Abstract
The effects of time-varying magnetic fields (MF) on humans have been actively investigated for the past three decades. One important unanswered question is the potential for MF exposure to have acute effects on human biology. Different strategies have been used to tackle this question using various physiological, neurophysiological and behavioral indicators. For example, researchers investigating electroencephalography (EEG) have reported that extremely low frequency (ELF, <300 Hz) MF can increase resting occipital alpha rhythm (8–12 Hz). Interestingly, other studies have demonstrated that human motricity can be modulated by ELF MF: a reduction of anteroposterior standing balance or a decrease of physiological tremor intensity have been reported as consequences of exposure. However, the main limitation in this domain lies in the lack of results replication, possibly originating from the large variety of experimental approaches employed. Therefore, the present study aimed to investigate the effects of a 60 Hz, 1,800 μT MF exposure on neurophysiological (EEG) and neuromotor (standing balance, voluntary motor function, and physiological tremor) aspects in humans using a single experimental procedure. Though results from this study suggest a reduction of human standing balance with MF exposure, as well as an increase of physiological tremor amplitude within the frequency range associated with central nervous system contribution, no exposure effect appeared on other investigated parameters (e.g., EEG or voluntary motor control). These results suggest that 1 h of 60 Hz, 1,800 μT MF exposure may modulate human involuntary motor control without being detected in the cortical electrical activity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recent studies have been trying to characterize the effects of extremely low frequency (ELF, below 300 Hz) magnetic fields (MF) on human biology and performance. Despite the amount of work conducted in this area, there is still no consensus regarding the effects of ELF MF exposure on humans. The main sources of ELF MF in our daily environment are domestic electrical appliances, distribution and transport power-lines, and residential wiring which are producing power-line frequency MF: 50 Hz in Europe and 60 Hz in North America. General public exposure to power-line frequency MF is on average <0.01 μT (Kaune et al. 2002). Although a few controversial epidemiological studies have reported an increased risk of developing childhood leukemia in populations chronically exposed to MF as low as 0.3–0.4 μT on average (Greenland and Kheifets 2006), no reliable acute effects of exposure have been reported for this type of MF. However, for higher intensities, researchers have recently suggested that MF exposure can modulate spontaneous electrical activity in the brain, and high-level cognitive processes (see Cook et al. 2002, 2006 for reviews). The general public is occasionally exposed to these MF intensities. For instance when one uses an electric shaver, a hairdryer, or a hair clipper, the MF generated on the surface of these devices can reach 1,500–2,000 μT (Gandhi et al. 2001; Gauger 1985). Moreover, workers of electricity companies are often exposed to MF levels above 1,000 μT (Gauger 1985; Maruvada and Jutras 1993) which raises specific questions; for example, is a worker operating on repairing a live power-line at a greater risk of committing an error, or creating a workplace hazard, than a worker repairing a deactivated power-line?
Organizations such as the International Commission on Non-Ionizing Radiation Protection (ICNIRP) or the Institute of Electrical and Electronics Engineers (IEEE) publish recommendations concerning the levels of exposure for the general public and for workers (ICNIRP 2010; IEEE 2001). When the present study has been initiated, before the latest update of ICNIRP guidelines, the basic restrictions of the ICNIRP guidelines were establishing that current density induced by MF occupational exposure “should be limited to fields that induce current densities less than 10 mA/m2” at the level of the head (ICNIRP 1998). According to the dosimetric calculations from Dimbylow (1998), it corresponded to a flux density of 1,800 μT at 60 Hz. This recommendation was including a security factor of 10 to prevent from “acute change in the central nervous system excitability and other acute effects”. Note that the new reference level of ICNIRP is now set at 1,000 μT in a controlled environment (ICNIRP 2010).
Magnetophosphenes are the first consistent and reproducible phenomenon of human exposure to ELF MF (ICNIRP 1998; IEEE 2001; WHO 2007). These flickering visual sensations are thought to result from the interaction between induced electric currents and electrically sensitive cells of the retina (described as “synaptic interactions” by IEEE 2001). It is generally agreed that, since they represent a direct and reproducible perception of the interaction with ELF MF, magnetophosphenes represent an appropriate model for induced electric field effects on central nervous system (Attwell 2003). Following the work from Lovsund et al. (1979, 1980a, b), Silny (1986) reported human perception of magnetophosphenes induced by an intermittent 1-h MF (1 s on–2 s off) between 10 and 20 mT depending on the stimulation frequency (5–60 Hz, Silny 1986). He reported a phosphene detection threshold of 5 mT at 18 Hz in a trained subject. In the same study, it is reported that there is an effect of the exposure on visual evoked potentials (VEP) lasting past the end of the exposure period. However, as noticed by Saunders and Jefferys (2007), the method and the data were not entirely described, and thus make this work “difficult to evaluate”, stressing the need for further experimental studies to confirm these results. The lowest estimate to date of the threshold for magnetophosphenes perception is 5 mT at 20 Hz and it is rising at higher frequencies (ICNIRP 2010). Note that this is much below the peripheral nerve stimulation threshold in humans, which is reported to be 47.9 mT at 60 Hz (median threshold, Bailey and Nyenhuis 2005).
Since a consensus regarding ELF MF acute exposure effects on living systems is still to be achieved, there is a need to replicate and provide further studies (WHO 2007). The effects of acute exposure to ELF MF below the threshold for magnetophosphenes perception have been studied on numerous aspects of human physiology, neurophysiology, and behavior. Electroencephalogram (EEG) or evoked potentials are examples of neurophysiological parameters that have received attention among researchers (Bell et al. 1992, 1994a, b; Cook et al. 2004, 2005a, b; Ghione et al. 2005; Heusser et al. 1997; Lyskov et al. 1993a, b; Marino et al. 2004). Although most conspicuous results seem to suggest a higher resting EEG in the alpha rhythm (8–13 Hz) after exposure in the occipital regions of the brain, the results are very heterogeneous between studies and no conclusion on the direction of these effects can be drawn.
Other studies analyzing the interaction between the cardiovascular system and ELF MF have shown that electrophysiological rhythms can be modified at the peripheral level. For example, it has been reported that an exposure to a 60 Hz MF could induce a slowing of the heart rate (HR), which may or may not be associated with changes in HR variability (Cook et al. 1992; Graham et al. 2000a; Maresh et al. 1988; Sastre et al. 1998, 2000). Again, other researchers have not confirmed these findings (Graham et al. 2000b, c; Kurokawa et al. 2003). McNamee et al. (2009) have published an exhaustive review on the topic. In a first round of analysis of the experimental data issued from the current experimental approach, we have not been able to confirm any effect of a 1,800 μT MF at 60 Hz on HR, HR variability and peripheral blood perfusion (McNamee et al. 2010).
Another strategy used by researchers to study ELF MFs was to focus on macroscopic neurophysiological indicators such as human motricity. Any modulation of the normal neurophysiological processing should have functional consequences that may be investigated with the study of neuromotor processes. Indeed, a few studies have demonstrated ELF MF effects on human standing balance and physiological tremor, but not on simple goal-directed movements (Lacy-Hulbert et al. 1998; Legros and Beuter 2005, 2006; Legros et al. 2006; Prato et al. 2001; Thomas et al. 2001a). Concerning standing balance (automatic control of the balance while standing), it has been reported that normal anteroposterior balance in healthy subjects can be decreased using a specific pulsed MF as low as 200 μT (Thomas et al. 2001a). Regarding human segmental micro movements (i.e. fine finger tracking performance and physiological tremor characteristics), though Legros et al. did not report any effect of a 50 Hz, 1,000 μT MF on index finger tracking performance (Legros and Beuter 2005), their results suggested that MF exposure facilitated postural tremor decrease in a relaxing situation (tremor recorded at the tip of the index finger, Legros and Beuter 2006). Interestingly, Cook et al. (2004) who demonstrated an effect of a pulsed 200 μT MF on EEG alpha activity also underlined the link between resting posterior alpha activity and the state of relaxed wakefulness.
Overall, these reported effects are often not replicated and sometimes contradictory. One of the reasons for these discrepancies could be due to the fact that some individuals may be more susceptible than others to MF exposure (as suggested by Saunders and Jefferys 2007, for example). However, although numerous studies testing for hypersensitivity to electromagnetic fields have been conducted, the results in this regard are still inconclusive (Levallois 2002; Rubin et al. 2005). As another objective (but non-exhaustive) potential reason for such discrepancies, it is often argued that the heterogeneity in the experimental procedures across studies is likely a cause for these observations. It is for example underlined by Crasson et al. (2003) in their review, who state that heterogeneity in intensity, shape, and frequency of the MF used, as well as the differences in exposure durations across the studies are introducing confounds which makes the interpretation of these results difficult. There are indeed evidences of frequency-dependant effects, such as with magnetophosphenes, which preferably occur at 20 Hz than at lower or higher frequencies (see for example ICNIRP 2010). Furthermore, results from the literature differ with continuous versus intermittent exposure, and depending on whether participant testing is performed during or after the exposure.
The main objective of this study was to evaluate the effects of a 60 Hz, 1,800 μT MF exposure on human physiology, neurophysiology, and motor functions in a single procedure [EEG, electrocardiogram (ECG), peripheral blood perfusion, rhythmic voluntary hand movements, physiological tremor, and standing balance]. Cardiovascular and blood perfusion results have already been reported (McNamee et al. 2010). This level of exposure was chosen referencing the ICNIRP basic restriction for ELF and to the calculation of Dimbylow in 1998 estimating the corresponding magnetic induction. According to Dimbylow (1998), ICNIRP’s (1998) basic restriction (10 mA/m2 of induced currents at the central nervous system level) corresponded to a calculated MF exposure value of 1,810 μT. Based on results from the literature, we hypothesized that this MF exposure level should increase EEG power in the alpha band (especially in the occipital regions), reduce standing balance, have no effect on the maximum frequency of rapid alternating hand movements, and decrease postural tremor amplitude at the tip of the index finger.
Methods
Subjects
Seventy-three participants (randomized recruitment, 46 males, 27 females, age = 28 ± 9) have completed two double blind counterbalanced sessions of testing (real/sham). Each session was given on a separate day and was composed of four sequences of testing (detailed in Fig. 1a). None of the participants had ever experienced an epileptic seizure, had motor limitations, suffered from chronic illness (e.g., diabetes, severe psychiatric, cardiovascular or neurological diseases), or had a cardiac or cerebral pacemaker. They had no history of head, eye or thorax injury involving metal fragments, and they did not wear metal braces on their teeth. Finally, women could not be pregnant, nor have an intrauterine device.
a Time course of the two exposure sessions (real and sham). The horizontal black line represents the MF status (OFF when down, ON when up). Note that during the sham exposure session, the MF is never ON. Vertical grey bands represent the four 15-min blocks of testing. b A zoomed-in view on the time course of a block of testing (the same for each block). White cells represent resting periods and grey cells represent testing periods (duration is displayed in seconds inside the cells). The table below the time course indicates the tests to which these periods correspond
Apparatus and procedure
A double blind computer driven procedure, controlling for variables, was used such that neither the participant nor the experimenter knew when the real or sham condition occurred (automatic audio directions through speakers, visual instructions on a LCD screen; programmed using LabView 8.0, NI Inc., USA). Each session lasted 1 h and 45 min and was composed of four 15-min blocks of testing spaced by 15 min of rest. One hour of MF exposure was given from minute 15 to minute 75 (active in the real exposure session, inactive in the sham session). The protocol was approved by the University of Western Ontario Health Sciences Research Ethics Board (#11956E).
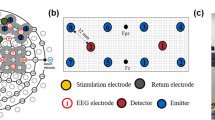
For the duration of the experiment, the participant was sitting in an elevated armchair located in the middle of our exposure system, which was composed of two octagonal coils positioned parallel to one another, spaced 1.2 m apart, and both with a diameter of 1.6 m. Each coil contained 80 turns of AWG-10 wire mounted with a nonconductive cooling/heating tubing system. The system was configured to generate a homogenous 60 Hz, 1,800 μT MF centered at the level of the head (see Fig. 2b for an illustration of the calculated MF distribution). Further details on the exposure system can be found in McNamee et al. (2010).
Spatial illustration in 3-D of the MF level (horizontal component) distribution within the exposure system (computed according to the Bio-Savart law). The grey volume in the middle of the two coils (thick black octagons) represents the 1,800 μT ± 5% homogeneity region. Each circular line in the sagittal plan represents 5% intervals. Actual MF measurements confirmed the computed values (Sentron, Sentron AG, Switzerland)
The room temperature was kept at 23°C. From the beginning of the testing session, both the subject and experimenter wore earplugs to make it impossible to detect the small buzz produced by the coils when the field is generated. During the testing time course, the MF generation and the data acquisition were entirely automated and computer driven (Labview 8.0 and Data acquisition card NI PCI-6289, National Instrument Inc., USA). EEG data were acquired continuously through the experiment (using the ambulatory 32 channels EEG/ECG Siesta system and the Profusion 3.7 software from Compumedics Inc., USA), but specific MF artifact-free periods were extracted for analysis (as illustrated in Fig. 1 and detailed below).
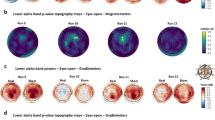
During both the EEG eyes open and eyes closed conditions (Fig. 2b), the MF was turned off during the second minute of recordings in order to keep 1 min of data free from MF artifact. In all the experimental EEG conditions (eight eyes open conditions and eight eyes closed conditions), only the 1-min free of MF artifact EEG data (measured in μV and sampled at 512 Hz) was kept for data analysis. EEG data of the frontal (F3, F4), central (C3, C4), parietal (P3, P4), and occipital (O1, O2) electrodes were saved into text files and then opened in Matlab (Matlab 7.0, The MatWorks. Inc, USA) for analysis. The first step consisted in removing the eyes movement artifacts if any, and in keeping 50 s of raw EEG data eyes movement artifact-free. The resulting 25,600 points time series were then band-pass filtered (using the Fast Fourier Transform and inverse Fast Fourier Transform algorithms—fft and ifft functions in Matlab) in the theta (3–7 Hz), alpha (8–13 Hz), beta (14–35 Hz) and gamma (36–50 Hz) ranges. Average amplitude over the periods of recording was then calculated for each frequency band as the root mean square of the corresponding band-pass filtered time series (in μV), and used for statistical analyses. Since the EEG alpha rhythm has been the EEG frequency band reported to be modulated by ELF MF exposure in several studies, we have focused in this paper on the results related to this particular frequency band.
The tremor tests consisted of 1 min of postural tremor recording with eyes open and then 1 min of postural tremor with eyes closed. Tremor was recorded at the tip of the dominant index finger using a Class II laser diode pointing down (Micro laser sensor LM10, series ARN11, Matsushita Electronic Work, Ltd., Osaka, Japan) located 8 cm above the piece of white cardboard fixed on the finger nail. This system enabled vertical displacement recording at 1,000 Hz with a 5 μm resolution (after filtering out frequencies >20 Hz). A feedback line representing the index finger position was displayed on the LCD screen facing the subject. In this test, the participant had to point the index finger (in extension at the level of the metacarpophalangeal joint) to keep the feedback line centered in the middle of the screen (“zero position”) for 1 min. As done for the EEG data, tremor time series (amplitude measured in mm with a sampling rate of 1,000 Hz) were exported to Matlab where ten specific tremor characteristics were computed in the time and frequency domains (Beuter and Edwards 1999; Edwards and Beuter 2000; Legros and Beuter 2005). Postural tremor data were analyzed using validated published characteristics (Beuter and Edwards 1999, 2002; Edwards and Beuter 2000) allowing the examination of temporal, frequential and morphological components of signals (Legros and Beuter 2005, 2006; Legros et al. 2006). The following indexes were computed on differentiated velocity time series for frequency characteristics (note that as for the EEG, the fft-ifft algorithms were used to filter the data in Matlab):
-
Amplitude: It is the root mean square of the filtered position series centered on their mean. Computed on position data filtered between 2 and 20 Hz. Larger values correspond to poorer performance.
-
Drift: It quantifies the amplitude of the drift of the finger (slow movements with frequencies <1 Hz). Computed on position data. Larger values correspond to poorer performance.
-
Median frequency: It is calculated on the power spectrum between 2 and 20 Hz and determines the value at which 50% of the power is below this frequency, and 50% is above. Computed on velocity data. Smaller values correspond to poorer performance.
-
Proportional power in 2–4 Hz: It is the proportion of the power contained in this range compared with the spectrum between 2 and 20 Hz. It represents the proportion of voluntary corrections of the posture. Computed on velocity data filtered between 2 and 20 Hz. Larger values correspond to poorer performance.
-
Proportional power in 4–7 Hz range: It is the proportion of the power contained in this range compared with the spectrum between 2 and 20 Hz. This range is typically increased in pathologies such as Parkinson’s disease. Computed on velocity data filtered between 2 and 20 Hz. Larger values correspond to poorer performance.
-
Proportional power in 7–12 Hz range: It is the proportion of the power contained in this range compared with the spectrum between 2 and 20 Hz. This range contains physiological tremor components. Computed on velocity data filtered between 2 and 20 Hz. Smaller values correspond to poorer performance.
-
Absolute power in 7–12 Hz: It is the sum of the power contained in this range. This range contains physiological tremor components. Computed on velocity data filtered between 2 and 20 Hz. Larger values correspond to poorer performance.
-
Proportional power in 12–20 Hz range: It is the proportion of the power contained in this range compared with the spectrum between 2 and 20 Hz. This range is typically modified by changes in the mechanical properties of the considered segment (here the index finger). Computed on velocity data filtered between 2 and 20 Hz. Larger values correspond to poorer performance.
-
Wobble: It characterizes the morphology of the oscillations. This is the ratio between the number of extrema in acceleration data and the number of extrema in displacement data. Larger values correspond to poorer performance.
-
Positive/negative asymmetry: It is the product of 2 ratios: (1) The ratio between the duration of the periods when values of the time series are positive and the duration of the periods when values of the time series are negative; (2) the ratio of the amplitude of oscillations in the positive values and in the negative values. Larger values correspond to poorer performance.
In the alternating movement tests, the participant was asked to extend his arms in front of him, parallel to the floor, and to bend them to a 90° angle at the elbow, with hands open, palms facing each other. From this starting position, he had to execute alternating rotating hand movements at the wrist axis: (1) with the right hand, natural rhythm; (2) with the left hand, natural rhythm; (3) with both hands, natural rhythm; (4) with the right hand, high frequency; (5) with the left hand, high frequency; and (6) both hands, high frequency. For the high frequency conditions, the participant was asked to rotate his hands “as fast and as far as possible”. Each testing condition lasted 5 s. Data were acquired using a Liberty 3-D tracking system (Liberty, Polhemus Inc., USA), with two transducers fixed like watches on the dorsal side of the wrists and allowing recording of movement kinematics with 6 degrees of freedom at 200 Hz (3 dimensions and 3 angles of rotation, accuracy of 0.076 cm RMS for X, Y, Z positions and 0.15° RMS for orientations). The Liberty system is an electromagnetic tracking system and therefore, during the MF exposure session, the exposure had to be turned off during each 5 s recording periods so as not to affect data. The rotation time series around the vertical axis were preprocessed with Matlab (low-pass filtered at 15 Hz, fft-ifft) and exported to a specifically designed analysis software (Neuro 2.1, Doco Microsystèmes, Canada). Eight validated indexes characterizing the subject performance were then calculated (Beuter et al. 1999a, b, 2004; Legros et al. 2004):
-
Duration: This parameter represents the mean duration of an oscillation in seconds. The final result is multiplied by a factor of ten. Larger values correspond to poor performance only in the rapid movement condition.
-
Velocity: The mean velocity of an oscillation in degrees per second. Under the maximum speed condition, larger values are considered better.
-
Maximum slope: The maximum slope of the oscillations (arbitrary unit, noted A.U.). It represents the averaged maximum velocity produced during the oscillations. Smaller values indicate a more regular performance.
-
Similarity in size: The weighted mean of “distances” between all pairs of cycles in which nearby cycles are weighted more heavily (A.U.). “Distances” between cycles are based on differences between 11 characteristics describing the shape of each cycle. Smaller values indicate a more regular performance.
-
Smoothness: The sum of all absolute values of negative accelerations where the velocity is generally increasing from zero to its maximum value and all positive accelerations where the velocity is generally decreasing from its maximum value to zero (A.U.). The larger the number, the more irregular the performance.
-
Sharpness: The mean absolute value of velocity relative to peak velocity for ascending and descending segments and averaged across all cycles (A.U.). The larger the number, the less difficulty there was at the turn (i.e. better performance).
-
Symmetry: The difference between the area under the ascending segment and the area under the descending segment relative to the total area under the cycle averaged over all cycles (A.U.). Larger numbers indicate larger deviations from symmetry.
-
Coherence: It is calculated from the spectral coherence between the signals recorded in the two hands (A.U.). We use the amplitude of the highest peak between 0 and 6 Hz. This reflects the similarity of the oscillation in the two hands. Larger values correspond to poorer performance.
Finally, the armchair with the subject was pulled backwards, opening the access to a force plate underneath the chair. The participant had to step onto it and take the standardized posture (standardized socks, feet parallel, 1 cm apart) and to relax before his postural sway was recorded in the eyes open and eyes closed conditions. The 3-D force plate, used in previous works (Prato et al. 2001; Thomas et al. 2001a, b, OR6-7-1000, AMTI, Watertown, MA), was mounted on the floor so that the subject had his head in the centre of the MF homogeneous region. It recorded the force and momentum applied by the subject’s feet at a sampling rate of 1,000 Hz. The data files were imported in Matlab, where the measurements were converted to centre of pressure (COP) values according to a conversion matrix provided by the force plate manufacturer (AMTI, Watertown MA). COP coordinates corresponded to the perpendicular projection of the centre of gravity through the force plate (expressed in terms of X and Y positions, measured in cm). Postural sway is represented as the change in COP over time (i.e. its trajectory). Seven validated postural sway characteristics were computed on COP trajectories (Despres et al. 2000; Thomas et al. 2001a):
-
Mean sway: Averaged distance between the geometric center of all recorded forces and each point visited by the COP during a recording. Larger values correspond to poorer performance.
-
Transversal mean sway: Averaged displacements on the X axis. Larger values correspond to poorer performance.
-
Sagittal mean sway: Averaged displacements on the Y axis. Larger values correspond to poorer performance.
-
Sway velocity: Averaged velocity of the COP displacements. Larger values correspond to poorer performance.
-
Sway velocity X: Averaged velocity of the COP displacements on the X axis (transversal). Larger values correspond to poorer performance.
-
Sway velocity Y: Averaged velocity of the COP displacements on the Y axis (sagittal). Larger values correspond to poorer performance.
-
Sway area: Area of the smallest polygon that includes the entire trajectory of the COP. Larger values correspond to poorer performance.
As detailed in Fig. 1b, this block of testing is given 4 times per session: 15 min before the beginning of the exposure period, 15 min after the beginning of the MF exposure, 45 min after the beginning of the MF exposure, and 15 min after the end of the MF exposure.
The skin temperature was monitored throughout the entire experiment. After each block, the participant answered the Field Status Questionnaire (FSQ, Cook et al. 1992) to assess his ability to detect the presence of the field. The ambient geomagnetic static field was measured in the chamber as 47.6 μT (±0.5%) using a fluxgate magnetometer (Fluxgate FGM 3D2, Walker Scientific Inc, USA). The ambient ELF MF was recorded as 0.139 μT (±0.5%), also measured with a fluxgate magnetometer (Mag-03, Bartington Instruments, England). Additional background vibration was recorded with a seismic accelerometer (Model 393A03, PCB Piezotronics, USA) between the coils. When the MF was turned off, the background vibration was 1.70 × 10−2 m/s2, compared to 1.74 × 10−2 m/s2 (±4.9 × 10−5 m/s2) when the MF was on. This difference is well below reported values for human linear velocity and acceleration thresholds (Kingma 2005) and thus we were satisfied that participants were not able to perceive vibrations as a result of the field generation.
Statistics
Repeated measures ANOVAs were conducted on each characteristic and were computed for each test using SPSS (SPSS 16.0, USA). The general linear model (GLM) for repeated measures implemented in SPSS was accounting for multiple comparisons, so the p values reported for the main and interaction effects were adjusted in this regard. Probability values were corrected for lack of sphericity using the Greenhouse–Geisser epsilon. Since it was an exploratory study, no a priori contrasts, neither pair-wise comparisons, were conducted to further test the interaction effects. Though the ANOVA is reported to be robust even if the assumption of normal distribution is not met (Ferguson and Takane 2005), it was decided to run the entire statistical analysis on the log-transformed data as well. Statistical results from the non-transformed data were kept when they met normal distribution; those from the log-transformed data were used otherwise. For the sake of clarity, only representative results are presented in this paper.
More specifically, regarding EEG, a repeated measure ANOVA 2 (session—sham/real) × 4 (block 1/2/3/4) × 2 (eyes—open/closed) has been conducted on the amplitude in the theta, alpha, and beta frequency bands for electrodes F3, F4, C3, C4, P3, P4, O1, and O2 for the 58 subjects with a complete EEG data set. For the physiological tremor test, a repeated measure ANOVA 2 (session) × 4 (block) × 2 (eyes) has been conducted on each of the ten computed tremor characteristics for the 68 subjects with a complete tremor data set. The alternating hand movements analysis consisted in a repeated measure ANOVA 2 (session) × 4 (block) × 2 (frequency) that has been conducted on each of the eight computed characteristics for the 48 subjects with a complete voluntary movements data set. Finally, a repeated measure ANOVA 2 (session) × 4 (block) × 2 (eyes) has been conducted on each of the seven standing balance computed characteristics for the 67 subjects with a complete standing balance data set.
Results
EEG
Results in the alpha frequency band are presented in this section (Fig. 3). Results from the electrode O2 have been selected as representative of overall EEG results concerning the alpha rhythm. As expected, higher EEG alpha activity was found with eyes closed (n = 58; F = 67.36, p < 0.001, η2 = 0.54) than with eyes open. No significant session (F = 0.53, p > 0.05, η2 < 0.01), block (F = 2.61, p > 0.05, η2 = 0.04), or interaction effects were found. No significant interaction involving the exposure condition was found in any electrode for any of the frequency bands analyzed.
Physiological tremor
Only results corresponding to tremor amplitude (i.e. average oscillations size in the 2–20 Hz frequency range) are presented here (Fig. 4). Tremor amplitude was found to be higher in the eyes closed than in the eyes open condition (n = 68; F = 96.58, p < 0.001, η2 = 0.59). No significant session (F = 2.52, p > 0.05, η2 = 0.04), block (F = 0.71, p > 0.05, η2 = 0.01) or interaction effects were found (Fig. 4a). However, a non-significant trend suggests an increase of tremor amplitude during exposure in the real session when compared to the sham session (block 3, Fig. 4a).
Because this effect was close to significance, and because the averaged tremor power spectrum from the 68 subjects produced for blocks 2 and 3 (those during which the MF was actually produced) in the sham and real sessions seems to indicate an increase of the power for the oscillations within the 7–12 Hz frequency band (consistent in the eyes open and eyes closed conditions), the absolute value of the power in this frequency band has been computed as the integral of the power spectrum on this interval and was used in the analyses.
As with the amplitude, the absolute power in the 7–12 Hz range was higher in the eyes closed than in the eyes open condition (F = 19.16, p < 0.001, η2 = 0.22). No session (F = 3.83, p > 0.05, η2 = 0.05) or block (F = 2.71, p > 0.05, η2 = 0.04) effect was found. However, results show a significant session × block interaction (F = 2.83, p < 0.05, η2 = 0.04) illustrated in Fig. 4b. This effect suggests an increase of tremor power in this frequency band during the block 3 in the real condition while it has a trend toward decrease in the sham condition.
Alternating hand movements
Only results corresponding to alternating hand movement duration (i.e. average time to make a back and forth wrist rotation with the hands) are presented here (Fig. 5). As expected, a higher duration was found in the natural frequency than in the high frequency condition (n = 48; F = 356.60, p < 0.001, η2 = 0.89). Also, a significant main block effect (F = 23.56, p < 0.001, η2 = 0.34) demonstrated a tendency to increase movement frequency over the course of the experiment. No significant effect involving the MF condition was found.
Standing balance
Only results corresponding to sway velocity (i.e. the average velocity of oscillations over a period of recording) are presented here (Fig. 6). As expected, a higher sway velocity was found in the eyes closed than in the eyes open condition (n = 67; F = 717.17, p < 0.001, η2 = 0.917). No session (F = 0.785, p > 0.05, η2 = 0.01) or block effect was found (F = 2.62, p > 0.05, η2 = 0.04). Interestingly, a significant session × block × eyes interaction (F = 3.38, p < 0.05, η2 = 0.05) demonstrated a lower sway velocity while exposed to the MF in the eyes closed condition only (during blocks 2 and 3, sway velocity was smaller in the real exposure than in the sham exposure session, illustrated in Fig. 6b). Interestingly, the same interaction was found for the mean sway (F = 3.69, p < 0.05, η2 = 0.05), the sway velocity X (F = 5.53, p < 0.05, η2 = 0.08) and the sway area (F = 3.84, p < 0.05, η2 = 0.06).
A χ2 test performed on the FSQ results confirmed that the subjects were not capable to detect the presence of the field when it was actually on. Their estimation was wrong more than 50% of the time (χ2 = 7.53, p > 0.05): they were wrong in 76% of the cases.
Discussion
This section summarizes the main results of this project with the general objective of determining if a 60 Hz, 1,800 μT magnetic exposure was capable of consistently modulating normal neurophysiological functions in humans. This objective implied the selection of functional variables capable of capturing this effect. Our results show that in humans, the 60 Hz, 1,800 μT MF exposure did not significantly modulate EEG activity in the theta, alpha, beta and gamma frequency bands. Previous studies using different exposure protocols have reported ELF MF effects on EEG alpha rhythms suggesting that EEG modulations may be stimulus specific (Bell et al. 1994a, b; Cook et al. 2004, 2005a, b; Ghione et al. 2005; Heusser et al. 1997; Lyskov et al. 1993a, 2001; Marino et al. 2004). The difference between protocols reporting effects seems to indicate that the underlying mechanism of action is associated with the frequency of MF exposure and is not the consequence of the field intensity alone. The absence of effect in resting EEG does not mean a total absence of effect: it is possible to speculate that if the 60 Hz sinusoidal stimulus used in this study does not seem to modulate the EEG frequency, it could modulate the phase of brain oscillations, which could be detected with other techniques such as the analysis of evoked potentials (Glover et al. 2007b), or with the use of non-linear analysis (Carrubba et al. 2007a, b).
As anticipated, results from the alternating wrist movement were not affected by the MF. This task was presented as a pilot trial to study the potential modulations of voluntary movements caused by ELF MF. However, although this task was designed to diagnose specific neurodegenerative impairments (Beuter et al. 1999a), it has shown its sensitivity to the current protocol by demonstrating a spontaneous tendency of individuals to produce faster rhythms over time within an experimental session. However, it may not be a sensitive enough task to pick up a small MF effect if it exists.
Our hypothesis of a reduction of tremor intensity as a result of MF exposure was not supported. On the contrary, our results suggest a small amplitude increase during exposure (after 50 min of MF “on”). This result contradicts findings from previous studies that suggest a decrease of tremor intensity with a short (a few minutes) 1,000 μT, 50 Hz MF exposure (Legros et al. 2006). This tendency, would it be reproduced, would support the aforementioned hypothesis that specific responses occur according to specific stimuli (in terms of amplitude, frequency and duration). Since in the present study physiological tremor is only modulated in the 7–12 Hz frequency band, which corresponds, according to certain authors, to the contribution of a central pacemaker to its generation (see Deuschl et al. 2001; McAuley and Marsden 2000 for reviews), it cannot be excluded that the 60 Hz stimulus might be capable of modulating the involved cerebral rhythms in deep structures such as the thalamus, which would not, however, interfere with the cortical EEG rhythms.
The most significant result attained from this study shows that standing balance oscillations produced during MF exposure were slower and smaller in amplitude as compared to the oscillations produced during the sham exposure. This result is in accordance with our hypotheses and supports results from previous studies conducted by our group, which demonstrated a reduction of anteroposterior natural oscillations with a specific pulsed 200 μT MF exposure (Thomas et al. 2001a). Normal standing balance is mainly controlled by three feedback loops: the visual loop, the proprioceptive loop, and the vestibular loop. Since the effect of the MF exposure was present only in the eyes closed condition, i.e. when the visual loop is not processing, we hypothesize that the ELF MF may act on either the vestibular or proprioceptive functions. Further research needs to be conducted in this area to study this effect in more details. Interestingly, the vestibular hypothesis is also proposed by Glover et al. in their study of vertigo induced by displacements in a 7 T static magnetic field (Glover et al. 2007a).
Conclusion
The general objective of this work was to provide a general view, from a multidimensional perspective, of the effects of a 60 Hz, 1,800 μT MF in humans. The large sample size, the counterbalanced statistical design with repeated measures, the implementation of positive control tests (eyes open/closed for tremor, postural sway and EEG; natural/maximum frequency in the alternating movements of the hands) and the diversity in the investigated functions and processes were the main strengths of this work.
In summary, the results from the current study show that 1 h of 60 Hz, 1,800 μT MF exposure does not modulate cardiovascular parameters (McNamee et al. 2010), neurophysiological processes involved in EEG rhythms generation, or voluntary rhythmic movement generation. However, our results indicate a MF effect leading to a small increase in tremor amplitude, localized in the frequency band associated with the contribution of central nervous system to its production (Elble and Koller 1990). Moreover, as already reported by our group (Thomas et al. 2001a), results show that MF exposure is associated with a decrease of standing balance oscillations. Since this effect appears only during the eyes closed condition, it suggests that the exposure acts on proprioceptive and/or vestibular functions (Glover et al. 2007a). This finding demonstrates that an acute global exposure of intensity slightly above the 2010 ICNIRP reference level in a controlled environment (1,000 μT) can be detected in humans. Would this result be reproduced, it would demonstrate a biological effect (not an adverse effect) of an ELF MF exposure encouraging future studies focusing on the involved mechanisms. Further studies aiming to detect and establish a threshold at which a MF exposure produces coherent and systematic effects on human physiology are needed to provide solid bases to better approach and characterize the underlying mechanisms of action.
References
Attwell D (2003) Interaction of low frequency electric fields with the nervous system: the retina as a model system. Radiat Prot Dosim 106:341–348
Bailey WH, Nyenhuis JA (2005) Thresholds for 60 Hz magnetic field stimulation of peripheral nerves in human subjects. Bioelectromagnetics 26:462–468
Bell GB, Marino AA, Chesson AL (1992) Alterations in brain electrical activity caused by magnetic fields: detecting the detection process. Electroencephalogr Clin Neurophysiol 83:389–397
Bell GB, Marino AA, Chesson AL (1994a) Frequency-specific blocking in the human brain caused by electromagnetic fields. Neuroreport 5:510–512
Bell GB, Marino AA, Chesson AL (1994b) Frequency-specific responses in the human brain caused by electromagnetic fields. J Neurol Sci 123:26–32
Beuter A, Edwards R (1999) Using frequency domain characteristics to discriminate physiologic and parkinsonian tremors. J Clin Neurophysiol 16:484–494
Beuter A, Edwards R (2002) Characterization and discrimination of kinetic tremor in Parkinson’s disease. Rev Neurol (Paris) 158:338–340
Beuter A, de Geoffroy A, Edwards R (1999a) Analysis of rapid alternating movements in Cree subjects exposed to methylmercury and in subjects with neurological deficits. Environ Res 80:64–79
Beuter A, de Geoffroy A, Edwards R (1999b) Quantitative analysis of rapid pointing movements in Cree subjects exposed to mercury and in subjects with neurological deficits. Environ Res 80:50–63
Beuter A, Legros A, Cif L et al (2004) Quantifying motion in dystonic syndromes: the bare essentials. J Clin Neurophysiol 21:209–214
Carrubba S, Frilot C 2nd, Chesson AL Jr et al (2007a) Evidence of a nonlinear human magnetic sense. Neuroscience 144:356–367
Carrubba S, Frilot C, Chesson AL et al (2007b) Nonlinear EEG activation evoked by low-strength low-frequency magnetic fields. Neurosci Lett 417:212–216
Cook MR, Graham C, Cohen HD et al (1992) A replication study of human exposure to 60-Hz fields: effects on neurobehavioral measures. Bioelectromagnetics 13:261–285
Cook CM, Thomas AW, Prato FS (2002) Human electrophysiological and cognitive effects of exposure to ELF magnetic and ELF modulated RF and microwave fields: a review of recent studies. Bioelectromagnetics 23:144–157
Cook CM, Thomas AW, Prato FS (2004) Resting EEG is affected by exposure to a pulsed ELF magnetic field. Bioelectromagnetics 25:196–203
Cook CM, Thomas AW, Keenliside L et al (2005a) Resting EEG effects during exposure to a pulsed ELF magnetic field. Bioelectromagnetics 26:367–376
Cook CM, Thomas AW, Prato FS (2005b) Changes in human EEG alpha activity following exposure to two different pulsed magnetic fields sequences. Bioelectromagnetics Annual Meeting, Dublin, Ireland
Cook CM, Saucier DM, Thomas AW et al (2006) Exposure to ELF magnetic and ELF-modulated radiofrequency fields: The time course of physiological and cognitive effects observed in recent studies (2001–2005). Bioelectromagnetics 27:613–627
Crasson M (2003) 50-60 Hz electric and magnetic field effects on congitive function in humans: a review. Radiat Prot Dosimetry 106(4):333–340
Despres C, Lamoureux D, Beuter A (2000) Standardization of a neuromotor test battery: the CATSYS system. Neurotoxicology 21:725–735
Deuschl G, Raethjen J, Lindemann M et al (2001) The pathophysiology of tremor. Muscle Nerve 24:716–735
Dimbylow PJ (1998) Induced current densities from low-frequency magnetic fields in a 2 mm resolution, anatomically realistic model of the body. Phys Med Biol 43:221–230
Edwards R, Beuter A (2000) Using time domain characteristics to discriminate physiologic and parkinsonian tremors. J Clin Neurophysiol 17:87–100
Elble RJ, Koller WC (1990) Tremor. The John Hopkins University press, London
Ferguson GA, Takane Y (2005) Statistical analysis in psychology and education. McGraw–Hill Ryerson Limited, Montréal
Gandhi OP, Kang G, Wu D et al (2001) Currents induced in anatomic models of the human for uniform and nonuniform power frequency magnetic fields. Bioelectromagnetics 22:112–121
Gauger JR (1985) Household appliance magnetic field survey. IEEE Trans Power Appar Syst PAS 104:2436–2444
Ghione S, Seppia CD, Mezzasalma L et al (2005) Effects of 50 Hz electromagnetic fields on electroencephalographic alpha activity, dental pain threshold and cardiovascular parameters in humans. Neurosci Lett 382:112–117
Glover PM, Cavin I, Qian W et al (2007a) Magnetic-field-induced vertigo: a theoretical and experimental investigation. Bioelectromagnetics 28:349–361
Glover PM, Eldeghaidy S, Mistry TR et al (2007b) Measurement of visual evoked potential during and after periods of pulsed magnetic field exposure. J Magn Reson Imaging 26:1353–1356
Graham C, Sastre A, Cook MR et al (2000a) Nocturnal magnetic field exposure: gender-specific effects on heart rate variability and sleep. Clin Neurophysiol 111:1936–1941
Graham C, Sastre A, Cook MR et al (2000b) Heart rate variability and physiological arousal in men exposed to 60 Hz magnetic fields. Bioelectromagnetics 21:480–482
Graham C, Sastre A, Cook MR et al (2000c) Exposure to strong ELF magnetic fields does not alter cardiac autonomic control mechanisms. Bioelectromagnetics 21:413–421
Greenland S, Kheifets L (2006) Leukemia attributable to residential magnetic fields: results from analyses allowing for study biases. Risk Anal 26:471–482
Heusser K, Tellschaft D, Thoss F (1997) Influence of an alternating 3 Hz magnetic field with an induction of 0.1 millitesla on chosen parameters of the human occipital EEG. Neurosci Lett 239:57–60
ICNIRP (1998) Guidelines for limiting exposure to time-varying electric, magnetic, and electromagnetic fields (up to 300 GHz). International Commission on Non-Ionizing Radiation Protection. Health Phys 74:494–522
ICNIRP (2010) Guidelines for limiting exposure to time-varying electric and magnetic fields (1 Hz to 100 kHz). Health Phys 99:818–836
IEEE (2001) IEEE P1555/D5 Draft Standard for safety levels with respect to human exposure to electric and magnetic fields 0 to 3 kHz. IEEE, New York
Kaune WT, Miller MC, Linet MS et al (2002) Magnetic fields produced by hand held hair dryers, stereo headsets, home sewing machines, and electric clocks. Bioelectromagnetics 23:14–25
Kingma H (2005) Thresholds for perception of direction of linear acceleration as a possible evaluation of the otolith function. BMC Ear Nose Throat Disord 5:5
Kurokawa Y, Nitta H, Imai H et al (2003) Can extremely low frequency alternating magnetic fields modulate heart rate or its variability in humans? Auton Neurosci 105:53–61
Lacy-Hulbert A, Metcalfe JC, Hesketh R (1998) Biological responses to electromagnetic fields. FASEB J 12:395–420
Legros A, Beuter A (2005) Effect of a low intensity magnetic field on human motor behavior. Bioelectromagnetics 26:657–669
Legros A, Beuter A (2006) Individual subject sensitivity to extremely low frequency magnetic field. Neurotoxicology 27:534–546
Legros A, Cif L, Sygiel M et al (2004) Kinematic evaluation of dystonic syndromes in patients treated with deep brain stimulation. Rev Neurol (Paris) 160:793–804
Legros A, Gaillot P, Beuter A (2006) Transient effect of low-intensity magnetic field on human motor control. Med Eng Phys 28:827–836
Levallois P (2002) Hypersensitivity of human subjects to environmental electric and magnetic field exposure: a review of the literature. Environ Health Perspect 110(Suppl 4):613–618
Lovsund P, Oberg PA, Nilsson SE (1979) Influence on vision of extremely low frequence electromagnetic fields. Industrial measurements, magnetophosphene studies volunteers and intraretinal studies in animals. Acta Ophthalmol (Copenh) 57:812–821
Lovsund P, Oberg PA, Nilsson SE (1980a) Magneto- and electrophosphenes: a comparative study. Med Biol Eng Comput 18:758–764
Lovsund P, Oberg PA, Nilsson SE et al (1980b) Magnetophosphenes: a quantitative analysis of thresholds. Med Biol Eng Comput 18:326–334
Lyskov EB, Aleksanian ZA, Iousmiaki V et al (1993a) Neurophysiologic effects of short-term exposure to ultra-low-frequency magnetic field. Fiziol Cheloveka 19:121–125
Lyskov EB, Juutilainen J, Jousmaki V et al (1993b) Effects of 45-Hz magnetic fields on the functional state of the human brain. Bioelectromagnetics 14:87–95
Lyskov E, Sandstrom M, Mild KH (2001) Provocation study of persons with perceived electrical hypersensitivity and controls using magnetic field exposure and recording of electrophysiological characteristics. Bioelectromagnetics 22:457–462
Maresh CM, Cook MR, Cohen HD et al (1988) Exercise testing in the evaluation of human responses to powerline frequency fields. Aviat Space Environ Med 59:1139–1145
Marino AA, Nilsen E, Chesson AL Jr et al (2004) Effect of low-frequency magnetic fields on brain electrical activity in human subjects. Clin Neurophysiol 115:1195–1201
Maruvada PS, Jutras P (1993) Études des sites susceptibles d’entraîner des expositions élevées des travailleurs d’Hydro-Québec aux champs électriques et magnétiques Hydro-Québec
McAuley JH, Marsden CD (2000) Physiological and pathological tremors and rhythmic central motor control. Brain 123(Pt 8):1545–1567
McNamee DA, Legros AG, Krewski DR et al (2009) A literature review: the cardiovascular effects of exposure to extremely low frequency electromagnetic fields. Int Arch Occup Environ Health 82:919–933
McNamee DA, Corbacio M, Weller JK et al (2010) The cardiovascular response to an acute 1800-microT, 60-Hz magnetic field exposure in humans. Int Arch Occup Environ Health 83:441–454
Prato FS, Thomas AW, Cook CM (2001) Human standing balance is affected by exposure to pulsed ELF magnetic fields: light intensity-dependent effects. Neuroreport 12:1501–1505
Rubin GJ, Das Munshi J, Wessely S (2005) Electromagnetic hypersensitivity: a systematic review of provocation studies. Psychosom Med 67:224–232
Sastre A, Cook MR, Graham C (1998) Nocturnal exposure to intermittent 60 Hz magnetic fields alters human cardiac rhythm. Bioelectromagnetics 19:98–106
Sastre A, Graham C, Cook MR (2000) Brain frequency magnetic fields alter cardiac autonomic control mechanisms. Clin Neurophysiol 111:1942–1948
Saunders RD, Jefferys JG (2007) A neurobiological basis for ELF guidelines. Health Phys 92:596–603
Silny J (1986) The Influence of the Time-Varying Magnetic Field in the Human Organism. In: Bernhardt J (ed) Biological effects of static and extremely low frequency magnetic fields. MMV Meizin Verlag München, Neuherberg, pp 105–112
Thomas AW, Drost DJ, Prato FS (2001a) Human subjects exposed to a specific pulsed (200 microT) magnetic field: effects on normal standing balance. Neurosci Lett 297:121–124
Thomas AW, White KP, Drost DJ et al (2001b) A comparison of rheumatoid arthritis and fibromyalgia patients and healthy controls exposed to a pulsed (200 microT) magnetic field: effects on normal standing balance. Neurosci Lett 309:17–20
WHO (2007) Extremely Low Frequency Fields Environmental Health Criteria Monograph No.238. WHO, Geneva. http://www.who.int/peh-emf/publications/Complet_DEC_2007.pdf
Acknowledgments
The authors thank Mr. Lynn Keenliside for this technical assistance; Ms. Samantha Brown, Julie Weller and Mr. David McNamee for their assistance in the data collection. This project was sponsored by Hydro-Québec, Électricité de France and Réseau de Transport d’Électricité. This study was also in part supported by Canadian Institutes of Health Research grants (MOP 43874 and FRN 85217), the Ontario Research and Development Challenge Fund (MAR-01- 0936), and the Canadian Foundation for Innovation (11358).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Fausto Baldissera.
Rights and permissions
About this article
Cite this article
Legros, A., Corbacio, M., Beuter, A. et al. Neurophysiological and behavioral effects of a 60 Hz, 1,800 μT magnetic field in humans. Eur J Appl Physiol 112, 1751–1762 (2012). https://doi.org/10.1007/s00421-011-2130-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-011-2130-x