Abstract
Background and aims
We analyzed the clinicopathologic features of node-positive gastric carcinoma patients who were long-term survivors (5 years or longer) and evaluated the predictive factors associated with long-term survival.
Patients and methods
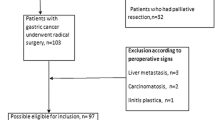
Of 554 node-positive gastric carcinoma patients with curative resection, 161 (29.1%) were long-term survivors, and 393 died of the disease before 5 years.
Results
The long-term survivor group had a recurrence rate of 16.1%, while the recurrence rate was 95.4% in the short-term survivor group (P < 0.05). The mean tumor size in the long-term survivors (4.5 cm) was significantly smaller than that in the short-term survivors (5.3 cm; P < 0.001). A depth of invasion greater than T3 was found more frequently in the short-term survivor group (88.1%) than in the long-term survivor group (70.1%; P < 0.001). Using Cox’s proportional hazard regression model, the only factor found to be an independent, statistically significant prognostic parameter was tumor size (risk ratio, 0.301; 95% confidence interval, 0.10–0.88; P < 0.05).
Conclusion
The tumor size emerged as the only independent, significant factor for the prediction of long-term survival in node-positive gastric carcinoma patients with curative resection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The presence or absence of lymph node metastasis is one of the most important clinicopathologic factors influencing the prognosis of patients with gastric carcinoma [1–4]. Several studies of the prognostic factors in patients with node-positive gastric carcinoma have been reported, but they have involved only a small number of patients with node-positive gastric carcinoma. We analyzed the clinicopathologic features of node-positive gastric carcinoma patients after resection with curative intent who were long-term survivors (5 years or longer) and compared them with the features of those who died before 5 years. We also evaluated the predictive factors associated with long-term survival.
Materials and methods
Patients and specimens
From 1986 to 2000, 2,848 patients with gastric carcinoma were treated by the Division of Gastroenterologic Surgery, Department of Surgery, Chonnam National University Medical School, Gwangju, Korea. Of these, 554 were in the node-positive group with curative resection. We analyzed the clinicopathologic features of the node-positive gastric carcinoma patients who were long-term survivors (5 years or longer).
Information on the patient’s age, gender, tumor size, tumor location, macroscopic appearance, depth of invasion, level of lymph node invasion, histologic type, hepatic metastasis, peritoneal dissemination, stage at initial diagnosis, operative type, and recurrence pattern was obtained from the hospital records. The American Joint Committee on Cancer (AJCC) tumor–node–metastasis (TNM) staging system was used for pathologic staging [5]. Histological evaluation was performed according to the Japanese General Rules for Gastric Cancer Study in Surgery and Pathology [6].
Patient follow-up lasted until death or until the cut-off date of December 31, 2000. At the time of the last follow-up, 108 patients (3.8%) had been lost to follow-up. Generally, patients return every 3 months for the first 2 years and then every 6 months until at least the end of the fifth year. We carried out dissection above the D2 lymph node in all patients in our department. An extended total gastrectomy with transhiatal resection of the distal esophagus was performed in patients with esophageal invasion.
Statistical analysis
The survival rates of the patients were calculated using the Kaplan–Meier method, and the relative prognostic importance of the parameters was investigated using Cox’s proportional hazards model. The chi-squared test was used to evaluate the statistical significance of differences, and P values <0.05 were considered statistically significant.
Results
Table 1 summarizes the clinical findings of 161 (29.1%) node-positive gastric carcinoma patients who lived longer than 5 years (long-term survivors, LTS) and 393 (70.9%) node-positive gastric carcinoma patients who died before 5 years (short-term survivors, STS). There was a significant difference in the mean age of patients between the two groups (LTS vs STS: 53.7 vs 57.1 years; P < 0.001). Of the 161 patients, 102 (63.4%) were male and 59 (36.6%) were female. There were 264 males (67.2%) and 129 females (32.8%) in the STS group of 393 patients. Although there were more males than females in each group, there was no statistically significant difference between the groups. Subtotal gastrectomy was the procedure most frequently performed in the LTS group (72.7% of cases). In contrast, total gastrectomy was performed more frequently in the STS group (36.4% of cases) than in the LTS group (25.5%; P < 0.05). There was a 16.1% recurrence rate in the LTS group and a 95.4% recurrence rate in the STS group; this rate was significantly different between the two groups (P < 0.05). Peritoneal recurrence was the predominant recurrence pattern in both groups.
The histopathologic findings are listed in Table 2 The mean tumor size in the LTS group (4.5 cm) was significantly smaller than that in the STS group (5.3 cm; P < 0.001). Most of the gastric carcinomas were located in the lower third of the stomach in both the LTS (100 cases; 62.2%) and STS groups (233 cases; 59.3%); the difference in the location was not significant. A depth of invasion greater than T3 was found more frequently in stomachs removed from the STS group (88.1%) than from the LTS group (70.1%; P < 0.001). A lymph node level above the N2 group was invaded more frequently in the STS group than in the LTS group (55.7 vs 37.9%, respectively; P < 0.001). There was no significant difference between the LTS and STS groups with respect to the number of hepatic metastases or peritoneal dissemination. There was also no significant difference in histologic type between the two groups. In the LTS group, 36 cases (22.3%) were classified as either stage I or II at the time of initial diagnosis. In contrast, 92.6% of the patients in the STS group were classified as advanced stage (≥stage III; P < 0.001). Using Cox’s proportional hazard regression model, only one factor was found to be an independent, statistically significant prognostic parameter, tumor size (risk ratio, 0.301; 95% confidence interval, 0.10–0.88; P < 0.05) (Table 3). The 10-year survival rate was lower for patients with tumor size more than 50 mm compared to that of patients with tumor size less than 50 mm (60.6 vs 77.7%) (P < 0.05) (Fig. 1).
Discussion and conclusion
The overall 5-year survival rate for all gastric carcinoma patients who undergo surgery is only 20–30%. Even after curative resection, only 30–50% of patients are still alive after 5 years [7]. Lymph node involvement is associated with a poor outcome in gastric carcinoma patients, even with curative resection. Therefore, identifying the factors associated with favorable survival in patients with node-positive gastric carcinoma is important. In this study, we compared node-positive gastric carcinoma patients who were long-term survivors and node-positive patients who died before 5 years to clarify the predictive factors associated with long-term survival.
Whether tumor size independently correlates with prognosis is still controversial. Some investigators stress that tumor size is an independent prognostic factor, but others claim that tumor size does not independently influence survival. Adachi et al. [8] reported that tumor size served as a simple prognostic indicator for long-term survival in gastric carcinoma, finding that patient survival showed a stepwise correlation with tumor size (<4 vs 4–10 vs ≥10 cm). By contrast, Maruyama [2] and Yokota et al. [9] concluded that the depth of invasion and tumor location were more important than tumor size as prognostic factors. In the present study, there was a significant difference in tumor size between the long-term and short-term survivors of node-positive gastric carcinoma; the LTS were characterized by a smaller tumor size (LTS vs STS: 4.5 vs 5.3 cm). Furthermore, tumor size emerged as the only independent significant factor for predicting long-term survival in node-positive gastric carcinoma patients with curative resection.
The use of age as a prognostic factor is another controversial subject [3, 10]. Moriguchi et al. [11] reported that the age at operation was a significant prognostic factor in patients with early gastric carcinoma. Adachi et al. [12] reported that patient age was the second most important prognostic indicator in patients with gastric carcinoma. Furthermore, some investigators found that survival rates decreased in elderly patients [13, 14]. We found that there was a statistical difference in the mean age of patients between the two groups (LTS vs STS: 53.7 vs 57.1 years). However, in contrast to other reports, our study showed that in patients with node-positive gastric carcinoma, age had no effect on long-term survival, according to Cox’s proportional hazard regression model.
In gastric carcinoma, the depth of wall invasion is another important prognostic factor in addition to lymph node invasion. Mori et al. [15] and Adachi et al. [12] demonstrated that the depth of wall invasion provided useful prognostic information in patients with gastric carcinoma. However, our results disagreed with their conclusions, as we found that serosal invasion did not emerge as an independent, statistically significant prognostic parameter for node-positive gastric carcinoma patients with curative resection who showed long-term survival according to the Cox’s proportional hazard regression model used in this study. Moriguchi et al. [16] also reported that it is unclear whether the degree of invasion correlates with prognosis in advanced carcinoma.
Although some investigators have reported that the histologic type of the gastric carcinoma correlated with patient survival [17], there was no prognostic value of tumor differentiation in node-positive gastric carcinoma patients who showed long-term survival in this study. Our results are in agreement with the data of previous studies [8, 18].
Some investigators have suggested that the extent of gastric resection was a significant independent predictor of survival in node-positive gastric carcinoma patients. Kim et al. [19] found that patients with subtotal gastrectomy showed a significantly better long-term prognosis than patients who had total gastrectomy. They interpreted their results in terms of indications for total gastrectomy and the relative risks for more distant and extensive lymph node metastasis. We also found that lymph nodes were invaded more extensively (≥N2) in node-positive gastric carcinoma patients who died before 5 years compared with those who showed long-term survival (STS vs LTS: 37.9 vs 55.1%; P < 0.001). But, operative type did not affect the survival rate in multivariate analysis.
Surgery is the only potentially curative modality for localized gastric carcinoma. It is reasonable to conclude that the extent of lymph node dissection did not influence the survival of patients without lymph node metastasis, but it is clear that curability is very important in achieving the long-term survival of gastric carcinoma patients with lymph node metastasis [20]. Thus, many investigators have recommended ≥D2 lymph node dissection for the curative treatment of node-positive gastric carcinoma [21]. We are in agreement with their recommendation and performed dissection above the D2 lymph node level in all patients with node-positive gastric carcinoma in our institution.
In conclusion, this study demonstrated that long-term survival of node-positive gastric carcinoma patients after curative resection is related to small tumor size. Thus, tumor size may be clinically useful for the prediction of survival after curative resection of node-positive gastric carcinoma patients.
References
Bozzetti F, Bonfanti G, Morabito A, Bufalino R, Menotti V, Andreola S, Doci R, Gennari L (1986) Multifactorial approach for the prognosis of patients with carcinoma of the stomach after curative resection. Surg Gynecol Obstet 162:229–234
Maruyama K (1987) The most important prognostic factors for gastric cancer patients. Scand J Gastroenterol 22:63–68
Adachi Y, Ogawa Y, Sasaki Y, Yukaya H, Mori M, Sugimachi K (1994) A clinicopathologic study of gastric carcinoma with reference to age of patients. J Clin Gastroenterol 18:287–290
Siewert JR, Bottcher K, Stein HJ, Roder JD (1998) Relevant prognostic factors in gastric cancer: ten-year results of the German Gastric Cancer Study. Ann Surg 228:449–461
Greene FL, Page DL, Fleming ID (2002) AJCC cancer staging manual, 6th ed. Springer, Berlin Heidelberg New York
Japanese Gastric Cancer Association (1998) Japanese classification of gastric carcinoma, 2nd English edn. Gastric Cancer 1:10–24
Neugut AI, Hayek M, Howe G (1996) Epidemiology of gastric cancer. Semin Oncol 28:281–291
Adachi Y, Mori M, Maehara Y, Sugimachi K (1995) Long-term survival after resection for advanced gastric carcinoma. J Clin Gastroenterol 21:208–210
Yokota T, Ishiyama S, Saito T, Teshima S, Yamada Y, Iwamoto K, Takahashi M, Murata K, Yamauchi H (2002) Is tumor size a prognostic indicator for gastric carcinoma? Anticancer Res 22:3673–3677
Mitsudomi T, Matsusaka T, Wakasugi K, Takenaka M, Kume K, Fujinaga Y, Teraoka H, Iwashita A (1989) A clinicopathological study of gastric cancer with special reference to age of the patients: an analysis of 1,630 cases. World J Surg 13:225–230
Moriguchi S, Odaka T, Hayashi Y, Nose Y, Maehara Y, Korenaga D, Sugimachi K (1991) Death due to recurrence following curative resection of early gastric cancer depends on age of the patient. Br J Cancer 64:555–558
Adachi Y, Mori M, Maehara Y, Kitano S, Sugimachi K (1997) Prognostic factors of node-negative gastric carcinoma: univariate and multivariate analyses. J Am Coll Surg 184:373–377
Houry S, Amenabar J, Rezvani A, Huguier M (1994) Should patients over 80 years old be operated on for colorectal or gastric cancer? Hepatogastroenterology 41:521–525
Takeda J, Tanaka T, Koufuji K, Kodama I, Tsuji Y, Kakegawa T (1994) Gastric cancer surgery in patients aged at least 80 years old. Hepatogastroenterology 41:516–520
Mori M, Sugimachi K (1990) Clinicopathologic studies of gastric carcinoma. Semin Surg Oncol 6:19–27
Moriguchi S, Maehara Y, Korenaga D, Sugimachi K, Hayashi Y, Nose Y (1992) Prediction of survival time after curative surgery for advanced gastric cancer. Eur J Surg Oncol 18:287–292
Noda S, Soejima K, Inokuchi K (1980) Clinicopathological analysis of the intestinal type and diffuse type of gastric carcinoma. Jpn J Surg 10:277–283
Nakamura K, Ueyama T, Yao T, Xuan ZX, Ambe K, Adachi Y, Yakeishi Y, Matsukuma A, Enjoji M (1992) Pathology and prognosis of gastric carcinoma. Findings in 10,000 patients who underwent primary gastrectomy. Cancer 70:1030–1037
Kim J, Cheong JH, Hyung WJ, Shen J, Choi SH, Noh SH (2004) Predictors of long-term survival in pN3 gastric cancer patients. J Surg Oncol 88:9–13
Adachi Y, Oshiro T, Mori M, Maehara Y, Sugimachi K (1997) Tumor size as a simple prognostic indicator for gastric carcinoma. Ann Surg Oncol 4:137–140
Harrison LE, Karpeh MS, Brennan MF (1998) Extended lymphadenectomy is associated with a survival benefit for node-negative gastric cancer. J Gastrointest Surg 2:126–131
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, D.Y., Joo, J.K., Park, Y.K. et al. Predictors of long-term survival in node-positive gastric carcinoma patients with curative resection. Langenbecks Arch Surg 392, 131–134 (2007). https://doi.org/10.1007/s00423-006-0114-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-006-0114-9