Abstract
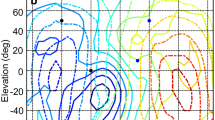
Animals, including humans, use interaural time differences (ITDs) that arise from different sound path lengths to the two ears as a cue of horizontal sound source location. The nature of the neural code for ITD is still controversial. Current models differentiate between two population codes: either a map-like rate-place code of ITD along an array of neurons, consistent with a large body of data in the barn owl, or a population rate code, consistent with data from small mammals. Recently, it was proposed that these different codes reflect optimal coding strategies that depend on head size and sound frequency. The chicken makes an excellent test case of this proposal because its physical prerequisites are similar to small mammals, yet it shares a more recent common ancestry with the owl. We show here that, like in the barn owl, the brainstem nucleus laminaris in mature chickens displayed the major features of a place code of ITD. ITD was topographically represented in the maximal responses of neurons along each isofrequency band, covering approximately the contralateral acoustic hemisphere. Furthermore, the represented ITD range appeared to change with frequency, consistent with a pressure gradient receiver mechanism in the avian middle ear. At very low frequencies, below 400Hz, maximal neural responses were symmetrically distributed around zero ITD and it remained unclear whether there was a topographic representation. These findings do not agree with the above predictions for optimal coding and thus revive the discussion as to what determines the neural coding strategies for ITDs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ashida G, Abe K, Funabiki K (2007a) Biophysical mechanism for forming sound-analogue membrane potentials by phase-locked synaptic inputs in owl’s auditory neuron. 30th Midwinter research meeting of the Association for Research in Otolaryngology, Denver, p 148
Ashida G, Abe K, Funabiki K and Konishi M (2007b). Passive soma facilitates submillisecond coincidence detection in the owl’s auditory system. J Neurophysiol 97: 2267–282
Batra R, Kuwada S and Fitzpatrick DC (1997). Sensitivity to interaural temporal disparities of low- and high-frequency neurons in the superior olivary complex. I. Heterogeneity of responses. J Neurophysiol 78: 1222–236
Batra R and Yin TCT (2004). Cross correlation by neurons of the medial superior olive: a reexamination. JARO 5: 238–52
Beckius GE, Batra R and Oliver DL (1999). Axons from anteroventral cochlear nucleus that terminate in medial superior olive of cat: observations related to delay lines. J Neurosci 19: 3146–161
Blauert J and Lindemann W (1986). Spatial mapping of intracranial auditory events for various degrees of interaural coherence. J Acoust Soc Am 79: 806–13
Brand A, Behrend O, Marquardt T, McAlpine D and Grothe B (2002). Precise inhibition is essential for microsecond interaural time difference coding. Nature 417: 543–47
Calford MB and Piddington RW (1988). Avian interaural canal enhances interaural delay. J Comp Physiol A 162: 503–10
Carr CE and Konishi M (1990). A circuit for detection of interaural time differences in the brain stem of the barn owl. J Neurosci 10: 3227–246
Carr CE and Köppl C (2004). Coding interaural time differences at low best frequencies in the barn owl. J Physiol (Paris) 98: 99–12
Chiappe LM and Dyke GJ (2002). The mesozoic radiation of birds. Annu Rev Ecol Sys 33: 91–24
Christensen-Dalsgaard J (2005). Directional hearing in nonmammalian tetrapods. In: Popper, AN and Fay, RR (eds) Sound source localization, pp 67–23. Springer, New York
Colburn HS and Kulkarni A (2005). Models of sound localization. In: Popper, AN and Fay, RR (eds) Sound source localization, pp 272–16. Springer, New York
Goldberg JM and Brown PB (1969). Response of binaural neurons of dog superior olivary complex to dichotic tonal stimuli: some physiological mechanisms of sound localization. J Neurophysiol 32: 613–36
Grantham DW and Wightman FL (1979). Detectability of a pulsed tone in the presence of a masker with time varying inter aural correlation. J Acoust Soc Am 65: 1509–517
Grothe B (2003). New roles for synaptic inhibition in sound localization. Nat Rev Neurosci 4: 540–50
Grothe B, Carr CE, Cassedy JH, Fritzsch B and Köppl C (2004). The evolution of central pathways and their neural processing patterns. In: Manley, GA, Popper, A, and Fay, RR (eds) Evolution of the vertebrate auditory system, pp 289–59. Springer, New York
Guinan JJ, Norris BE and Guinan SS (1972). Single auditory units in the superior olivary complex. II: locations of unit categories and tonotopic organization. Int J Neurosci 4: 147–66
Hancock KE and Delgutte B (2004). A physiologically based model of interaural time difference discrimination. J Neurosci 24: 7110–117
Harper NS and McAlpine D (2004). Optimal neural population coding of an auditory spatial cue. Nature 430: 682–86
Hyson RL, Overholt EM and Lippe WR (1994). Cochlear microphonic measurements of interaural time differences in the chick. Hear Res 81: 109–18
Jeffress LA (1948). A place theory of sound localization. J Comp Physiol Psychol 41: 35–9
Jeffress LA, Blodgett HC, Sandel TT and Wood CL (1956). Masking of tonal signals. J Acoust Soc Am 28: 416–26
Jeffress LA, Blodgett HC and Deatherage BH (1962). Effect of interaural correlation on the precision of centering a noise. J Acoust Soc Am 34: 1122–123
Joris PX, Yin TCT (2007) A matter of time: internal delays in binaural processing. Trends Neurosci 30
Joris PX, Van de Sande B, Louage DH and Van der Heijden M (2006). Binaural and cochlear disparities. Proc Natl Acad Sci USA 103: 12917–2922
Klump GM (2000). Sound localization in birds. In: Dooling, RJ, Fay, RR and Popper, AN (eds) Comparative hearing: birds and reptiles, pp 249–07. Springer, New York
Konishi M (1986). Centrally synthesized maps of sensory space. Trends Neurosci 9: 163–68
Konishi M (2003). Coding of auditory space. Ann Rev Neurosci 26: 31–5
Köppl C (1997). Phase locking to high frequencies in the auditory nerve and cochlear nucleus magnocellularis of the barn owl, Tyto alba. J Neurosci 17: 3312–321
Köppl C and Carr CE (1997). Low-frequency pathway in the barn owl’s auditory brainstem. J Comp Neurol 378: 265–82
Köppl C and Carr CE (2003). Computational diversity in the cochlear nucleus angularis of the barn owl. J Neurophysiol 89: 2313–329
Kuba H, Ishii TM and Ohmori H (2006). Axonal site of spike initiation enhances auditory coincidence detection. Nature 444: 1069–072
Kubke MF, Massoglia DP and Carr CE (2004). Bigger brains or bigger nuclei? Regulating the size of auditory structures in birds. Brain Behav Evol 63: 169–80
Larsen ON, Dooling RJ and Ryals BM (1997). Roles of intracranial air pressure in bird audition. In: Lewis, ER, Long, GR, Lyon, RF, Narins, PM, Steele, CR, and Hecht-Poinar, E (eds) Diversity in auditory mechanics, pp 11–7. World Scientific, Singapore
Larsen ON, Dooling RJ and Michelsen A (2006). The role of pressure difference reception in the directional hearing of budgerigars (Melopsittacus undulatus). J Comp Physiol A 192: 1063–072
MacLeod KM, Soares D and Carr CE (2006). Interaural timing difference circuits in the auditory brainstem of the emu (Dromaius novaehollandiae). J Comp Neurol 495: 185–01
Marquardt T and McAlpine D (2007). A pi-limit for coding ITDs: implications for binaural models. In: Kollmeier, B, Klump, G, Hohmann, V, Langemann, U, Mauermann, M, Uppenkamp, S and Verhey, J (eds) Hearing—from sensory processing to perception, pp 407–16. Springer, Heidelberg
McAlpine D (2005). Creating a sense of auditory space. J Physiol London 566: 21–8
McAlpine D, Jiang D and Palmer AR (2001). A neural code for low- frequency sound localization in mammals. Nature Neurosci 4: 396–01
Overholt EM, Rubel EW and Hyson RL (1992). A circuit for coding interaural time differences in the chick brainstem. J Neurosci 12: 1698–708
Palmer AR (2004). Reassessing mechanisms of low-frequency sound localisation. Curr Opin Neurobiol 14: 457–60
Parks TN and Rubel EW (1975). Organization and development of brain stem auditory nuclei of the chicken: Organization of projections from N. magnocellularis to N. laminaris. J Comp Neurol 164: 435–48
Pena JL, Viete S, Albeck Y and Konishi M (1996). Tolerance to sound intensity of binaural coincidence detection in the nucleus laminaris of the owl. J Neurosci 16: 7046–054
Pena JL, Viete S, Funabiki K, Saberi K and Konishi M (2001). Cochlear and neural delays for coincidence detection in owls. J Neurosci 21: 9455–459
Rosowski JJ and Saunders JC (1980). Sound transmission through the avian interaural pathways. J Comp Physiol 136: 183–90
Rubel EW and Parks TN (1975). Organization and development of brain stem auditory nuclei of the chicken: Tonotopic organization of N. magnocellularis and N. laminaris. J Comp Neurol 164: 411–33
Saberi K, Takahashi Y, Konishi M, Albeck Y, Arthur BJ and Farahbok H (1998). Effects of interaural decorrelation on neural and behavioral detection of spatial cues. Neuron 21: 789–98
Salvi RJ, Saunders SS, Powers NL and Boettcher FA (1992). Discharge patterns of cochlear ganglion neurons in the chicken. J Comp Physiol A 170: 227–41
Schwarz DWF (1992). Can central neurons reproduce sound waveforms? An analysis of the neurophonic potential in the laminar nucleus of the chicken. J Otolaryngol 21: 30–8
Scott LL, Mathews PJ and Golding NL (2005). Posthearing developmental refinement of temporal processing in principal neurons of the medial superior olive. J Neurosci 25: 7887–895
Scott LL, Hage TA and Golding NL (2007). Weak action potential backpropagation is associated with high-frequency axonal firing capability in principal neurons of the gerbil medial superior olive. J Physiol London 583: 647–61
Shamma SA, Shen N and Gopalaswamy P (1989). Stereausis: binaural processing without neural delays. J Acoust Soc Am 86: 989–006
Smith PH, Joris PX and Yin TCT (1993). Projections of physiologically characterized spherical bushy cell axons from the cochlear nucleus of the cat: evidence for delay lines to the medial superior olive. J Comp Neurol 331: 245–60
Spitzer MW and Semple MN (1995). Neurons sensitive to interaural phase disparity in gerbil superior olive: Diverse monaural and temporal response properties. J Neurophysiol 73: 1668–690
Sullivan WE and Konishi M (1986). Neural map of interaural phase difference in the owl’s brainstem. Proc Natl Acad Sci USA 83: 8400–404
Takahashi TT, Bala ADS, Spitzer MW, Euston DR, Spezio ML and Keller CH (2003). The synthesis and use of the owl’s auditory space map. Biol Cybern 89: 378–87
Thompson SK, von Kriegstein K, Deane-Pratt A, Marquardt T, Deichmann R, Griffiths TD and McAlpine D (2006). Representation of interaural time delay in the human auditory midbrain. Nature Neurosci 9: 1096–098
van Hemmen JL (2005). What is a neuronal map, how does it arise, and what is it good for? In: Van Hemmen, JL and Sejnowski, TJ (eds) 23 problems in systems neuroscience, pp 83–02. Oxford University Press, Oxford
Viete S, Pena JL and Konishi M (1997). Effects of interaural intensity difference on the processing of interaural time difference in the owl’s nucleus laminaris. J Neurosci 17: 1815–824
Wagner H, Brill S, Kempter R and Carr CE (2005). Microsecond precision of phase delay in the auditory system of the barn owl. J Neurophysiol 94: 1655–658
Wagner H, Asadollahi a, Bremen P, Endler F, Vonderschen K and Von Campenhausen M (2007). Distribution of interaural time difference in the barn owl’s inferior colliculus in the low- and high-frequency ranges. J Neurosci 27: 4191–200
Yin TCT (2002). Neural mechanisms of encoding binaural localization cues in the auditory brainstem. In: Oertel, D, Fay, RR and Popper, AN (eds) Integrative functions in the mammalian auditory pathway, pp 99–59. Springer, New York
Yin TCT and Chan JCK (1990). Interaural time sensitivity in the medial superior olive of the cat. J Neurophysiol 64: 465–88
Yin TCT and Kuwada S (1983). Binaural interaction in low-frequency neurons in inferior colliculus of the cat. III. Effects of changing frequency. J Neurophysiol 50: 1020–042
Young SR and Rubel EW (1983). Frequency-specific projections of individual neurons in chick brainstem auditory nuclei. J Neurosci 3: 1373–378
Yin TCT, Chan JCK and Carney LH (1987). Effects of interaural time delays of noise stimuli on low-frequency cells in the cat’s inferior colliculus III. Evidence for cross-correlation. J Neurophysiol 58: 562–83
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Köppl, C., Carr, C.E. Maps of interaural time difference in the chicken’s brainstem nucleus laminaris. Biol Cybern 98, 541–559 (2008). https://doi.org/10.1007/s00422-008-0220-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00422-008-0220-6