Abstract
Purpose
Intraoperative parathyroid hormone (ioPTH) measurement has facilitated a move to minimally invasive parathyroidectomy. Patients are referred for surgery earlier with milder hypercalcaemia and smaller tumours. Whilst previous research has shown that glands size can affect ioPTH kinetics in patients with multiple gland disease, the dynamics of ioPTH in patients with mild hyperparathyroidism (HPT) has not been studied. We therefore investigated the relationship between biochemical parameters and parathyroid adenoma weight, and determined the dynamics and accuracy of ioPTH assay in patients with milder hypercalcaemia undergoing parathyroidectomy.
Methods
Patients undergoing parathyroidectomy for single gland disease from January 2004 to March 2011 were divided prospectively into two groups according to preoperative serum calcium: patients with a preoperative calcium ≥2.85 mmol/L (11.4 mg/dL) and <2.85 mmol/L were grouped as severe and mild hypercalcaemia, respectively. Correlation coefficients were calculated to assess the relationship between biochemical markers of calcium homeostasis and ioPTH measurements with respect to parathyroid gland weight.
Results
There was a weak correlation of preoperative serum calcium (r = 0.248, r = 0.207), PTH (r = 0.392, r = 0.275), and baseline ioPTH (r = 0.516, r = 0.244) with parathyroid gland weight in severe (n = 113) and mild groups (n = 190), respectively. No correlation between the magnitude in ioPTH drop with parathyroid gland weight at 5 or 10 min post-excision for either group was observed. Success rates (post-operative normocalcaemia) were similar for each group (99.1 % severe, 98.9 % mild).
Conclusion
This prospective study provides evidence that ioPTH assay is a valuable tool in predicting adequate tissue removal in patients with milder and more severe hypercalcaemia due to single gland primary HPT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Primary hyperparathyroidism (HPT) is a common endocrine disorder with an incidence of around 3/1,000 in Europe [1]. Most (80–85 %) cases are due to a solitary parathyroid adenoma [2–4] with excision of the pathological gland resulting in cure. Surgery carried out by bilateral neck exploration was and still is used with success rates of 90–95 % [5] when performed by experienced surgeons. A move to targeted minimally invasive unilateral parathyroidectomy (PTX) has now been permitted by the accuracy of preoperative localisation techniques such as sestamibi scintigraphy (MIBI) and high-resolution ultrasound (US) that have been shown to have a sensitivity in excess of 95 % [6] when the scan findings are concordant.
Intraoperative parathyroid hormone (ioPTH) can be used as an adjunct to preoperative localisation techniques enabling the surgeon to predict the adequate removal of hyperfunctioning parathyroid gland(s) in 94–98 % of cases [7, 8]. Furthermore, ioPTH has been shown to be an accurate predictor of post-operative success [9, 10]. Limiting to a unilateral neck exploration as opposed to a more invasive bilateral exploration, particularly when used in conjunction with preoperative localisation, reduces operation time and post-operative hypercalcaemia [11–13] and may also reduce cost [14]. During PTX PTH samples are assayed at baseline, pre-excision and 5 and 10 min post-excision. Results can be available to the surgeon in as little as 8 min depending on the assay used [15]. Many surgeons use the Miami criteria whereby a 50 % reduction from the highest of either the baseline or pre-excision ioPTH value predicts adequate excision of hyperfunctioning parathyroid tissue [16].
According to NIH guidelines for parathyroid surgery, a corrected serum plasma calcium equal or greater than 2.85 mmol/L (11.4 mg/dL) is the recommended threshold for PTX in patients with asymptomatic primary hyperparathyroidism [0.25 mmol/L (1 mg/dL) above the upper limit of the normal reference range] [17]. Patients with serum calcium less than 2.85 mmol/L could be considered to have a milder form of hypercalcaemia.
It is recognised that patients currently undergoing PTX for HPT may have a milder form of hypercalcaemia compared with the past [18]. Given that several studies have shown that biochemical values such as preoperative serum calcium and PTH levels correlate with parathyroid gland weight [3, 19–24], it follows that patients with milder hypercalcaemia are more likely to have smaller tumours. Previous research has also demonstrated that the size of the abnormal gland may influence the operative pattern of PTH decay [25]. Therefore in patients with milder hypercalcaemia, smaller adenoma size could potentially influence the kinetics of ioPTH: an aspect that has not previously been investigated. We were therefore interested to determine if there was a difference in the dynamics of ioPTH for patients with milder hypercalcaemia and indeed whether ioPTH was less or equally reliable for patients with mild HPT.
Materials and methods
This was a prospective study of patients undergoing PTX for single gland disease from January 2004 to March 2011 at a tertiary centre. Once a decision to operate was made, patients underwent a neck US and a MIBI scintiscan. Clinical, biochemical and radiology data were prospectively entered into a database for all patients undergoing parathyroid surgery. A focused approach, in which only the targeted glands were inspected, was used when either study was positive. A bilateral approach was employed when (a) localisation was negative, (b) no tumour was found or (c) no drop in ioPTH at 10 min was recorded. The highest recorded preoperative corrected serum calcium and PTH concentrations were used. For ioPTH; samples were taken at baseline, pre-excision, 5 and 10 min post-excision using a two-site chemiluminescent assay (Cambridge Diagnostics Ltd, Galway, Ireland). The percentage reduction in ioPTH at 5 and 10 min post-excision was calculated from the highest of either baseline or pre-excision values. A 50 % reduction was used as a predictor of adequate excision of hyperfunctioning parathyroid tissue [16]. False-positive results occur when ioPTH decreases by >50 % despite all hyperfunctioning gland(s) not being removed. False-negative results occur when ioPTH fails to decrease by >50 % despite adequate removal of the hyperfunctioning gland(s).
A total of 482 patients with primary HPT underwent surgery during this time period in our unit. We excluded 90 patients for whom ioPTH was either not used or the data were incomplete. Patients with parathyroid carcinoma (n = 1), MEN-1 (n = 2) and those with missing data: no recorded preoperative corrected calcium (n = 1) adenoma weight (n = 4), and 3 month post-operative corrected serum calcium (n = 18) were also excluded from the study. Patients were divided into two groups according to the preoperative corrected calcium level: group A with a preoperative calcium ≥2.85 mmol/L (11.4 mg/dL) and group B with a preoperative calcium <2.85 mmol/L. More than one gland was excised in 63 of the remaining 366 patients, 43 (17.8 %) in group A and 20 (14.4 %) in group B. Patients in whom more than one gland was excised were also excluded leaving a cohort of 303 patients (group A, n = 113; group B, n = 190). We also recognise that a cut-off at 2.85 mmol/L (11.4 mg/dL) might seem too high nowadays. It was chosen according to NIH criteria in the absence of a standard definition of ‘mild’ HPT. However to allow application of these data to wider clinical setting, we performed further analysis for a lower cut-off of 2.75 mmol/L (11 mg/dL).
The correlation between parathyroid gland weight and preoperative serum calcium levels, preoperative PTH levels, baseline PTH levels, the absolute PTH reduction and the PTH percentage reduction during surgery were calculated with Pearson’s correlation. Pearson’s Chi squared test was used to assess statistical difference between localisation results and the Mann–Whitney U test was used for baseline variables. All statistical analysis was carried out using SPSS 18.0.
Results
The baseline variables for groups A and B are shown in Table 1. Most patients (n = 298; 98 %) underwent both US and MIBI scans (Table 2). Three patients (0.9 %) had no localisation. There is a significant difference between the rate of successful localisation and concordance between the two groups; overall, 79 % of patients were correctly localised by at least one scan (group A, 85 %; group B, 76 %) (P = 0.05) and the overall rate of concordant scans was 57 % (group A, 64 %; group B, 51 %) (P < 0.05). The accuracy of concordant scans was 97.7 %. Unilateral neck exploration was undertaken in a greater proportion of patients in group A (86 %) compared with group B (80 %).
The median parathyroid gland weight of [severe] group A (870 mg) was over double that of the [mild] group B (430 mg) (P < 0.001). Preoperative calcium and parathyroid gland weight showed a weak positive correlation in group A (r = 0.248) and group B (r = 0.207). Preoperative PTH levels also showed a weak positive correlation with parathyroid gland weight in group A (r = 0.392), group B (r = 0.275) as well as baseline ioPTH group A (r = 0.516) and group B (r = 0.244).
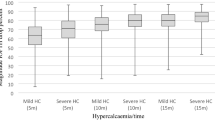
The per-operative pattern of ioPTH is remarkably similar for each group (Fig. 1). There was no statistically significant correlation between parathyroid gland weight and percentage reduction in ioPTH at either 5 or 10 min in either the severe or mild groups, although absolute reduction in ioPTH at 5 and 10 min showed a weak correlation in both groups (Table 3). The overall success rate for surgery (defined as normocalcaemia at 3 months) was 99.0 %; 99.1 % in group A and 98.9 % in group B. The 1 % (n = 3) of patients with persistent hypercalcaemia had very mild hypercalcaemia [3-month post-operative corrected calcium = 2.61 (n = 1) − 2.62 (n = 2)]. For all patients, ioPTH correctly predicted cure in 291 (96 %; positive predictive value 99.3 %). The false-positive rate was 0.7 % (n = 2) and was the same in both groups (0.3 %; n = 1). The false-negative rate was 2.9 % (n = 9) and was lower in group A (1.8 %; n = 2) compared with group B (3.8 %; n = 7), leading to nine unnecessary conversions to a BNE.
Further analysis for a lower cut-off of 2.75 mmol/L (11 mg/dL) is displayed in Table 4. No correlation existed between parathyroid gland weight and ioPTH percentage reduction in either group at 5 min. At 10 min, a weak correlation between adenoma weight and ioPTH percentage reduction was observed in the mild (r = 0.191) and severe group (r = 0.180).
Although renal function influences biochemical parameters, there was no difference in renal function between the two groups in terms of creatinine levels (data not shown).
Discussion
In this study, the median parathyroid gland weight of [severe] group A (870 mg) was over double that of the [mild] group B (430 mg) suggesting that tumour weight influences preoperative serum calcium and serum PTH levels. This is the most likely explanation for the significantly lower success rate of localisation in the milder group as it is known that accuracy of localisation drops as the weight of the gland falls [26]. This together with a significantly lower proportion of concordant scans infers that ioPTH may be more valuable for patients with mild hypercalcaemia.
The findings of this study showed no correlation between parathyroid gland weight and ioPTH percentage reduction in either group. Two previous studies have investigated the correlation between parathyroid gland weight and percentage reduction in ioPTH. Biertho et al. found no correlation between parathyroid gland weight and ioPTH percentage reduction [24] whereas Moretz et al. reported a weak negative correlation between parathyroid gland weight and percentage reduction at 10 min [23].
The similar success (normocalcaemia) rates and weak of correlation between adenoma weight and ioPTH percentage reduction between the two groups suggest that the kinetics of ioPTH and other biochemical values are similar irrespective of the tumour mass. We chose to analyse only those patients with single adenoma in order to simplify the interpretation of tumour weight as well as minimise the confounding issues regarding the dynamics of ioPTH in patients with multiple gland disease where high false-positive rates are experienced [27]. Furthermore, previous research on the kinetics of ioPTH in patients with MEN-1 has suggested that the size of the abnormal gland may influence the operative pattern of PTH decay [25]. All patients were followed up for a minimum 3-month period, and although the focus of this paper is the intraoperative dynamics of PTH, we acknowledge that the weakness of our study is the short follow-up period to be certain of biochemical cure.
The effectiveness of ioPTH in severe and mild hypercalcaemia has not previously been reported. The definition of ‘mid HPT’ is complex and we selected serum calcium as an objective surrogate variable to define this. In the absence of an agreed standard definition, we chose a cut-off from the consensus guidelines. However, nowadays, a cut-off of 2.85 mmol/L seems too high, and to allow application of these data to wider clinical setting, we performed further analysis for a lower cut-off of 2.75 mmol/L (11 mg/dL) (Table 4). We found that there was no correlation between parathyroid gland weight and ioPTH percentage reduction in either group at 5 min. At 10 min, a weak correlation between adenoma weight and ioPTH percentage reduction was observed in the mild (r = 0.191) and severe groups (r = 0.180). This sub-analysis provides further reassurance that ioPTH kinetics are similar in patients with milder as well as more severe hypercalcaemia.
Our results provide new evidence supporting the value of ioPTH in patients with mild primary HPT. This is in line with the results of the Scandinavian audit which showed that ioPTH favourably influenced the outcome of parathyroidectomy in patients with smaller tumours [28].
Conclusion
The absence of any difference in the dynamics of ioPTH with mild and severe hypercalcaemia and equivalent normocalcaemia rates in each group provides reassurance that ioPTH measurement is a valuable tool for patients with milder hypercalcaemia.
References
Inabet WB, Lee JA, Henry J-F, Sebag F (2006) Parathyroid disease. In: Lennard TWJ (ed) Endocrine surgery: a companion to specialist surgical practice, 3rd edn. Elsevier Saunders, Amsterdam, pp 1–42
Randhawa PS, Mace AD, Nouraei SAR, Stearns MP (2007) Primary hyperparathyroidism: do perioperative biochemical variables correlate with parathyroid adenoma weight or volume? Clin Otolaryngol 32:179–184
Williams JG, Wheeler MH, Aston JP, Brown RC, Woodhead JS (1992) The relationship between adenoma weight and intact (l-84) parathyroid hormone. Am J Surg 163:301–304
Norton JA, Brennan MF, Wells SA Jr (1994) Surgical management of hyperparathyroidism. In: Bilezikian JP, Levine MA, Marcus R (eds) The parathyroids, 1st edn. Raven, New York, pp 531–551
Stasio ED, Carrozza C, Lombardi CP, Raffaelli M, Traini E, Bellantone B et al (2007) Parathyroidectomy monitored by intra-operative PTH: the relevance of the 20 min end-point. Clin Biochem 40:595–603
Patel CN, Salahudeen HM, Lansdown M, Scarsbrook AF (2010) Clinical utility of ultrasound and 99mTc sestamibi SPECT/CT for preoperative localization of parathyroid adenoma in patients with primary hyperparathyroidism. Clin Radiol 65:278–287
Irvin GL, Deriso GT (1994) A new, practical intraoperative parathyroid hormone assay. Am J Surg 168:466–468
Carneiro DM, Irvin GL (2002) New point-of-care intraoperative parathyroid hormone assay for intraoperative guidance in parathyroidectomy. World J Surg 26:1074–1077
Mandell DL, Genden EM, Mechanick JI, Bergman DA, Diamond EJ, Urken ML (2001) The influence of intraoperative parathyroid hormone monitoring on the surgical management of hyperparathyroidism. Arch Otolaryngol Head Neck Surg 127:821–827
Burkey SH, van Heerden JA, Farley DR, Thompson GB, Grant CS, Curlee KJ (2002) Directed parathyroidectomy utilizing the gamma probe or intraoperative parathyroid hormone assay replace bilateral cervical exploration as the preferred operation for primary hyperparathyroidism? World J Surg 26:914–920
Vignali E, Picone A, Materazzi G, Steffe S, Berti P, Cianferotti L et al (2002) A quick intraoperative parathyroid hormone assay in the surgical management of patients with primary hyperparathyroidism: a study of 206 consecutive cases. Eur J Endocrinol 146:783–788
Patel PC, Pellitteri PK, Patel NM, Fleetwood MK (1998) Use of a rapid intraoperative parathyroid hormone assay in the surgical management of parathyroid disease. Arch Otolaryngol Head Neck Surg 124:559–562
Carneiro-Pla D (2011) Contemporary and practical uses of intraoperative parathyroid hormone monitoring. Endocr Pract 17(1):44–53
Johnson LR, Doherty G, Lairmore T, Moley JF, Brunt LM, Koenig J et al (2001) Evaluation of the performance and clinical impact of a rapid intraoperative parathyroid hormone assay in conjunction with preoperative imaging and concise parathyroidectomy. Clin Chem 47(5):919–925
Irvin GL III, Carneiro DM (2005) Intraoperative parathyroid hormone assay as a surgical adjunct in patients with sporadic primary hyperparathyroidism. In: Clark O, Duh Q-Y, Kebebew E (eds) Textbook of endocrine surgery, 2nd edn. Elsevier Saunders, Philadelphia, pp 472–480
Carneiro DM, Solorzano CC, Nader MC, Ramirez M, Irvin GL III (2003) Comparison of intraoperative iPTH assay (QPTH) criteria in guiding parathyroidectomy: Which criterion is the most accurate? Surgery 134(6):973–979
Bilezikian JP, Khan AA, Potts JRJT (2009) Guidelines for the management of asymptomatic primary hyperparathyroidism: summary from the third international workshop. J Clin Endocrinol Metab 94(2):335–339
Sitges-Serra A, Rosa P, Valero M, Membrilla E, Sancho JJ (2008) Surgery for sporadic primary hyperparathyroidism: controversies and evidence-based approach. Langenbecks Arch Surg 393:239–244
Hamidi S, Aslani A, Nakhjavani M, Pajouhi M, Hedayat A, Kamalian N (2006) Are biochemical values predictive of adenoma’s weight in primary hyperparathyroidism? ANZ J Surg 76:882–885
Bindlish V, Freeman JL, Witterick IJ, Asa SL (2002) Correlation of biochemical parameters with single parathyroid adenoma weight and volume. Head Neck 24(11):1000–1003
Mozes G, Curlee KJ, Rowland CM, van Heerden JA, Thompson GB, Grant CS et al (2002) The predictive value of laboratory findings in patients with primary hyperparathyroidism. J Am Coll Surg 194(2):126–130
Rutledge R, Stiegel M, Thomas CGJ, Wild RE (1985) The relation of serum calcium and immunoparathormone levels to parathyroid size and weight in primary hyperparathyroidism. Surgery 98(6):1107–1112
Moretz WH, Watts TL, Virgin FW, Chin E, Gourin CG, Terris DJ (2007) Correlation of intraoperative parathyroid hormone levels with parathyroid gland size. Laryngoscope 117:1957–1960
Biertho LD, Kim C, Wu H-S, Unger P, Inabnet WB (2004) Relationship between sestamibi uptake, parathyroid hormone assay, and nuclear morphology in primary hyperparathyroidism. J Am Coll Surg 199(2):229–233
Tonelli T, Spini S, Tommasi M, Gabbrielli G, Amorosi A, Brocchi A et al (2000) Intraoperative parathormone measurement in patients with multiple endocrine neoplasia type I syndrome and hyperparathyroidism. World J Surg 24(5):556–563
Pattou F, Huglo D, Proye C (1998) Radionuclide scanning in parathyroid diseases. Br J Surg 85(12):1605–1616
Stiges Serra A, Diaz-Aguirregoitia FJ, de la Qunitana A, Gil-Sanchez J, Jimeno J, Prieto R et al (2010) Weight difference between double parathyroid adenomas is the cause of false-positive IOPTH test after a resection of the first lesion. World J Surg 34(6):1337–1342
Bergenfelz AOJ, Wallin G, Jansson S, Eriksson H, Martensson H, Christianson P et al (2011) Results of surgery for sporadic hyperparathyroidism in patients with preoperatively negative sestamibi and ultrasound. Langenbecks Arch Surg 396:83–90
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hathaway, T.D., Jones, G., Stechman, M. et al. The value of intraoperative PTH measurements in patients with mild primary hyperparathyroidism. Langenbecks Arch Surg 398, 723–727 (2013). https://doi.org/10.1007/s00423-013-1080-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-013-1080-7