Abstract
Muscle mass and strength are well known to decline in response to actual and simulated microgravity exposure. However, despite the considerable knowledge gained on the physiological changes induced by spaceflight, the mechanisms of muscle atrophy and the effectiveness of in-flight countermeasures still need to be fully elucidated. The present review examines the effects and mechanisms of actual and simulated microgravity on single fibre and whole muscle structural and functional properties, protein metabolism, tendon mechanical properties, neural drive and reflex excitability. The effects of inflight countermeasures are also discussed in the light of recent advances in resistive loading techniques, in combined physical, pharmacological and nutritional interventions as well as in the development of artificial gravity systems. Emphasis has been given to the pioneering work of Pietro Enrico di Prampero in the development of artificial gravity systems and in the progress of knowledge on the limits of human muscular performance in space.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The loss of skeletal muscle mass in response to microgravity exposure has been a medical and physiological concern since the early Gemini, Soyuz and Skylab missions (Convertino 1990) and, together with changes in neural drive, is a main determinant of the decline in muscle strength and power (di Prampero and Narici 2003). Since Yuri Gagarin’s flight in 1961, more than 500 human beings have flown into space according to records of the Fédération Aéronautique Internationale (FAI). However, despite the considerable knowledge gained in space physiology, the mechanisms of muscle atrophy and the effectiveness of in-flight countermeasures still need to be fully elucidated. Progress in the knowledge of the mechanism of muscle atrophy and weakness has not been simple because of, (a) the small sample size of astronauts/cosmonauts involved in the same flight mission, (b) differences in mission duration, (c) type of and adherence to inflight countermeasures, (d) sex-related differences, and, (e) individual variability. A full understanding of the mechanisms of disuse-muscle atrophy is even incomplete for Earth-bound individuals (Rennie et al. 2010) and this problem, together with the constraints imposed by spaceflight, makes the task of identifying causes and remedies a major challenge. Nevertheless, also with the use of simulated microgravity paradigms such as bed rest and lower limb suspension, a wealth of useful data have been obtained. This information is of fundamental importance not only for being able to sustain human presence in space and extend the exploration of our Solar system, but also for enhancing the understanding of disuse-atrophy in bed-ridden patients and its contribution to sarcopenia in older individuals. Although full prevention of muscle atrophy and weakness due to spaceflight has not yet been fully achieved through the use of the available inflight countermeasure programmes, recent advances in resistive loading techniques, the use of combined physical, pharmacological and nutritional interventions (Pavy-Le Traon et al. 2007), as well as the development of artificial gravity systems (di Prampero et al. 2009; di Prampero 2000; di Prampero and Antonutto 1997), seem particularly promising as suggested by several ground-testing studies.
Effect of actual and simulated microgravity on muscle size
Whole muscle atrophy
It is well established that skeletal muscle undergoes substantial atrophy in response to actual and simulated microgravity exposure. Data from animal models show that postural muscles, generally containing a higher percentage of slow fibres, are more prone to atrophy than non-postural ones (Gardetto et al. 1989; Ohira et al. 1992; Roy et al. 1987).
However, the existing data on the effect of spaceflight on human skeletal muscle size (Table 1) show considerable heterogeneity in terms of size and rate of atrophy. This may be the result of various factors, such as age, pre-flight fitness level and nutritional status, and the content of and adherence to onboard countermeasures. For example, Kozlovskaya et al. (1981) observed that after 140 and 175-day Mir missions, the reduction in leg circumference (which underestimates changes in muscle CSA as it includes subcutaneous fat and bone) was relatively small when compared to those found after short-term spaceflight (Table 1). As Table 1 suggests, irrespective of the type of in-flight countermeasure, losses of lower limb muscle CSA, or volume, between 6 and 24% should be expected for spaceflight durations from 8 to 197 days.
Amongst the several paradigms used to simulate microgravity (Table 1), bed rest is the most commonly utilised since it can be performed under strictly controlled conditions and, when combined with a −6° head-down tilt, it mimics the fluid shift that occurs in space (Montgomery 1993). With this model, the loss of muscle mass is very rapid, since a 3% decrease in thigh volume is observed after only 7 days of recumbency (Ferrando et al. 1995) and after 90–120 days reductions in quadriceps and calf muscle volume approximating 30% have been found (Alkner and Tesch 2004a; Shackelford et al. 2004). Similar findings have recently been reported by Belavy et al. (2009) after 56 days of bed rest, confirming the original observations of Leblanc et al. (1997) that, amongst the muscles of the lower limbs, the calf muscles are those most prone to atrophy. Although Belavy et al. (2009) claimed as novel the observation of a heterogeneous pattern of atrophy amongst the muscles of the lower limb in response to unloading, this was earlier described by Narici and Capodaglio (1998). Despite the heterogeneity in their degree of atrophy, the muscles of the lower limbs are far more affected by unloading than those of the upper limb since no changes in human deltoideus muscle fibre morphology and enzymatic activities of oxidative and glycolytic enzymes were found after 37-day bed rest (Desplanches et al. 1998).
Unilateral limb suspension (ULLS) has also been shown to be an effective disuse paradigm for inducing muscle atrophy. In a 4-week ULLS investigation that pioneered the use of this model, Berg et al. (1991) reported a decrease in knee extensors peak torque of 22% and cross-sectional area of 7%. Several subsequent studies (Tables 1, 2) confirmed the validity of ULLS in inducing losses in muscle mass and function; these have been found to be quantitatively and qualitatively similar to those produced by spaceflight and bed rest (Adams et al. 1994).
More recently, de Boer et al. (2007a, b) studied the time course of the musculo-skeletal adaptations to ULLS and reported decreases in quadriceps CSA of 5.2 and 10% after 14 and 23 days of ULLS, equivalent to an approximate rate of loss ~0.4%/day (assuming that over 3 weeks the decline is almost linear).
Immobilization by casting of the lower limb has also been used to induce muscle atrophy (Table 1). For example, a 12% reduction in leg volume was reported after immobilization (Sargeant et al. 1977) accompanied by 46 and 37% decreases in VL type I and type II fibre CSA, respectively.
When the data on the relative loss of plantarflexor and knee extensor CSA or volume extracted from studies performed in actual and simulated microgravity are plotted against time (Fig. 1), the following general observations may be made: (1) over the course of time, and for both muscle groups, atrophy proceeds in a non-linear fashion, (2) in both environments, atrophy of the plantarflexors is greater than that of the knee extensors, (3) up to about 50 days, the degree of atrophy observed in actual and simulated microgravity are similar for both muscles, (3) for spaceflight of 180–200 days the degree of atrophy is comparable, if not smaller, than that observed in simulated microgravity models of 85–120 days. This apparent paradox is probably due to insufficient sample size in spaceflight studies and also to the effect of inflight countermeasures. One limitation of this graph is that muscle CSA and volumes are plotted together. However, it can be seen from Table 1 that up to about 20 days the decreases in muscle CSA and volume are similar. Instead, for longer durations, the decline in volume tends to be smaller than that of CSA as changes in volume will average changes in CSA along the muscle belly. Despite these limitations, the observation that the plantarflexors are more prone to atrophy than the knee extensors seems particularly noteworthy. Although the exact reasons for this phenomenon have not been fully understood, they may reflect different loading patterns during habitual locomotor activities that are likely to influence protein turnover of these muscles. During locomotion, as body mass is propelled forward, the plantarflexors are likely to experience a proportionally greater loading than the knee extensors because their muscle mass is about 50% smaller (c.f. muscle volume data in Alkner and Tesch 2004b). Thus, given these differences in loading patterns, the unloading effects on the plantarflexors would be expected to be greater than those on the knee extensors.
Muscle size versus duration of unloading for knee extensor and plantarflexor muscles. Data are combined from the different unloading paradigms and contain the results from the references presented in Tables 1 and 2. PF plantarflexors, KE knee extensors, SF spaceflight, BR bed rest, ULLS unilateral lower limb suspension
Single fibre atrophy
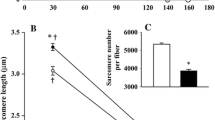
The decline in muscle mass induced by spaceflight is associated with a decrease in fibre size, rather than with a reduction in fibre number (Roy et al. 1987; Templeton et al. 1984). Although disuse atrophy in animals is by-and-large fibre specific, there seems to be a clear difference in the response of rat and human calf muscles to microgravity. This is because, (1) disuse-atrophy in rodents tends to be greater, over a given time, than in humans (on average 1.5–3.0%/day vs. 0.4–0.75%/day, respectively (Rennie et al. 2010), (2) rodents are much less metabolically stable than humans, (3) they display a clear pattern of selective atrophy of slow fibres while in humans the evidence of this seems weaker, (4) data on rodents is mainly on immature animals, still growing whereas data for humans are on mature beings (Rennie et al. 2010). For instance, when fibres of the soleus muscle from the two species are compared, there seems to be a preferential atrophy of the slow antigravity type I fibres in rats, but not in humans (Fitts et al. 2000). It has even been suggested that atrophy of fast type II fibres is comparable, if not greater than, that of slow type I fibres (Fitts et al. 2000). In agreement with this, 11 days of human spaceflight resulted in a greater reduction in fibre size of vastus lateralis (VL) type IIx fibres than in type I fibres (Edgerton et al. 1995). Similar observations were made in the human soleus muscle after 17 days of spaceflight, as type IIa fibre cross-sectional area decreased by 26% compared to a 15% decline in the slow type I fibre cross-sectional area (Widrick et al. 1999). However, no changes were observed in the gastrocnemius fibres after a 17-day flight (Trappe et al. 2001). The inconsistency of the data of human fibre atrophy with spaceflight is likely due to various factors such as the confounding effect of in-flight countermeasures and activities, mission duration, and nutrition. The combination of these factors, together with the limited data so far available, does not enable to draw any objective conclusions on differences in fibre type susceptibility to atrophy due to microgravity.
As far as simulated microgravity is concerned, some evidence of preferential type I fibre atrophy for human muscle, seems to emerge from various studies using different unloading paradigms. However, there are also reports of greater atrophy of type II fibres (Fitts et al. 2001).
In fact, contrary with the pattern seen with spaceflight, Rudnick et al. (2004) reported greater atrophy of type I than of type II fibres in the soleus and VL muscles after 12 weeks of bed rest. Similarly, 84 days of bed rest resulted in a reduction in VL type I fibre diameter of 15%, whereas type II fibre diameter was only reduced by 8% (Trappe et al. 2004), although gender differences might exist in this response (Trappe et al. 2007). Also, after 21 days of immobilization VL fibre type I CSA declined by 13%, whereas both type IIa and IIx CSA was reduced by 10% (Hortobagyi et al. 2000). This seems consistent with the findings of a reduction of 7% in soleus fibre diameter without any changes in the gastrocnemius muscle following 12 days of ULLS (Widrick et al. 2002).
Greater susceptibility of slow muscle to atrophy has also been recently observed by Haus et al. (2007) comparing myofibrillar and sarcoplasmic protein content of the human soleus and VL muscles in response to 35 day ULLS and 90-day bed rest. They found that the general (mixed, sarcoplasmic and myofibrillar) as well as force-specific (myosin, actin and collagen) protein fractions were maintained in samples of the VL in both unloading paradigms, whereas in the soleus the general protein fraction was reduced at the expense of the force-specific proteins. Although this study did not specifically assess fibre size, protein content probably gives a more robust information on fibre atrophy than fibre size alone and, as the results show, the evidence points to a greater susceptibility of type I fibres to atrophy.
In contrast, Veldhuizen et al. (1993) reported that VL fibre type IIx showed the greatest atrophy, followed by type I and type IIa. Greater atrophy of fast fibres was also reported by Jaweed (1994) following 42 days of ULLS as the decrease in VL fibre CSA was 12% for type I and 15% for type II fibres, respectively.
Thus, although the issue seems unsettled, there seems to be stronger evidence of greater susceptibility of human type I fibres to atrophy in response to simulated microgravity.
Protein synthesis and breakdown
The loss of muscle mass observed in actual and simulated microgravity is the result of a disruption in the balance between protein synthesis and breakdown (Gamrin et al. 1998). A reduction in muscle protein anabolism has indeed been observed in both animal and human models following immobilization (Gibson et al. 1987), and actual spaceflight (Ferrando et al. 2002). In rodents, this fall in muscle protein synthesis is accompanied by an increase in degradation (Thomason and Booth 1990). However, this does not seem to be the case in humans (Ferrando et al. 1996; Paddon-Jones et al. 2006) as little or no changes in protein breakdown were observed after 14 and 28 days of bed rest and, more recently, after 10 and 21 days of a 23-day unilateral lower limb suspension (ULLS) study in humans. In fact, de Boer et al. (2007a, b) showed a 50% fall of myofibrillar protein synthesis in the human VL after just 10 days of ULLS with no marked increase in genes for proteolytic enzymes throughout the 21-day ULLS period. It seems that an increase in protein breakdown through activation of the ubiquitin–proteasome system (UPS) mainly occurs in inflammation-mediated muscle atrophy such as in cancer, COPD, critical illness, severe trauma, amyotrophic lateral sclerosis or AIDS (Murton et al. 2008). Instead, the data on changes in MAFbx/atrogin-1 and MuRF1 mRNAs and protein expression in non-inflammatory muscle atrophy are inconsistent and do not show a clear role of protein breakdown in disuse-atrophy in healthy individuals (Murton et al. 2008).
Growth factors and cytokines
Intricate signalling relationships, believed to drive these adaptations in protein synthesis and breakdown, exist between anabolic (e.g. IGF-1, -II, GH and testosterone) and catabolic (e.g. IL-1, IL-6 and TNF-α) pathways. The insulin-like growth factors (IGF)-I and -II have been implicated in several phases of muscle development mediating myoblast proliferation, survival and terminal differentiation. Furthermore, IGFs have been suggested to increase myotube diameter, increase amino acid uptake and stimulate protein synthesis as well as suppress protein degradation in cultured cells (Rommel et al. 2001). Elevated levels of several cytokines, such as IL-1, IL-6, and TNF-α (Payette et al. 2003; Zimmers et al. 2002), and myostatin (Zimmers et al. 2002) have been associated with reduced muscle strength and function. In addition, these cytokines are related to a loss of muscle mass through several pathways. Lower concentrations of circulating and intramuscular IGF-1 have been shown to be associated with increased levels of circulatory cytokines in cachexia (Stewart 2004), since these can interfere with IGF-1 gene and protein expression (Ferrucci and Guralnik 2003) in addition to hindering the interaction between IGF-1 and its receptor which results in a synthesis of muscle proteins leading to muscle loss. However, there is scanty evidence that disuse in healthy humans induced by actual or simulated microgravity leads to significant changes in these growth factors and cytokines. Instead, these seem to be confined to clinical conditions in which inflammation plays a primary role in muscle atrophy (Murton et al. 2008).
Mechanotransduction
Integrins, the major transmembrane components of the link between the extracellular matrix (ECM) and the cytoskeleton, are vital for transduction of mechanical forces from and to the cell (Shyy and Chien 1997). It seems clear that signalling pathways originating from ECM and integrin interplay are mediated by the non-receptor protein kinases focal adhesion kinase (FAK) and particular members of the mitogen-activated protein kinases (MAPKs) including extracellular-signal-regulated kinases (ERKs) (Fluck et al. 1999). Phosphorylation of FAK leads to the subsequent activation of the downstream MAPK-pathway that ultimately regulates transcription of genes imperative for cell growth and differentiation (Boudreau and Jones 1999). In a recent pioneering experiment on the effects of chronic unloading in humans, obtained by ULLS, FAK activity was found to decrease by 30% (Fig. 2) and content by 20% after just 10 days of ULLS (de Boer et al. 2007b). This fall in FAK activity was accompanied by a 50% decrease in the fractional rate of myofibrillar protein synthesis confirming the role of FAK as an upstream modulator of protein synthesis (Klossner et al. 2009).
Changes in FAK phosphorylation between days 0 and 10 and 10 and 21 in the two in the two groups of subjects of four subjects studied twice (reprinted from de Boer et al. 2007b, reproduced with Authors’ permission). Values are means and standard errors. *P < 0.05
Effect of unloading on muscle architecture
The structural arrangement of muscle fibres within human skeletal muscle is an important factor contributing to the mechanical functioning of the muscle–tendon unit as a whole. Previous studies making use of ultrasonography have found that after periods of disuse, changes in muscle size are accompanied by alterations in fascicle length (Lf) and pennation angle (θ) (Kawakami et al. 2000; Narici and Cerretelli 1998; Reeves et al. 2002). Indeed, in the gastrocnemius medialis (GM) muscle Lf and θ were found to decrease by 13 and 10% respectively after 90 days of bed rest (Reeves et al. 2002) and in the VL muscle both Lf and θ decreased by about 8% after 23 days of ULLS (de Boer et al. 2007a). The greater susceptibility to atrophy and to architectural changes of the GM compared to the VL in response to unloading was recently shown by de Boer et al. (2008) in a 5-week bed rest (BR) study. After BR, muscle thickness and Lf decreased by 12 and 6% in the GM and by 8 and 5% in the VL. Consistent with the notion that disuse-atrophy mainly affects the postural muscles, no changes in muscle thickness or architecture were found in the upper limb muscle biceps brachii (de Boer et al. 2008). Reductions in muscle fascicle length and pennation angle respectively reflect a loss of sarcomeres in series and in parallel (Gans and Bock 1965). This rapid remodelling of muscle architecture with unloading seems mediated by a fall in FAK activity since this was found to decrease by 30% of just 14 days of lower limb suspension in humans, concomitantly with a significant decrease in fascicle length (de Boer et al. 2007b).
Effect of actual and simulated microgravity on muscle function
Muscle strength
In both human beings and animals, considerable losses in muscle strength have been found after spaceflight. This phenomenon was observed after only 2–5 days of spaceflight in 12 cosmonauts of the Soyuz-3 to Soyuz-8 missions (Convertino 1990) and also in astronauts participating to Skylab missions lasting 28, 56, and 84 days who experienced a 5–26% decline in knee extensor and flexor muscle strength (Rummel et al. 1975). Also, Soviet cosmonauts showed a significant loss in ankle extensor muscles’ strength after long-duration (110–237 days) space missions (Grogor’eva and Kozlovskaia 1987).
These earlier pioneering observations were confirmed by later studies reporting reductions in maximum force of the plantarflexor, knee extensor and trunk flexor muscles during spaceflights of different duration (Table 2). As can be inferred from this table, variation in response exists amongst these studies; this can be likely ascribed to differences in flight duration, the muscle group tested, the method of testing, the type of contraction used, the joint angle at which the measurements were performed, and adherence to the onboard countermeasure program (Kozlovskaya and Grigoriev 2004). The term strength entails quite a broad spectrum of parameters concerning the force output of the muscle–tendon complex. Where available, the values of isometric maximum voluntary contractions (MVC) are displayed in Table 2, and when muscle force was tested at multiple joint angles, the angle closest to the optimum angle for force production was chosen.
All of the above investigations involved the use of voluntary contractions which are influenced by fluctuations of neural drive, known to be affected by spaceflight. To bypass this problem, electrically evoked contractions were used to study changes in plantarflexion muscle function during the 17-day LMS Spaceshuttle (STS-78) mission by Narici et al. (2003). The results showed a decline in tetanic force (−22%), in force/CSA (−19.5%) and an increase in fatiguability (+16%) that persisted and reached a nadir in the recovery phase, suggesting the presence of muscle damage due to reloading in 1 g. Hence independently of neural changes, muscle force undergoes a significant decline in microgravity and since the loss of force is greater than that of muscle size, this is evidence of a decrease in muscle intrinsic force (F/CSA). The loss of muscle force persisting during the recovery phase suggests the presence of muscle damage due to reloading in 1 g. MRI measurements performed in the same crew members after the flight revealed an increase in signal intensity of proton relaxation time, known to be associated with muscle damage (LeBlanc et al. 2000). Also, muscle biopsies obtained in these astronauts revealed a selective loss of thin filaments after the flight which has been proposed to increase the vulnerability of muscle fibres to damage because of an increased load per thin filament (Riley et al. 2000). Although muscle damage as a result of spaceflight has sofar not been directly investigated in humans, its presence is consistent with complaints of persistent muscle soreness by astronauts for several months after the flight (Fitts et al. 2001). Also, simulated microgravity experiments performed in humans showed various ultrastructural alterations such as disorganised myofibrils, cellular oedema, irregular Z-bands and fibre necrosis after 30 days of bed rest (Hikida et al. 1989).
Deficits in muscle strength after exposure to simulated microgravity are reported in Table 2. Similar to spaceflight, considerable strength losses already become apparent after short term unloading. As with the spaceflight data, strength data originating from several different types of measurement are reported that range from isometric MVC to averages of maximal concentric and eccentric contractions (Ploutz-Snyder et al. 1995), to a reduction in 1 repetition maximum on a knee extension resistance exercise device (Thom et al. 2001). Interestingly, regardless of this methodological diversity, the data from the different unloading paradigms show a remarkable similarity over time, which is especially evident in the early phases of unloading (Alkner and Tesch 2004b; Reeves et al. 2005).
After longer periods (28–42 days) of ULLS, the fall in KE performance apparently begins to level off while most of these investigations report decreases of ~20% in KE MVC (Berg et al. 1991; Dudley et al. 1992; Ploutz-Snyder et al. 1996). However, it is difficult to estimate the reductions in strength after longer durations of ULLS, since there are simply no data available to support this hypothesis. A better picture is drawn by the bed rest data, which seem to suggest that the fall in muscle performance approaches a plateau after 90–120 days of unloading (Alkner and Tesch 2004b; LeBlanc et al. 1992).
Muscle force per unit cross-sectional area
The tetanic force developed by a muscle normalized to its cross-sectional area, or specific force, has also been shown to adapt to unloading, indicating a qualitative adaptation of the muscle to unloading (Berg and Tesch 1996; Dudley et al. 1992; LeBlanc et al. 1988). Several factors have been called upon to explain this finding: (a) a reduction in the specific tension of single fibres, which in turn is believed to be due to a reduction in myofibrillar density (Larsson et al. 1996) and a selective loss of actin (Riley et al. 2000), (b) by a lower motor drive to the muscles (Berg and Tesch 1996; Duchateau 1995; Koryak 1998), (c) by a reduction in electromechanical coupling efficiency (Milesi et al. 2000), and (d) by an increase in the quantity of intramuscular non-contractile tissue (Ryan et al. 2002).
Muscle fibre force and specific tension
At the single fibre level, changes in contractile properties are also evident after exposure to actual or simulated microgravity. Peak force (P 0) and peak force normalized to fibre cross-sectional area (P 0/CSA), have been found to be reduced in both animals and humans. For example, a 25 and 45% reduction in P 0 have been observed in rat soleus muscle fibres after 14 (Stevens et al. 1993) and 18.5 (Rapcsak et al. 1983) days spaceflight, respectively. Also, as few as 7 days of hind limb suspension in rats resulted in a fall in P 0/CSA of 17% (McDonald and Fitts 1995). In humans, similar results have been found, as soleus fibre P 0/CSA decreased by 4% after 17 days of spaceflight with countermeasures (Widrick et al. 1999). These results have been confirmed during and after prolonged bed rest as P 0 was reduced by as much as 47% after 84 days (Trappe et al. 2004) and P 0/CSA by as much as 40% after 42 days (Larsson et al. 1996). Other studies have also confirmed the observed decreases in P 0 and P 0/CSA with bed rest in humans (Widrick et al. 1997; Yamashita-Goto et al. 2001). The reduction in P 0/CSA is mainly attributed to a lower myofibrillar density (Larsson et al. 1996), which suggests a decline in cross-bridge number rather than a lower force exerted by each cross-bridge (D’Antona et al. 2003). A role for the selective loss of actin, observed in rats with spaceflight (Fitts et al. 2000), in the fall of P 0/CSA seems improbable, since P 0/CSA was preserved when a disproportionate loss of actin occurred (Widrick et al. 1999). However, a decline in the amount of actin has been implicated in the loss of single fibre maximal unloaded shortening velocity, as will be discussed in the following sections (Riley et al. 2000).
Whole muscle power
The generation of muscle power is notably compromised with long term unloading, and to a greater extent than muscle force alone. This is especially evident after spaceflight, as shown by the experiments of di Prampero 2000; di Prampero and Narici 2003 using the multipurpose ergometer dynamometer “MED” (Fig. 3) during the Euromir 94 and 95 missions. In these missions, maximal explosive power of the lower limbs, measured on five astronauts/cosmonauts during maximal “all-out” pushes on a force platform, fell to about 67% after 31 days and to about 45% of pre-flight values after 180 days. At variance with these data, the maximal power developed during 6–7 s all-out bouts on a cycloergometer was reduced to a lesser extent, attaining ~75% of preflight value, regardless of the flight duration. Since in the same subjects, the muscle mass of the lower limbs decreased only 9–13%, irrespective of the flight duration, the authors suggested that a large fraction of the decline of the maximal power was due to the effects of weightlessness on motor unit recruitment pattern, electromechanical efficiency and predisposition to muscle damage (Antonutto et al. 1999).
Schematic representation of the multipurpose ergometer dynamometer (MED), reprinted from Antonutto et al. (1999). Subject sitting on carriage seat (C-S) pushes with both feet on force platforms (FP). Velocity (V) of consequent backward movement of C-S is determined by means of a wire tachometer (WT). Force (F) exerted by subject is measured by two load cells indwelling into FP. Instantaneous power \( \left( {\dot{W}} \right) \) is calculated as \( \dot{W} \) = F · V. Hinges (Hi) allowing tilting up of MED’s mobile frame, by action of hydraulic jack (HJ), and isokinetic cycloergometer (Cy), are also indicated. Reproduced with Authors’ permission
Also in long-term spaceflight (90–180 days) experiments on a total of 15 cosmonauts performed during the EuroMir ‘94, ‘95, ‘98-E and in Mir EO 19-24 missions, Lambertz et al. (2001) reported that despite countermeasures practiced aboard, spaceflight induced a decrease in maximal isometric torque (17%) and an increase in an index of maximal shortening velocity (31%) of the plantarflexors. Although muscle power was not assessed in this experiment, the results suggest that an increase in maximum shortening may in part compensate for the loss in muscle power (being the product of force and velocity), consistent to what observed in single fibres by Widrick et al. (1999). In these cosmonauts, Lambertz et al. (2001) also reported a 39% decrease on muscle activation after the flight, indicating a decreased ability in motor unit recruitment which is consistent with the observations of Antonutto et al. (1999).
A decline in muscle power, but of smaller amplitude, has also been found in simulated microgravity. Vertical jumping power was indeed found to decrease by 24% after 42 days (Ferretti et al. 2001) and by 30% after 90 days of bed rest without countermeasures but only by 10% in subjects that underwent flywheel resistive countermeasures (Rittweger et al. 2007).
The effects of unloading on mechanical power appear to be velocity-specific. After exposure to actual spaceflight (Kozlovskaya et al. 1981) or to any of the ground-based models described in this work (Adams et al. 1994; Berg et al. 1993; Dudley et al. 1992, 1989; LeBlanc et al. 1992), power at low velocities seems more severely compromised than at high velocities. This could possibly be due to an increase in shortening velocity (as described below), which may act to mitigate the effect of atrophy on mechanical power at higher velocities.
Single fibre power
The loss in peak power with microgravity can be translated to the single fibre level. For example, decreases of 16–20% in rat soleus single fibre peak power have been found after 6–14 days of spaceflight (Caiozzo et al. 1994, 1996). This loss of power occurred with a concomitant increase in single fibre maximum unloaded shortening velocity (V 0). This is interesting since power is the product of force and velocity, and these results suggest that the loss in muscle fibre peak force must be the main element responsible for the fall in peak power. Results from human studies are not so clear-cut, probably due to scant data. Soleus type I fibre peak power decreased by ~20% in two of the crewmembers, but in another two the increase in V 0 was enough to preserve power by compensating for the reduction in fibre force (Widrick et al. 1999). With bed rest (Trappe et al. 2004), the composite soleus muscle single fibre power was reduced by 23%. However, in the subjects undergoing bed rest in combination with flywheel exercise as a countermeasure, peak power was preserved. These results indicate that exercise, as countermeasure or during in-flight tasks, is imperative for the preservation of power by increasing peak force. Instead, changes in V 0 may be significant at single fibre level but of little consequence at phenotypic level.
Myosin heavy chains and maximum shortening velocity
Increases in the maximal shortening velocity of the calf muscles have consistently been found after spaceflight in humans and animals. The increases of 14 and 20% in maximal shortening velocity (V max)Footnote 1 after 6 (Caiozzo et al. 1994) and 14 days (Caiozzo et al. 1996) of spaceflight observed in rats, have been associated with a higher type IIx myosin heavy chain (MHC) expression and a lower expression of type I MHC. In humans, similar results have been found, as calf muscle V max increased by 4–75% in seven out of eight cosmonauts after 17 days of spaceflight (Lambertz et al. 2001). A study on human single fibre maximum unloaded shortening velocity showed that soleus type I fibre V 0 and V max increased by 30 and 44% respectively after a 17-day spaceflight, and that soleus type IIa fibre V 0 increased by 55% (Widrick et al. 1999). These results suggest that the higher shortening velocity of the plantarflexor muscles can be attributed both to an elevated maximal shortening velocity (V 0) of the individual slow and fast fibres and to an increased expression of fibres containing fast myosin (Fitts et al. 2001). It is unknown why V 0 and V max increase with microgravity, but a reduction in thin filament density has been proposed as one of the potential contributors. A selective loss of actin relative to myosin has indeed been found with spaceflight (Riley et al. 2000) and is thought to increase shortening velocity by increasing the space between myosin and actin (lattice spacing). This is suggested to detach cross-bridges earlier, reducing the internal drag during the final stage of the cross-bridge stroke (Riley et al. 2000).
The MHC shift from slow to fast isoforms mentioned above is a common observation with unloading in rat skeletal muscle (Baldwin and Haddad 2001). A 2.8-fold increase in the ratio of type I/IIa MHC was observed in humans after 84 days of bed rest (Trappe et al. 2004), in combination with an increase in the proportion of hybrid fibres co-expressing more than one MHC isoform. Similar observations were made in humans suffering form spinal cord injury (SCI) (Andersen et al. 1996) and in rats after spinal cord transsection (Talmadge 2000). Hence, these data suggest that fast fibre types are the default phenotypes.
Effect of unloading on neural drive and reflex excitability
The general suggestion from the available data on neural adaptations during and after exposure to microgravity is that the removal of weight bearing has a degenerative effect on neural drive and muscle activation capacity. For example, plantarflexor electromyographic activity (EMG) activity fell by ~35–40% after 90–180 days exposure to microgravity (Lambertz et al. 2001). In accordance with this, EMG activity recorded in the plantarflexor muscles is diminished rapidly with hind limb suspension in rats (Edgerton and Roy 1994), but is gradually increased to pre-unloading levels as unloading continues (Ohira et al. 1992). The available data on chronic EMG activity in humans shows that tibialis anterior and soleus muscles total EMG activity is increased with 17 days of spaceflight, without any change in the gastrocnemius medialis’ activity (Edgerton et al. 2001). Maximal EMG activity decreased by 19% during maximal contractions of the knee extensor muscles after 42 days of bed rest in humans (Berg et al. 1997). A reduction in EMG activity was also observed after 90 days bed rest in both the plantarflexor and knee extensor muscles during maximal contractions on either a flywheel ergometer or isokinetic dynamometer (Alkner and Tesch 2004b). These observations give support to the hypothesis of a reduction in motor unit recruitment with spaceflight put forward by Antonutto et al. (1999).
Data from a study on one subject show that 5 weeks of bed rest resulted in a deficit of 33% in central activation of the plantarflexor muscles and a reduced EMG activity during MVC in both the gastrocnemius lateralis (−51%) and soleus (−32%) muscles (Duchateau 1995). Another 20-day bed rest study resulted in 6.5% a fall in maximal voluntary activation of the knee extensors determined by twitch interpolation (Kawakami et al. 2001). Also, after 6 weeks of immobilisation of the human forearm, a decreased EMG activity of the thumb muscles was observed (Duchateau and Hainaut 1987). A very detailed study by Clark et al. (2006) investigated the neural and muscular parameters that were partly responsible for the fall in plantarflexor muscle strength after 4 weeks of ULLS and concluded that deficits in central activation are the primary contributors to strength loss based on a multiple regression analysis. These authors observed a slowing in the compound muscle action potential (M max) by 30%, a decrease in evoked doublet force and an increase in the ratio of twitch-to-doublet force, an altered postactivation potentiation response and a reduction in rate of electrically evoked force development in the initial phase of contraction. In addition, an increase in Hoffmann reflex to M max ratio (an indication of spinal α-motoneuron excitability), together with a reduction in nerve conduction velocity, were found (Clark et al. 2006). This confirmed previous Russian observations made during short-term Soyuz flights (2–18 days) of an increased reflex excitability (Cherepakhin and Pervushin 1970; Kakurin et al. 1971). Also, more recently, an increase in soleus H-reflex (normalised to M max) was described by Seynnes et al. (2008) after 23 days of ULLS in humans. Particular noteworthy of this study was a decreased sensitivity of the T-reflex evidenced by an increase in the threshold necessary for evoking this reflex (Seynnes et al. 2008).
Tendon adaptations
In response to chronic unloading, not only muscles but also tendons, undergo deconditioning. A reduction in tensile stiffness of the VL deep aponeurosis of 32% (Kubo et al. 2000) and 28% (Kubo et al. 2004) have been found after 20 days of bed rest. Marked changes in tendon mechanical properties were also found by Reeves et al. (2005) who reported a reduction in Achilles tendon stiffness and Young’s modulus of 58 and 57%, respectively, after 90 days of bed rest. This loss of tendon stiffness induced by disuse is very rapid, corresponding to 9.8% after 14 days and 29.3% after 23 days of ULLS (de Boer et al. 2007a). Since the decrease in tendon stiffness was of the same magnitude of that of Young’s modulus (i.e. stiffness normalized for tendon dimensions), the observed fall in stiffness was attributed to changes in tendon material properties. These may be influenced by alterations in the structure and packing of the collagen fibres (Danielsen and Andreassen 1988) as a result of differences either in the cross-link pattern of the collagen (Prockop and Kivirikko 1995). The decrease in tendon stiffness is reflected by an increase in tendon extensibility at a given level of force exerted by the muscle and this has implications on the rate of force development (RFD) (Wilkie 1949). Indeed, after 23-day ULLS, RFD was found to decrease by 38% (de Boer et al. 2007a).
Countermeasures
Several approaches, based mainly on muscular and cardiovascular countermeasures, have been proposed to combat the atrophy and deteriorated function of skeletal muscle that occurs in space owing to the lack of weight bearing. It is fair to say that none of these methods has been thoroughly evaluated with regard to their efficacy to prevent or ameliorate muscle atrophy and function impairment in humans exposed to chronic microgravity. However, and regardless of the mechanism(s) responsible for the negative impact of spaceflight on skeletal muscle, countermeasures to this effect are imperative for maintaining crew-health and mobility during long-duration space missions.
Most astronauts/cosmonauts, have carried out in-flight countermeasures which have varied considerably in type, duration and intensity (Convertino 1990; di Prampero and Narici 2003; Greenleaf et al. 1989a; Tesch and Berg 1997), but full prevention of muscle and strength loss has not been yet achieved. In addition to physical, also nutritional and pharmacological countermeasures have been tested in simulated microgravity studies and the combination of these three different types of intervention seems particularly promising.
Aerobic exercise
In-flight cycle ergometer exercise has been used mainly to maintain cardiovascular function (Chase et al. 1966). However, such exercises have been shown to be ineffective for maintaining musculo-skeletal function in space simply because the mechanical load provided by aerobic exercise programmes is too low to prevent muscle atrophy or to induce muscle hypertrophy in space or in 1 g. For instance, results from a 30-day bed rest study show that 30 min of cycle ergometer exercise performed 5 days per week, failed to prevent muscle atrophy and weakness when compared to non-exercising controls (Greenleaf et al. 1989b). Similarly, daily supine cycle ergometry exercise for 60 min at 40% of maximal aerobic power did not protect against muscle or strength loss in individuals subjected to 20 days bed rest (Suzuki et al. 1994).
Resistive exercise
A protective effect of resistive training on protein synthesis has been shown by several studies performed on animals and humans. Healthy men who were confined to bed rest for 2 weeks and performed knee extensor and ankle extensor resistance exercise every other day using a five set, 6–10 repetition regimen at about 80% of maximum, were able to maintain muscle protein synthesis rate. Subjects who performed no training, showed decreased muscle strength, mass and protein synthesis rate. The protocol was also sufficient to maintain dynamic strength; yet isometric strength and neural activation were reduced (Bamman et al. 1998; Ferrando et al. 1997). A protective effect of resistive training on protein synthesis has also been shown in rats in which 4 weeks of flywheel training significantly attenuated the reduction in soleus muscle mass and protein synthesis when compared to non-exercising rats (Fluckey et al. 2002). This mode of training based on concentric and contractions against a flywheel, introduced by Berg and Tesch (1998), has been used for the prevention of muscle atrophy and weakness in two models of simulated microgravity, ULLS and bed rest. For instance, quadriceps muscle atrophy, induced by 5 weeks of lowerlimb suspension, was prevented by four sets of seven maximal concentric and eccentric knee extensions, performed twice or thrice weekly and using flywheel training (Tesch et al. 2004). However, in humans, the soleus muscle shows limited predisposition to work-induced hypertrophy, especially when compared to the quadriceps. For instance, whereas high-intensity flywheel exercised performed twice per week during a 90-day bed rest study fully prevented quadriceps atrophy, it only partially mitigated atrophy of the soleus muscle (Alkner and Tesch 2004b). This limited predisposition to hypertrophy of the human soleus is also confirmed by findings of a poor increase in protein synthesis of this muscle in response to an acute resistive exercise bout (Trappe et al. 2004). Taken together, these results suggest that resistive exercise is presently the method of choice for mitigating or even preventing the negative effects of unloading on skeletal muscle. However, significant differences in the response to training exist among muscles and the causes thereof warrant further investigation.
Penguin suit exercise
This type of exercise, introduced by Russian space scientists, consists of an all-body suit with sewn-in elastic bands for maintaining a stretching load on antigravity muscles. It is difficult to judge whether this type of exercise is really effective in combating muscle atrophy and weakness during disuse in space or on Earth. However, in a small group of subjects (n = 4), who were bedridden for about 120 days, fibre size of the soleus muscle appeared maintained after performance of a single daily 10 h bout of modest loading, i.e. about 10 kg, using the Penguin suit. Instead, three subjects, who did not load the ankle extensor muscles, showed soleus atrophy (Ohira et al. 1999). Although these results may sound promising, no hard conclusions may be drawn from observations made on such small sample and it seems unlikely that such low loads may prove effective in preventing muscle atrophy and weakness of large antigravity muscles.
Pharmacological and nutritional interventions
Pharmacological interventions have been proposed for preventing or mitigating muscle loss associated with spaceflight. For some time clenbuterol (a beta-2 adrenergic agonist) has been used on farm animals to increase muscle mass and it has been shown that chronic administration of this drug induces muscle hypertrophy by increasing protein synthesis (Wineski et al. 2002). Although its precise mode of action is not clear, clenbuterol has been found to reduce muscle atrophy in hind-limb suspended rats (Apseloff et al. 1993). It has also proved successful in reducing disuse muscle atrophy in orthopaedic patients (Maltin et al. 1993) and has been proposed as a therapeutic tool to combat muscle atrophy in patients with neuromuscular disorders (Lynch et al. 2001). The use of albuterol (another beta-2 receptor agonist) combined with resistive exercise (flywheel) has been recently tested on 21 men (11 treated with albuterol, 10 with placebo) and 15 women (9 treated with albuterol, 6 with placebo) during a 40-day ULLS period (Caruso et al. 2005). The results showed that the combined resistive training-albuterol treatment increased muscle strength in the women group, whereas the placebo group lost strength.
Nutritional supplementations associated with resistive exercise have also been shown to protect from disuse atrophy induced by bed rest. Administration of essential amino acids (AA) with resistive training during 23-day bed rest has indeed been shown to attenuate the loss of muscle mass by about 2/3 compared to AA administration alone (Brooks et al. 2008). By contrast administration of a mixture of AA and carbohydrate without exercise mitigated but did not prevent muscle atrophy during 28 days of bed rest (Fitts et al. 2007).
Testosterone administration has also been proposed as a pharmacological countermeasure against muscle atrophy and weakness since it is known to increase protein synthesis in young and elderly adults (Ferrando et al. 1998; Urban et al. 1995). However, in a 28-day bed rest study testosterone administration to ten healthy young men was found to preserve protein balance but not muscle strength. The authors concluded that in the absence of daily ambulatory activity, testosterone administration does not increase or, in the case of this bed rest model, preserve muscle strength (Zachwieja et al. 1999). The observation that this hormone maintained protein turnover but failed to prevent knee extensors strength loss seems to suggest that either the force per unit area of muscle fibres declined with bed rest, as observed by Larsson et al. (1996), or/and a decrease in motor unit recruitment occurred, as shown by previous studies (Alkner and Tesch 2004b; Berg et al. 1997). However, the results of this study seem at odd with the observation that resistive exercise during 30-day bed rest actually depresses testosterone level (Wade et al. 2005) suggesting that a synergistic action of exercise plus testosterone may not exist during bed rest or that exercise levels at a much lower intensity may be needed to prevent muscle loss in these conditions. Hence more studies are needed before conclusive statements may be drawn.
Considerable attention has also been given to insulin-like growth factor-1 (IGF-1) as a possible remedy against muscle atrophy in actual and simulated microgravity. Although low IGF-1 levels are associated with muscle atrophy in various human conditions (Wang et al. 2005) no protective effects on muscle mass have been found in transgenic mice overexpressing human IGF-I since these exhibited similar atrophy to normal mice during 14-day hind limb suspension (Criswell et al. 1998). Also, intramuscular IGF-1 mRNA levels have been found not to differ between space-flown rats and ground-control rats whereas IGF-II levels were depressed. In the same study, myostatin levels were significantly increased by spaceflight. This suggests that the levels of negative regulators of muscle mass are increased with spaceflight while those of the positive regulators are depressed (Lalani et al. 2000). Hence further studies are needed before growth factors may be considered for the prevention of muscle atrophy in space.
Electrical stimulation
The use of transcutaneous electrical muscle stimulation (EMS) has been tested as a countermeasure against muscle atrophy and weakness during a 30-day bed rest study (Duvoisin et al. 1989). In this investigation, three healthy subjects were treated unilaterally with EMS (60 Hz frequency, 0.30 ms pulse width, 4 s train duration) twice daily every third day. The results showed a smaller decrease in strength and muscle mass in limb treated with EMS than in the non-stimulated limb. Although these results show a mitigation of muscle atrophy and weakness by EMS, they do not show a prevention of these conditions and do not provide a comparison between the effects of EMS and those of voluntary contractions. Also, in a hind-limb suspension (HS) study in rats, daily transcutaneous EMS plus HS did not provide protection against muscle atrophy when compared to rats exposed to HS only (Yoshida et al. 2003). Although EMS has the advantage of standardising muscle activation, contraction level and duration, to compare in terms of efficacy to voluntary contractions, it requires high stimulation currents, and these are quite uncomfortable.
Artificial gravity: centrifuges and cycling in space
This concept of countermeasure is probably the most innovative but also the most challenging. Since chronic exposure to gravitational force on Earth is sufficient to prevent muscle atrophy, it has been hypothesised that the introduction of Earth-like gravity in a space station may prove useful for preventing muscle deconditioning. Currently three methods have been proposed for exercising with Earth-like gravity: (a) conditioning with centrifuges, (b) cycling along the inner wall of the space module, and (c) cycling in a ‘track tunnel’ on the Lunar or Martian surface.
Countermeasures based on centrifuges
In humans, the application of artificial gravity combined with intensive aerobic training has been found to maintain muscle size during 20 days of bed rest (Akima et al. 2005). In this study five healthy men were assigned to a countermeasure group and five to a non-exercising control group. The countermeasure group undertook intensive cycle training (to 90% of maximum HR) with short-arm centrifuge-induced artificial gravity on alternate days. The results showed that the volume of the total thigh muscles was maintained in the countermeasure group, whereas it decreased by 9% in the non-exercising controls. While knee extensors maximum voluntary contraction (MVC) decreased by 7% in the countermeasure group, in the non-exercising controls MVC decreased by as much as 23%. In a more comprehensive and recent study by Caiozzo et al. (2009), daily 1 h exposure to artificial gravity by centrifugation (2.5 G measured at the feet), resulted in a full/partial prevention of torque loss of the plantarflexor and knee extensor muscles, and in a full prevention of VL and soleus muscle fibre atrophy during 21-day bed rest (Caiozzo et al. 2009).
The results of this pilot study seem promising as they suggest that the minimum intensity and duration of the gravitational acceleration necessary to prevent muscle atrophy and torque loss in space may require a daily hourly exposure to about 2.5 G. Whether this type of countermeasure is effective in preventing the loss of muscle mass and function in mission lasting several months, or years, remains to be established.
Cycling in space, on the Moon and Mars
The concept of cycling in space as a countermeasure against cardiovascular and muscular deconditioning has been originally introduced by Antonutto et al. (1991) who proposed to simulate gravity on a space vehicle denominated Twin Bikes System (TBS). Recently, this concept has been further elaborated by di Prampero et al. (2009) who proposed the construction of a Lunar or Martian cycling track tunnel. A description of this project and of the TBS concept can be found in the paper of Lazzer et al. (2010) in the present issue of this Journal.
The prosaic argument could be made that these innovative concepts for space countermeasures are pure fiction. However, such argument would be scientifically and philosophically flawed since any new research idea is actually fiction until realised. The main achievements in terrestrial and space exploration have indeed been achieved through the bold spirit and vision of explorers, scientists and funders. Most people thought that Kubrick’s Film 2001 Space Oddessy was pure fiction when released in 1968. However, in 2004 NASA announced plans for building a fully functional lunar station with rotating crews and extend human presence across the Solar system. If we are to succeed as scientists in these goals, bold projects of innovative vision, such as the Lunar and Martian “track-tunnel” of Pietro Enrico di Prampero and his team, are exactly what are needed.
Notes
Maximum unloaded shortening velocity (V 0) is estimated in isolated preparations using the so-called ‘slack’ test (Edman (1979). A contracting muscle is rapidly shortened by a known distance (at a velocity greater than V max) so that the fibres become slack and will then shorten without any external load. The time for the fibre to take up the slack and start pulling on the force transducer gives a measure of the velocity of unloaded shortening. Maximum shortening velocity (V max) is estimated from the force–velocity relationship, which in turn is determined from a series of consecutive isotonic steps. V max is defined as the intercept of the force–velocity curve with the velocity axis.
References
Adams GR, Hather BM, Dudley GA (1994) Effect of short-term unweighting on human skeletal muscle strength and size. Aviat Space Environ Med 65:1116–1121
Akima H, Kawakami Y, Kubo K, Sekiguchi C, Ohshima H, Miyamoto A, Fukunaga T (2000) Effect of short-duration spaceflight on thigh and leg muscle volume. Med Sci Sports Exerc 32:1743–1747
Akima H, Kubo K, Imai M, Kanehisa H, Suzuki Y, Gunji A, Fukunaga T (2001) Inactivity and muscle: effect of resistance training during bed rest on muscle size in the lower limb. Acta Physiol Scand 172:269–278
Akima H, Katayama K, Sato K, Ishida K, Masuda K, Takada H, Watanabe Y, Iwase S (2005) Intensive cycle training with artificial gravity maintains muscle size during bed rest. Aviat Space Environ Med 76:923–929
Alkner BA, Tesch PA (2004a) Efficacy of a gravity-independent resistance exercise device as a countermeasure to muscle atrophy during 29-day bed rest. Acta Physiol Scand 181:345–357
Alkner BA, Tesch PA (2004b) Knee extensor and plantar flexor muscle size and function following 90 days of bed rest with or without resistance exercise. Eur J Appl Physiol 93:294–305
Andersen JL, Mohr T, Biering-Sorensen F, Galbo H, Kjaer M (1996) Myosin heavy chain isoform transformation in single fibres from m. vastus lateralis in spinal cord injured individuals: effects of long-term functional electrical stimulation (FES). Pflugers Arch 431:513–518
Antonutto G, Capelli C, Di Prampero PE (1991) Pedalling in space as a countermeasure to microgravity deconditioning. Microgravity Q 1:93–101
Antonutto G, Capelli C, Girardis M, Zamparo P, di Prampero PE (1999) Effects of microgravity on maximal power of lower limbs during very short efforts in humans. J Appl Physiol 86:85–92
Apseloff G, Girten B, Walker M, Shepard DR, Krecic ME, Stern LS, Gerber N (1993) Aminohydroxybutane bisphosphonate and clenbuterol prevent bone changes and retard muscle atrophy respectively in tail-suspended rats. J Pharmacol Exp Ther 264:1071–1078
Baldwin KM, Haddad F (2001) Effects of different activity and inactivity paradigms on myosin heavy chain gene expression in striated muscle. J Appl Physiol 90:345–357
Bamman MM, Clarke MS, Feeback DL, Talmadge RJ, Stevens BR, Lieberman SA, Greenisen MC (1998) Impact of resistance exercise during bed rest on skeletal muscle sarcopenia and myosin isoform distribution. J Appl Physiol 84:157–163
Belavy DL, Miokovic T, Armbrecht G, Richardson CA, Rittweger J, Felsenberg D (2009) Differential atrophy of the lower-limb musculature during prolonged bed-rest. Eur J Appl Physiol 107:489–499
Berg HE, Tesch PA (1996) Changes in muscle function in response to 10 days of lower limb unloading in humans. Acta Physiol Scand 157:63–70
Berg HE, Tesch PA (1998) Force and power characteristics of a resistive exercise device for use in space. Acta Astronaut 42:219–230
Berg HE, Dudley GA, Haggmark T, Ohlsen H, Tesch PA (1991) Effects of lower limb unloading on skeletal muscle mass and function in humans. J Appl Physiol 70:1882–1885
Berg HE, Dudley GA, Hather B, Tesch PA (1993) Work capacity and metabolic and morphologic characteristics of the human quadriceps muscle in response to unloading. Clin Physiol 13:337–347
Berg HE, Larsson L, Tesch PA (1997) Lower limb skeletal muscle function after 6 wk of bed rest. J Appl Physiol 82:182–188
Berg HE, Eiken O, Miklavcic L, Mekjavic IB (2007) Hip, thigh and calf muscle atrophy and bone loss after 5-week bedrest inactivity. Eur J Appl Physiol 99:283–289
Boudreau NJ, Jones PL (1999) Extracellular matrix and integrin signalling: the shape of things to come. Biochem J 339(Pt 3):481–488
Brooks N, Cloutier GJ, Cadena SM, Layne JE, Nelsen CA, Freed AM, Roubenoff R, Castaneda-Sceppa C (2008) Resistance training and timed essential amino acids protect against the loss of muscle mass and strength during 28 days of bed rest and energy deficit. J Appl Physiol 105:241–248
Caiozzo VJ, Baker MJ, Herrick RE, Tao M, Baldwin KM (1994) Effect of spaceflight on skeletal muscle: mechanical properties and myosin isoform content of a slow muscle. J Appl Physiol 76:1764–1773
Caiozzo VJ, Haddad F, Baker MJ, Herrick RE, Prietto N, Baldwin KM (1996) Microgravity-induced transformations of myosin isoforms and contractile properties of skeletal muscle. J Appl Physiol 81:123–132
Caiozzo VJ, Haddad F, Lee S, Baker M, Paloski W, Baldwin KM (2009) Artificial gravity as a countermeasure to microgravity: a pilot study examining the effects on knee extensor and plantar flexor muscle groups. J Appl Physiol 107:39–46
Caruso J, Hamill J, Yamauchi M, Mercado D, Cook T, Higginson B, O’Meara S, Elias J, Siconolfi S (2005) Albuterol aids resistance exercise in reducing unloading-induced ankle extensor strength losses. J Appl Physiol 98:1705–1711
Chase GA, Grave C, Rowell LB (1966) Independence of changes in functional and performance capacities attending prolonged bed rest. Aerosp Med 37:1232–1238
Cherepakhin MA, Pervushin VI (1970) Space flight effect on the neuromuscular system of cosmonauts. Kosm Biol Aviakosm Med 4:46–49
Clark BC, Fernhall B, Ploutz-Snyder LL (2006) Adaptations in human neuromuscular function following prolonged unweighting: I. Skeletal muscle contractile properties and applied ischemia efficacy. J Appl Physiol 101:256–263
Clark BC, Pierce JR, Manini TM, Ploutz-Snyder LL (2007) Effect of prolonged unweighting of human skeletal muscle on neuromotor force control. Eur J Appl Physiol 100:53–62
Convertino VA (1990) Physiological adaptations to weightlessness: effects on exercise and work performance. Exerc Sport Sci Rev 18:119–166
Convertino VA, Doerr DF, Stein SL (1989) Changes in size and compliance of the calf after 30 days of simulated microgravity. J Appl Physiol 66:1509–1512
Criswell DS, Booth FW, DeMayo F, Schwartz RJ, Gordon SE, Fiorotto ML (1998) Overexpression of IGF-I in skeletal muscle of transgenic mice does not prevent unloading-induced atrophy. Am J Physiol 275:E373–E379
D’Antona G, Pellegrino MA, Adami R, Rossi R, Carlizzi CN, Canepari M, Saltin B, Bottinelli R (2003) The effect of ageing and immobilization on structure and function of human skeletal muscle fibres. J Physiol 552:499–511
Danielsen CC, Andreassen TT (1988) Mechanical properties of rat tail tendon in relation to proximal-distal sampling position and age. J Biomech 21:207–212
Davies CT, Rutherford IC, Thomas DO (1987) Electrically evoked contractions of the triceps surae during and following 21 days of voluntary leg immobilization. Eur J Appl Physiol Occup Physiol 56:306–312
de Boer MD, Maganaris CN, Seynnes OR, Rennie MJ, Narici MV (2007a) Time course of muscular, neural and tendinous adaptations to 23 day unilateral lower-limb suspension in young men. J Physiol 583:1079–1091
de Boer MD, Selby A, Atherton P, Smith K, Seynnes OR, Maganaris CN, Maffulli N, Movin T, Narici MV, Rennie MJ (2007b) The temporal responses of protein synthesis, gene expression and cell signalling in human quadriceps muscle and patellar tendon to disuse. J Physiol 585:241–251
de Boer MD, Seynnes OR, di Prampero PE, Pisot R, Mekjavic IB, Biolo G, Narici MV (2008) Effect of 5 weeks horizontal bed rest on human muscle thickness and architecture of weight bearing and non-weight bearing muscles. Eur J Appl Physiol 104:401–407
Deschenes MR, Giles JA, McCoy RW, Volek JS, Gomez AL, Kraemer WJ (2002) Neural factors account for strength decrements observed after short-term muscle unloading. Am J Physiol 282:R578–R583
Desplanches D, Hoppeler H, Mayet MH, Denis C, Claassen H, Ferretti G (1998) Effects of bedrest on deltoideus muscle morphology and enzymes. Acta Physiol Scand 162:135–140
di Prampero PE (2000) Cycling on Earth, in space, on the Moon. Eur J Appl Physiol 82:345–360
di Prampero PE, Antonutto G (1997) Cycling in space to simulate gravity. Int J Sports Med 18(Suppl 4):S324–S326
di Prampero PE, Narici MV (2003) Muscles in microgravity: from fibres to human motion. J Biomech 36:403–412
di Prampero P, Lazzer S, Antonutto G (2009) Human power centrifuges on the Moon and Mars. Microgravity Sci Technol 21:209–215
Duchateau J (1995) Bed rest induces neural and contractile adaptations in triceps surae. Med Sci Sports Exerc 27:1581–1589
Duchateau J, Hainaut K (1987) Electrical and mechanical changes in immobilized human muscle. J Appl Physiol 62:2168–2173
Dudley GA, Duvoisin MR, Convertino VA, Buchanan P (1989) Alterations of the in vivo torque-velocity relationship of human skeletal muscle following 30 days exposure to simulated microgravity. Aviat Space Environ Med 60:659–663
Dudley GA, Duvoisin MR, Adams GR, Meyer RA, Belew AH, Buchanan P (1992) Adaptations to unilateral lower limb suspension in humans. Aviat Space Environ Med 63:678–683
Duvoisin MR, Convertino VA, Buchanan P, Gollnick PD, Dudley GA (1989) Characteristics and preliminary observations of the influence of electromyostimulation on the size and function of human skeletal muscle during 30 days of simulated microgravity. Aviat Space Environ Med 60:671–678
Edgerton VR, Roy RR (1994) Neuromuscular adaptation to actual and simulated weightlessness. Adv Space Biol Med 4:33–67
Edgerton VR, Zhou MY, Ohira Y, Klitgaard H, Jiang B, Bell G, Harris B, Saltin B, Gollnick PD, Roy RR et al (1995) Human fiber size and enzymatic properties after 5 and 11 days of spaceflight. J Appl Physiol 78:1733–1739
Edgerton VR, McCall GE, Hodgson JA, Gotto J, Goulet C, Fleischmann K, Roy RR (2001) Sensorimotor adaptations to microgravity in humans. J Exp Biol 204:3217–3224
Edman KA (1979) The velocity of unloaded shortening and its relation to sarcomere length and isometric force in vertebrate muscle fibres. J Physiol 291:143–159
Ferrando AA, Stuart CA, Brunder DG, Hillman GR (1995) Magnetic resonance imaging quantitation of changes in muscle volume during 7 days of strict bed rest. Aviat Space Environ Med 66:976–981
Ferrando AA, Lane HW, Stuart CA, Davis-Street J, Wolfe RR (1996) Prolonged bed rest decreases skeletal muscle and whole body protein synthesis. Am J Physiol 270:E627–E633
Ferrando AA, Tipton KD, Bamman MM, Wolfe RR (1997) Resistance exercise maintains skeletal muscle protein synthesis during bed rest. J Appl Physiol 82:807–810
Ferrando AA, Tipton KD, Doyle D, Phillips SM, Cortiella J, Wolfe RR (1998) Testosterone injection stimulates net protein synthesis but not tissue amino acid transport. Am J Physiol 275:E864–E871
Ferrando AA, Paddon-Jones D, Wolfe RR (2002) Alterations in protein metabolism during space flight and inactivity. Nutrition 18:837–841
Ferretti G, Berg HE, Minetti AE, Moia C, Rampichini S, Narici MV (2001) Maximal instantaneous muscular power after prolonged bed rest in humans. J Appl Physiol 90:431–435
Ferrucci L, Guralnik JM (2003) Inflammation, hormones, and body composition at a crossroad. Am J Med 115:501–502
Fitts RH, Riley DR, Widrick JJ (2000) Physiology of a microgravity environment invited review: microgravity and skeletal muscle. J Appl Physiol 89:823–839
Fitts RH, Riley DR, Widrick JJ (2001) Functional and structural adaptations of skeletal muscle to microgravity. J Exp Biol 204:3201–3208
Fitts RH, Romatowski JG, Peters JR, Paddon-Jones D, Wolfe RR, Ferrando AA (2007) The deleterious effects of bed rest on human skeletal muscle fibers are exacerbated by hypercortisolemia and ameliorated by dietary supplementation. Am J Physiol Cell Physiol 293:C313–C320
Fluck M, Carson JA, Gordon SE, Ziemiecki A, Booth FW (1999) Focal adhesion proteins FAK and paxillin increase in hypertrophied skeletal muscle. Am J Physiol 277:C152–C162
Fluckey JD, Dupont-Versteegden EE, Montague DC, Knox M, Tesch P, Peterson CA, Gaddy-Kurten D (2002) A rat resistance exercise regimen attenuates losses of musculoskeletal mass during hindlimb suspension. Acta Physiol Scand 176:293–300
Gamrin L, Berg HE, Essen P, Tesch PA, Hultman E, Garlick PJ, McNurlan MA, Wernerman J (1998) The effect of unloading on protein synthesis in human skeletal muscle. Acta Physiol Scand 163:369–377
Gans C, Bock WJ (1965) The functional significance of muscle architecture—a theoretical analysis. Ergeb Anat Entwicklungsgesch 38:115–142
Gardetto PR, Schluter JM, Fitts RH (1989) Contractile function of single muscle fibers after hindlimb suspension. J Appl Physiol 66:2739–2749
Gibson JN, Halliday D, Morrison WL, Stoward PJ, Hornsby GA, Watt PW, Murdoch G, Rennie MJ (1987) Decrease in human quadriceps muscle protein turnover consequent upon leg immobilization. Clin Sci (Lond) 72:503–509
Gogia P, Schneider VS, LeBlanc AD, Krebs J, Kasson C, Pientok C (1988) Bed rest effect on extremity muscle torque in healthy men. Arch Phys Med Rehabil 69:1030–1032
Gopalakrishnan R, Genc KO, Rice AJ, Lee SM, Evans HJ, Maender CC, Ilaslan H, Cavanagh PR (2010) Muscle volume, strength, endurance, and exercise loads during 6-month missions in space. Aviat Space Environ Med 81:91–102
Goubel F (1997) Changes in mechanical properties of human muscle as a result of spaceflight. Int J Sports Med 18(Suppl 4):S285–S287
Greenisen MC, Edgerton VR (1994) Human capabilities in the space environment. In: Nicogossian AE, Huntoon CL, Pool SL (eds) Space physiology and medicine. Lea & Febiger, Philadelphia, PA, pp 194–210
Greenleaf JE, Bernauer EM, Ertl AC, Trowbridge TS, Wade CE (1989a) Work capacity during 30 days of bed rest with isotonic and isokinetic exercise training. J Appl Physiol 67:1820–1826
Greenleaf JE, Bulbulian R, Bernauer EM, Haskell WL, Moore T (1989b) Exercise-training protocols for astronauts in microgravity. J Appl Physiol 67:2191–2204
Grogor’eva LS, Kozlovskaia IB (1987) Effect of weightlessness and hypokinesia on the velocity-strength properties of human muscles. Kosm Biol Aviakosm Med 21:27–30
Hather BM, Adams GR, Tesch PA, Dudley GA (1992) Skeletal muscle responses to lower limb suspension in humans. J Appl Physiol 72:1493–1498
Haus JM, Carrithers JA, Carroll CC, Tesch PA, Trappe TA (2007) Contractile and connective tissue protein content of human skeletal muscle: effects of 35 and 90 days of simulated microgravity and exercise countermeasures. Am J Physiol 293:R1722–R1727
Hespel P, Op’t Eijnde B, Van Leemputte M, Urso B, Greenhaff PL, Labarque V, Dymarkowski S, Van Hecke P, Richter EA (2001) Oral creatine supplementation facilitates the rehabilitation of disuse atrophy and alters the expression of muscle myogenic factors in humans. J Physiol 536:625–633
Hikida RS, Gollnick PD, Dudley GA, Convertino VA, Buchanan P (1989) Structural and metabolic characteristics of human skeletal muscle following 30 days of simulated microgravity. Aviat Space Environ Med 60:664–670
Hortobagyi T, Dempsey L, Fraser D, Zheng D, Hamilton G, Lambert J, Dohm L (2000) Changes in muscle strength, muscle fibre size and myofibrillar gene expression after immobilization and retraining in humans. J Physiol 524(Pt 1):293–304
Jaweed MM (1994) Muscle structure and function. In: Nicogossian AE, Huntoon CL, Pool SL (eds) Space physiology and medicine. Lea & Febiger, Philadelphia, pp 317–326
Kakurin LI, Cherepakhin MA, Pervushin VI (1971) Effect of brief space flights on the human neuromuscular system. Kosm Biol Aviakosm Med 5:53–56
Kawakami Y, Muraoka Y, Kubo K, Suzuki Y, Fukunaga T (2000) Changes in muscle size and architecture following 20 days of bed rest. J Gravit Physiol 7:53–59
Kawakami Y, Akima H, Kubo K, Muraoka Y, Hasegawa H, Kouzaki M, Imai M, Suzuki Y, Gunji A, Kanehisa H, Fukunaga T (2001) Changes in muscle size, architecture, and neural activation after 20 days of bed rest with and without resistance exercise. Eur J Appl Physiol 84:7–12
Klossner S, Durieux AC, Freyssenet D, Flueck M (2009) Mechano-transduction to muscle protein synthesis is modulated by FAK. Eur J Appl Physiol 106:389–398
Koryak Y (1995) Contractile properties of the human triceps surae muscle during simulated weightlessness. Eur J Appl Physiol Occup Physiol 70:344–350
Koryak Y (1998) Effect of 120 days of bed-rest with and without countermeasures on the mechanical properties of the triceps surae muscle in young women. Eur J Appl Physiol Occup Physiol 78:128–135
Koryak Y (1999) The effects of long-term simulated microgravity on neuromuscular performance in men and women. Eur J Appl Physiol Occup Physiol 79:168–175
Koryak YU (2001) Electrically evoked and voluntary properties of the human triceps surae muscle: effects of long-term spaceflights. Acta Physiol Pharmacol Bulg 26:21–27
Kozlovskaya IB, Grigoriev AI (2004) Russian system of countermeasures on board of the International Space Station (ISS): the first results. Acta Astronaut 55:233–237
Kozlovskaya IB, Kreidich Yu V, Oganov VS, Koserenko OP (1981) Pathophysiology of motor functions in prolonged manned space flights. Acta Astronaut 8:1059–1072
Kubo K, Akima H, Kouzaki M, Ito M, Kawakami Y, Kanehisa H, Fukunaga T (2000) Changes in the elastic properties of tendon structures following 20 days bed-rest in humans. Eur J Appl Physiol 83:463–468
Kubo K, Akima H, Ushiyama J, Tabata I, Fukuoka H, Kanehisa H, Fukunaga T (2004) Effects of 20 days of bed rest on the viscoelastic properties of tendon structures in lower limb muscles. Br J Sports Med 38:324–330
Lalani R, Bhasin S, Byhower F, Tarnuzzer R, Grant M, Shen R, Asa S, Ezzat S, Gonzalez-Cadavid NF (2000) Myostatin and insulin-like growth factor-I and -II expression in the muscle of rats exposed to the microgravity environment of the NeuroLab space shuttle flight. J Endocrinol 167:417–428
Lambertz D, Perot C, Kaspranski R, Goubel F (2001) Effects of long-term spaceflight on mechanical properties of muscles in humans. J Appl Physiol 90:179–188
Larsson L, Li X, Berg HE, Frontera WR (1996) Effects of removal of weight-bearing function on contractility and myosin isoform composition in single human skeletal muscle cells. Pflugers Arch 432:320–328
Lazzer S, Plaino L, Antonutto G (2010) The energetics of cycling on Earth, Moon and Mars. Eur J Appl Physiol (Epub)
LeBlanc A, Gogia P, Schneider V, Krebs J, Schonfeld E, Evans H (1988) Calf muscle area and strength changes after five weeks of horizontal bed rest. Am J Sports Med 16:624–629
LeBlanc AD, Schneider VS, Evans HJ, Pientok C, Rowe R, Spector E (1992) Regional changes in muscle mass following 17 weeks of bed rest. J Appl Physiol 73:2172–2178
LeBlanc A, Rowe R, Schneider V, Evans H, Hedrick T (1995) Regional muscle loss after short duration spaceflight. Aviat Space Environ Med 66:1151–1154
LeBlanc A, Rowe R, Evans H, West S, Shackelford L, Schneider V (1997) Muscle atrophy during long duration bed rest. Int J Sports Med 18(Suppl 4):S283–S285
LeBlanc A, Lin C, Shackelford L, Sinitsyn V, Evans H, Belichenko O, Schenkman B, Kozlovskaya I, Oganov V, Bakulin A, Hedrick T, Feeback D (2000) Muscle volume, MRI relaxation times (T2), and body composition after spaceflight. J Appl Physiol 89:2158–2164
Lynch GS, Hinkle RT, Faulkner JA (2001) Force and power output of diaphragm muscle strips from mdx and control mice after clenbuterol treatment. Neuromuscul Disord 11:192–196
Maltin CA, Delday MI, Watson JS, Heys SD, Nevison IM, Ritchie IK, Gibson PH (1993) Clenbuterol, a beta-adrenoceptor agonist, increases relative muscle strength in orthopaedic patients. Clin Sci (Lond) 84:651–654
McDonald KS, Fitts RH (1995) Effect of hindlimb unloading on rat soleus fiber force, stiffness, and calcium sensitivity. J Appl Physiol 79:1796–1802
Milesi S, Capelli C, Denoth J, Hutchinson T, Stussi E (2000) Effects of 17 days bedrest on the maximal voluntary isometric torque and neuromuscular activation of the plantar and dorsal flexors of the ankle. Eur J Appl Physiol 82:197–205
Montgomery LD (1993) Body volume changes during simulated microgravity. II: comparison of horizontal and head-down bed rest. Aviat Space Environ Med 64:899–904
Murton AJ, Constantin D, Greenhaff PL (2008) The involvement of the ubiquitin proteasome system in human skeletal muscle remodelling and atrophy. Biochim Biophys Acta 1782:730–743
Narici M, Capodaglio P (1998) Changes in muscle size and architecture in disuse atrophy. In: Capodaglio P, Narici M (eds) Muscle atrophy: disuse and disease. Le Collane della Fondazione Salvatore Maugeri, PI-ME Press, Pavia, Italy, pp 55–64
Narici M, Cerretelli P (1998) Changes in human muscle architecture in disuse-atrophy evaluated by ultrasound imaging. J Gravit Physiol 5:P73–P74
Narici M, Kayser B, Barattini P, Cerretelli P (2003) Effects of 17-day spaceflight on electrically evoked torque and cross-sectional area of the human triceps surae. Eur J Appl Physiol 90:275–282
Ohira Y, Jiang B, Roy RR, Oganov V, Ilyina-Kakueva E, Marini JF, Edgerton VR (1992) Rat soleus muscle fiber responses to 14 days of spaceflight and hindlimb suspension. J Appl Physiol 73:51S–57S
Ohira Y, Yoshinaga T, Ohara M, Nonaka I, Yoshioka T, Yamashita-Goto K, Shenkman BS, Kozlovskaya IB, Roy RR, Edgerton VR (1999) Myonuclear domain and myosin phenotype in human soleus after bed rest with or without loading. J Appl Physiol 87:1776–1785
Paddon-Jones D, Sheffield-Moore M, Cree MG, Hewlings SJ, Aarsland A, Wolfe RR, Ferrando AA (2006) Atrophy and impaired muscle protein synthesis during prolonged inactivity and stress. J Clin Endocrinol Metab 91:4836–4841
Pavy-Le Traon A, Heer M, Narici MV, Rittweger J, Vernikos J (2007) From space to Earth: advances in human physiology from 20 years of bed rest studies (1986–2006). Eur J Appl Physiol 101:143–194
Payette H, Roubenoff R, Jacques PF, Dinarello CA, Wilson PW, Abad LW, Harris T (2003) Insulin-like growth factor-1 and interleukin 6 predict sarcopenia in very old community-living men and women: the Framingham Heart Study. J Am Geriatr Soc 51:1237–1243
Ploutz-Snyder LL, Tesch PA, Crittenden DJ, Dudley GA (1995) Effect of unweighting on skeletal muscle use during exercise. J Appl Physiol 79:168–175
Ploutz-Snyder LL, Tesch PA, Hather BM, Dudley GA (1996) Vulnerability to dysfunction and muscle injury after unloading. Arch Phys Med Rehabil 77:773–777
Prockop DJ, Kivirikko KI (1995) Collagens: molecular biology, diseases, and potentials for therapy. Annu Rev Biochem 64:403–434
Rapcsak M, Oganov VS, Szoor A, Skuratova SA, Szilagyi T, Takacs O (1983) Effect of weightlessness on the function of rat skeletal muscles on the biosatellite “Cosmos-1129”. Acta Physiol Hung 62:225–228
Reeves ND, Maganaris CN, Ferretti G, Narici MV (2002) Influence of simulated microgravity on human skeletal muscle architecture and function. J Gravit Physiol 9:P153–P154
Reeves ND, Maganaris CN, Ferretti G, Narici MV (2005) Influence of 90-day simulated microgravity on human tendon mechanical properties and the effect of resistive countermeasures. J Appl Physiol 98:2278–2286
Rennie MJ, Selby A, Atherton P, Smith K, Kumar V, Glover EL, Philips SM (2010) Facts, noise and wishful thinking: muscle protein turnover in aging and human disuse atrophy. Scand J Med Sci Sports 20:5–9
Riley DA, Bain JL, Thompson JL, Fitts RH, Widrick JJ, Trappe SW, Trappe TA, Costill DL (2000) Decreased thin filament density and length in human atrophic soleus muscle fibers after spaceflight. J Appl Physiol 88:567–572
Rittweger J, Felsenberg D, Maganaris C, Ferretti JL (2007) Vertical jump performance after 90 days bed rest with and without flywheel resistive exercise, including a 180 days follow-up. Eur J Appl Physiol 100:427–436
Rommel C, Bodine SC, Clarke BA, Rossman R, Nunez L, Stitt TN, Yancopoulos GD, Glass DJ (2001) Mediation of IGF-1-induced skeletal myotube hypertrophy by PI(3)K/Akt/mTOR and PI(3)K/Akt/GSK3 pathways. Nat Cell Biol 3:1009–1013
Roy RR, Bello MA, Bouissou P, Edgerton VR (1987) Size and metabolic properties of fibers in rat fast-twitch muscles after hindlimb suspension. J Appl Physiol 62:2348–2357
Rozier CK, Elder JD, Brown M (1979) Prevention of atrophy by isometric exercise of a casted leg. J Sports Med Phys Fitness 19:191–194
Rudnick J, Puttmann B, Tesch PA, Alkner B, Schoser BG, Salanova M, Kirsch K, Gunga HC, Schiffl G, Luck G, Blottner D (2004) Differential expression of nitric oxide synthases (NOS 1-3) in human skeletal muscle following exercise countermeasure during 12 weeks of bed rest. FASEB J 18:1228–1230
Rummel JA, Sawin CF, Michel EL, Buderer MC, Thornton WT (1975) Exercise and long duration spaceflight through 84 days. J Am Med Womens Assoc 30:173–187
Ryan AS, Dobrovolny CL, Smith GV, Silver KH, Macko RF (2002) Hemiparetic muscle atrophy and increased intramuscular fat in stroke patients. Arch Phys Med Rehabil 83:1703–1707
Sargeant AJ, Davies CT, Edwards RH, Maunder C, Young A (1977) Functional and structural changes after disuse of human muscle. Clin Sci Mol Med 52:337–342
Schulze K, Gallagher P, Trappe S (2002) Resistance training preserves skeletal muscle function during unloading in humans. Med Sci Sports Exerc 34:303–313
Seynnes OR, Maffiuletti NA, Maganaris CN, de Boer MD, Pensini M, di Prampero PE, Narici MV (2008) Soleus T reflex modulation in response to spinal and tendinous adaptations to unilateral lower limb suspension in humans. Acta Physiol (Oxf) 194:239–251
Shackelford LC, LeBlanc AD, Driscoll TB, Evans HJ, Rianon NJ, Smith SM, Spector E, Feeback DL, Lai D (2004) Resistance exercise as a countermeasure to disuse-induced bone loss. J Appl Physiol 97:119–129
Shyy JY, Chien S (1997) Role of integrins in cellular responses to mechanical stress and adhesion. Curr Opin Cell Biol 9:707–713
Stevens L, Mounier Y, Holy X (1993) Functional adaptation of different rat skeletal muscles to weightlessness. Am J Physiol 264:R770–R776
Stewart CE (2004) The physiology of stem cells: potential for the elderly patient. J Musculoskelet Neuronal Interact 4:179–183
Suzuki Y, Kashihara H, Takenaka K, Kawakubo K, Makita Y, Goto S, Ikawa S, Gunji A (1994) Effects of daily mild supine exercise on physical performance after 20 days bed rest in young persons. Acta Astronaut 33:101–111
Talmadge RJ (2000) Myosin heavy chain isoform expression following reduced neuromuscular activity: potential regulatory mechanisms. Muscle Nerve 23:661–679
Templeton GH, Padalino M, Manton J, Glasberg M, Silver CJ, Silver P, DeMartino G, Leconey T, Klug G, Hagler H et al (1984) Influence of suspension hypokinesia on rat soleus muscle. J Appl Physiol 56:278–286
Tesch PA, Berg HE (1997) Resistance training in space. Int J Sports Med 18(Suppl 4):S322–S324
Tesch PA, Trieschmann JT, Ekberg A (2004) Hypertrophy of chronically unloaded muscle subjected to resistance exercise. J Appl Physiol 96:1451–1458
Tesch PA, Berg HE, Bring D, Evans HJ, LeBlanc AD (2005) Effects of 17-day spaceflight on knee extensor muscle function and size. Eur J Appl Physiol 93:463–468
Thom JM, Thompson MW, Ruell PA, Bryant GJ, Fonda JS, Harmer AR, De Jonge XA, Hunter SK (2001) Effect of 10-day cast immobilization on sarcoplasmic reticulum calcium regulation in humans. Acta Physiol Scand 172:141–147
Thomason DB, Booth FW (1990) Atrophy of the soleus muscle by hindlimb unweighting. J Appl Physiol 68:1–12
Thorton WE, Rummel JA (1977) Muscular deconditioning and its prevention in space flight. In: Johnson RS, Deitlein LF (eds) Biomedical results from skylab. NASA, Washington, DC, pp 191–197
Trappe SW, Trappe TA, Lee GA, Widrick JJ, Costill DL, Fitts RH (2001) Comparison of a space shuttle flight (STS-78) and bed rest on human muscle function. J Appl Physiol 91:57–64
Trappe S, Trappe T, Gallagher P, Harber M, Alkner B, Tesch P (2004) Human single muscle fibre function with 84 day bed-rest and resistance exercise. J Physiol 557:501–513
Trappe TA, Burd NA, Louis ES, Lee GA, Trappe SW (2007) Influence of concurrent exercise or nutrition countermeasures on thigh and calf muscle size and function during 60 days of bed rest in women. Acta Physiol (Oxf) 191:147–159
Urban RJ, Bodenburg YH, Gilkison C, Foxworth J, Coggan AR, Wolfe RR, Ferrando A (1995) Testosterone administration to elderly men increases skeletal muscle strength and protein synthesis. Am J Physiol 269:E820–E826
Veldhuizen JW, Verstappen FT, Vroemen JP, Kuipers H, Greep JM (1993) Functional and morphological adaptations following four weeks of knee immobilization. Int J Sports Med 14:283–287
Wade CE, Stanford KI, Stein TP, Greenleaf JE (2005) Intensive exercise training suppresses testosterone during bed rest. J Appl Physiol 99:59–63
Wang H, Casaburi R, Taylor WE, Aboellail H, Storer TW, Kopple JD (2005) Skeletal muscle mRNA for IGF-IEa, IGF-II, and IGF-I receptor is decreased in sedentary chronic hemodialysis patients. Kidney Int 68:352–361
White MJ, Davies CT, Brooksby P (1984) The effects of short-term voluntary immobilization on the contractile properties of the human triceps surae. Q J Exp Physiol 69:685–691
Widrick JJ, Romatowski JG, Bain JL, Trappe SW, Trappe TA, Thompson JL, Costill DL, Riley DA, Fitts RH (1997) Effect of 17 days of bed rest on peak isometric force and unloaded shortening velocity of human soleus fibers. Am J Physiol 273:C1690–C1699
Widrick JJ, Knuth ST, Norenberg KM, Romatowski JG, Bain JL, Riley DA, Karhanek M, Trappe SW, Trappe TA, Costill DL, Fitts RH (1999) Effect of a 17 day spaceflight on contractile properties of human soleus muscle fibres. J Physiol 516(Pt 3):915–930
Widrick JJ, Trappe SW, Romatowski JG, Riley DA, Costill DL, Fitts RH (2002) Unilateral lower limb suspension does not mimic bed rest or spaceflight effects on human muscle fiber function. J Appl Physiol 93:354–360
Wilkie DR (1949) The relation between force and velocity in human muscle. J Physiol 110:249–280
Wineski LE, von Deutsch DA, Abukhalaf IK, Pitts SA, Potter DE, Paulsen DF (2002) Muscle-specific effects of hindlimb suspension and clenbuterol in mature male rats. Cells Tissues Organs 171:188–198
Yamashita-Goto K, Okuyama R, Honda M, Kawasaki K, Fujita K, Yamada T, Nonaka I, Ohira Y, Yoshioka T (2001) Maximal and submaximal forces of slow fibers in human soleus after bed rest. J Appl Physiol 91:417–424
Yoshida N, Sairyo K, Sasa T, Fukunaga M, Koga K, Ikata T, Yasui N (2003) Electrical stimulation prevents deterioration of the oxidative capacity of disuse-atrophied muscles in rats. Aviat Space Environ Med 74:207–211
Zachwieja JJ, Smith SR, Lovejoy JC, Rood JC, Windhauser MM, Bray GA (1999) Testosterone administration preserves protein balance but not muscle strength during 28 days of bed rest. J Clin Endocrinol Metab 84:207–212
Zange J, Muller K, Schuber M, Wackerhage H, Hoffmann U, Gunther RW, Adam G, Neuerburg JM, Sinitsyn VE, Bacharev AO, Belichenko OI (1997) Changes in calf muscle performance, energy metabolism, and muscle volume caused by long-term stay on space station MIR. Int J Sports Med 18(Suppl 4):S308–S309
Zimmers TA, Davies MV, Koniaris LG, Haynes P, Esquela AF, Tomkinson KN, McPherron AC, Wolfman NM, Lee SJ (2002) Induction of cachexia in mice by systemically administered myostatin. Science 296:1486–1488
Acknowledgments
A significant portion of this Review paper makes direct reference to published literature of Prof. PE di Prampero and co-workers and indirectly, to work performed through the close and most enjoyable collaboration we shared over the last 30 years. We are also grateful to the funding agencies that partly supported this work (ESA, ASI and the Slovenian Ministry of Defence) and to Prof. Rado Pisot (University of Primorska, Science and Research Centre Koper, Institute for Kinesiology Research, Slovenia) for creating the opportunity for performing some of the ground-based studies cited in this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Susan Ward.
This article is published as part of the Special Issue dedicated to Pietro di Prampero, formerly Editor-in-Chief of EJAP.
Rights and permissions
About this article
Cite this article
Narici, M.V., de Boer, M.D. Disuse of the musculo-skeletal system in space and on earth. Eur J Appl Physiol 111, 403–420 (2011). https://doi.org/10.1007/s00421-010-1556-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-010-1556-x