system. Active NaCl transport in the TAL appears to be enhanced by perfusion flow rate or luminal NaCl concentration. Immunohistochemical and molecular studies indicate that aquaporin 2 (AQP2) is present in the collecting duct (CD), regulating water transport across the CDs. Right hypothetical cascade transport of NaCl in medullary cone, showing that more NaCl extrusion occurs near the tip of the cone, which may help to enhance an osmotic gradient. PT proximal tubule AVT arginine vasotocin. Reproduced with modification from Nishimura et al. [58] and Osono and Nishimura [62]
In birds, as in mammals, an aquaporin-2 (AQP2) water channel is present at the apical membrane of the cortical and medullary collecting duct (CD) [55, 78]. Synthesis, secretion, and activity of avian AQP2 appear to be regulated by arginine vasotocin (AVT), an avian antidiuretic hormone (ADH) [21, 78]. This brief review summarizes the structural and functional basis of urine concentration and the role of the AQP water channels in birds. Birds live in a variety of habitats and the role of the kidney in the maintenance of body fluid homeostasis varies in accordance with adaptation to environment. Moreover, osmoregulatory organs other than the kidney (such as the gastrointestinal tract, cloaca, and nasal gland) that complement or interact with the kidneys play an important role in ion and water balance in birds [8, 9].
Functional morphology of avian kidney and urine concentration
Architectural organization of the medullary cone
Avian kidneys are composed of numerous medullary cones comprising grossly separated cortical and medullary regions [11, 16]. The cortical reptilian-type nephrons consist of proximal tubule, intermediate segment, distal tubule, and collecting tubule and CD [9, 13]. Since they lack the loop structure (loopless nephron) and their collecting tubules open to the CD at a right angle, reptilian-type nephrons do not directly contribute to urine concentration [9, 16]. The early distal tubule of loopless nephrons reveals a lumen-positive transepithelial voltage (inhibitable by ouabain or Na cyanide), whereas the late distal segment shows a deep lumen-negative potential, suggesting that the early distal segment may function as a diluting segment [57]. Proximal tubules absorb Na, Cl, and water isosmotically [42].
In looped nephrons of avian kidneys, the straight segment of the proximal tubule runs into the narrower descending limb (DL) rather abruptly. This transition from DL to thick limb occurs prior to the hairpin turn; thus, avian looped nephrons, differing from the long-looped nephrons of mammalian kidneys, lack the thin ascending limb (thin AL); and, accordingly, bird kidneys lack the inner medulla [32] (Fig. 1). The thick ascending limb moves into the distal tubule, collecting tubules, and CDs; the CDs run parallel to Henle’s loop, indicating that looped nephrons have a structural basis for producing concentrated urine by a countercurrent multiplier mechanism [12]. There is no clear positive correlation, however, between morphometrical indices of the medulla and urine-concentrating ability [22]. Furthermore, the hairpin turns occur in a graded fashion at various levels of the medullary cone (Fig. 1) [13, 58].
Descending and ascending limbs of Henle
The DL of avian looped nephrons shows morphometrical and functional heterogeneity of epithelial cells, and three types of cells (A, B, and C) have been identified [12, 58]. Type A cells, located near the proximal tubule-DL junction, are relatively tall and have numerous apical microvilli, a moderate number of mitochondria, and lateral interdigitation with neighboring cells, forming a labyrinthine structure; whereas type B and C cells are more flat [58]. The upper segment of the DL (type A cells) has no net volume flux, has low diffusional and osmotic water permeability, and has zero transepithelial voltage [58]. Type A cells have high and nearly equal permeability to Na and Cl ions, suggesting that NaCl is likely to enter the DL unaccompanied by water (Table 1).
Mammalian kidneys show structural and functional heterogeneity of thin limbs within nephrons, between short-looped and long-looped nephrons, and among species [4, 38; Table 1]. The epithelia of thin limbs are classified into four types (types I–IV) based on cell thickness, abundance of luminal microvilli, mitochondria and interdigitation, and the depth of tight junctions [38]. Although the heterogeneity of thin limbs appears less complex in avian nephrons, the upper part of the DL of avian looped nephrons and the type II segment of mammalian long-looped nephrons share some common features. This similarity suggests that avian looped nephrons may evolutionarily correspond to the outer medullary part of mammalian long-looped nephrons rather than to the cortical short-looped nephrons. The major difference in transport properties, however, is that in the upper part of the DL (the lower part has not been examined) of avian looped nephrons, water permeability is low and permeabilities to Na+ and Cl− are high and nearly equal; whereas very high water permeability (both upper and lower part) and cation selectivity (upper part) are present in the DL of mammalian long-looped nephrons (Table 1). These transport characteristics appear important for the difference between countercurrent urine-concentrating mechanisms in avian and mammalian kidneys.
Thick limbs and collecting ducts
In looped nephrons of bird kidneys, the thick AL shows characteristics of diluting segments, including (1) lumen-positive transepithelial voltage, (2) furosemide- or bumetanide-inhibitable Na+–2Cl−K+ cotransport (stoichiometry of membrane transporters has not been examined) and an ouabain-sensitive basolateral Na+–K+ pump, and (3) low volume flux and osmotic permeability [46, 54, 57]. In the isolated perfused thick AL of the quail kidney, the lumen-to-bath Na+ and Cl− flux increased proportionately to perfusion flow rate and NaCl concentration, suggesting that increased luminal loading of NaCl evoked by increased volume flow or NaCl concentration facilitates NaCl extrusion (reabsorption), which may help enhance the imposition of the medullary osmotic gradient [62].
The CD cells in the cortical zone contain dark electron-dense cells that have apical microvilli and vesicles, resembling intercalated cells, and light mucus-secreting cells that have large vacuoles, particularly in the apical side of the nucleus [50, 59]. Abundant interdigitation and tight deep junctions are seen between mucus-secreting cells and also between intercalated cells and mucus-secreting cells. Regulation of avian kidney function by AVT and biological evidence for AVT receptors on preglomerular vasculature and renal tubules have been established in birds (for review, see [21]). In isolated perfused medullary CDs of Coturnix quail kidneys, the volume flux is nearly zero [59]. When medullary CDs are acutely treated with AVT, the volume flux increases only in water-deprived quail. Furthermore, the basal diffusional water permeability (tritiated water flux coefficient) in medullary CDs is considerable, whereas the diffusional water permeability is increased slightly by AVT and only modestly by forskolin, which stimulates adenylate cyclase, whereas a forskolin analogue that does not stimulate adenylate cyclase shows no effect [59]. Thus AVT’s direct action on water transport in CDs appears less marked in avian kidneys [23, 59] than in mammalian kidneys [18, 35]. In medullary cones, cAMP levels are also increased only slightly by AVT but markedly by forskolin, suggesting that the relatively low response to AVT is not due to insufficient adenylate cyclase activity [59]. cAMP response to AVT also varies among species and ages [23]. An AVT-sensitive AQP water channel is present in CDs of medullary cones (see below). It is thus possible that the number/affinity of AVT receptors or cellular signal pathway distal to cAMP may not have fully developed in birds.
Countercurrent multiplier mechanism for urine
There are three basic requirements for the DL and thick AL of the loop of Henle to act as a single-effect countercurrent multiplier: (1) a source of energy to create a transverse difference in osmotic pressure, (2) differences in epithelial permeability, and (3) countercurrent flow. First, the transport properties of the thick AL of Coturnix quail agree with a luminal Na+-dependent Cl− transport driven by a basolateral Na+–K+–ATPase-operated pump without water accompaniment [46, 57]. The thick AL is thus able to provide an energy source. Second, the epithelial permeabilities of the DL and thick AL differ [58]. Third, the architectural organization of the medullary cone indicates that there is a counterflow between the descending and ascending limbs of Henle’s loop. Thus, the Coturnix kidney satisfies the basic requirements mentioned above [52, 58]. The DL is highly and nearly equally permeable to Na and Cl with no active transport, whereas the osmotic and diffusional permeabilities to water are low [58]; AQP1 is not expressed in DLs from Coturnix quail [79 and an unpublished manuscript]. Therefore, tubular urine may be concentrated by the addition of NaCl while it passes through the DL. In contrast, urine dilution occurs by active NaCl absorption in the thick AL. Net NaCl flux in the thick AL is further facilitated by increased luminal NaCl delivery evoked by increased flow rate or NaCl concentration [62]. Therefore, in avian kidneys, urine concentration and dilution occur by a combination of passive diffusion and active transport and the recycling of a single solute (NaCl). A mathematical model of the urine-concentrating mechanism for avian kidneys supports this concept [41]. This single effect may be multiplied by counterflow and the graded elongation of hairpin structures, forming an osmotic gradient along the medullary cones (Fig. 1). The low water permeability of the DL helps to maintain the bulk of volume flow from the proximal tubules, which in turn facilitates NaCl transport from the luminal to the interstitial side via the perfusion flow-dependent mechanism [62]. In rodent kidneys, while tubular urine descends along the DL of Henle, equilibration between tubular fluid and the interstitium can be more efficiently accomplished by subtraction of water [27–29]; this mechanism is apparently lacking in quail kidneys. It will be interesting to investigate whether DLs from avian species that show higher urine-concentrating capacity express AQP1 and are water permeable.
In mammalian kidneys, urea and a unique urea recirculation system significantly contribute to urine concentration (for review, see [5]). Furthermore, in rodent kidneys, the DL of Henle from the short-looped nephron runs in parallel to the ascending vasa recta in the vascular bundle [4, 38]. High urea permeability in DLs of short-looped nephrons appears important for recapturing urea escaped from the inner medulla via the ascending vasa recta and delivering it to distal nephrons [4, 27]. Urea is subsequently recycled to the inner medulla via urea-permeable CD segments. High osmolarity of the inner medulla/papilla is therefore maintained in a “sealed” compartment [4]. Avian kidneys lack an efficient vasa recta mechanism to effectively remove the water from the tip of the medullary cone and transport it to the cortex and systemic circulation. These differences may limit the urine-concentrating capacity of avian kidneys. In most birds examined, the maximum osmolal urine to plasma (U/P) ratio is below 2.5 after dehydration [8, 16].
In avian kidneys, the hyposmotic urine that drains from the cortical loopless nephrons to the medullary CDs decreases the final urine concentration. Considering the smaller number of looped nephrons, usually 10–30% [16], and the fact that only a limited number of nephrons in Henle’s loop reach the tip of the medullary cone, the looped nephrons of avian kidneys must work efficiently to double the urine osmolality. Furthermore, in birds, the plasma osmolality increases considerably during water deprivation; the higher plasma osmolality apparently reduces the osmolal U/P ratio. Thus, caution is needed for the use of the osmolal U/P ratio as an index of urine-concentrating ability.
Renal responses to osmotic challenges
Although it is difficult to generalize because of the numerous variations among species, there seem to be two patterns of avian renal response to osmotic challenges [17]: (1) an acute reduction in glomerular filtration rate (GFR) and urine flow to retain water and prevent an increase in plasma osmolality, a response pattern seen in desert quail; and (2) excretion of excess salt at the expense of water, as seen in starlings. Similarly, excess water may be excreted at the expense of salt [17, 70], or it may perhaps be retained in the body to prevent acute salt loss [10]. During dehydration or salt loading, the inulin U/P ratio or creatinine U/P ratio increases in reptiles and birds, indicating that renal tubules effectively absorb water [11, 70]. In contrast, significant tubular water absorption continues during water loading in desert quail; only 79% of loaded water is excreted, despite the increase in single nephron GFR and the number of filtering nephrons [10]. This may suggest that avian kidneys cannot efficiently turn on and off the control mechanism for renal tubule water permeability, unlike the case in mammalian CDs, in which ADH effectively serves this function via AQP2 [34]. On the other hand, birds have, in general, a higher tolerance and adaptability than mammals to a wide range of plasma osmolalities. Furthermore, some avian species, such as domestic fowl, are able to shift tissue fluid to the vascular compartment when their blood volume/blood pressure is acutely reduced; and blood volume/blood pressure is thus promptly restored [53, 74]. Such a unique compensatory mechanism may help in the maintenance of avian body fluid homeostasis. There is no marked difference in GFR when compared on a weight basis between birds and mammals, but the percentage of filtered water excreted after water deprivation is higher in birds [55]. A larger urine flow may be required for birds to excrete uric acid and other metabolic products. In nonmammalian vertebrates, GFR is controlled intermittently [11, 16]. In this context, caution is needed in interpreting the percentage of filtered water excreted/reabsorbed.
Aquaporin water channels in avian kidneys
AQP and glyceroporin homologues are widely found in plants, microorganisms, invertebrates, and vertebrate animals [2, 73, 75]. Although it is anticipated that all vertebrate kidneys possess AQPs, in nonmammalian vertebrates, AQP isoforms have been cloned from only limited species of fish, amphibians, and birds [15, 37, 55] (Fig. 2). Studies on the expression, function, and regulation of AQPs in animals that live in different salinities and during osmotic challenges will elucidate fundamental mechanisms of AQP water channels in vertebrates.
Evolutionary analysis of aquaporins (AQPs) in vertebrates using the Clustal X program based on similarity scores [72, 77]; the resulting multiple alignments were refined by visual inspection. Neighbor-joining analysis [68] of the amino acid alignment was conducted to reconstruct phylogenetic trees based on mean character distance matrices. The principle of this method is to find pairs of operational taxonomic units (OTUs, neighbors) that minimize the total branch length at each stage of clustering of OTUs, starting with a starlike tree [68]. The outgroup to construct a rooted tree was not applied in our analysis. AQPs and aquaglyceroporins (AQGPs) from plants and invertebrates are not included. AQP and AQGP isoform structures reported in GenBank (reference number in parenthesis) were used for analysis. a Phylogenetic comparison of vertebrate AQPs. AQPs exhibit two major groups and three clades (see text for details). The length of the horizontal connecting bars is inversely proportional to the pair-wise similarity scores calculated from the alignment of sequences with use of the Clustal algorithm for PC/GENE. b The unrooted phylogenetic tree of AQPs in vertebrates. It suggests a divergence (dichotomy) from the ancestor of the AQP superfamily [2, 73]. The colors of the three clades correspond to those of a. AQPs identified in nonmammalian vertebrates are in bold. r rat; m mouse; h human; z zebrafish; k killifish; e eel; x Xenopus; f frog, t toad; q quail; ck chicken; eb Eptatretus burgeri (inshore hagfish); hyla Hyla chrysoscelis (southern gray tree frog). Reproduced with modification from Yang et al. [77] (see also for references). Horizontal bars (length of branch) indicate 0.05 for a and 0.1 for b
AQP2-homologue channel
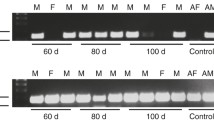
An AQP2-homologue water channel has been identified in the medullary cones of Japanese quail, Coturnix coturnix (qAQP2) by reverse transcription-polymerase chain reaction (RT-PCR)-based cloning techniques [78]. This full-length cDNA contains an 822-bp open reading frame that encodes a 274-amino acid sequence with 75.5% identity to rat AQP2. The qAQP2 has six transmembrane domains, two asparagine-proline-alanine (NPA) sequences, and putative N-glycosylation (asparagine-124) and phosphorylation sites (serine-257) for cAMP-dependent protein kinase [78]. qAQP2 is expressed in the membrane of Xenopus laevis oocytes and significantly increases its osmotic water permeability (P f), inhibitable by mercury chloride. qAQP2 mRNA was detected in the kidney; medullary mRNA levels were higher than cortical levels [40]. A qAQP2 protein that binds to rabbit anti-rat AQP2 antibody is present in the apical/subapical regions of both cortical and medullary CDs from normally hydrated quail, and the intensity of staining increased in the medullary CDs after water deprivation or AVT treatment (Fig. 3) [78]. The relative density of the ∼29-kDa protein band detected by immunoblot, and the in situ hybridization signals of the riboprobe of qAQP2 mRNA [60] increased after water deprivation/AVT treatment, suggesting that an AQP2 mechanism regulated by ADH/AVT via cAMP exists in quail. A partial amino acid sequence of AQP2 has also been identified in chickens and the transcript of chicken AQP2 was detected only in the kidney [64].
Immunohistochemical localization of quail AQP2 (qAQP2) in the kidney. Normally hydrated (a, b, and c), water-deprived (48 h; d), and arginine vasotocin-treated (AVT, 50 ng/kg/day, for 3 days; e) Coturnix quail (20 weeks of age) are shown. Tissues were collected from the cortical (superficial) area (a), midsection (b), and medullary cones (c, d, e, and f). Frozen tissue slices were processed with rabbit anti-rat AQP antisera (1:1,000) (a–e), or normal rabbit serum (f), using an immunoperoxidase-antiperoxidase complex. Arrows in b indicate immunoreactive qAQP2 staining in the apical membrane in normally hydrated birds. The arrow in d indicates AQP2 negative cells (possibly intercalated cells). Horizontal bar 20 μm; G glomerulus; P proximal tubule. Reproduced from Yang et al. [78]
AQP4-homologue channel
Two distinct qAQP4 cDNAs were isolated from quail medullary cones; long (L, open reading frames) and short (S) cDNA encoded 335 (qAQP4-L) and 301 (qAQP4-S) amino acids with, respectively, 80% and 87% identity to human long- and short-form AQP4 [77]. qAQP4-S is identical to qAQP4-L from the second initiation site. Both isoforms have two NPA motifs, but lack cysteine at the known mercury-sensitive site. qAQP4-L and qAQP4-S are expressed in membranes of X. laevis oocytes, but both failed to increase the water permeability (P f) of oocytes exposed to a hypotonic solution. When glutamate (Q242) was replaced with histidine, a known mammalian structure, qAQP4 did not increase P f [77]. With conventional RT-PCR and real-time PCR, qAQP4-L/S mRNA signals were detected in the brain, lung, heart, intestine, adrenal gland, skeletal muscle, liver, and kidney (higher in medulla than in cortical region). qAQP4-L mRNA was detected only in the brain and adrenal gland. Orthogonal arrays of intramembranous particles seen in mammalian AQP4 [26, 69] were not detected in quail CDs, suggesting that although qAQP4-L and qAQP4-S have high homology to mammalian AQP4, their physiological function may be different [77].
Madrid and coworkers [44] identified in epithelial MDCK (Madin–Darby canine kidney) cells two independent C-terminal signals that regulate a sorting of AQP4 molecules from the Golgi apparatus to the basolateral membrane: (1) a signal that encodes a dileucine-like motif preceded by a cluster of acidic residues (ETEDLIL) and (2) a signal consisting of a tyrosine-based motif (GSYMEV). The latter also appears to regulate AQP4 endocytosis and cell surface expression through serial interactions with different clathrin-adaptor protein complexes. Mutation of these motifs significantly impairs basolateral targeting of AQP4 [44]. In contrast, the two motifs in the equivalent positions in qAQP4 are ETDDLIL and GKYIEV, respectively. It will be of interest to investigate whether alteration in these sequences impairs basolateral sorting and expression of qAQP4, leading to the lack of water channel function.
Chicken AQP4 (cAQP4) [67] has 99% amino acid homology with qAQP4 [77]. cAQP4 mRNAs have been detected in the hypothalamus, proventriculus, kidney, muscle, and other organs [64, 67]. It is interesting to note that chicken AQP4 mRNA levels increased in the hypothalamus, whereas they decreased in the kidney, after 2 d water deprivation [67]. Immunoreactive AQP4 has been detected in circumventricular organs of chicken brain [24]. Recent evidence suggests that AQP4 may have a role in cell migration [63]. AQP4 deletion slows migration of astrocytes in cell culture and in vitro wound assays [66]. The evolution of AQP4 appears phylogenetically old (Fig. 2) and an AQP4 homologue has been reported in cyclostomes [51]. Elucidation of the role of AQP4 in birds and other nonmammalian animals may shed light on functions of AQP4 other than the water channel.
AQP1 and other AQP homologues
A 271-amino acid homologue to AQP1 has been isolated from the kidney and lower intestine of the house sparrow, Passer domesticus [14]. Sparrow AQP1 protein is expressed in both the cortex and medulla at the proximal and distal tubules, and in podocytes of the glomerulus. The distribution appears broader than the characteristic area reported for mammalian AQP1 [2, 75]. A full-length cDNA encoding a 270-amino-acid sequence that shares specific structure of AQPs with, respectively, 82%, 77%, and 60% identity to rat AQP1, frog FA-CHIP [1], and eel AQP1 [3] has been cloned from quail kidney [79 and an unpublished manuscript]. Phylogenetic analysis (Clustal X 2.0) of the AQP superfamily revealed that this clone is clustered into the AQP1 subset. The qAQP1 protein shows high water permeability and is selectively inhibitable by HgCl2 in a Xenopus expression system [79]. qAQP1 mRNA was detected (real time-PCR) in the brain, intestine, heart, adrenal glands, liver, and kidney. qAQP1 mRNA levels are, however, rather low in both the cortex and medulla in quail kidneys [79]. Immunoreactive qAQP1 protein is expressed in proximal tubule brush-borders, but not in CDs. In ducks given hypertonic saline for drinking water, nasal gland AQP1 (endothelial cells) and AQP5 (epithelial cells) are downregulated, possibly to enhance water retention while preventing dilution of secreted salt [49]. AQP5 mRNA and protein have been also detected in intestines from chickens [64], although their function remains to be determined. The presence of immunoreactive AQPs 2, 3, and 9 has been reported in gonadal accessory organs in turkeys [82, 83].
AQPs during development and maturation
During vertebrate evolution, three types of kidneys (pronephros, mesonephros, and metanephros) evolved that can serve as developmental models [39]. At embryonic day 5 (E5) of Japanese quail, mesonephric kidneys with larger glomeruli are seen; at E10, both meso- and metanephric kidneys are present. In E15 embryos, mesonephric kidneys are scarce; and after hatch, only metanephric kidneys are seen. In metanephros, glomeruli are larger in deeper regions than in superficial regions and the number of glomeruli per kidney increases exponentially during the later embryonic stage and early postnatal days (∼10 days) [60]. Elongation of medullary cones continues ∼4 weeks. Throughout the surface area of developing metanephric kidneys, condensed areas similar to nephrogenic zone of mammalian kidneys are seen that contain mesenchymal cells; nephrogenic zones decrease with kidney maturation.
qAQP2 mRNA hybridization signals were not detectable in mesonephric kidneys at E5 from Japanese quail, while positive hybridization signals were seen in the CDs/ureteric bud-like structures in the deeper area of metanephric kidneys on E10 [60] (Fig. 4). Expression continued to increase during the rest of the embryonic and postnatal period in quail kidneys until ∼3 weeks of age [60]. During development, rat kidneys express AQP mRNAs 1, 2, 3, and 4 [76]. The mRNA levels of rat AQPs 1, 3, and 4 reached maximum at the time of birth, whereas the level of rat AQP2 mRNA continued to increase postnatally for 10 days [6] and 4 weeks [76]. AQP1 homologue is also expressed at ectodermal and endodermal epithelia and chorioallantoic membrane blood vessels (endothelium and smooth muscle) of chick embryos [65].
qAQP2 mRNA expression detected by in situ hybridization (ISH) of a digoxigenin-labeled riboprobe (qAQP2 cDNA fragment riboprobes of 515 bp). a Kidneys from intact embryonic day 10 (E10) embryos exhibit both mesonephric and metanephric regions. qAQP mRNA signals were detected in some epithelial cells of the collecting ducts (CDs)/ureteric bud branch of deeper regions (arrows) of metanephric kidney. b Medullary cones from E15 embryo (metanephric kidney). ISH signals are more clearly detectable and are mainly localized in the large medullary CD stem and major branches of the CDs. Reproduced from Nishimura et al. [60]
Increasing evidence suggests that the onset of diseases in adulthood may originate in adverse events in fetal life [19, 30]. Reduced nutritional supply and hypoxia in the perinatal period result in smaller birth size and may predispose humans and experimental animals to various health problems after maturation, including hypertension, type 2 diabetes, and cardiovascular disorders [25]. The kidney is a primary organ for regulating blood pressure and blood volume homeostasis and may have an essential role in fetal programming of adult diseases [47]. We investigated whether under-nutrition during the embryonic period (5–10% egg white withdrawn) or 48 h food deprivation in newborn Japanese quail retards kidney and AQP2 development and programs impaired volume regulation in adults [60]. In egg white-withdrawn embryos, qAQP2 mRNA expression is initially delayed, then restored; birth weight and hatching rate are lower than in controls. Adults from egg white-withdrawn embryos and food-deprived chicks have significantly fewer glomeruli (Fig. 5) [60]. Water deprivation reduces body weight more in egg white-withdrawn birds than in controls. The results suggest that qAQP2 evolved in metanephric kidneys and that under-nutrition may retard nephrogenesis, leading to impaired adult water homeostasis.
Effect of under-nutrition on kidney and water balance in quail. a The effects of under-nutrition treatments (5–10% egg white withdrawn, EwW; food deprivation, FD) on the number of glomeruli in superficial (cortical) and deeper (juxtamedullary) regions of 4.5-week-old quail. Food was removed during postnatal days 4–6 for 48 h. a P < 0.01 from superficial region; b P < 0.01 (one-way ANOVA) and c P < 0.05 (Student’s t test) from control. b The effects of under-nutrition treatments on body fluid balance. Birds were exposed to 72 h water deprivation or remained on normal hydration. The loss of body weight was expressed as % decrease from pretreatment body weight. P < 0.01 by Student’s t test. Reproduced from Nishimura et al. [60]
Diabetes insipidus strain of quail
It has been shown that mutation in the AQP2 water channel gene causes autosomal recessive nephrogenic diabetes insipidus (nephrogenic DI) [48]. A Coturnix japonica strain with clinical signs of DI has been developed after screening for polydipsia and polyuria and has revealed homozygous autosomal recessive mutation with an unaffected vasotocin system [45]. To determine the mechanism behind DI manifestation, we examined kidney morphology, molecular expression of AQP2 water channels, and responses to water restriction [80 and an unpublished manuscript]. DI quail kidneys have poorly developed medullary cones with a significantly smaller medullary area per kidney (longitudinal section); the number of glomeruli is similar in intact controls and DI quail. The AQP2 genes and amino acid sequences are nearly identical in the DI and control quail, including two NPA motifs, a mercury-sensitive structure, and a putative phosphorylation site. AQP2 mRNA levels measured by in situ riboprobe hybridization and real-time PCR are low in DI quail. Hematocrit and plasma osmolality are also lower in DI than in control quail; both increased after 24 h water restriction in DI and control groups, but DI quail lost more body weight [80]. These findings suggest that the renal medulla plays a major role in causing DI manifestation. The mechanism of possible blood volume expansion remains to be elucidated.
Phylogeny of aquaporins
Figure 2 shows an unrooted tree for AQP phylogeny prepared with the Clustal X program based on similarity scores [72, 77]; the resulting multiple alignments were refined by visual inspection. Neighbor-joining analysis [68] of the amino acid alignment for reconstructing phylogenetic trees was based on mean character distance matrices. AQPs from plants and bacteria were not included in the analysis. The dendrogram shows that members of the AQP family appear to be divided into two major groups: subcellular AQPs (top part), and membrane-bound AQPs. AQP11 and AQP12 from the first group are localized inside the cell [31], but the functions and biological roles of these cellular AQPs are not well understood. The rest of the dendrogram contains three major clades. The first clade (Fig. 2a, upper area, rose color) includes AQPs 0, 2, 5, and 6 and their isoforms. Within the first clade, the branch leading to AQP2 and AQP5 appears to have evolved later than the branch leading to AQP0 isoforms. The second clade (Fig. 2a, middle area, aquamarine color) shows two clusters, AQP1 and AQP4, although the homology of these AQPs is only 48%. The third clade (Fig. 2a, lower area, blue color) includes aquaglyceroporins that are permeable to both water and glycerol (AQPs 3, 7, 9, and 10).
Judging from the current dendrogram, the evolutionary lines leading to AQP1 and AQP4 appear to have diverged phylogenetically earlier than those leading to AQP2. This agrees with the ontogenic appearance of AQPs, in which AQP1, AQP3, and AQP4 appear to mature earlier than AQP2 [76]. The expression of AQP2 mRNA continues to increase after birth both in rats [76] and quail [60]. AQP water channels are evolutionarily old and are present in plants, invertebrates, and vertebrates [2, 73, 84]. We speculate that AQP1, AQP3, and AQP4 may be fundamental in water handling in the kidney and other water-transport epithelia, whereas AQP2 may have co-evolved with vasotocin’s effect on the collecting ducts. Evolution of AQPs in amphibians and mammals has been reviewed recently in detail by Krane and Goldstein [37]. The phylogenetic tree of the aquaporin superfamily, drawn by the same program, is shown in Fig. 2b.
Evolutionary perspectives
Water handling by the kidney is essential for both aquatic and terrestrial life for maintaining body fluid osmolality and volume homeostasis; but the role of the kidney differs, depending on habitats and the phylogenetic stage of the vertebrates. However, when we compare structures and functions of renal corpuscles and renal tubules among animals that belong to various stages of phylogeny, we find a number of similarities in glomerular filtration and tubular transport properties, such as cellular mechanisms of filtration, active ion pumps, cotransporters, and exchangers. During an early stage of vertebrate phylogeny, the evolution of distal nephrons, particularly, the diluting segment [56, 61, 71], enabled vertebrates to adapt to hyposmotic environments by selectively absorbing ions from tubular urine without the accompaniment of water, and forming dilute urine. In contrast, the same diluting segment plays a major role in countercurrent urine concentration mechanisms, providing an energy source for water conservation, because development of the loop structure with CDs running in parallel provides the structural basis for the countercurrent multiplier mechanism. The factors that make kidney functions diverse are: (1) architectural organization, such as increases in complexity in nephron segments, development of the vasa recta, and the interaction of vasa recta with tubules, such as those in the countercurrent exchange mechanism, thereby increasing the efficiency of ion and water circulation; and (2) intrarenal and systemic regulatory systems involving hemodynamic, humoral, and neural mechanisms. (3) Furthermore, communication between preglomerular/glomerular structures and tubules, such as glomerulo-tubular balance or tubuloglomerular feedback, and the integration of systemic and intrarenal structures by autoregulation, make the entire fluid-ion balance more complex. It is therefore very important to investigate the role of the kidney in body fluid homeostasis from both aspects: elucidation of cellular/molecular mechanisms and integration at the in vivo level as to how the various systems work in concert.
In this context, a comparison of phylogeny and ontogeny may provide useful information. Liu, Kondo, and coworkers [36, 43] compared medullary nephrons from the kidneys of (1) quail (looped nephron) [46, 57–59], (2) neonatal rats (long-looped nephron) [36, 43], and (3) mature adult rats (long-looped nephron) [27–29, 36] and found several structural and functional similarities between the first two (Fig. 6). First, both lack the thin AL and the entire AL shows characteristics of diluting segments. During maturation of rat kidneys, apoptosis occurs in the lower segment of the AL, where the Na+–K+–2Cl− cotransporter is replaced by a thin AL-specific Cl channel. Second, the DLs from quail and neonatal rats show low water permeability; AQP1 expression is absent in DLs from neonatal rat and quail kidneys. In contrast, both show high Na and Cl permeability, suggesting that an increase in the osmotic concentration of tubular urine in the DL of Henle is likely to occur by addition of solute, but not subtraction of water. Third, AQP2 is present in the CDs of both neonatal rat and quail kidneys, but the AQP2 responses to vasopressin (rat)/AVT (quail) are only modest [43, 78]. Fourth, the CDs of neonatal rat (and presumably quail) kidneys lack urea permeability and urea transporters (UT), whereas UTA1 mRNA is expressed in mature rat kidneys [35]. Thus, the contribution of urea to the urine-concentrating mechanism is unlikely in neonatal rats, supporting the fact that urine-concentrating abilities are low in neonatal mammals, including humans. AQP expression and cAMP response to ADH are only modest in immature kidneys [7]. In mature mammalian kidneys, urea recirculation utilizing exchange mechanisms between the vasa recta and tubule segments contributes to the development of an osmotic gradient, and this urea recirculation mechanism may not have developed in immature kidneys.
Comparison of transport properties in avian kidney and neonatal rat kidney. Looped nephron of quail (left), long-looped nephron of neonatal rat kidney (middle), and long-looped nephron of mature rat kidney (right) are shown; the first two show many similarities. See text (“Evolutionary perspective” section) for details. AQP aquaporin; AVT arginine vasotocin; AVP arginine vasopressin. Slightly modified and reproduced with permission from Liu et al. [43]
Zardoya and Villalba [84] conducted comprehensive phylogenetic analysis of AQPs (defined as a family of major intrinsic proteins) that have a water channel function to identify homologous groups (orthologues and paralogues). Water channel proteins are classified into six major paralogous groups in which vertebrate AQPs belong to either group 1, glycerol-transporting proteins, or group 2, AQPs that regulate water transport in specialized tissues. The most basic orthologue in this group is AQP4; and the next gene duplication gave rise to AQP1, which is a highly ubiquitous water channel protein, and to the ancestor of a group of tissue-specific AQPs that include AQP0, AQP6, AQP2, and AQP5. The latter two appear to be the most recent duplications within the AQP gene family [84]. This evolutionary analysis agrees with the order of appearance of AQPs within the phylogenetic advancement of vertebrates and perhaps during ontogeny [6, 76].
Recent evidence indicates that AQPs show functions other than water channel or glycerol transport, including cell adhesion/migration [26, 63, 66]. Those issues are discussed in detail in other sections of this book. Comparative analysis of AQPs in primitive animals may provide useful information as to the fundamental function. The importance of AQP2 in developing kidneys to fluid homeostasis needs to be pursued. Fetal programming of adult disease is an important human health issue in which multiple inducing factors and mechanisms are involved. Utilizing avian eggs, we can study the direct effects of environmental factors, such as temperature, oxygen levels, humidity, and nutritional supplies, on embryonic growth. Such studies will provide insight for understanding possible mechanisms of developmental programming in adults.
References
Abrami L, Simon M, Rousselet G, Berthonaud V (1994) Sequence and functional expression of an amphibian water channel, FA-CHIP: a new member of the MIP family. Biochim Biophys Acta 1192:147–151
Agre P (2000) Aquaporin water channels in kidney. J Am Soc Nephrol 11:764–777
Aoki M, Kaneko T, Katoh F, Hasegawa S, Tsutsui N, Aida K (2003) Intestinal water absorption through aquaporin 1 expressed in the apical membrane of mucosal epithelial cells in seawater-adapted Japanese eel. J Exp Biol 206:3495–3505
Bankir L, de Rouffignac C (1985) Urinary concentrating ability: insights from comparative anatomy. Am J Physiol 249:R643–R666
Bankir L, Trinh-Trang-Tan MM (2000) Renal urea transporters. Direct and indirect regulation by vasopressin. Exp Physiol 85S:243S–252S
Baum MA, Ruddy MK, Hosselet CA, Harris HW (1998) The perinatal expression of aquaporin-2 and aquaporin-3 in developing kidney. Pediatr Res 43:783–790
Bonilla-Felix M (2004) Development of water transport in the collecting duct. Am J Physiol 287:F1093–F1101
Braun EJ (1980) Renal osmoregulation. In: Epple A, Stetson MH (eds) Avian endocrinology. Academic, New York
Braun EJ (1993) Renal function in birds. In: Brown JA, Balment RJ, Rankin JC (eds) New insights in vertebrate kidney function. Cambridge University Press, Cambridge
Braun EJ, Dantzler WH (1975) Effects of water load on renal glomerular and tubular function in desert quail. Am J Physiol 229:222–228
Braun EJ, Dantzler WH (1997) Vertebrate renal system. In: Dantzler WH (ed) Handbook of physiology, section 13: comparative physiology, vol 1. Oxford University Press, New York
Braun EJ, Reimer PR (1988) Structure of avian loop of Henle as related to countercurrent multiplier system. Am J Physiol 255:F500–F512
Casotti G, Lindberg KK, Braun EJ (2000) Functional morphology of the avian medullary cone. Am J Physiol 279:R1722–R1730
Casotti G, Waldron T, Misquith G, Powers D, Slusher L (2007) Expression and localization of an aquaporin-1 homologue in the avian kidney and lower intestinal tract. Comp Biochem Physiol, Part A: Mol Integr Physiol 147(2):355–362
Cutler CP, Cramb G (2000) Water transport and aquaporin expression in fish. In: Hohmann S, Nielsen S (eds) Molecular biology and physiology of water and solute transport. Academic, New York, pp 433–441
Dantzler WH (1989) Comparative physiology of the vertebrate kidney. In: Farner DS (ed) Zoophysiology, vol 22. Springer, Berlin
Dantzler WH, Braun EJ (1980) Comparative nephron function in reptiles, birds, and mammals. Am J Physiol 239:R197–R213
de Rouffignac C, Roinel N, Elalouf JM (1993) Comparative effects of peptide hormones on water and electrolyte transport along the proximal and distal tubules of the mammalian nephron. In: Brown JA, Balment RJ, Rankin JC (eds) New insights in vertebrate kidney function, Soc Exp Biol Seminar Series 52. Cambridge University Press, Cambridge
Fowden AL, Giussani DA, Forhead AJ (2006) Intrauterine programming of physiological systems: causes and consequences. Physiology 21:29–37
Fujimori M (2000) Comparative physiology of body fluid homeostasis. In: Fujimori M, Imai M (eds) Handbook of physiological sciences: physiology of kidney and body fluid regulation, vol 19. Igakushoin, Tokyo, pp 1–28
Goldstein DL (2006) Regulation of the avian kidney by arginine vasotocin. Gen Comp Endocrinol 147:78–84
Goldstein DL, Braun EJ (1989) Structure and concentrating ability in the avian kidney. Am J Physiol 256:R501–R509
Goldstein DL, Reddy V, Plaga K (1999) Second messenger production in avian medullary nephron segments in response to peptide hormones. Am J Physiol 276:R847–R854
Goren O, Adorián I, Kálmán M (2006) Heterogeneous occurrence of aquaporin-4 in the ependyma and in the circumventricular organs in rat and chicken. Anat Embryol (Berl) 211:155–172
Harding JE (2001) The nutritional basis of the fetal origins of adult disease. Int J Epidemiol 30:15–23
Hiroaki Y, Tani K, Kamegawa A, Gyobu N, Nishikawa K, Suzuki H, Walz T, Sasaki S, Mitsuoka K, Kimura K, Mizoguchi A, Fujioshi Y (2006) Implications of the aquaporin-4 structure on array formation and cell adhesion. J Mol Biol 355:628–639
Imai M (1984) Functional heterogeneity of the descending limbs of Henle’s loop. II. Interspecies differences among rabbits, rats, and hamsters. Pflügers Arch 402:393–401
Imai M, Hayashi M, Araki M (1984) Functional heterogeneity of the descending limbs of Henle’s loop. I. Internephron heterogeneity in the hamster kidney. Pflügers Arch 402:385–392
Imai M, Taniguchi J, Yoshitomi K (1988) Transition of permeability properties along the descending limb of long-loop nephron. Am J Physiol 254:F323–F328
Ingelfinger JR, Schnaper HW (2005) Renal endowment: developmental origins of adult disease. J Am Soc Nephrol 16:2533–2536
Ishibashi K (2006) Aquaporin subfamily with unusual NPA boxes. Biochim Biophys Acta 1758:989–993
Johnson OW, Mugaas JN (1970) Some histological features of avian kidneys. Am J Anat 127:423–436
King AS, King DZ (1979) Avian morphology: general principles. In: King AS, McLelland J (eds) Form and function in birds, vol 1. Academic, New York
Knepper MA (1997) Molecular physiology of urinary concentrating mechanism: regulation of aquaporin water channels by vasopressin. Am J Physiol 272(41):F3–F12
Kondo Y, Imai M (1987) Effects of glutaraldehyde fixation on renal tubular function. I. Preservation of vasopressin-stimulated water and urea pathways in rat papillary collecting duct. Pflügers Arch 408:479–483
Kondo Y, Morimoto T, Nishio T, Aslanova UF, Nishino M, Farajov EI, Sugawara N, Kumagai N, Ohsaga A, Maruyama Y, Takahashi S (2006) Phylogenetic, ontogenetic, and pathological aspects of the urine-concentrating mechanism. Clin Exp Nephrol 10:165–174
Krane CM, Goldstein DL (2007) Comparative functional analysis of aquaporins/glyceroporins in mammals and anurans. Mamm Genome 18:452–462
Kriz W, Schiller A, Kaissling B, Taugner R (1980) Comparative and functional aspects of thin loop limb ultrastructure. In: Maunsbach AB, Olsen TS, Christensen EI (eds) Functional ultrastructure of the kidney. Academic, London, pp 241–250
Kuure S, Vuolteenaho R, Vainio S (2000) Kidney morphogenesis: cellular and molecular regulation. Mech Dev 92:31–45
Lau K, Yang Y, Cook G, Wyatt R, Nishimura H (2006) Differential control by ADH of looped and loopless nephrons in quail kidneys. FASEB J 20:A1219 (Abstract #754.4)
Layton HE, Davies JM, Casotti G, Braun EJ (2000) Mathematical model of an avian urine concentrating mechanism. Am J Physiol 279:F1139–F1160
Laverty G, Dantzler WH (1982) Micropuncture of superficial nephrons in avian (Sturnus vulgaris) kidney. Am J Physiol 243:F561–F569
Liu W, Morimoto T, Kondo Y, Iinuma K, Uchida S, Imai M (2001) “Avian-type” renal medullary tubule organization causes immaturity of urine-concentrating ability in neonates. Kidney Int 60:680–693
Madrid R, Le Maout S, Barrault MB, Janvier K, Benichou S, Merot J (2001) Polarized trafficking and surface expression of the AQP4 water channel are coordinated by serial and regulated interactions with different clathrin-adaptor complexes. EMBO J 20:7008–7021
Minvielle F, Grossmann R, Gourichon D (2007) Development and performances of a Japanese quail line homozygous for the diabetes insipidus (DI) mutation. Poult Sci 86:249–254
Miwa T, Nishimura H (1986) Diluting segment in avian kidney. II. Water and chloride transport. Am J Physiol 250:R341–R347
Moritz KM, Dodic M, Wintour EM (2003) Kidney development and the fetal programming of adult disease. BioEssays 25:212–220
Mulders SM, Bichet DG, Rijss JPL, Kamsteeg E, Arthus M, Lonergan M, Fujiwara M, Morgan K, Leijendekker R, van der Sluijs P, van Os CH, Deen PM (1998) An aquaporin-2 water channel mutant which causes autosomal dominant nephrogenic diabetes insipidus is retained in the Golgi complex. J Clin Invest 102:57–66
Müller C, Sendler M, Hildebrandt JP (2006) Downregulation of aquaporins 1 and 5 in nasal gland by osmotic stress in ducklings, Anas platyrhynchos: implications for the production of hypertonic fluid. J Exp Biol 209(Pt 20):4067–4076
Nicholson JK, Kendall MD (1983) The fine structure of dark or intercalated cells from the distal and collecting tubules of avian kidneys. J Anat 136:145–156
Nishimoto G, Sasaki G, Yaoita E, Nameta M, Li H, Furuse K, Fujinaka H, Yoshida Y, Yamamoto T (2007) Molecular characterization of water-selective AQP (EbAQP4) in hagfish: insight into ancestral form of AQP4. Am J Physiol 292:R644–R651
Nishimura H (1993) Countercurrent urine concentration in birds. In: Brown JA, Balment RJ, Rankin JC (eds) New insight in vertebrate kidney function. Soc Exper Biol Seminar Series 52. Cambridge University Press, Cambridge
Nishimura H, Bailey JR (1982) Intrarenal renin-angiotensin system in primitive vertebrates. Kid Int 22(Suppl 12):185–192
Nishimura H, Fan Z (2002) Sodium and water transport and urine concentration in avian kidney. In: Hazon N, Flik G (eds) Osmoregulation and drinking in vertebrates. BIOS Scientific Publishers Ltd, Oxford
Nishimura H, Fan Z (2003) Regulation of water movement across vertebrate renal tubules. Comp Biochem Physiol Part A 136:479–498
Nishimura H, Imai M, Ogawa M (1983) Sodium chloride and water transport in the renal distal tubule of the rainbow trout. Am J Physiol 244:F247–F254
Nishimura H, Imai M, Ogawa M (1986) Diluting segment in avian kidney. I. Characterization of transepithelial voltages. Am J Physiol 250:R333–R340
Nishimura H, Koseki C, Imai M, Braun EJ (1989) Sodium chloride and water transport in the thin descending limb of Henle of the quail. Am J Physiol 257(26):F994–F1002
Nishimura H, Koseki C, Patel TB (1996) Water transport in collecting ducts of Japanese quail. Am J Physiol 271(40):R1535–R1543
Nishimura H, Yang Y, Lau K, Kuykindoll RJ, Fan Z, Yamaguchi K, Yamamoto T (2007) Aquaporin-2 water channel in developing quail kidney: possible role in programming adult fluid homeostasis. Am J Physiol 293:R2147–2158
Oberleithner H, Guggino W, Giebisch G (1982) Mechanism of distal tubular chloride transport in Amphiuma kidney. Am J Physiol 242:F331–F339
Osono E, Nishimura H (1994) Control of sodium and chloride transport in the thick ascending limb in the avian nephron. Am J Physiol 267(36):R455–R462
Papadopoulos MC, Saadoun S, Verkman AS (2008) Aquaporins and cell migration. Pflügers Arch-Eur J Physiol DOI 10.1007/s00424-007-0357-5
Ramírez-Lorca R, Muñoz-Cabello A-M, Toledo-Aral JJ, Ilundáin AA, Echevarría M (2006) Aquaporins in chicken: localization of ck-AQP5 along the small and large intestine. Comp Biochem Physiol Part A 143:269–277
Ribatti D, Frigeri A, Nico B, Nicchia GP, De Giorgis M, Roncali L, Svelto M (2002) Aquaporin-1 expression in the chick embryo chorioallantoic membrane. Anat Rec 268:85–89
Saadoun S, Papadopoulos MC, Watanabe H, Yan D, Manley GT, Verkman AS (2005) Involvement of aquaporin-4 in astroglial cell migration and glial scar formation. J Cell Sci 118:5691–5698
Saito N, Ikegami H, Shimada K (2005) Effect of water deprivation on aquaporin 4 (AQP4) mRNA expression in chickens (Gallus domesticus). Mol Brain Res 141:193–197
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Silberstein C, Bouley R, Huang Y, Fang P, Pator-Soler P, Brown D, Van Hoek AN (2004) Membrane organization and function of M1 and M23 isoforms of aquaporin-4 in epithelial cells. Am J Physiol 287:F501–F511
Skadhauge E, Schmidt-Nielsen B (1967) Renal function in domestic fowl. Am J Physiol 212:793–798
Stoner LC (1977) Isolated, perfused amphibian renal tubules: the diluting segment. Am J Physiol 233:F438–F444
Thompson JD, Higgins DG, Gibson TJ (1994) Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Verkman AS, Mitra AK (2000) Structure and function of aquaporin water channels. Am J Physiol 278:F13–F28
Wyse DC, Nickerson M (1971) Studies on hemorrhage hypotension in domestic fowl. Can J Physiol Pharmacol 49:919–926
Yamamoto T, Sasaki S (1998) Aquaporins in the kidney: emerging new aspects. Kidney Int 54:1041–1051
Yamamoto T, Sasaki S, Fushimi K, Ishibashi K, Yaoita E, Kawasaki K, Fujinaka H, Marumo F, Kihara I (1997) Expression of AQP family in rat kidneys during development and maturation. Am J Physiol 272(41):F198–F204
Yang Y, Cui Y, Fan Z, Cook GA, Nishimura H (2007) Two distinct aquaporin-4 cDNAs isolated from medullary cone of quail kidney. Comp Biochem Physiol, Part A 147:84–93
Yang Y, Cui Y, Wang W, Zhang L, Bufford L, Sasaki S, Fan Z, Nishimura H (2004) Molecular and functional characterization of a vasotocin-sensitive aquaporin water channel in quail kidney. Am J Physiol 287:R915–R924
Yang Y, Nishimura H (2007) Molecular and functional characterization of aquaporin 1 in Japanese quail kidneys. FASEB J 20:A905
Yang Y, Minvielle F, Kuykindoll RJ, Gasc J-M, Yamamoto T, Nishimura H (2008) “Diabetes Insipidus” strain quail show poorly developed medullary cones and low aquaporin 2 water channel expression. FASEB J (in press)
Young JZ (1981) The origin and evolution of birds. The life of vertebrates, (3rd edn). Clarendon Press, Oxford, pp 386–401
Zaniboni L, Akuffo V, Bakst MR (2004) Aquaporins are observed in the duct epithelia of the epididymal region of the large white turkey. Poult Sci 83(11):1917–1920
Zaniboni L, Bakst MR (2004) Localization of aquaporins in the sperm storage tubules in the turkey oviduct. Poult Sci 83(7):1209–1212
Zardoya R, Villalba S (2001) A phylogenetic framework for the aquaporin family in eukaryotes. J Mol Evol 52:391–404
Acknowledgment
Supported by Japan Society Promotion of Science fellowship grant L-005573, NSF grant IOB0615359, and NIH grant HL52881.
Author information
Authors and Affiliations
Corresponding author
Additional information
Proceedings for 2007 Aquaporin Conference, July 13–16, 2007, Kyoto, Japan.
Rights and permissions
About this article
Cite this article
Nishimura, H. Urine concentration and avian aquaporin water channels. Pflugers Arch - Eur J Physiol 456, 755–768 (2008). https://doi.org/10.1007/s00424-008-0469-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-008-0469-6