Abstract
Background and aims
Intra-operative parathyroid hormone (IOPTH) results are not known in the setting of tertiary hyperparathyroidism (HPT) after renal transplantation.
Materials and methods
A retrospective analysis of 35 tertiary HPT patients who all underwent subtotal parathyroidectomy and IOPTH monitoring was conducted.
Results
The mean follow-up time was 2.2±1.4 years. Thirty-four patients were cured; one patient (2.8%) had a persistent disease and was cured after reoperation. Median parathyroid hormone (PTH) (median percent decrease from highest) at baseline and at 5, 10, 20, and 30 min were 244, 78 (69%), 63 (75%), 53 (79%), and 49 pg/ml (83%), respectively. Four patients who were cured had a decrease of <50% at 5 min and two of them had a decrease of <50% at 10 min. The patient with persistent disease had a decrease of >50% at 10 min. The sensitivity of the test was 94% at 10 min using the Miami criteria.
Conclusion
This study shows that IOPTH in tertiary hyperparathyroidism has a high sensitivity. However, because of the low risk of persistent hyperparathyroidism when a subtotal parathyroidectomy is performed, its potential impact on the overall success rate is very small. We therefore do not recommend the routine use of IOPTH in tertiary hyperparathyroidism.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In patients with tertiary hyperparathyroidism (HPT), even when renal function returns to normal after kidney transplantation, the secretion of parathyroid hormone (PTH) does not. The reason for this is because PTH secretion has become autonomous and no longer responses to calcium and calcitriol levels. The association of normal renal function and high PTH level leads then to hypercalcemia. When hypercalcemia persists for more than 1 year after kidney transplantation or when patients develop complication related to hyperparathyroidism, parathyroidectomy is currently the only available curative treatment. The standard surgical approach for patients with tertiary hyperparathyroidism is subtotal parathyroidectomy or total parathyroidectomy with autotransplantation [1–7]; however, some authors have advocated a less-than-subtotal resection in some patients, describing up to one third of their patients with limited gland involvement (single or double ‘adenoma’) [8–11]. Because it is sometimes difficult to find all parathyroid glands, particularly when patients have more than four glands, and to leave a remnant of adequate size (big enough to avoid hypoparathyroidism but small enough to avoid persistent and recurrent disease), it would be helpful to have a reliable intra-operative indicator of cure. Intra-operative parathyroid hormone (IOPTH) has been proven to be helpful in primary HPT, particularly when a unilateral approach is used, and it has an accuracy of >95% in this setting [12–17]. If IOPTH has the same sensitivity and specificity in tertiary HPT, it could help define the extent of parathyroidectomy. The accuracy of IOPTH in renal HPT is not clearly described and different authors have described conflicting results [18–20]. Moreover, many papers describe IOPTH independently of the renal function (in patients under dialysis and patients after kidney transplantation) [18, 20] and few have focused their attention on patients with tertiary HPT after kidney transplantation.
Moreover, because tertiary hyperparathyroidism is a unique situation in which a multiglandular disease is associated with normal renal function in most of the patients, the results of IOPTH in this situation could help understand how to interpret the IOPTH results in patients with primary hyperparathyroidism and multiglangular disease. The aim of this study was to assess the accuracy of IOPTH in tertiary hyperparathyroidism and to describe the results according to the renal function.
Materials and methods
Between January 1998 and December 2003, 35 consecutive patients were operated in our center for tertiary hyperparathyroidism, as defined by persistently high calcium and PTH levels after kidney transplantation. The criteria for surgery were asymptomatic persisting hypercalcemia (>10.5 mg/dl) with an inappropriately high PTH level or the presence of signs or symptoms of tertiary HPT with elevated PTH, usually more than 1 year after kidney transplantation.
All patients underwent a complete bilateral neck parathyroid exploration, a transcervical thymectomy, and a subtotal parathyroidectomy, leaving a single remnant of the size of two normal parathyroid glands, as previously described [7]. All patients underwent IOPTH monitoring. Peripheral blood samples were withdrawn at anesthesia induction, before excision of the first parathyroid gland and 5, 10, 20, and 30 min after excision of the last parathyroid gland. Intra-operative intact PTH levels were measured using a commercially available two-site assay (Nichols Institute Diagnostics, San Juan Capistrano, CA, USA; normal range 10–60 pg/ml). This assay is known to detect 1–84 PTH and other fragments, including 7–84 PTH [21]. The percentage of decrease was calculated using the highest of the two pre-excision values, and the Miami criteria were used to define a positive or negative IOPTH result (decrease of >50% from highest value 10 min after resection of the last gland).
The post-operative PTH level was always intact PTH, measured by a standard radioimmunometric assay (reference range 10–60 pg/ml). The serum calcium levels were corrected to the protein level using standard formulas. Hypoparathyroidism was defined as hypocalcemia and/or infranormal PTH level (<10 pg/ml). Persistent or recurrent disease was defined as hypercalcemia (serum calcium level >10.5 mg/dl) and/or PTH level above the recommended K/DOQI target range according to renal function [22] during the first 6 months after parathyroidectomy and after 6 months, respectively. Glomerular filtration rate (GFR) was calculated using the Cockroft and Gault formula.
The results are presented as mean values±standard deviation unless otherwise stated. Statistical analysis was performed with SPSS 11.0.1 (SPSS, Chicago, IL, USA). Graphs were created with SPSS and legends were added with Adobe Photoshop Elements (Adobe Systems, San Jose, CA, USA) when needed. Comparisons between pre- and post-operative serum calcium and PTH level were made with paired-samples t test. A p value was considered statistically significant when <0.05.
Results
Surgical results
There were 35 patients, 18 men and 17 women. The duration of dialysis before kidney transplantation was 2.7±2.8 years. The time between kidney transplantation and parathyroidectomy was 5.3±5.1 years. The age at parathyroidectomy was 47±11 years. GFR at parathyroidectomy was 53±21 ml/min. The preoperative calcium level was 11.2±1.0 mg/dl and PTH level was 306±340 pg/ml.
All patients underwent a bilateral neck exploration with subtotal parathyroidectomy and transcervical bilateral thymectomy. During the operation, four cervical glands were found in all patients. Hyperplastic glands were found in all patients at microscopic examination. Parathyroid hyperplasia was defined as an increased parenchymal mass in the absence of a rim of compressed parathyroid tissue [23]. The total excised parathyroid tissue weighed 1,073±775 mg.
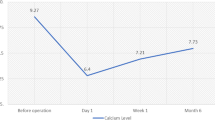
There was no post-operative death. Two patients under anticoagulant therapy were reoperated for hemorrhage and eight patients had transient ‘simple’ hypocalcemia (without hypocalcemia-related complications). At day 1, the calcium level was 8.3±1.0 mg/dl and the PTH level was 28±14 pg/ml. Follow-up was available for all patients and was 2.2±1.4 years. One patient died of cardiovascular complication; one patient was on dialysis again. At follow-up, GFR was 49±25 ml/, serum calcium level was 9.1±0.7 mg/dl, and PTH level was 66±48 pg/ml. One patient presented with a persistent disease and underwent a successful re-excision of the parathyroid remnant. At follow-up, no patient was hypoparathyroid and all had PTH level in the target range recommended by the K/DOQI [22].
IOPTH results
As shown in Fig. 1, less than 50% of the patients reached normal PTH level 10 min after the resection of the last gland (median value at 10 min, 63 pg/ml). At 30 min, 12 patients (34%) still had PTH values above the normal range. When the percentage of decrease is studied, IOPTH did not decrease by >50% in two patients who were cured at 10 min, while all patients had a decrease of >50% at 20 min, including the patient with a persistent disease (Fig. 2). The sensitivity of IOPTH was 94% and the positive predictive value was 97% using the Miami criteria. However, the specificity and the negative predictive value could not be calculated because only one patient had a persistent disease and no patient had a true negative IOPTH result (less than 50% decrease with persistent disease).
Taking the renal function into account did not add any precision at 10 min; in effect, more than 50% of the patients in both groups had a PTH value above the normal range (the median PTH value was 70 pg/ml in the group with a GFR <60 ml/min and was 61 pg/ml in the group with a GFR >60 ml/min). However, at 30 min, only 2 (13%) of the 15 patients in the ≥60 ml/min group did not have PTH level in the normal range, while 11 (55%) of the 20 patients in the group <60 ml/min were in that situation. (Fig. 3). One patient who was cured in each group had a <50% decrease at 10 min.
Discussion
This study shows that IOPTH during parathyroidectomy for tertiary hyperparathyroidism after renal transplantation has a high sensitivity (94%) and positive predictive value (97%) using the Miami criteria for cure (decrease of >50% from highest value 10 min after excision of the last parathyroid gland). However, because only one patient had a persistent or recurrent hyperparathyroidism and no patient had a true negative test, the specificity and negative predictive value could not be calculated.
Thanks to the short half-life of PTH (1 to 3 min) in patients with normal renal function, IOPTH has been developed as a reliable tool to predict the success of parathyroidectomy in primary hyperparathyroidism [12, 13, 24]. However, PTH half-life is increased in patients with renal insufficiency, due not only to a prolonged half-life of the 1–84 PTH but also to the particularly prolonged half-life of the 7–84 fragment, co-detected by most of the standard PTH assays [18, 19, 21, 25, 26]. Therefore, the accuracy of IOPTH in patients with renal insufficiency is debated on and its ability to predict cure or failure is highly variable according to the parameters studied [18–20, 25, 27]. After renal transplantation, most of the patients retrieve good renal function; however, the accuracy of IOPTH in patients with tertiary hyperparathyroidism is not known. Previous reports have shown that IOPTH changed the management in 5 of 32 patients studied with only one false positive result; however, the long-term cure rate is given by the calcium level only and not the PTH level in this study [28]. Because of the retrospective nature of our study, the IOPTH did not change the management of any of the 35 patients. However, even if performed in a prospective way, it could have led to an unnecessary, more aggressive resection in two patients who had a false-negative IOPTH result and it would not have permitted to cure the patient who had persistent disease as he had a false-positive result. Taking renal function into account increases the accuracy of the IOPTH; however, it was only at 30 min after the resection of the last gland in this study, rendering this test less useful because of the waiting time associated to such a long delay.
In this study, 20 of the 35 patients (57%) had GFR <60 ml/min, showing a significant decrease in renal function despite the renal graft, which is in accordance with the relatively long time interval between renal transplantation and parathyroidectomy (5.3 years in this study) and with the natural history of renal transplantation [29]. At this stage of renal insufficiency, bone resistance to PTH increases, PTH secretion is stimulated, and PTH and its fragments begin to accumulate [22]. The increase in PTH levels makes it difficult to reach the true normal range during parathyroidectomy because of the sometimes very high preoperative PTH levels (maximum of 3,190 pg/ml in this study). Moreover, the increased PTH half-life (whole and fragments) seen in renal insufficiency [18, 19, 30] makes it difficult to achieve a significant and reproducible decrease in a reasonable amount of time. Different criteria for cure have been proposed for IOPTH in renal hyperparathyroidism such as >50% decrease at 20 min [18], >82% at 15 min [20], or >60% at 10 min [19]. However, none of them could predict cure or failure in 100% of the patients. Our study in patients with tertiary hyperparathyroidism confirms this and no criteria had an accuracy of 100%.
Two recent reports show an increased accuracy when the newest whole-PTH assay was used. Yamashita et al. [31] had a decline of >50% in 17 of 18 patients with renal hyperparathyroidism and Kaczirek et al. [30] showed that IOPTH fell in the normal range only after adequate resection in 30 of 30 patients (total or subtotal parathyroidectomy) and could predict insufficient resection in four of the five patients who had persistent hyperparathyroidism. In our study, we have used a first-generation IOPTH assay and further investigations are needed to confirm the increased accuracy of whole-PTH assay in the management of tertiary hyperparathyroidism after renal transplantation.
In conclusion, this study shows that IOPTH has a high sensitivity and positive predictive value in tertiary hyperparathyroidism, meaning that it is able to correctly predict cure when the patient is cured. However, such a test is most useful when the patient is not cured and, with a systematic subtotal parathyroidectomy and cervical thymectomy, the risk appears to be small and the potential benefit of IOPTH seems to be very low, particularly with the assays co-detecting PTH fragments. We therefore do not recommend the routine use of IOPTH in tertiary hyperparathyroidism when a systematic subtotal parathyroidectomy is performed.
References
Rothmund M, Wagner PK, Schark C (1991) Subtotal parathyroidectomy versus total parathyroidectomy and autotransplantation in secondary hyperparathyroidism: a randomized trial. World J Surg 15(6):745–750
Kerby JD, Rue LW, Blair H, Hudson S, Sellers MT, Diethelm AG (1998) Operative treatment of tertiary hyperparathyroidism: a single-center experience. Ann Surg 227(6):878–886
Tominaga Y, Uchida K, Haba T, Katayama A, Sato T, Hibi Y, Numano M, Tanaka Y, Inagaki H, Watanabe I, Hachisuka T, Takagi H (2001) More than 1,000 cases of total parathyroidectomy with forearm autograft for renal hyperparathyroidism. Am J Kidney Dis 38(4 Suppl 1):S168–S171
Gasparri G, Camandona M, Abbona GC, Papotti M, Jeantet A, Radice E, Mullineris B, Dei Poli M (2001) Secondary and tertiary hyperparathyroidism: causes of recurrent disease after 446 parathyroidectomies. Ann Surg 233(1):65–69
Sancho JJ, Sitges-Serra A (1997) Surgical approach to secondary hyperparathyroidism. In: Clark OH, Duh QY (eds) Textbook of endocrine surgery, vol 50. Saunders, San Francisco, pp 403–409
Kebebew E, Duh QY, Clark OH (2004) Tertiary hyperparathyroidism: histologic patterns of disease and results of parathyroidectomy. Arch Surg 139(9):974–977
Triponez F, Dosseh D, Hazzan M, Noel C, Vanhille P, Proye CA (2005) Subtotal parathyroidectomy with thymectomy for autonomous hyperparathyroidism after renal transplantation. Br J Surg 92(10):1282–1287
Nichol PF, Starling JR, Mack E, Klovning JJ, Becker BN, Chen H (2002) Long-term follow-up of patients with tertiary hyperparathyroidism treated by resection of a single or double adenoma. Ann Surg 235(5):673–678
Nichol PF, Mack E, Bianco J, Hayman A, Starling JR, Chen H (2003) Radioguided parathyroidectomy in patients with secondary and tertiary hyperparathyroidism. Surgery 134(4):713–717
Kilgo MS, Pirsch JD, Warner TF, Starling JR (1998) Tertiary hyperparathyroidism after renal transplantation: surgical strategy. Surgery 124(4):677–683
Cintin C, Karstrup S, Ladefoged SD, Joffe P (1994) Tertiary hyperparathyroidism treated by ultrasonically guided percutaneous fine-needle ethanol injection. Nephron 68(2):217–220
Nussbaum SR, Thompson AR, Hutcheson KA, Gaz RD, Wang CA (1988) Intraoperative measurement of parathyroid hormone in the surgical management of hyperparathyroidism. Surgery 104(6):1121–1127
Irvin GL 3rd, Deriso GT 3rd (1994) A new, practical intraoperative parathyroid hormone assay. Am J Surg 168(5):466–468
Johnson LR, Doherty G, Lairmore T, Moley JF, Brunt LM, Koenig J, Scott MG (2001) Evaluation of the performance and clinical impact of a rapid intraoperative parathyroid hormone assay in conjunction with preoperative imaging and concise parathyroidectomy. Clin Chem 47(5):919–925
Udelsman R (2002) Six hundred fifty-six consecutive explorations for primary hyperparathyroidism. Ann Surg 235(5):665–670
Fahy BN, Bold RJ, Beckett L, Schneider PD (2002) Modern parathyroid surgery: a cost–benefit analysis of localizing strategies. Arch Surg 137(8):917–922
Westerdahl J, Bergenfelz A (2004) Sestamibi scan-directed parathyroid surgery: potentially high failure rate without measurement of intraoperative parathyroid hormone. World J Surg 28(11):1132–1138
Lokey J, Pattou F, Mondragon-Sanchez A, Minuto M, Mullineris B, Wambergue F, Foissac-Geroux P, Noel C, de Sagazan HL, VanHille P, Proye CA (2000) Intraoperative decay profile of intact (1–84) parathyroid hormone in surgery for renal hyperparathyroidism—a consecutive series of 80 patients. Surgery 128(6):1029–1034
Chou FF, Lee CH, Chen JB, Hsu KT, Sheen-Chen SM (2002) Intraoperative parathyroid hormone measurement in patients with secondary hyperparathyroidism. Arch Surg 137(3):341–344
Seehofer D, Rayes N, Klupp J, Steinmuller T, Ulrich F, Muller C, Schindler R, Frei U, Neuhaus P (2005) Predictive value of intact parathyroid hormone measurement during surgery for renal hyperparathyroidism. Langenbecks Arch Surg 390(3):222–229
Lepage R, Roy L, Brossard JH, Rousseau L, Dorais C, Lazure C, D’Amour P (1998) A non-(1–84) circulating parathyroid hormone (PTH) fragment interferes significantly with intact PTH commercial assay measurements in uremic samples. Clin Chem 44(4):805–809
National Kidney Foundation (2003) K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis 42(4 Suppl 3):1–201
Krause MW, Hedinger CE (1985) Pathologic study of parathyroid glands in tertiary hyperparathyroidism. Hum Pathol 16(8):772–784
Libutti SK, Alexander HR, Bartlett DL, Sampson ML, Ruddel ME, Skarulis M, Marx SJ, Spiegel AM, Simmonds W, Remaley AT (1996) Kinetic analysis of the rapid intraoperative parathyroid hormone assay in patients during operation for hyperparathyroidism. Surgery 126(6):1145–1150
Kaczirek K, Riss P, Wunderer G, Prager G, Asari R, Scheuba C, Bieglmayer C, Niederle B (2005) Quick PTH assay cannot predict incomplete parathyroidectomy in patients with renal hyperparathyroidism. Surgery 137(4):431–435
Boudou P, Ibrahim F, Cormier C, Chabas A, Sarfati E, Souberbielle JC (2005) Third- or second-generation parathyroid hormone assays: a remaining debate in the diagnosis of primary hyperparathyroidism. J Clin Endocrinol Metab 90(12):6370–6372
Lorenz K, Dralle H (2005) Will intra-operative measurement of parathyroid hormone alter the surgical concept of renal hyperparathyroidism? Langenbecks Arch Surg 390(4):277–279
Haustein SV, Mack E, Starling JR, Chen H (2005) The role of intraoperative parathyroid hormone testing in patients with tertiary hyperparathyroidism after renal transplantation. Surgery 138(6):1066–1071
US Renal Data System (USRDS) (2004) Annual data report: atlas of end-stage renal disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD
Kaczirek K, Prager G, Riss P, Wunderer G, Asari R, Scheuba C, Bieglmayer C, Niederle B (2006) Novel parathyroid hormone (1–84) assay as basis for parathyroid hormone monitoring in renal hyperparathyroidism. Arch Surg 141(2):129–134
Yamashita H, Gao P, Cantor T, Noguchi S, Uchino S, Watanabe S, Ogawa T, Kawamoto H, Fukagawa M (2004) Comparison of parathyroid hormone levels from the intact and whole parathyroid hormone assays after parathyroidectomy for primary and secondary hyperparathyroidism. Surgery 135(2):149–156
Acknowledgements
We thank Dr. R. Azar, Dr. F. Dequiedt, Dr. P. Duchatelle, Dr. F. Dumont, Dr. D. Fleury, Dr. F. Lamotte, Dr. H. Lemonies de Sagazan, Dr. A. Tacquet, Dr. V. Lemaitre, Dr. P. Vanhille, and Dr. F. Wambergue for their help in the clinical follow-up of these patients. F.T. was supported by a grant from the University Hospital of Geneva for his fellowship in Lille.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Triponez, F., Dosseh, D., Hazzan, M. et al. Accuracy of intra-operative PTH measurement during subtotal parathyroidectomy for tertiary hyperparathyroidism after renal transplantation. Langenbecks Arch Surg 391, 561–565 (2006). https://doi.org/10.1007/s00423-006-0070-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-006-0070-4