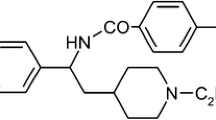
Abstract
The role of the slow delayed rectifier K+ current (I Ks) in cardiac repolarization seems to be largely influenced by the experimental conditions including the species and tissue studied. The aim of this study was to determine the contribution of I Ks to repolarization in canine ventricular myocytes by measuring the frequency dependent action potential lengthening effect of 10 μM chromanol 293B using sharp microelectrodes. Pretreatment with isoproterenol (2 nM), E-4031 (1 μM), and injection of inward current pulses were applied to modify action potential configuration. Chromanol alone caused moderate but statistically significant lengthening of action potentials at cycle lengths longer than 500 ms. The lengthening effect of chromanol, which was strongly enhanced in the presence of either isoproterenol or E-4031, was proportional to the amplitude of plateau, whereas poor correlation was found with action potential duration. Similar results were obtained when action potential configuration was modified by injection of depolarizing current pulses. Computer simulations revealed that activation of I Ks is a sharp function of the plateau amplitude within the physiological range, while elongation of repolarization may enhance I Ks only when it is excessive. It was concluded that the effect of I Ks on ventricular repolarization critically depends on the level of action potential plateau; however, other factors, like action potential duration, cycle length, or suppression of other K+ currents can also influence its contribution.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The delayed rectifier K+ current is composed of two components in most mammalian species including dog and human [7, 13, 20]. The rapid component (I Kr) is considered to be a major repolarizing current responsible for governing terminal repolarization of the action potential and is a classical target for class 3 antiarrhytmics. The role of the slow component (I Ks) in cardiac repolarization is largely influenced by the experimental conditions including the species and tissue studied. One may find data supporting the hypothesis that I Ks plays a crucial role in cardiac repolarization [3, 14, 15]. Others found little, if any, contribution of I Ks to normal repolarization [12, 23]. In the majority of the studies devoted to examine the role of I Ks in repolarization, I Ks was suppressed with specific inhibitors and the importance of I Ks was assessed from the blocker-induced prolongation of the action potential. In some of these studies, I Ks blockade was reported to induce significant prolongation of the action potential [3, 6, 14, 15], while in other reports the lengthening was negligible under baseline conditions [12, 23]. These discrepancies were initially attributed to interspecies differences, but the experimental conditions also seem to be important. Varro et al. [23] suggested that contribution of I Ks to repolarization in canine ventricular myocytes and Purkinje fibers increases with increasing action potential duration. Others found that the importance of I Ks increased after beta-adrenergic stimulation in canine [25] and human [10] ventricular myocardium; however, significant lengthening of action potential could be induced by inhibition of I Ks in anesthetized dogs even after blocking of beta-adrenoceptors [15].
The goal of the present study was twofold. First, we aimed to determine the influence of action potential duration and plateau height on activation of I Ks, which was monitored by measuring the action potential lengthening effect of the selective I Ks blocker drug, chromanol 293B (10 μM) under various experimental conditions. Secondly, the lengthening effect of chromanol was studied in the presence of low concentration (2 nM) of isoproterenol and 1 μM E-4031. Isoproterenol was used to mimic the effect of increased sympathetic tone, while E-4031 was applied to mimic the compromised repolarization reserve capacity [18]. Finally, computer simulations were performed to visualize the influence of action potential duration and plateau height on the profile of I Ks.
Materials and methods
Isolation of single canine ventricular myocytes
Adult mongrel dogs of either sex were anesthetized with intravenous injections of 10 mg/kg ketamine hydrochloride (Calypsolvet, SelBruHa Kft., Hungary) + 1 mg/kg xylazine hydrochloride (Rometar, Alfasan, The Netherlands). The hearts were quickly removed in deep anesthesia and placed in Tyrode solution. Single myocytes were obtained by enzymatic dispersion using the segment perfusion technique [1]. Briefly, a wedge-shaped section of the ventricular wall supplied by the left anterior descending coronary artery was dissected, cannulated, and perfused with oxygenated Tyrode solution containing: NaCl 144, KCl 5.6, CaCl2 2.5, MgCl2 1.2, HEPES 5, and dextrose 11 mM at pH 7.4. Perfusion was maintained until the removal of blood from the coronary system and then switched to a nominally Ca2+-free Joklik solution (Minimum Essential Medium Eagle, Joklik Modification, Sigma) for 5 min. This was followed by 30 min perfusion with recirculated Joklik solution supplemented with 1 mg/ml collagenase (Type II. Worthington, Chemical) and 0.2% bovine serum albumine (Fraction V., Sigma) containing 50 μM Ca2+. Portions of the left ventricular wall were cut into small pieces and the cell suspension obtained from the midmyocardial region was washed with Joklik solution and the Ca2+ concentration was gradually increased to 2.5 mM. The cells were stored in Minimum Essential Medium Eagle supplemented with taurine (20 mM), pyruvic acid (2 mM), ribose (5 mM), allopurinol (0.1 mM), NaHCO3 (26 mM), and NaH2PO4 (1.5 mM) until use. The cell yield (ratio of the rectangular cells showing clear cross striation) varied typically from 40 to 70%.
Recording of action potentials from single canine myocytes
The viable cells were sedimented in a plexiglass chamber allowing continuous superfusion with oxygenated Tyrode solution having pH adjusted to 7.4±0.05. Transmembrane potentials were recorded at 37°C using sharp glass microelectrodes filled with 3 M KCl and having tip resistance between 20 and 40 MΩ. These electrodes were connected to the input of an Axoclamp-2B amplifier (Axon Instruments). The cells were paced through the recording electrode at steady cycle length of 1 s using 1 ms wide rectangular current pulses with 120% threshold amplitude. Because the cytosol was not dialyzed, time-dependent changes in action potential duration were negligible under these experimental conditions (Fig. 1).
Stability of action potential duration (measured at 50 and 95% level of repolarization: APD50 and APD95, respectively) when recorded at 1 Hz from isolated canine ventricular cells (n=9) using sharp microelectrodes. The 60-min period of recording, shown in panel a, started after 5 min after the impalement. Superimposed action potentials recorded from the same cell at the beginning (a) and the end (b) of the recording period are presented in panel b. Symbols and bars represent mean±SEM values
When performing frequency-dependent measurements, the cycle length was set to 5 s, and after equilibration, at least for 5 min, the cycle length was continuously varied to the shorter values. In some experiments, inward current pulses, having properly designed parameters, were delivered into the cell through the recording electrode. Action potentials were digitized at 100 kHz using Digidata 1200 A/D card (Axon Instruments) and stored for later analysis. Duration of the action potential was measured at 50 and 95% levels of repolarization (APD50 and APD95, respectively), while the amplitude of the plateau was measured from the resting potential to the plateau level determined at 20% duration of action potential. This value was used for characterization of the plateau height because it was not distorted by the voltage shift often observed when using high resistance sharp microelectrodes.
The myocytes were superfused with chromanol, E-4031 (both from Tocris Bioscience, Bristol, UK), and isoproterenol (Sigma-Aldrich, St. Louis, MI, USA) at least for 5 min, allowing full development of drug effects. Chromanol was dissolved in dimethylsulfoxide (DMSO) (10 mM stock solution) and was diluted to the final concentration of 10 μM with Tyrode solution. E-4031 was dissolved in distilled water to produce 1 mM stock solution. Isoproterenol was freshly dissolved before the experiment.
Computer simulations
We used computer simulation to investigate the effects of plateau height and action potential duration on the kinetic properties of I Ks. The numerical model, describing the voltage and time dependence of I Ks, published by Viswanathan et al. [24], was modified and used to predict the profile of the current during the action potential. Action potentials, recorded previously from canine ventricular myocytes predominantly of midmyocardial origin, were used to drive the model to compute the profile of I Ks. To test the effect of the altered action potential duration or plateau height, we developed a computer program, which could set the plateau height or action potential duration of the recorded action potential to the required values. Thus, a series of action potentials having differences only in the studied parameter were generated from a single action potential.
Mathcad 2000i (MathSoft Engineering & Education, Surrey, UK) was used to run the model. Equations describing the time-dependent behavior were entered to Mathcad without modification. All parameters of the model were obtained from Viswanathan et al. [24] except for G max, which was multiplied with a factor of 0.084. Using this factor the amplitude of I Ks current tails measured earlier in canine ventricular cells matched those obtained with the model. A 10-ms period of lag at the resting potential was applied before the action potential to reach steady state conditions for the current before activation. The integration time step was 1 ms. The model included two gating constants; thus, their product was used in computing the current density. Because the model involves changes of the cytosolic calcium concentration, an artificially constructed calcium transient was inserted into the model. The diastolic and systolic calcium concentrations were set to 200 nM and 2 μM, respectively.
Statistics
All values presented are arithmetic means±SEM. Statistical significance was determined by using Student’s t test for paired and unpaired data as appropriate. Differences were considered significant when the P value was less than 0.05.
The investigation conforms to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996), and the principles outlined in the Declaration of Helsinki. The experimental protocol was also approved by the local ethical committee.
Results
Effect of chromanol on action potential duration
Chromanol caused a frequency dependent lengthening of action potentials without changing their other parameters. APD50 and APD90 were increased by 10 μM chromanol from 152.0±7.4 to 158.0±10.9 ms and from 200.5±8.2 to 209.7±10.8 ms, respectively, at the cycle length of 1 s. This effect, corresponding to 6.0±1.6 and 9.2±3.5 ms lengthening of APD50 and APD90, respectively, was moderate but statistically significant (P<0.05, n=6), and increased with increasing the cycle length (Fig. 2). No significant effect was observed on the resting potential (−81.0±2.8 vs −82.7±2.1 mV), action potential amplitude (99.4±2.9 vs 101.2±3.2 mV), plateau amplitude (88.1±3.3 vs 90.2±3.5 mV), or maximal rate of depolarization (272±23 vs 286±31 V/s) of the myocytes (n=6) measured in the absence and presence of 10 μM chromanol, respectively.
Effect of 10 μM chromanol alone on action potential configuration. a Representative superimposed records obtained at 1 Hz before and 5 min after chromanol treatment. b Effect of chromanol on action potential duration measured at 50 and 95% level of repolarization (APD50 and APD95, respectively) as a function of the pacing cycle length. Symbols and bars represent mean±SEM values, asterisks indicate significant changes (determined using Student’s t test for paired data) induced by chromanol treatment in the six myocytes studied
Effect of chromanol in the presence of isoproterenol and E-4031
The effect of 10 μM chromanol on action potential duration was measured after pretreatment with 2 nM isoproterenol and in the presence of 1 μM E-4031. Isoproterenol increased the amplitude of the plateau with no significant effect on APD95. E-4031 increased both APD95 and the amplitude of plateau. These effects (i.e., elevation of the plateau and lengthening of APD95) were statistically significant at all cycle lengths longer than 0.5 s, and were enhanced at longer cycle lengths (Fig. 3a–c). As shown in Fig. 3d, the chromanol-induced lengthening of APD was significantly augmented by the presence of isoproterenol, as well as by E-4031, in a reverse rate-dependent manner.
a Representative superimposed pairs of action potentials recorded at 1 Hz before and 5 min after superfusion with 2 nM isoproterenol (upper panel) and 1 μM E-4031 (lower panel). Frequency dependent effects of isoproterenol (n=7) and E-4031 (n=6) on the amplitude of plateau (b) and action potential duration (c). Asterisks indicate significant changes from control determined with Student’s t test for paired data. d Chromanol-induced lengthening of action potential duration (APD95) measured in control (n=6), in the presence of isoproterenol (n=7) and E-4031 (n=6) as a function of the pacing cycle length. In this figure, asterisks indicate significant differences comparing to control obtained with Student’s t test for unpaired data
One of the main goals of this study was to identify the determinant parameter(s) of the action potential responsible for activation of I Ks, which was assessed from the magnitude of the chromanol-induced lengthening of APD. Therefore, the lengthening effect of chromanol was correlated with the amplitude of the plateau and with APD95, both measured in the presence of isoproterenol or E-4031 (Fig. 4). The lengthening effect of chromanol showed good correlation with the amplitude of plateau: correlation coefficients of 0.902 and 0.937 were obtained in the presence of isoproterenol and E-4031, respectively, whereas no correlation was found with APD95 in isoproterenol (r=0.119) and E-4031 (r=0.424). These results indicate that the amplitude of the plateau is an important determinant of the activation of I Ks during the action potential independently of the way of plateau elevation.
Correlation of the chromanol-induced lengthening of action potential duration (APD95) with the amplitude of plateau (a, b) and action potential duration (c, d) obtained in the presence of isoproterenol (a, c) or E-4031 (b, d) at various pacing cycle lengths. Data are taken from experiments shown in Fig. 3. Correlation coefficient (r) values were determined using linear regression
Effect of chromanol in myocytes injected with inward current pulses
Because isoproterenol was shown to alter the kinetics of I Ks in canine cardiac preparations [8, 25], a strategy allowing to study the effect of plateau elevation without altering the phosphorylation state of the I Ks channel was the application of rectangular inward current pulses in current clamp mode (Fig. 5). The beginning of these pulses was synchronized to action potential upstroke, their duration was set to 170 ms, and their amplitude was increased up to 500 pA in 100 pA steps. The series of current injection was repeated before and after chromanol treatment in the same cell allowing to asses the lengthening effect of chromanol in a relatively wide range of plateau amplitude and APD95 values. The current pulses increased both the amplitude of plateau and duration of action potentials. These effects were greater in the presence than in the absence of 10 μM chromanol (Fig. 3b,c). Similarly, to results obtained with isoproterenol and E-4031, the chromanol-induced lengthening was highly proportional to the amplitude of plateau (r=0.954, Fig. 5d), while no correlation was found with the duration of the action potential (r=0.401, Fig. 5e). Both parameters were measured from cells injected with inward current of various amplitudes.
Effect of chromanol in myocytes injected with inward current pulses. a Superimposed pairs of action potentials recorded at 1 Hz before (left panel) and 5 min after superfusion with 10 μM chromanol (middle panel). Upper records were taken with injection of rectangular inward current pulses having amplitudes of 200 pA; these records are directly compared in the right panel. b, c Effect of current injection on the amplitude of plateau (b) and action potential duration (c) measured in the presence and absence of 10 μM chromanol. Amplitude of the current was increased in 100 pA steps. Asterisks indicate significant differences between respective data obtained before and after the chromanol treatment, determined with Student’s t test for paired data. Correlation of the chromanol-induced lengthening of action potential duration (APD95) with the amplitude of plateau (d) and action potential duration (e) obtained from myocytes injected with various amplitudes of inward current (n=6). Data are taken from experiments shown in b and c. Correlation coefficient (r) values were determined using linear regression
Simulation of the profile of I Ks during the action potential
Using the modified model of Viswanathan et al. [24] the effect of the plateau height and action potential duration on the density of I Ks was studied. Series of action potentials, generated from a single action potential recorded previously so as they differed from one another only in their plateau height or APD95, were used to drive the model to compute the profile of I Ks. Values of plateau height and APD95 were chosen to cover the range obtained with isoproterenol, E-4031, and current injection. As presented in Fig. 6, the peak amplitude of the computed I Ks was a sharp function of the amplitude of plateau growing up close to fourfold when the plateau height was increased from 72 to 107 mV (Fig. 6e). I Ks was proportional also to APD95; however, doubling APD95 (changing it from 180 to 350 ms) increased the maximum of I Ks by less than 50% (Fig. 6f).
Effect of the plateau height (a, c, e) and action potential duration (b, d, f) on the profile of slow delayed rectifier K+ current (I Ks) calculated using the modified Viswanathan model. Series of action potentials, differing from one another only in the plateau height (a) or APD95 (b) were used to drive the models to compute the corresponding I Ks profiles (c, d). Normal action potentials, recorded at 1 Hz in Tyrode solution, with the respective I Ks current traces are shown bold. Maximum values of the computed I Ks were plotted as a function of the plateau amplitude (e) or APD95 (f) of the input action potential
Discussion
Contribution of I Ks to repolarization
Based on the results obtained from studying the APD lengthening effect of chromanol in canine ventricular myocytes two important statements can be made. Firstly, we have demonstrated that I Ks does contribute to normal ventricular repolarization because chromanol caused a moderate but statistically significant lengthening of APD in this species at cycle lengths longer than 0.5 s. Secondly, we have shown that contribution of I Ks to repolarization is primarily determined by the plateau height. The augmentation of the chromanol induced lengthening of APD observed in the presence of isoproterenol or E-4031 confirms the results of previous studies [10, 25].
Regarding the first statement, it must be noted that those investigators who concluded that I Ks failed to contribute to repolarization in canine myocytes under baseline conditions have also observed some APD lengthening with chromanol, as well as with other selective I Ks blockers, like HMR 1556, or L-735,821 [12, 23, 25]. In our experiments, however, these differences were statistically significant, although the magnitude of lengthening was moderate. On the other hand, the lengthening effect of chromanol was dramatically increased by all interventions resulting in the elevation of action potential plateau (pretreatment with isoproterenol, E-4031, or application of inward current). These results were corroborated by computer simulations indicating that elevation of the plateau amplitude within the range of 72–107 mV (corresponding to −8 to +27 mV of plateau potential considering an approximately −80 mV resting potential) caused an almost fourfold increase in the maximal amplitude of I Ks. As indicated in Fig. 6c, the sharp increase in I Ks amplitude observed in response to plateau elevation is clearly due to the increased voltage-dependent activation of I Ks and may account for the apparent discrepancies in the literature. For instance, in guinea pig ventricular cells the plateau potential is well above +30 mV, in contrast with canine Purkinje fibers having their plateau potentials in the negative voltage range. Accordingly, prominent APD lengthening was observed in the former preparation [3], while practically none in the latter one [23] in response to I Ks blockade. In our experiments, the plateau potential was around +10 mV, which is congruent with a moderate effect of chromanol at baseline. Nakashima et al. [15] came to the same conclusion reporting delayed ventricular repolarization in response to inhibition of I Ks in the presence of beta-adrenoceptor blockade.
In contrast to the good correlation found between the chromanol-induced APD lengthening and the plateau height (see the correlation coefficients of 0.902, 0.937, and 0.954, obtained in the presence of isoproterenol, E-4031, and with current injection, respectively), virtually no correlation was observed between the lengthening effect of chromanol and APD95 (the respective correlation coefficients were 0.119, 0.424, and 0.401). This seems to be in conflict with the linear relationship demonstrated by the simulation study in Fig. 6f. The contradiction can be resolved considering that in the presence of E-4031, and also in the case of current injection, both APD95 and the plateau amplitude were simultaneously increased. The low, but nonzero, values of the correlation coefficients obtained for APD95 can be explained by the higher steepness of the plateau height-lengthening relationship than of the APD95-lengthening relationship, presented in Fig. 6e,f, respectively. Varro et al. [23] observed strong enhancement of the chromanol-induced APD lengthening in canine ventricular myocardium, when the action potential was prolonged by E-4031 or veratrine, suggesting that APD is an important determinant of the chromanol-induced lengthening. Because both drugs are known to increase the plateau height substantially, contribution of this latter effect to the observed lengthening cannot be ruled out. It must be mentioned, however, that the largest chromanol-induced lengthening in our experiments was obtained in the presence of E-4031—in spite of the less pronounced elevation of the plateau induced by E-4031 comparing to isoproterenol. This result could have been interpreted as an evidence supporting the role of action potential duration. Considering the flat APD95-lengthening relationship, it seems to be more likely that suppression of I Kr increased the relative contribution of I Ks to repolarization, and this might cause the increased lengthening effect of chromanol. The same argumentation may be used to explain the reverse rate dependent nature of the chromanol-induced lengthening shown in Fig. 2. I Kr is much stronger at shorter than at longer cycle lengths due to its frequency dependent accumulation [17] diminishing the relative significance of I Ks at fast heart rates. Such accumulation of I Ks was not observed in canine myocytes at cycle length longer than 500 ms [22]. Based on the speculation above one may predict an enhanced lengthening effect of chromanol under all circumstances when the repolarization reserve is compromised [2].
Because configuration of the cardiac action potential critically depends on the fine balance between several inward and outward currents, all manipulations leading to elevation of the plateau will necessarily modify all the ion currents contributing to ventricular repolarization. For instance, in addition to enhancement of the two delayed rectifier K+ currents (I Kr and I Ks) the driving force for Ca2+ entry through the L-type Ca2+ channel will also be diminished by elevation of the plateau potential resulting in a smaller density of inward current, with the concomitant outward shift in the net membrane current, which, in turn, will increase the repolarization reserve. The opposite can be anticipated in the presence of isoproterenol, which is known to increase Ca2+ current several fold. Of course, modification of the other currents has also to be considered, which may distort the evaluation of the role of I Ks in repolarization.
Clinical implications
The original concept for development of I Ks blocker drugs, such as chromanol, as potential antiarrhythmic agents was based on the assumption that they would be less proarrhytmic comparing to I Kr blockers because their effect was anticipated to be less affected by the disadvantageous reverse rate dependent mode of action, a common characteristic of pure I Kr blockers [9, 11]. Several studies—including this one—suggest, however, that this is not fully the case [3, 25].
In this study, the lengthening effect of chromanol was tested in two situations occurring most typically under pathological conditions. Low concentration (2 nM) of isoproterenol was used to mimic the effect of strongly increased sympathetic tone, which is associated with several cardiac disorders including heart attack. The results indicate that the APD lengthening effect of chromanol (the desired class 3 antiarrhytmic action) was markedly enhanced in the presence of isoproterenol. This suggests—in spite of the limited contribution of I Ks to normal repolarization—that a substantial APD lengthening effect can be expected in vivo, especially when the sympathetic tone is elevated. Similar conclusions were drawn by Volders et al. [25] and Jost et al. [10] when studying canine and human ventricular myocardium. One might argue that suppression of I Ks in this situation may potentially be beneficial because it may counterbalance the shortening of APD evoked by the sympathetic activation resulting in prolongation of the refractory period. Although I Ks blockade was shown to decrease dispersion of repolarization in failing canine hearts [16], marked heterogeneities in action potential duration with resultant arrhythmias were observed when the I Ks blockade was applied after stimulation of beta-adrenoceptors [21].
The I Kr blocker E-4031 was applied to simulate the compromised repolarization reserve, which can be either inherited or acquired [19]. This latter form, very often caused by noncardiac medication, sometimes remains hidden. In these patients the already compromised repolarization reserve would further be reduced by the I Ks blockade, increasing the risk of torsades de pointes type of arrhythmias [5]. If the I Ks blockade is introduced at low heart rate or increased sympathetic activity combined with partially suppressed I Kr, the risk of fatal outcome may be even higher. These data suggest that I Ks blockade may carry a significant risk of proarrhythmia. In conclusion, the strategy of I Ks blockade as a potential therapeutic intervention seems to be rather harmful than beneficial, however, conclusive clinical investigations are required to clarify the therapeutic value of I Ks blockers [4].
References
Bányász T, Magyar J, Körtvély Á, Szigeti Gy, Szigligeti P, Papp Z, Mohácsi A, Kovács L, Nánási PP (2001) Different effects of endothelin-1 on calcium and potassium currents in canine ventricular cells. Naunyn Schmiedeberg’s Arch Pharmacol 363:383–390
Biliczki P, Virág L, Iost N, Papp JGy, Varró A (2002) Interaction of different potassium channels in cardiac repolarization in dog ventricular preparations: role of repolarization reserve. Br J Pharmacol 137:361–368
Bosch RF, Gaspo R, Busch AE, Lang HJ, Li GR, Nattel S (1998) Effects of the chromanol 293B, a selective blocker of the slow, component of the delayed rectifier K+ current, on repolarization in human and guinea pig ventricular myocytes. Cardiovasc Res 38:441–450
Curtis MJ (2004) Is cardiac IKs a relevant drug target? Cardiovasc Res 61:651–652
El-Sherif N, Turitto G (1999) The long QT syndrome and torsade de pointes. Pacing Clin Electrophysiol 22:91–110
Finley MR, Li Y, Hua F, Lillich J, Mitchell KE, Ganta S, Gilmour RF Jr, Freeman LC (2002) Expression and coassociation of ERG1, KCNQ1, and KCNE1 potassium channel proteins in horse heart. Am J Physiol 283:H126–H138
Gintant GA (1996) Two components of delayed rectifier current in canine atrium and ventricle. Does IKs play a role in the reverse rate dependence of class III agents? Circ Res 78:26–37
Han W, Wang Z, Nattel S (2001) Slow delayed rectifier current and repolarization in canine cardiac Purkinje cells. Am J Physiol 280:H1075–H1080
Hondeghem LM, Snyders DJ (1990) Class III antiarrhythmic agents have a lot of potential but a long way to go. Reduced effectiveness and dangers of reverse use dependence. Circulation 81:686–690
Jost N, Virag L, Bitay M, Takacs J, Lengyel C, Biliczki P, Nagy Z, Bogats G, Lathrop DA, Papp JG, Varro A (2005) Restricting excessive cardiac action potential and QT prolongation: a vital role for IKs in human ventricular muscle. Circulation 112:1392–1399
Jurkiewicz NK, Sanguinetti MC (1993) Rate-dependent prolongation of cardiac action potentials by a methanesulfonanilide class III antiarrhythmic agent. Specific block of rapidly activating delayed rectifier K+ current by dofetilide. Circ Res 72:75–83
Lengyel C, Iost N, Virag L, Varro A, Lathrop DA, Papp JG (2001) Pharmacological block of the slow component of the outward delayed rectifier current (IKs) fails to lengthen rabbit ventricular muscle QTc and action potential duration. Br J Pharmacol 132:101–110
Li GR, Feng J, Yue L, Carrier M, Nattel S (1996) Evidence for two components of delayed rectifier K+ current in human ventricular myocytes. Circ Res 78:689–696
Lu Z, Kamiya K, Opthof T, Yasui K, Kodama I (2001) Density and kinetics of IKr and IKs in guinea pig and rabbit ventricular myocytes explain different efficacy of IKs blockade at high heart rate in guinea pig and rabbit: implications for arrhythmogenesis in humans. Circulation 104:596–951
Nakashima H, Gerlach U, Schmidt D, Nattel S (2004) In vivo electrophysiological effects of a selective slow delayed-rectifier potassium channel blocker in anesthetized dogs: potential insights into class III actions. Cardiovasc Res 61:705–714
Pajouh M, Wilson LD, Poelzing S, Johnson NJ, Rosenbaum DS (2005) IKs blockade reduces dispersion of repolarization in heart failure. Heart Rhythm 2:731–738
Rocchetti M, Besana A, Gurrola GB, Possani LD, Zaza A (2001) Rate dependency of delayed rectifier currents during the guinea-pig ventricular action potential. J Physiol 534:721–732
Roden DM (1998) Taking the “idio” out of “idiosyncratic”: predicting torsades de pointes. Pacing Clin Electrophysiol 21:1029–1034
Roden DM, Viswanathan PC (2005) Genetics of acquired long QT syndrome. J Clin Invest 115:2025–2032
Sanguinetti MC, Jurkiewicz NK (1990) Two components of cardiac delayed rectifier K+ current. Differential sensitivity to block by class III antiarrhythmic agents. J Gen Physiol 96:195–215
Shimizu W, Antzelevitch C (1998) Cellular basis for the ECG features of the LQT1 form of the long-QT syndrome: effects of beta-adrenergic agonists and antagonists and sodium channel blockers on transmural dispersion of repolarization and torsade de pointes. Circulation 98:2314–2322
Stengl M, Volders PGA, Thomsen MB, Spatjens RLHMG, Sipido KR, Vos MA (2003) Accumulation of slowly activating delayed rectifier potassium current (IKs) in canine ventricular myocytes. J Physiol 551:777–786
Varro A, Balati B, Iost N, Takacs J, Virag L, Lathrop DA, Lengyel Cs, Tálosi L, Papp JGy (2000) The role of the delayed rectifier component IKs in dog ventricular muscle and Purkinje fibre repolarization. J Physiol 523:67–81
Viswanathan PC, Shaw RM, Rudy Y (1999) Effects of IKr and IKs heterogeneity on action potential duration and its rate dependence: a simulation study. Circulation 99:2466–2474
Volders PGA, Stengl M, van Opstal JM, Gerlach U, Spatjens RLHMG, Beekman JDM, Sipido KR, Vos MA (2003) Probing the contribution of IKs to canine ventricular repolarization: key role for β-adrenergic receptor stimulation. Circulation 107:2753–2760
Acknowledgements
Financial support for the studies was provided by grants from the Hungarian Research Found (OTKA-T043182), and the National Research and Development Programs (NKFP-1A/0011/2002, NKFP-1A/008/2004). The experiments comply with the current laws of Hungary.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Horváth, B., Magyar, J., Szentandrássy, N. et al. Contribution of I Ks to ventricular repolarization in canine myocytes. Pflugers Arch - Eur J Physiol 452, 698–706 (2006). https://doi.org/10.1007/s00424-006-0077-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-006-0077-2