Abstract
The mechanism of activation and repression of apoptosis has been a central focus of many studies examining the role of programmed cell death in both normal and pathological conditions. Despite intensive research efforts, the precise cellular and molecular mechanisms that trigger and/or prevent apoptosis remain undefined. A universal characteristic of apoptosis is the loss of cell volume or cell shrinkage, recently termed apoptotic volume decrease. While cell shrinkage has traditionally been viewed as a passive event during apoptosis, recent work from several laboratories has shown that the loss of cell volume, or more specifically the flux of ions associated with the change in cell size, play a critical role in the regulation of the cell death machinery. On going studies continue to support the hypothesis that the change in intracellular ions can alter a cells decision to die by apoptosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Apoptosis is a physiological cell death process set apart from other modes of cell death by a distinct set of morphological, biochemical, and genetic characteristics. These characteristics uniquely define apoptosis where a cell can be removed or eliminated from a tissue without disruption of its surrounding environment. Several classical features are used to define apoptosis that include cell shrinkage, nuclear condensation, internucleosomal DNA fragmentation, and eventual blebbing of the cell membrane known as apoptotic body formation. Over the past 10 years we have only begun to understand the far-reaching role this programmed cell death process plays, not only during embryonic development and maintaining adult cellular homeostasis, but also in how apoptosis impinges on human health. Much of our current understanding of apoptosis has come about through numerous comprehensive and elegant studies defining key genes and proteins in the programmed process. However, recently a role for alterations in intracellular ions, particularly intracellular potassium and sodium, has become apparent as a critical component of the cell death process.
The loss of cell volume or cell shrinkage that occurs during apoptosis has been the key morphological characteristic separating this physiological cell death process from an accidental cell death process known as necrosis. In the very first reports of a physiological mode of cell death, the term “shrinking necrosis” was used to highlight the unique property of cell volume loss in these dying cells [18, 19], but the mechanisms controlling this volume loss are not understood. Cells respond to osmotic changes in their extracellular environment through the activation of various cell volume regulatory responses [1, 15]. Briefly, under hypotonic conditions cells swell. However, most cells compensate for this increase in cell size by the activation of regulatory volume decrease (RVD) response. Conversely, under hypertonic conditions cells shrink. Again, most cells compensate for this decrease in cell size by the activation of a regulatory volume increase (RVI) response. The goal of both these responses is to return the cell to a near normal cell size and maintain cellular homeostasis. Interestingly, during apoptosis the loss of cell volume or cell shrinkage occurs in the absence of extracellular osmotic challenge. Therefore, cell shrinkage during apoptosis occurs under normotonic conditions and has been termed apoptotic volume decrease (AVD; [25, 26]). Additionally, it has been suggested that these inherent volume regulatory responses that normally protect the cell from aberrant changes in cell size are either inhibited or overridden during the apoptotic process [4].
Most cells contain all the required components to invoke the programmed cell death process given an appropriate apoptotic stimulus. Therefore a cell must control or regulate the apoptotic machinery to ensure that inappropriate activation of the cell death process does not occur. In contrast upon receiving an apoptotic signal, the cell must quickly activate the cell death program, ensuring that the events leading to cellular destruction are carried out in proper order. Over the past several years, numerous proteins have been reported which impact the cell death program in both a positive and negative way. Proteolytic enzymes such as caspases, pro-apoptotic Bcl-2 proteins, along with the release of cytochrome c from the mitochondria impact the cell death program in a positive way. On the contrary, proteins such as anti-apoptotic Bcl-2 family members and IAPs repress the apoptotic program. We examine here the current evidence that ions also play an important role in both the activation and repressions of apoptosis, and may also impact many of the proteins that are known to regulate the programmed cell death process.
The importance of maintaining ionic homeostasis
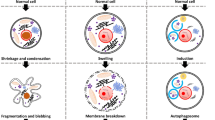
Cells cannot live without an appropriate balance of ions across their cell membrane. Proper functioning of cellular proteins, gene expression, second messenger cascades, and hormone release are all extremely dependent on a homeostatic intracellular ionic environment [22, 23, 40]. Therefore, cells use multiple ion transport mechanisms such as channels, co-transporters, and exchangers to maintain a consistent ionic balance. Of particular importance is the constitutive activity of the Na+/K+-ATPase, which exchanges three intracellular sodium ions for two extracellular potassium ions in an energy-dependent manner. This protein uses substantial amounts of cellular ATP to maintain a homeostatic balance of intracellular sodium and potassium, along with preserving the plasma membrane potential. In fact, the primary consumers of energy in a cell are these homeostatic ionic transport mechanisms. Minor variations in cell volume occur in accordance with the normal cellular activity of the cells. However, as outlined above, inherit volume regulatory mechanisms exist that compensate for these minor variations. As shown in Fig. 1, the problem arises when these compensatory mechanisms are inactivated or overridden resulting in an uncontrollable change in cell volume. Under this condition, the loss of cell volume can lead to the activation of the AVD process resulting in apoptosis. For example, it has been shown in cells that lack an RVI response that hypertonic conditions result in sustained cell shrinkage leading to the activation of the apoptotic process, whereas cells that have an RVI response were resistant to hypertonic-induced cell death [4]. In contrast, excessive cell swelling can lead to necrosis if not compensated by inherent cellular volume regulatory mechanisms [11, 30].
A schematic representation for the role of apoptotic volume decrease (AVD) and ionic homeostatsis in the activation and repression of apoptosis. Apoptotic stimulation results in an early increase in intracellular sodium, plasma membrane depolarization, and AVD leading to cell death. The depolarization of the plasma membrane is denoted by the change in color of the cell membrane. Inherent volume regulatory mechanisms, specifically regulatory volume increase (RVI) can be activated to protect the cell from apoptosis. However, inhibited or overridden RVI can result in AVD resulting in apoptosis. The presence of high extracellular potassium can protect the cell from AVD by maintaining a homeostatic balance of ions along with holding in check the apoptotic machinery. At the point of or prior to AVD, the presence or absence of extracellular sodium controls the morphological occurrence of either cell shrinkage or cell swelling, respectively. However, in either condition the activation of the apoptotic machinery still occurs resulting in death of the cell
Potassium ions in the activation and repression of apoptosis
Cell shrinkage during apoptosis has been shown to occur on conjunction with the loss of intracellular ions [5, 8, 25, 26, 28, 43]. Since cells maintain a high concentration of intracellular potassium through the continuous activity of the Na+/K+-ATPase, the loss of this ion has been the primary focus for a majority of these studies [12, 20, 21, 28, 35, 44, 45]. It has been shown that the loss of intracellular ions, particularly potassium plays an active role in the activation of the apoptotic machinery. Early studies by Walev et al. [41] showed in human monocytes that low intracellular potassium levels permit the processing of proIL-1β to its mature form by the interleukin-1β converting enzyme (ICE), also known as caspase-1. Additionally, the activation of ICE has been shown to occur in response to hypotonic conditions, where potassium ions are lost from cells via the RVD response [34]. Shortly after these studies, Hughes et al. [16] showed that the loss of intracellular potassium not only enhances caspase activation by allowing efficient conversion of the procaspase to the active cleavage form, but the loss of potassium also results in the optimal catalytic activity of apoptotic nucleases.
These initial studies demonstrating how the loss of intracellular potassium plays a key role in the activation of apoptosis are supported by numerous studies that describe how maintaining a homeostatic intracellular potassium concentration prevents programmed cell death. We have previously shown that high extracellular potassium prevents cell shrinkage and DNA degradation in anti-Fas treated Jurkat cells [8]. Additionally, Hughes et al. [16] showed that physiological concentrations of potassium prevent caspase activation and apoptotic nuclease activity. Recently, Thompson et al. [39] and Cain et al. [10] reported the inhibition of apoptosis by high extracellular potassium in both extrinsic and intrinsic models of programmed cell death. These latter studies revealed how potassium has a regulatory and inhibitory effect at various levels of the apoptotic process including the formation of the apoptosome and release of cytochrome c. Furthermore, high extracellular potassium may also prevent apoptosis under conditions where the inherent RVI response is either inhibited or overridden (Fig. 1), again highlighting the importance of maintaining ionic homeostatic for cellular viability.
Conversely, in certain apoptotic model systems, the presence of high extracellular potassium has been reported to have no effect in preventing cell death. For example, in primary rat cortical cultures, high extracellular potassium did not prevent staurosporin-induced apoptosis [37]. These authors however defined high extracellular potassium as only 25 mM, as compared to previous studies that used a more physiological intracellular potassium concentration such as 100–120 mM. In a second study, no anti-apoptotic effect of high extracellular potassium (126 mM) or increased organic osmolytes was observed in serum-deprived vascular smooth muscle cells transfected with the E1A adenovirus [33]. These studies suggest that differences in osmotic sensitivity may exist depending on the specific cell type and model system being examined. This is not unexpected given that specific cell types employ different mechanisms for their transport of ions. A critical point to remember is that the loss of intracellular potassium while facilitating the loss of cell volume also provides for an intracellular environment where optimal activation of apoptosis can occur.
The exact nature for this loss of intracellular potassium during apoptosis is not completely understood. Various potassium channels and transport mechanisms have been suggested to play a role during AVD; however, many of these appear to be either stimulus and/or cell type specific. The human ether-a go-go (HERG) K+ channel, IKCa1 K+ channel, and the Kv1.3 K+ channel have all recently been suggested as molecular targets for potassium loss during apoptosis [3, 13, 38, 42]. Additionally, it has been shown that along with the efflux of intracellular potassium during apoptosis, the inability to take up potassium through the early inhibition of the Na+/K+-ATPase also plays a central role during cell shrinkage in certain cell types [9, 27, 31]. With regard to the nature of the loss of intracellular potassium during apoptosis, one has to keep in mind the overall goal of this physiological cell death process. The elimination of cells at a defined time or in response to a given stimulus is an inherent, homeostatic response. Thus, cells that are signaled to die must complete the apoptotic process to ensure survival of the remaining cells. This implies that multiple mechanisms may exist for the loss of intracellular potassium, AVD, and optimal activation of the apoptotic machinery. The inability to completely prevent apoptosis with a single potassium channel inhibitor suggests that secondary or even tertiary ionic pathways exist that adds to the complexity of the programmed cell death process [6].
The role of sodium ions during apoptosis
Interestingly, studies examining the role of intracellular sodium during apoptosis have lagged behind those of intracellular potassium. A primary reason for this hesitation has been the initial low intracellular sodium concentration compared to potassium in normal cells. However, it is well known that sodium does play a primary and pivotal role in controlling and maintaining a homeostatic cell volume. It has been shown that a loss of intracellular sodium accompanies the loss of intracellular potassium during AVD. However, recently we have shown an early increase in intracellular sodium upon anti-Fas induced apoptosis in Jurkat T-cells [6]. This early rise in intracellular sodium appears to be transient, and was shown to occur prior to the loss in intracellular potassium and AVD. The increase in intracellular sodium upon apoptotic stimulation correlates with plasma membrane depolarization, which was due in part to an early inhibition of the Na+/K+-ATPase [6]. Additionally, we showed that inhibition of the Na+/K+-ATPase with ouabain enhanced apoptosis in this model system. Furthermore, several other studies have also alluded to an early increase in intracellular sodium during programmed cell death [14, 17, 32, 36]. While these early studies have suggested an increase in intracellular sodium during apoptosis, only recently has a role for this ion been defined.
Inhibition of this early increase in intracellular sodium using the sodium channel blocker saxitoxin in anti-Fas treated Jurkat cells prevented the loss of intracellular potassium, cell shrinkage, and DNA degradation [7]. This suggests that the early movement of sodium during apoptosis plays a critical role in the activation of the cell death process. To further identify a role of sodium, we substituted either choline or NMDG for sodium in normal isotonic culture media for Jurkat T-cells and assessed the progression of apoptosis upon anti-Fas treatment [7]. Interestingly, by inhibiting the increase in intracellular sodium in this manner, anti-Fas-treated cells did not shrink, but swelled. However, other characteristics classically associated with apoptosis still occurred including chromatin condensation, externalization of phosphatidylserine, caspase activity, and internucleosomal DNA degradation. The occurrence of these apoptotic characteristics was shown to be associated with a loss of intracellular potassium in these swollen cells and inhibition of potassium loss by high extracellular potassium prevented apoptosis in both choline and NMDG media. The most surprising aspect of this study was when these swollen “apoptotic” cells were placed back into normal media containing the standard level of sodium. After 30 min of exposure to normal media, the swollen cells shrank, showing the typical morphology of classical apoptosis. This suggests that the ionic pathways for sodium remain open and active during apoptosis, and only the presence of sodium is required for the orchestration of the downstream ionic fluxes resulting in AVD.
Does cell size matter during the cell death process?
An important question to consider is whether the actual size of a cell is of any significant importance during apoptosis? As outlined above, cells have the inherit ability to regulate their cell volume upon exposure to anisotonic conditions through the activation of various ionic pathways, including the accumulation and removal of organic osmolytes [22]. While the ionic composition inside the cell may change under these anisotonic conditions, the overall objective of the inherent volume regulatory response is to return the cell to a near normal cell size. This ability in itself suggests that maintaining a proper cell size is a critical feature for the overall health of the cells. Additionally, in both necrosis and apoptosis, the cells change size when they die: swelling during necrosis and shrinking during apoptosis. This again suggests that a proper cell size is harmonious with cellular homeostasis and life. Therefore, sustained changes in cell size are detrimental to the viability of the cell.
The observation of cell shrinkage during apoptosis and cell swelling during necrosis have been a defining feature distinguishing these two modes of cell death. However in light of our recent study showing that in the absence of extracellular sodium [7], cells swell in conjunction with numerous apoptotic characteristics challenges this dogma (see Fig. 1). In retrospect, the morphological observation of changes in cell size may not be a necessary aspect of apoptosis. The critical component appears to be the specific movement of ions that is associated with either necrosis or apoptosis. Therefore, it is these ionic fluxes that define the specific mode of cell death a cell ensues under non-homeostatic conditions.
A second crucial aspect in defining cell death in regards to the impact of cell size is the concept of the programmed process of apoptosis; one event leading to another in an orchestrated manner leading to the individual demise of the cell. As described above, an increase in intracellular sodium has been shown to occur during apoptosis. Additionally, an increase in intracellular sodium has been suggested to be a key determinant during necrotic cell death. For example, ATP depletion in isolated hepatocytes exposed to oxidative stress resulted in an increase in intracellular sodium and necrotic cell death [11]. In this manner, many biological intermediates such as cellular ATP, calcium, and reactive oxygen species have been shown to play a role in both apoptosis and necrosis [29]. However, it is the timing of events that appears to be a decision point in whether a cell shrinks or swells, thus undergoing apoptosis or necrosis. A strong link between cellular ATP levels and ionic changes has been reported to confer a cells ability to either shrink or swell in response to lethal injury [24]. Maintenance of high cellular ATP levels appears to be an absolute requirement for apoptosis. Thus, maintaining high cellular ATP levels in the presence of a transient increase in intracellular sodium may orchestrate the downstream ionic changes resulting in apoptosis, whereas an immediate loss of cellular ATP in the presence of increased intracellular sodium results in necrosis [2].
Perspectives
Apoptosis has the unique characteristic of cell shrinkage that has separated this mode of physiological cell death from other cell death processes. Our ultimate goal is to understand how changes in cell volume and the associated movement of ions work in concert to achieve either the activation or repression of apoptosis. While we have described some of the recent studies pertaining to the role of sodium and potassium during apoptosis, clearly we have only just begun to shed light on what has emerged to be a rather complex and well-orchestrated process. The contribution of anions such as chloride and osmolytes such as taurine undoubtedly play a significant role in cell volume and activation/repression of the cell death. Our continual understanding of the interplay between ions and the apoptotic process would be of tremendous benefit in the therapeutic intervention of disease related to disregulation of apoptosis.
References
Al-Habori M (1994) Cell volume and ion transport regulation. Int J Biochem 26:319–334
Barros LF, Hermosilla T, Castro J (2001) Necrotic volume increase and the early physiology of necrosis. Comp Biochem Physiol 130:401–409
Bock J, Szabó I, Jekle A, Gulbins E (2002) Actinomycin D-induced apoptosis involves the potassium channel Kv1.3. Biochem Biophys Res Commun 295:526–531
Bortner CD, Cidlowski JA (1996) Absence of volume regulatory mechanisms contributes to the rapid activation of apoptosis in thymocytes. Am J Physiol 271:C950–C961
Bortner CD, Cidlowski JA (1998) A necessary role for cell shrinkage in apoptosis. Biochem Pharmacol 56:1549–1559
Bortner CD, Cidlowski JA (2002) Apoptotic volume decrease and the incredible shrinkage cell. Cell Death Differ 9:1307–1310
Bortner CD, Cidlowski JA (2003) Uncoupling cell shrinkage from apoptosis reveals that Na+ influx is required for volume loss during programmed cell death. J Biol Chem 278:39176–39184
Bortner CD, Hughes FM Jr, Cidlowski JA (1997) A primary role for K+ and Na+ efflux in the activation of apoptosis. J Biol Chem 272:32436–32442
Bortner CD, Gomez-Angelats M, Cidlowski JA (2001) Plasma membrane depolarization without repolarization is an early molecular event in anti-Fas-induced apoptosis. J Biol Chem 276:4304–4314
Cain K, Langlais C, Sun XM, Brown DG, Cohen GM (2001) Physiological concentrations of K+ inhibit cytochrome c-dependent formation of the apoptosome. J Biol Chem 276:41985–41990
Carini R, Autelli R, Bellomo G, Albano E (1999) Alterations of cell volume regulation in the development of hepatocyte necrosis. Exp Cell Res 248:280-293
Colom LV, Diaz ME, Beers DR, Neely A, Xie WJ, Appel SH (1998) Role of potassium channels in amyloid-induced cell death. J Neurochem 70:1925–1934
Elliott JI, Higgins CF (2003) IKCa1 activity is required for cell shrinkage, phosphatidylserine translocation and death in T lymphocyte apoptosis. EMBO Rep 4:189–194
Fernandez-Segura E, Canizares FJ, Cubero MA, Warley A, Campos A (1999) Changes in elemental content during apoptotic cell death by electron probe X-ray microanalysis. Exp Cell Res 253:454–462
Hoffmann EK (1987) Volume regulation in cultured cells. Curr Top Membr Trans 30:125–180
Hughes FM Jr, Bortner CD, Purdy GD, Cidlowski JA (1997) Intracellular K+ suppresses the activation of apoptosis in lymphocytes. J Biol Chem 272:30567–30576
Jonas D, Walev I, Berger T, Liebetrau M, Palmer M, Bhakdi S (1994) Novel path to apoptosis: small transmembrane pores created by staphylococcal alpha-toxin in T lymphocytes evokes internucleosomal DNA degradation. Infect Immunol 62:1304–1312
Kerr JFR (1965) A histochemical study of hypertrophy and ischaemic injury of rat liver with special reference to changes in lysosomes. J Pathol Bacteriol 90:419–435
Kerr JFR (1971) Shrinkage necrosis: a distinct mode of cellular death. J Pathol 105:13–20
Krick S, Platoshyn O, McDaniel SS, Rubin LJ, Yuan JX (2001) Augmented K(+) currents and mitochondrial membrane depolarization in pulmonary artery myocyte apoptosis. Am J Physiol 281:L887–L894
Krick S, Platoshyn O, Sweeney M, Kim H, Yuan JX (2001) Activation of K+ channels induces apoptosis in vascular smooth muscle cells. Am J Physiol 280:C970–C979
Lang F, Busch GL, Ritter M, Volkl H, Waldegger S, Gulbins E, Haussinger D (1998) Functional significance of cell volume regulatory mechanisms. Physiol Rev 78:247–306
Lang F, Ritter M, Gamper N, Huber S, Fillon S, Tanneur V, Lepple-Wienhues A, Szabo I, Gulbins E (2000) Cell volume in the regulation of cell proliferation and apoptotic cell death. Cell Physiol Biochem 10:417–428
Leist M, Single B, Castoldi AF, Kühnle S, Nicotera P (1997) Intracellular adenosine triphosphate (ATP) concentrations: a switch in the decision between apoptosis and necrosis. J Exp Med 185:1481–1486
Maeno E, Ishizaki Y, Kanaseki T, Hazama A, Okada Y (2000) Normotonic cell shrinkage because of disordered volume regulation is an early prerequisite to apoptosis. Proc Natl Acad Sci USA 97:9487–9492
Okada Y, Maeno E, Shimizu T, Dezaki K, Wang J, Morishima S (2001) Receptor-mediated control of regulatory volume decrease (RVD) and apoptotic volume decrease (AVD). J Physiol 532:3–16
Mann CL, Bortner CD, Jewell CM, Cidlowski JA (2001) Glucocorticoid-induced plasma membrane depolarization during thymocyte apoptosis: association with cell shrinkage and degradation of the Na+/K+-adenosine triphosphatase. Endocrinology 142:5059–5068
McCarthy JV, Cotter TG (1997) Cell shrinkage and apoptosis: a role for potassium and sodium ion efflux. Cell Death Differ 4:756–770
McConkey DJ (1998) Biochemical determinants of apoptosis and necrosis. Toxicol Lett 99:157–168
Nieminen AL (2003) Apoptosis and necrosis in health and disease: role of mitochondria. Int Rev Cytol 224:29–55
Nobel, CS, Aronson JK, Dobbelsteen DJ van den, Slater AF (2000) Inhibition of Na+/K+-ATPase may be one mechanism contributing to potassium efflux and cell shrinkage in CD95-induced apoptosis. Apoptosis 5:153–163
Offen D, Ziv I, Gorodin S, Barzilai A, Malik Z, Melamed E (1995) Dopamine-induced programmed cell death in mouse thymocytes. Biochem Biophys Acta 1268:171–177
Orlov SN, Pchejetski D, Taurin S, Thorin-Trescases N, Maxomov GV, Pshezhetsky AV, Rubin AB, Hamet P (2004) Apoptosis in serum-deprived vascular smooth muscle cells: evidence for cell volume-independent mechanism. Apoptosis 9:55–66
Perregaux DG, Laliberte RE, Gabel CA (1996) Human monocyte interleukin-1β posttranslational processing: evidence of a volume-regulated response. J Biol Chem 271:29830–29838
Platoshyn O, Zhang S, McDaniel SS, Yuan JX (2002) Cytochrome c activates K+ channels before inducing apoptosis. Am J Physiol 283:C1298–C1305
Skepper JN, Karydis I, Garnett MR, Hegyi L, Hardwick SJ, Warley A, Mitchinson MJ Cary NR (1999) Changes in elemental concentrations are associated with early stages of apoptosis in human monocyte-macrophages exposed to oxidized low-density lipoprotein: an X-ray microanalytical study. J Pathol 188:100–106
Small DL, Tauskela J, Xia Z (2002) Role for chloride but not potassium channels in apoptosis in primary rat cortical cultures. Neurosci Lett 334:95–98
Storey NM, Gómez-Angelats M, Bortner CD, Armstrong DL, Cidlowski JA (2003) Stimulation of Kv1.3 potassium channels by death receptors during apoptosis in Jurkat T lymphocytes. J Biol Chem 278:33319–33326
Thompson GJ, Langlais C, Cain K, Conley EC, Cohen GM (2001) Elevated extracellular [K+] inhibits death-receptor- and chemical-mediated apoptosis prior to caspase activation and cytochrome c release. Biochem J 357:137–145
Waldegger S, Lang F (1998) Cell volume and gene expression. J Membr Biol 162:95–100
Walev I, Reske K, Pamer M, Valeva A, Bhakdi S (1995) Potassium-inhibited processing of IL-1B in human monocytes. EMBO J 14:1607–1614
Wang H, Zhang Y, Cao L, Han H, Wang J, Yang B, Nattel S, Wang Z (2002) HERG K+ channel, a regulator of tumor cell apoptosis and proliferation. Cancer Res 62:4843–4848
Yu SP, Choi DW (2000) Ions, cell volume, and apoptosis. Proc Natl Acad Sci USA 97:9360–9362
Yu SP, Yeh CH, Sensi SL, Gwag BJ, Canzoniero LM, Farhangrazi ZS, Ying HS, Tian M, Dugan LL, Choi DW (1997) Mediation of neuronal apoptosis by enhancement of outward potassium current. Science 278:114–117
Yu SP, Yeh C, Strasser U, Tian M, Choi DW (1999) NMDA receptor-mediated K+ efflux and neuronal apoptosis. Science 284:336–339
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bortner, C.D., Cidlowski, J.A. The role of apoptotic volume decrease and ionic homeostasis in the activation and repression of apoptosis. Pflugers Arch - Eur J Physiol 448, 313–318 (2004). https://doi.org/10.1007/s00424-004-1266-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-004-1266-5