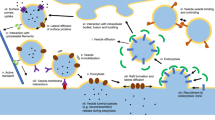
Abstract
The release of transmitter from the vesicle, its diffusion through the fusion pore, and the cleft and its interaction with the carbon electrode were simulated using the Monte Carlo method. According to the simulation the transmitter release is largely determined by geometric factors – the ratio of the fusion pore cross-sectional and vesicular areas, if the diffusion constant is as in the aqueous solution – but the speed of transmitter dissociation from the gel matrix plays an important role during the rise phase of release. Transmitter is not depleted near the entrance to the fusion pore and there is no cleft-to-vesicle feedback, but the depletion becomes evident if the diffusion constant is reduced, especially if the pore is wide. In general, the time course of amperometric currents closely resembles the time course of the simulated transmitter concentration in the cleft and the time course of release. Surprisingly, even a tenfold change of the electrode efficiency has only a marginal effect on the amplitude or the time course of amperometric currents. Greater electrode efficiency however lowers the cleft concentration, but only if the cleft is narrow. As the cleft widens the current amplitudes diminish and rise times lengthen, but the decay times are less affected. Moreover, the amplitude dependence of the rise and decay times becomes steeper as the cleft widens and/or as the release kinetics slows. Finally, lower diffusion constant of transmitter in the narrow cleft does not further prolong the amperometric currents, whose slow time course reflects slow release kinetics.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amatore C, Bouret Y, Travis ER, Wightman RM (2000) Interplay between membrane dynamics, diffusion and swelling pressure governs individual vesicular exocytotic events during release of adrenaline by chromaffin cells. Biochimie 82:481–496
Anderson BB, Zerby SE, Ewing AG (1999) Calculations of transmitter concentration in individual PC12 cell vesicles with electrochemical data and a distribution of vesicle size obtained by electron microscopy. J Neurosci Methods 88:163–170
Aspinwall CA, Brooks SA, Kennedy RT, Lakey JRT (1997) Effects of intravesicular H+ and extracellular H+ and Zn2+ on insulin secretion in pancreatic beta cells. J Biol Chem 272:31308–31314
Bennett MR, Farnell L, Gibson WG (2004) The facilitated probability of quantal secretion within an array of calcium channels of an active zone at the amphibian neuromuscular junction. Biophys J 86:2674–2690
Brozoski TJ, Brown RM, Rosvold HE, Goldman PS (1979) Cognitive deficit caused by regional depletion of dopamine in prefrontal cortex of rhesus monkey. Science 205:929–932
Coupland RE (1968) Determining sizes and distribution of sizes of spherical bodies such as chromaffin granules in tissue sections. Nature 217:384–388
Crank J (1984) Free and moving boundary problems. Clarendon Press, Oxford, UK
Di Chiara G, Bassareo V, Fenu S, De Luca MA, Spina L, Cadoni C, Acquas E, Carboni E, Valentini V, Lecca D (2004) Dopamine and drug addiction: the nucleus accumbens shell connection. Neuropharmacol 47:227–241
Eccles JC, Jaeger JC (1958) The relationship between the mode of operation and the dimensions of the junctional regions at synapses and motor end-organs. Proc Royal Soc B 148:38–56
Finnegan JM, Pihel K, Cahill PS, Huang L, Zerby SE, Ewing AG,Kennedy RT, Wightman RM (1996) Vesicular Quantal Size Measured by Amperometry at Chromaffin, Mast, Pheochromocytoma, and Pancreatic-Cells. J Neurochem 66:1914–1923
Frucht SJ (2004) Parkinson disease: an update. Neurologist 10:185–194
Garris PA, Ciolkowski EL, Pastore P, Wightman RM (1994) Efflux of dopamine from the synaptic cleft in the nucleus accumbens of the rat brain. J Neurosci 14:6084–6093
Gil A, Viniegra S, Gutierrez LM (2001) Temperature and PMA affect different phases of exocytosis in bovine chromaffin cells. Eur J Neurosci 13:1380–1386
Glavinović MI (1999) Monte Carlo simulation of vesicular release, spatio-temporal distribution of glutamate in synaptic cleft, and generation of postsynaptic currents. Pflugers Archiv Eur J Physiol 437:462–470
Glavinović MI, Rabie HR (1997) Monte Carlo simulation of release of content of individual vesicles in secretory cells. Abstr Soc Neurosci 23:366
Glavinović MI, Vitale ML, Trifaro JM (1998) Comparison of vesicular volume and quantal size in bovine chromaffin cells. Neuroscience 85:957–968
Hafez I, Kisler K, Berberian K, Dernick G, Valero V, Yong MG, Craighead HG, Lindau M (2005) Electrochemical imaging of fusion pore openings by electrochemical detector arrays. Proc Natl Acad Sci USA 102:13879–13884
Jena BP, Cho SJ, Jeremic A, Stromer MH, Hamdah RA (2003) Structure and composition of the fusion pore. Biophys J 84:1337–1343
Karunanithi S, Marin L, Wong K, Atwood HL (2002) Quantal size and variation determined by vesicle size in normal and mutant Drosophila glutamatergic synapses. J Neurosci 22:10267–10276
Kebir S, Aristizabal F, Maysinger D, Glavinović MI (2005) Rapid change of quantal size in PC-12 cells detected by neural networks. J Neurosci Methods 142:231–242
Kelly RB (1993) Storage and release of neurotransmitters. Cell 72:45–53
Khanin R, Parnas H, Segel L (1994) Diffusion cannot govern the discharge of neurotransmitter in fast synapses. Biophys J 67:966–972
Lee PI (1980) Diffusion release of a solute from a polymeric matrix: approximate analytical solutions. J Membr Sci 7:255–275
Lee PI (1985) Kinetics of drug release from hydrogel matrices. J Contr Rel 2:277–288
Monck JR, Fernandez JM (1992) The exocytotic fusion pore. J Cell Biol 119:1395–1404
Ritger PL, Peppas NA (1987a) A simple equation for description of solute release. I. Fickian and non-Fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. J Contr Rel 5:23–36
Ritger PL, Peppas NA (1987b) A simple equation for description of solute release. I. Fickian and anomalous release from swellable devices. J Contr Rel 5:37–42
Saulle E, Centonze D, Martin AB, Moratalla R, Bernardi G, Calabresi P (2002) Endogenenous dopamine amplifies ischemic long-term potentiation via D1 receptors. Stroke 33:2978–2984
Schikorski T, Stevens CF (1997) Quantitative, ultrastructural analysis of hippocampal excitatory synapses. J Neurosci 17:5858–5867
Schroeder TJ, Jankowski JA, Senyshyn J, Holz RW, Wightman RM (1994) Zones of exocytotic release on bovine adrenal medullary cells in culture. J Biol Chem 269:17215–17220
Schroeder TJ, Borges R, Finnegan JM, Pihel K, Amatore C, Wightman RM (1996) Temporally resolved, independent stages of individual exocytotic secretion events. Biophys J 70:1061–1068
Spruce AE, Breckenridge LJ, Lee AK, Almers W (1990) Properties of the fusion pore that forms during exocytosis of a mast cell secretory vesicle. Neuron 4:643–654
Sulzer D, Pothos EN (2000) Regulation of quantal size by presynaptic mechanisms. Rev Neurosci 11:159–212
Tanaka S (2005) Dopaminergic control of working memory and its relevance to schizophrenia: a circuit dynamics perspective. Neuroscience (in press). DOI:10.1016/J.neuroscience.20 05.08.070
Tang KS, Tse A, Tse FW (2005) Differential regulation of multiple populations of granules in rat adrenal chromaffin cells by culture duration and cyclic AMP. J Neurochem 92:1126–1139
Terakawa S, Kumakura K, Duchen M (1995) Spatiotemporal analysis of quantal secretory events from bovine adrenal chromaffin cells in culture. J Physiol (Lond) 487:59–60
Tsuboi T, Kikuta T, Sakurai T, Terakawa S (2002) Water secretion associated with exocytosis in endocrine cells revealed by micro forcemetry and evanescent wave microscopy. Biophys J 83:172–183
Uvnas B, Aborg CH (1989) Role of ion exchange in release of biogenic amines. News Physiol Sci 4:68–71
Ventriglia F (2004) Saturation in excitatory synapses of hippocampus investigated by computer simulations. Biol Cybern 90:349–359
Wahl LM, Pouzat C, Stratford KJ (1996) Monte Carlo Simulation of fast excitatory synaptic transmission at a hippocampal synapse. J Neurophysiol 75:597–608
Walker A, Glavinović MI, Trifaro JM (1996) Time course of release of content of single vesicles in bovine chromaffin cells. Pflugers Archiv Eur J Physiol 431:729–735
Westerink, RHS, de Groot A, Vijverberg HPM (2000) Heterogeneity of catecholamine-containing vesicles in PC12 cells. Biochem Biophys Res Comm 270:625–630
Wightman RM, Jankowski JA, Kennedy RT, Kawagoe KT, Schroeder TJ, Leszczyszyn DJ, Near JA, Diliberto EJ, Viveros OH (1991) Temporally resolved catecholamine spikes correspond to single vesicle release from individual chromaffin cells. Proc Natl acad Sci USA 88:10754–10758
Winkler H, Apps DK, Fischer-Colbrie R (1986) The molecular function of adrenal chromaffin granules: established facts and unresolved topics. Neuroscience 18:261–290
Yoo SH, Lewis MS (1993) Dimerization and tetramerization properties of the C-terminal region of chromogranin A: a thermodynamic analysis. Biochemistry 32:8816–8219
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rabie, H.R., Rong, J. & Glavinović, M.I. Monte Carlo Simulation of Release of Vesicular Content in Neuroendocrine Cells. Biol Cybern 94, 483–499 (2006). https://doi.org/10.1007/s00422-006-0061-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00422-006-0061-0