Abstract
Background
Adrenal tumors are a common manifestation of the multiple endocrine neoplasia type 1 (MEN-1) syndrome. Prevalence in recent studies varies between 9 and 45%. A genotype–phenotype correlation has been described as well as the development of adrenocortical carcinomas. Long-term prospective data are still lacking.
Materials and methods
Thirty-eight MEN-1 patients with proven germline mutations have been prospectively observed in a regular screening program in our hospital. Adrenal glands have been screened by biochemical analysis and either by endoscopic ultrasound (EUS) or computed tomography (CT) or both. Median follow-up was 48 months (12–108 months). Age at diagnosis of MEN-1, type of adrenal tumor, genotype, therapy, and clinical characteristics have been analyzed.
Results
In 21 (55%) patients, adrenal involvement of the disease was detected. Adrenal lesions were detected in average 6.9 years after the initial diagnosis of MEN-1. Median tumor size was 12 mm (5–40 mm). Tumor size smaller than 10 mm was observed in 11 patients. Twelve patients had unilateral while nine had bilateral adrenal lesions. EUS detected all adrenal tumors, whereas CT failed in seven cases. In three patients, functioning tumors (one pheochromocytoma, one bilateral Cushing adenoma, and one adrenocortical carcinoma) and one nonfunctioning adenoma were diagnosed by histology and biochemical assessment. Two laparoscopic adrenalectomies and one laparoscopic subtotal resection were performed. Nonfunctioning adrenal lesions, not characterized by histology yet, were found in 18 patients. There was no statistical difference with regard to adrenal involvement between patients with germline mutations in exons 2 and 10 (12/21) and those with mutations in exons 3–9 (6/11).
Conclusion
MEN-1-associated adrenal tumors are mostly small, benign, and nonfunctioning and much more common than previously reported. EUS was the most sensitive imaging procedure. The genotype–pheotype correlation previously suggested by our group could not be confirmed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multiple endocrine neoplasia type 1 (MEN-1) is an autosomal dominant tumor syndrome caused by germline mutations of the Menin gene on chromosome 11q13 [1, 2]. Classical manifestations are pituitary adenomas, most often prolactinomas, parathyroid hyperplasia causing primary hyperparathyroidism, and pancreaticoduodenal endocrine tumors (PETs). Different tumor entities have also been reported to be associated with this hereditary disease, such as lipoma, foregut-, thymic-, and bronchial carcinoids, ependymomas, and adrenal lesions [3–8]. An overall incidence of 0.25% for MEN-1 seems to be apparently higher in autopsy studies [9, 10] than clinically observed so far. Prevalence of adrenal lesions ranged from 9 to 45% [11–17], and clinical features varied. Nonfunctional enlargement of adrenal glands was most common and described to occur uni- or bilaterally. Also, pheochromocytomas, primary hyperaldosteronism, Cushing’s syndrome, and even adrenocortical carcinomas (ACC) have been observed [11–17] and may lead to premature death in MEN-1 patients.
It has been proposed that long-term follow-up including yearly imaging studies may reveal an increased frequency of adrenal lesions [17, 18]. Long-term prospective data are still lacking. A higher frequency of adrenal tumors in MEN-1 patients with PETs was observed and is eventually caused by overexpressed growth factors like proinsulin or insulin [12]. The absence of loss of heterozygosity of the MEN-1 gene in adrenal lesions may indicate that inactivation of the Menin gene does not rule tumorigenesis in the adrenal gland [12].
In this study, we assessed prevalence and clinical characteristics of adrenal lesions in 38 genetically confirmed MEN-1 patients by a prospective screening program at our hospital. Functional tests as well as imaging by either computed tomography (CT) or endoscopic ultrasound (EUS) or both have been performed.
Materials and methods
Since 1998, 7 asymptomatic germline mutation carriers and 31 MEN-1 patients out of 24 unrelated kindred were followed up in this study by an annual screening program. Diagnosis of MEN-1 was based on germline mutation status.
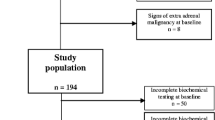
A total of 38 patients were included in the study. The group consisted of 20 men and 18 women with a mean age of 44.2 years (18–71 years). Median follow-up was 48 months (12–108 months). Adrenal lesions were scheduled for surgery when exceeding 50 mm or secreting hormones. Since 2001, we emphasized a more aggressive strategy and performed either total adrenalectomy or partial resection if tumors were larger than 30 mm [15]. We analyzed age of diagnosis of MEN-1 and adrenal involvement, clinical characteristics, genotype, therapy, and follow-up.
All patients were examined clinically with special regard to symptoms known to be associated with hormone-secreting adrenal tumors.
At diagnosis and then annually, patients underwent biochemical screening including measurement of serum concentration of dehydroepiandrosterone sulphate, cortisol, renin, aldosterone, and routine markers. Output of metanephrine, normetanephrine, epinephrine, norepinephrine, and vanillylmandelic acid as well as cortisol were determined in 24-h urine.
Adrenal lesions were visualized either by EUS or thin-sliced CT or both.
Nonfunctional lesions were diagnosed if enlargement or nodular changes of the adrenal gland were present in imaging modalities without causing symptoms or abnormal hormone levels. We considered any lesion larger than 5 mm, detected by CT or EUS, which was reproducibly visualized, either as adenoma or as nodular hyperplasia. Adrenal Cushing’s syndrome was diagnosed if suppressed ACTH levels were combined with positive dexamethasone suppression tests. Elevated urinary catecholamine levels indicated pheochromocytoma.
Histologic confirmation was obtained in one pheochromocytoma, one ACC, one Cushing adenoma, one nonfunctional adenoma, and one adrenal cyst.
All patients had undergone genetic testing for germline mutations within the Menin gene. Statistical analysis was performed using the chi-square test. A p-value <0.05 was considered to be statistically significant.
Results
Twenty-one (55%) of 38 patients included in this study revealed adrenal involvement (see also Table 1). Among them were two asymptomatic mutation carriers. The mean age of these 11 women and 10 men was 47.0 years (19–70 years). Adrenal manifestation of the disease was diagnosed at a mean age of 42.7 years (19–69 years), while MEN-1 was initially diagnosed by a mean age of 35.8 years (18–49 years).
Clinical presentation
In three patients, functioning adrenal tumors were found: one unilateral pheochromocytoma in a patient having high blood pressure, bilateral adrenal adenomas causing Cushing’s syndrome in a women and one ACC in another patient causing a mild virilization. The remaining adrenal tumors were nonfunctioning.
Laboratory tests
In the case of the pheochromocytoma, fourfold elevated urinary katecholamines were present. Cushing’s syndrome was detected by elevated cortisol in urine and serum as well as a suppressed ACTH level in combination with a positive dexamethason suppression test. ACC demonstrated elevated serum levels of cortisol and testosterone. A false positive result of biochemical markers was detected in 15 patients in 22 assessments.
Imaging
Adrenal enlargement was detected in 21/21 (100%) cases by EUS, whereas CT was nondiagnostic in seven of the cases. Median tumor diameter at diagnosis was 10 mm (5–50 mm). In 9/21 (42%) cases, the tumor diameter did not exceed 10 mm. Seven (33%) patients demonstrated bilateral adrenal abnormalities. All patients with adrenal lesions had also PETs.
Surgery
Adrenalectomy was performed in two patients with Cushing adenomas and pheochromocytoma. Due to metastatic disease (Lee Stage IV), the patient with the ACC was not scheduled for surgery. Two patients who had nonfunctional tumors also underwent surgery because of tumor size larger than 30 mm. A subtotal resection was done in one patient who had an adenoma measuring 50 mm in diameter. In the other patient, close follow-up by EUS showed an annual tumor progression of 10 mm, a diameter of 42 mm, and a suspicious cystic part. Therefore, adrenalectomy was performed (see also Fig. 1).
a Computed tomography showing a nodular hyperplastic left adrenal gland in 2002. b Note the large cystic tumor of the left adrenal demonstrated in the computed tomography in 2006. c Endoscopic ultrasound in 2002 demonstrating the nodular characteristics of the enlarged adrenal gland more in detail; also shown: nonfunctioning tumor of pancreatic tail (arrow). d Corresponding endoscopic ultrasound in 2006 revealed a cystic tumor 42 mm in diameter. e Gross appearance of the pseudocystic tumor after adrenalectomy. The adjacent adrenal tissue impressed by its macronodular texture. Histology showed a pseudocyst of the adrenal gland. Asterisks indicate the left adrenal gland. P Pancreas, K kidney, S spleen, ST stomach
Histology
The pheochromocytoma was confirmed histologically. In the patient with Cushing’s syndrome, one solitary adenoma and one macronodular hyperplasia were found. A cortical adenoma measuring 50 mm was found in the patient where parenchyma-preserving resection was performed. The specimen of the remaining patient with nonfunctioning enlargement revealed a pseudocyst of the adrenal gland.
Follow-up
At conclusion of the study, all patients were alive except one. As described previously, one patient with a nonfunctional adrenal tumor measuring 35 mm in diameter developed a cortisol and testosterone-secreting ACC within 9 months. She received mitotane and succumbed 3 months after diagnosis to progressive disease.
Sixteen patients with nonfunctional adrenal lesions had stable disease without significant change of size and function and without other indications for malignancy. They were followed up for a median period of 42 months (24–108 months).
Genetics
Molecular analysis confirmed diagnosis of MEN-1 in all 38 patients. Mutations were predominantly found in exons 2, 3, 9, and 10 (36/38, 95%). Altogether, 21 different mutations in 29 unrelated kindred were observed [P529X, E116X, Q554X, Y90X, K119X, K120X, E530X, F448X, R171Q, L168P, E26K, W126K, R436Y, nt894-1 (G→A), nt1390 (del6), nt1507 (del14), nt310 (ins5), nt742 (del4), T193I, nt1651 (InsC), nt894-9 (G→A)].
Twelve patients out of 21 with adrenal abnormalities carried mutations in exons 2 and 10, whereas 9 of 17 patients with adrenal lesions had mutations in exons 3–9. There was no statistic significant difference between the two groups (p = 0.6417) (see also Table 2).
Discussion
Adrenal tumors associated with MEN-1 had already been reported in the 1960s. The first reports referred to autopsy studies or to incidental findings at laparotomy, and the impact of adrenal involvement was underestimated. In series published later, the prevalence of adrenal involvement ranged from 9 to 45% [11–13, 19, 20]. First studies on genetically confirmed MEN-1 patients and mutation carriers showed the high incidence and malignant potential of adrenal tumors in this special clinical setting [15]. Autopsy studies revealed adrenocortical lesions in about one-third of MEN-1 patients [19, 20]. It is noteworthy that adrenal enlargement is also observed in about 1–8% of cases at necropsy and is supposed to be due to the adrenal aging process [21–23]. A definition of adrenocortical lesions and a discrimination between adenomas and adrenal nodules in non-MEN-1 patients are not defined. There were no criteria for size of adrenal incidentaloma during the NIH consensus conference in 2002 [24]. Thompson and Young [25] suggested a limit of 10 mm, which is in the range of the minimal size that one could detect by CT scan. However, this limit of 10 mm is more of a personal consideration than of a valid and established limit for an adrenal incidentaloma. Kloos et al. [26] reported a series of 21 small adrenal lesions (<10 mm), which were classified as small adenomas. Commons and Callaway observed adenomas larger than 3 mm in only 2.8% of adrenal glands at autopsy [27]. Higher incidences were reported by Reinhard et al. who observed adrenocortical nodules in 53% of 500 autopsies [28]. However, 80% of these lesions were between 1 and 2 mm, which would be consequently missed by EUS in our study. Granger and Genest [29] proposed to differentiate between adenoma and adrenal nodular by the size: nodule if the lesions are smaller than 8 mm; adenoma if they exceed 8 mm. According to their definition, 7/21 patients had adrenal nodules, whereas 14/21 revealed adenomas. When comparing our results with results of autopsy studies, one should be aware that the median age of our patients was 44 years, which is about 20–30 years less than in most autopsy studies. This underscores the meaning of adrenal lesions in MEN-1 patients as a part of the syndrome; thus, in older patients, adrenocortical nodules or adenomas are associated with arteriopathy [28]. It still remains as a matter of debate whether adrenal lesions are primary or secondary lesions in MEN-1 patients.
The present prospective study over 7 years shows a frequency of 55%, which is obviously higher than reported by other groups and our own data published recently, which found adrenal involvement in 9–45% [13–17]. It has been proposed earlier that extensive screening strategies and long-term follow-up may lead to increased frequency of adrenal manifestation as part of MEN-1 [17, 18]. The clinical impact as well as the therapeutic strategy of adrenal lesions are still not defined [30].
In none of the patients in the present study the adrenal manifestation was the initial finding. On average, MEN-1 syndrome was diagnosed 6.9 years before adrenal lesions. The most common initial finding was primary hyperparathyroidism in 67% (21/31) of our patients, which is consistent with the data reported in the literature [31–33]. Because adrenal involvement was asymptomatic in almost all cases in the present study, imaging studies contributed to the diagnosis in every patient. EUS was the superior imaging modality in our study because 33% of adrenal lesions detected by EUS were missed by CT. In cases missed by CT, EUS was the only proof for adrenal lesions, and all lesions were smaller than 10 mm. One could now argue whether the visualized tumors have pathologic impact or are real adrenal tumors or macronodular hyperplasia. The incidence of pathological findings like macronodular changes in EUS in healthy persons is unknown. On the other hand, in about 1,200 EUS investigations performed by the same investigator of our group for other indications than the suspected adrenal tumors, only one patient revealed an adrenal adenoma. The total number of non-MEN-1 patients was evidently higher than our cohort of MEN-1 patients although they could not be suggested as “healthy controls”. Although this may not represent incidence in a normal population, pathologic adrenal findings in EUS seem to be rare in non-MEN-1 patients.
Nonfunctional nodular hyperplasia or adenomas were present in most of the patients demonstrating adrenal abnormalities, which confirms the previously reported data [10, 12, 15]. Our observation of unilateral adrenal enlargement in 77% (14/21) was in consensus with the reported 55–79% in three other studies [15, 17, 34]. Nevertheless, it was controversial to the 60% bilateral adrenal involvement reported by Skogseid et al. [12].
Functioning tumors like pheochromocytoma (2.6%), Cushing adenoma (2.6%), and ACC (2.6%) are rare manifestations of MEN-1. Gibril et al. [17] reported similar results in their studies, with 48 MEN-1 patients having Zollinger–Ellison syndrome.
The incidence of ACC in 68 and 43 MEN-1 patients reported by our group and Skogseid, respectively, was about 6% [13, 15]. Both studies emphasized the observation that patients with initially small and nonfunctional adenoma may instantly develop ACC. Our patient, already reported in a former study, developed ACC with testosterone and cortisol excess with liver and lung metastases 9 months after being diagnosed to have a 35-mm nonfunctional adenoma. Therefore, we recommended surgery in MEN-1 patients with adrenal tumors exceeding 30 mm and a closer follow-up by EUS in patients with newly diagnosed adrenal lesions. The present study shows that development of ACC seems to be rare although it has tremendous clinical impact. The patient with the growing cystic tumor reported in the present study belongs to the same kindred as the ACC patient reported previously. Therefore, and due to the facts that the tumor was growing and showed multicystic appearance, we performed surgery (see also Fig. 1). Postoperative histology report revealed an adrenal pseudocyst without any evidence for malignancy or hyperplasia.
Skogseid et al. [12] pointed out that the release of growth factors like proinsulin or insulin by PETs may contribute to a higher rate of adrenal tumors in MEN-1 patients with PETs. Gibril et al. [17] reported a higher incidence of adrenal lesions in ZES/MEN-1 patients, which might support this idea. In vitro studies showed evidence for stimulation of cell proliferation by pancreatic hormones [37]. Thirty-six of our 38 (94.7%) patients had PETs, but both patients without PETs did not reveal adrenal lesions so far. This might also support the hypothesis of Skogseid et al. It seems to be unlikely that pituitary lesions may contribute to an adrenal enlargement. In the present study, 9/21 (42%) of the patients with adrenal lesions demonstrated pituitary tumors, including five patients with prolactinoma.
The tumorigenesis in MEN-1 patients is still not well understood. As a tumor-suppressor gene, the Menin gene is likely to be involved in the maintenance of genetic stability and/or regulation pathways of cell growth. Several proteins have been shown to interact with menin, which plays a fundamental role in the regulation of transcription [35–37]. Somatic loss of the menin wild-type allele was neither not detected in benign adrenal tumors of MEN-1 patients [12] nor in sporadic adrenocortical tumors [38–41], whereas heterozygote knock out mice developed PETs, parathyroid hyperplasia, pituitary adenoma, as well as ACC [42]. Both, normal and tumor cells in MEN-1 patients, are known to have an elevated level of chromosome alterations besides 11q13 [43–45]. Therefore, the impact of LOH on 11q13 in MEN-1 patients might be lower than previously suspected with regard to tumorigenesis. Subsequent chromosomal aberrations may lead to neoplasms like in other cancer predisposition syndromes.
In a previous study, we described a potential genotype–phenotype correlation for adrenal involvement in MEN-1. Patients with truncating mutations in exons 2 and 10 had a higher risk of adrenal tumors [15, 46]. We could not confirm this observation in 38 patients reported in this study. Large-scale studies are required to confirm or disprove this possible genotype–phenotype correlation.
Conclusion
Adrenal tumors in MEN-1 occur in more than half of MEN-1 patients and are therefore much more frequent than previously reported. Most of the tumors are benign, small, and nonfunctioning. Although the development of ACC seems to be the exception, we still recommend a close follow-up by EUS or CT at least annually. EUS is the most sensitive imaging modality especially in small lesions. The genotype–phenotype correlation previously reported by our group could not be confirmed in the present study.
References
Chandrasekharappa SC et al (1997) Positional cloning of the gene for multiple endocrine neoplasia-type 1. Science 276(5311):404–407
Wermer P (1954) Genetic aspects of adenomatosis of endocrine glands. Am J Med 16:363–371
Burgess JR, Greenaway TM, Shepherd JJ (1998) Expression of the MEN-1 gene in a large kindred with multiple endocrine neoplasia type 1. J Intern Med 243(6):465–470
Darling TN et al (1997) Multiple facial angiofibromas and collagenomas in patients with multiple endocrine neoplasia type 1. Arch Dermatol 133(7):853–857
Duh QY et al (1987) Carcinoids associated with multiple endocrine neoplasia syndromes. Am J Surg 154(1):142–148
Gibril F et al (2003) Prospective study of thymic carcinoids in patients with multiple endocrine neoplasia type 1. J Clin Endocrinol Metab 88(3):1066–1081
Hofmann M et al (1998) Multiple endocrine neoplasia associated with multiple lipomas. Med Klin (Munich) 93(9):546–549
Marx S et al (1998) Multiple endocrine neoplasia type 1: clinical and genetic topics. Ann Intern Med 129(6):484–494
Lips CJ, Vasen HF, Lamers CB (1984) Multiple endocrine neoplasia syndromes. Crit Rev Oncol Hematol 2(2):117–184
Thakker RV (2000) Multiple endocrine neoplasia type 1. Endocrinol Metab Clin North Am 29(3):541–567
Carty SE et al (1998) The variable penetrance and spectrum of manifestations of multiple endocrine neoplasia type 1. Surgery 124(6):1106–1113; discussion 1113–4
Skogseid B et al (1992) Clinical and genetic features of adrenocortical lesions in multiple endocrine neoplasia type 1. J Clin Endocrinol Metab 75(1):76–81
Skogseid B et al (1995) Adrenal lesion in multiple endocrine neoplasia type 1. Surgery 118(6):1077–1082
Burgess JR et al (1996) Adrenal lesions in a large kindred with multiple endocrine neoplasia type 1. Arch Surg 131(7):699–702
Langer P et al (2002) Adrenal involvement in multiple endocrine neoplasia type 1. World J Surg 26(8):891–896
Barzon L et al (2001) Multiple endocrine neoplasia type 1 and adrenal lesions. J Urol 166(1):24–27
Gibril F et al (2004) Multiple endocrine neoplasia type 1 and Zollinger–Ellison syndrome: a prospective study of 107 cases and comparison with 1009 cases from the literature. Medicine (Baltimore) 83(1):43–83
Cadiot G et al (1999) Prognostic factors in patients with Zollinger–Ellison syndrome and multiple endocrine neoplasia type 1. Groupe d’Etude des Neoplasies Endocriniennes Multiples (GENEM and groupe de Recherche et d’Etude du Syndrome de Zollinger–Ellison (GRESZE). Gastroenterology 116(2):286–293
Ballard HS, Fame B, Hartstock RJ (1964) Familial endocrine adenoma-peptic ulcer complex. Medicine (Baltimore) 43:481–516
Croisier JC, Azerad E, Lubetzki J (1971) L’adenomatose polyendocrinienne (syndrome de Wermer). Semin Hop Paris 47:494–525
Dobbie JW (1969) Adrenocortical nodular hyperplasia: the ageing adrenal. J Pathol 99:1–18
Kokko JP, Brown TC, Berman MM (1967) Adrenal adenoma and hypertension. Lancet 1(7488):468–470
Hedeland H, Ostberg G, Hokfelt B (1968) On the prevalence of adrenocortical adenomas in an autopsy material in relation to hypertension and diabetes. Acta Med Scand 184(3):211–214
Anonymous (2002) NIH state-of-the-science statement on management of the clinically inapparent adrenal mass (“incidentaloma”). NIH Consens State Sci Statements 19(2):1–25
Thompson GB, Young WF Jr (2003) Adrenal incidentaloma. Curr Opin Oncol 15(1):84–90
Kloos RT et al (1997) Diagnostic dilemma of small incidentally discovered adrenal masses: role for 131I-6beta-iodomethyl-norcholesterol scintigraphy. World J Surg 21(1):36–40
Commons RR, Callaway CP (1948) Adenomas of the adrenal cortex. Arch Intern Med 81:37–41
Reinhard C, Saeger W, Schubert B (1996) Adrenocortical nodules in post-mortem series. Development, functional significance, and differentiation from adenomas. Gen Diagn Pathol 141(3–4):203–208
Granger P, Genest J (1970) Autopsy study of adrenals in unselected normotensive and hypertensive patients. Can Med Assoc J 103(1):34–36
Brandi ML et al (2001) Guidelines for diagnosis and therapy of MEN type 1 and type 2. J Clin Endocrinol Metab 86(12):5658–5671
Skogseid B et al (1996) Surgery for asymptomatic pancreatic lesion in multiple endocrine neoplasia type I. World J Surg 20(7):872–876; discussion 877
Brandi ML et al (1987) Familial multiple endocrine neoplasia type I: a new look at pathophysiology. Endocr Rev 8(4):391–405
Trump D et al (1996) Clinical studies of multiple endocrine neoplasia type 1 (MEN1). Q J Med 89(9):653–669
Chico A et al (2000) Genetic, clinical, and biochemical analysis of unrelated Spanish families with multiple endocrine neoplasia type I. Endocr Pract 6(1):13–19
Agarwal SK et al (1999) Menin interacts with the AP1 transcription factor JunD and represses JunD-activated transcription. Cell 96(1):143–152
Kim YS et al (1999) Stable overexpression of MEN1 suppresses tumorigenicity of RAS. Oncogene 18(43):5936–5942
Heppner C et al (2001) The tumor suppressor protein menin interacts with NF-kappaB proteins and inhibits NF-kappaB-mediated transactivation. Oncogene 20(36):4917–4925
Zwermann O et al (2000) Multiple endocrine neoplasia type 1 gene expression is normal in sporadic adrenocortical tumors. Eur J Endocrinol 142(6):689–695
Schulte KM et al (1999) MEN I gene mutations in sporadic adrenal adenomas. Hum Genet 105(6):603–610
Gortz B et al (1999) MEN1 gene mutation analysis of sporadic adrenocortical lesions. Int J Cancer 80(3):373–379
Heppner C et al (1999) MEN1 gene analysis in sporadic adrenocortical neoplasms. J Clin Endocrinol Metab 84(1):216–219
Crabtree JS et al (2001) A mouse model of multiple endocrine neoplasia, type 1, develops multiple endocrine tumors. Proc Natl Acad Sci USA 98(3):1118–1123
Tomassetti P et al (1995) Chromosomal instability in multiple endocrine neoplasia type 1. Cytogenetic evaluation with DEB test. Cancer Genet Cytogenet 79(2):123–126
Scappaticci S et al (1992) Cytogenetics of multiple endocrine neoplasia syndrome. II. Chromosome abnormalities in an insulinoma and a glucagonoma from two subjects with MEN1. Cancer Genet Cytogenet 63(1):17–21
Pannett AA, Thakker RV (2001) Somatic mutations in MEN type 1 tumors, consistent with the Knudson “two-hit” hypothesis. J Clin Endocrinol Metab 86(9):4371–4374
Bartsch DK et al (2000) Pancreaticoduodenal endocrine tumors in multiple endocrine neoplasia type 1: surgery or surveillance? Surgery 128(6):958–966
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Waldmann, J., Bartsch, D.K., Kann, P.H. et al. Adrenal involvement in multiple endocrine neoplasia type 1: results of 7 years prospective screening. Langenbecks Arch Surg 392, 437–443 (2007). https://doi.org/10.1007/s00423-006-0124-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-006-0124-7