Abstract
This study investigated the relationship between \(\dot{V}\hbox{O}_{2\max}\) and repeated-sprint ability (RSA), while controlling for the effects of initial sprint performance on sprint decrement. This was achieved via two methods: (1) matching females of low and moderate aerobic fitness (\(\dot{V}\hbox{O}_{2\max}\): 36.4 ± 4.7 vs 49.6 ± 5.5 ml kg−1 min−1 ; p < 0.05) for initial sprint performance and then comparing RSA, and (2) semi-partial correlations to adjust for the influence of initial sprint performance on RSA. Tests consisted of a RSA cycle test (5×6-s max sprints every 30 s) and a \(\dot{V}\hbox{O}_{2\max}\) test. Muscle biopsies were taken before and after the RSA test. There was no significant difference between groups for work (W 1, 3.44 ± 0.57 vs 3.58 ± 0.49 kJ; p = 0.59) or power (P 1, 788.1 ± 99.2 vs 835.2 ± 127.2 W; p = 0.66) on the first sprint, or for total work (W tot, 15.2 ± 2.2 vs 16.6 ± 2.2 kJ; p = 0.25). However, the moderate \(\dot{V}\hbox{O}_{2\max}\) group recorded a smaller work decrement across the five sprints (W dec, 11.1 ± 2.5 vs 7.6 ± 3.4%; p = 0.045). There were no significant differences between the two groups for muscle buffer capacity, muscle lactate or pH at any time point. When a semi-partial correlation was performed, to control for the contribution of W 1 to W dec, the correlation between \(\dot{V}\hbox{O}_{2\max}\) and W dec increased from r = −0.41 (p > 0.05) to r = −0.50 (p < 0.05). These results indicate that \(\dot{V}\hbox{O}_{2\max}\) does contribute to performance during repeated-sprint efforts. However, the small variance in W dec explained by \(\dot{V}\hbox{O}_{2\max}\) suggests that other factors also play a role.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Team-sports such as football, hockey and basketball require participants to perform a number of short sprints, interspersed with periods of rest or low-to-moderate intensity activity. Many of these sprints are separated by rest periods long enough (> 1 min) to allow complete or near complete recovery and therefore subsequent sprint performance is not significantly impaired. However, recent game analysis has shown that some of these sprints are separated by short rest periods (< 30 s; Spencer et al. 2004), which have been shown to negatively affect subsequent sprint performance (Balsom et al. 1992). Therefore, one of the fitness requirements of team-sport athletes is the ability to perform short-duration sprints (< 10 s) with a short recovery time (< 30 s), and this has been termed repeated-sprint ability (RSA). However, despite the importance of RSA for many sports, more research is required to investigate the physiological characteristics that determine RSA.
It is generally accepted that during a single, short-duration (< 10 s) sprint, the contribution of aerobic metabolism to ATP resynthesis during the sprint is negligible. However, when this type of exercise is repeated with short recovery intervals, the relative contribution from aerobic sources increases and may be important to maintain power output (Christensen et al. 1960). This is supported by research showing that a decreased oxygen availability (as a result of hypoxia) results in a greater accumulation of blood lactate and impaired RSA (Balsom et al. 1994b). In contrast, greater oxygen availability (induced via EPO administration) increases the relative contribution of aerobic metabolism to ATP resynthesis during repeated-sprint exercise (Balsom et al. 1994a). Furthermore, the correlation between \(\dot{V}\hbox{O}_{2\max}\) and the aerobic contribution to repeated-sprint exercise (r = 0.78–0.83; Hamilton et al. 1991; Tomlin and Wenger 2002) suggests that the aerobic contribution to repeated-sprint exercise is related to \(\dot{V}\hbox{O}_{2\max}.\) Consequently, many athletes, coaches and sports scientists believe that a high level of aerobic fitness (as assessed by \(\dot{V}\hbox{O}_{2\max}\)) is important for RSA.
The research, however, is less than convincing. Low to non-significant correlation coefficients (0.42 < r < 0.56) are typically reported between \(\dot{V}\hbox{O}_{2\max}\) and repeated-sprint performance (McMahon and Wenger 1998; Tomlin and Wenger 2002; Bishop et al. 2003, 2004b). Furthermore, while endurance-trained athletes are better able to maintain repeated-sprint performance than team-sport athletes (Hamilton et al. 1991; Bishop and Spencer 2004; Edge et al. 2006), endurance-trained athletes also produce lower initial sprint performances. This is important, as it has been reported that initial sprint performance is strongly correlated with the sprint decrement that occurs during a repeated-sprint test (Hamilton et al. 1991; Wadley and Le Rossignol 1998; Bishop et al. 2003) and is inversely related to \(\dot{V}\hbox{O}_{2\max}\) (Bogdanis et al. 1996). Thus, the greater RSA reported in endurance-trained subjects may actually be associated with their lower initial sprint performance, rather than their greater \(\dot{V}\hbox{O}_{2\max}.\)
The purpose of this study was to investigate the relationship between \(\dot{V}\hbox{O}_{2\max}\) and RSA, while attempting to control for the effects of initial sprint performance on sprint decrement. This was achieved via two methods: (1) matching subjects of different aerobic fitness \(\dot{V}\hbox{O}_{2\max})\) for initial sprint performance and then comparing RSA, and (2) the use of semi-partial correlations to adjust for the influence of initial sprint performance on RSA. Muscle buffer capacity was also examined as this has previously been reported to be related to RSA (Bishop et al. 2004b).
Methods
Subjects
Sixteen female, recreational, team-sport athletes (netball, basketball and field hockey) were matched on single, 6-s sprint performance (J). Subjects competed in local competitions and were tested at the start of their respective seasons. Weekly training typically consisted of two to three endurance-training sessions (30–45 min at 70–95% HRmax) and two skills-based sessions in which HR was maintained between 80 and 95% HRmax for 40–50 min.
The mean ± SD difference between matched pairs was 224 ± 130 J. Within each matched pair, subjects with \(\dot{V}\hbox{O}_{2\max}\) scores < 40 ml kg−1 min−1 were assigned to the untrained (UT) group and subjects with \(\dot{V}\hbox{O}_{2\max}\) scores >45 ml kg−1 min−1 were assigned to the moderately trained (MT) group. There were no significant differences between the groups for age (20 ± 1 vs 19 ± 1 y; p > 0.05) or body mass (63.0 ± 4.0 kg vs 57.9 ± 5.6 kg; p > 0.05). After being informed of the risks associated with participation, each subject gave her written consent. Approval for the study’s procedures was granted by the Institutional Ethics’ Committee.
Experimental overview
In addition to a familiarisation session for all tests, the experiment required the subjects to be tested on two separate occasions. On day one, subjects performed a graded exercise test (GXT) to determine \(\dot{V}\hbox{O}_{2\max}.\) At least 48 h later, subjects performed the 5×6-s test of RSA. Muscle biopsies from the vastus lateralis were also taken prior to (n = 16) and immediately following each 5×6-s cycle test (n = 12). Subjects were asked to maintain their normal diet and training throughout the study. Subjects were required to consume no food or beverages (other than water) 2 h prior to testing and were asked not to consume alcohol or perform vigorous exercise in the 24 h prior to testing.
Graded exercise test
The GXT was performed on an air-braked track-cycle ergometer (Evolution Pty Ltd., Adelaide, Australia) and consisted of graded exercise steps (4-min stages), using an intermittent protocol (1-min passive rest between stages). The test commenced at 50 W and thereafter, intensity was increased by 30 W every 4 min until volitional exhaustion. Subjects were required to maintain the set power output, which was displayed on a computer screen in front of them. The test was stopped when the subject could no longer maintain the required power output. Strong verbal encouragement was provided to each subject as they came to the end of the test, and was consistent between subjects.
Gas analysis (GXT)
During the GXT, expired air was continuously analysed for O2 and CO2 concentrations using Ametek gas analysers (Applied Electrochemistry, SOV S-3A11 and COV CD-3A, Pittsburgh, PA). Ventilation was recorded every 15 s using a turbine ventilometer (Morgan, 225A, Kent, England). The gas analysers were calibrated immediately before and verified after each test using three certified gravimetric gas mixtures (BOC Gases, Chatswood, Australia); the ventilometer was calibrated pre-exercise and verified post-exercise using a 1-l syringe in accordance with the manufacturer’s instructions. The ventilometer and gas analysers were connected to an IBM PC that measured and displayed variables every 15 s. The sum of the four highest consecutive 15-s values was recorded as the subject’s \(\dot{V}\hbox{O}_{2\max}.\)
5×6-s cycle test
Subjects performed a pre-test warm up consisting of 5 min of cycling at approximately 80 W, followed by three practice sprint starts. The practice starts required the subject to pedal close to maximal for 2–3 s, interspersed with 20 s of slow pedaling. A 90-s rest followed this. Each subject then performed a 6-s maximal sprint test on a front-access cycle ergometer (Model Ex-10, Repco, Australia). The total work recorded during the 6-s sprint was used as the criterion score during the subsequent 5×6-s cycle test. Upon completion of the 6-s test, subjects rested for 5 min before performing the 5×6-s cycle test.
The 5×6-s cycle test consisted of 5, 6-s maximal sprints departing every 30 s. During the first sprint, subjects were required to achieve at least 95% of their criterion score, as a check on pacing. If 95% of the criterion score was not achieved, subject were told that they would have to rest for a further 5 min and then recommence the 5×6-s cycle test (this never occurred). During the 24-s recovery between sprints, subjects had an inactive rest while seated on the cycle. Five seconds before starting each sprint, subjects were asked to assume the ready position and await the start signal. Strong verbal encouragement was provided to each subject during all sprints. This test has previously been reported to be both a valid (Bishop et al. 2001) and reliable (FitzSimons et al. 1993) test of RSA. For each 6-s sprint, work (W) and peak power output (P) were calculated. The total work (W tot) and percentage decrement over the repeated sprints for work (W dec) and power (P dec) were also calculated (Bishop et al. 2004a).
Muscle sampling and analysis
On the day of the 5×6-s cycle test, one incision was made under local anaesthesia (2.5 ml, 1% Xylocaine) into the skin, surrounding fascia and sheath of the vastus lateralis of each subject. The incision was used for the pre-test biopsy and then closed with a steri strip and subsequently used for the post-test biopsy. The first muscle sample (50–80 mg wet mass per sample) was taken (prior to warm up) during supine rest. The second muscle sample (50–80 mg) was taken immediately following the cessation of the 5×6-s test, while the subject remained on the cycle ergometer. Mean time for the pre-exercise and post-exercise (time from cessation of pedaling) muscle sample to be placed in liquid nitrogen was 6.9 ± 1.7 s and 17.5 ± 5.8 s, respectively. The samples were then removed from the biopsy needle and stored at −80°C until subsequent analysis. The frozen muscle samples were weighed on a microbalance (HM202 Lab Supply Pty Ltd., Sydney, Australia) and freeze dried. The freeze-dried muscle was then dissected free from visible blood and connective tissue.
Muscle buffering capacity; titration method (βmin vitro)
Freeze-dried, resting muscle samples (1.8–2.3 mg) were homogenised on ice for 2 min in a solution containing sodium fluoride (NaF; 10 mM) at a dilution of 30 mg dry muscle/ml of homogenising solution (Mannion et al. 1993). The muscle homogenate was then placed in a circulating water bath at 37°C for 5 min prior to and during the measurement of pH. The pH measurements were made with a microelectrode (MI-415, Microelectrodes, Inc., Bedford, NH, USA) connected to a pH meter (SA 520, Orion Research Inc., Cambridge, MA, USA). After initial pH measurement, muscle homogenates (rest samples) were adjusted to a pH of ∼7.2 with a sodium hydroxide (NaOH; 0.02 M) solution and then titrated to a pH of ∼6.2 by the serial addition of 2 μl of hydrochloric acid (HCl; 10 mM). From the fitted titration trendline, the number of moles of H+ (per gram of dry muscle) required to change the pH from 7.1–6.5 were interpolated. This value was then normalised to the whole pH unit for final display as micromoles H+ per gram of dry muscle per unit pH (μmol H+ gram muscle dw−1 pH−1) and determined as the subjects βmin vitro. The coefficient of variation for the measurement of βmin vitro using this technique in our laboratory is 5.4% and is comparable to that reported elsewhere (Mannion et al. 1993; Weston et al. 1996)
Muscle lactate concentration and βmin vivo determination
Freeze-dried, rest and post exercise muscle samples (3.5–4.5 mg) were enzymatically assayed for [La−] according to the methods of Harris et al. (1974). Muscle buffer capacity (βmin vivo) was estimated from changes in [La−]i and pHi; βmin vivo = Δ [La−] i /Δ pH i (Sahlin et al. 1976).
Statistical analysis
Analysis of the performance and physiological data was conducted using a one-way ANOVA. Where appropriate, post-hoc comparisons (Newman–Keuls) were employed. Least-squares linear regression analysis was used to calculate correlation coefficients between independent variables and repeated-sprint performance variables, using Pearson’s product moment (r). Correlations between W dec and other variables were re-examined by the use of semi-partial correlations (Thomas and Nelson 1990) to adjust for the contribution of the first sprint to decrement scores. Sigma Stat software (Jandel Scientific, San Rafael, CA, USA) was used for all statistical analyses and the level of statistical significance was set at p < 0.05.
Results
The results for peak aerobic power and RSA are summarised in Table 1. There was no significant difference between the groups for work (W 1; p = 0.59) or power (P 1; p = 0.66) on the first sprint, or for total work (W tot; p = 0.25). However, the MT subjects had significantly higher scores for \(\dot{V}\hbox{O}_{2\max}\) (p = 0.0001) and a significantly smaller work decrement across the five sprints (W dec; p = 0.045); differences in power decrement (P dec) approached significance (p = 0.09). Work scores for both groups for each sprint of the RSA test are shown in Fig. 1. There were no significant differences between the two groups for any of the first four sprints; however, differences in work performed during sprint 5 were significant (3.17 ± 0.54 vs 2.71 ± 0.36 kJ; p = 0.05). There were no significant differences between the two groups for βmin vitro (148.0 ± 26.9 vs 147.6 ± 27.2 μmol g dw−1 pH unit−1; p = 0.98), βmin vivo (238.0 ± 48.0 vs 230.4 ± 20.5 μmol g dw−1 pH unit−1; p = 0.98), or muscle lactate concentration and pH at any time point (Table 2).
Correlations
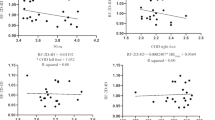
There was a significant correlation between W tot (J) and both \(\dot{V}\hbox{O}_{2\max}\) (ml kg−1 min−1; r = 0.64; p < 0.05), and βmin vivo (r = 0.52; p < 0.05), but not βmin-vitro (r = 0.41; p > 0.05). There were no significant correlations between W tot (J) and any of the other muscle variables. There was not a significant correlation between work decrement (W dec) and \(\dot{V}\hbox{O}_{2\max}\) (ml kg−1 min−1; r = −0.41; p > 0.05). However, when a semi-partial correlation was performed, to control for the contribution of W 1 to W dec, the correlation between W dec and \(\dot{V}\hbox{O}_{2\max}\) improved slightly and reached significance (r = −0.50; p < 0.05). There were also significant correlations between W dec and change in [H+] i (r = 0.66; p < 0.05), and βmin vivo (r = −0.61; p<0.05). These correlations were not altered when a semi-partial correlation was performed, to control for the contribution of W 1 to W dec. There was no significant correlation between W dec and β m in vitro (r = −0.22; p > 0.05).
Discussion
The aim of the present study was to investigate the relationship between \(\dot{V}\hbox{O}_{2\max}\) and RSA, while attempting to control for the previously reported effect of initial sprint performance on sprint decrement. The main finding was that, in subjects matched for initial sprint performance, the moderate \(\dot{V}\hbox{O}_{2\max}\) group recorded a smaller work decrement across five sprints and performed significantly more work on the final sprint. In addition, the semi-partial correlation between \(\dot{V}\hbox{O}_{2\max}\) and W dec (controlling for the contribution of first sprint power to W dec) was significant (r = −0.50; p = 0.04). Given no differences for initial sprint performance, muscle buffer capacity and metabolic responses, these results suggest that the smaller W dec in the MT group was associated with their higher \(\dot{V}\hbox{O}_{2\max}.\) However, the small variance (25%) in W dec explained by \(\dot{V}\hbox{O}_{2\max}\) suggests that other factors also play a role.
There were no significant differences between groups for age, body mass or muscle buffer capacity. In addition, subjects were matched for initial sprint performance, which has not been addressed in previous studies investigating the relationship between \(\dot{V}\hbox{O}_{2\max}\) and RSA. It has been reported that initial sprint performance is strongly correlated with sprint decrement during repeated-sprint efforts (Hamilton et al. 1991; Bogdanis et al. 1996; Wadley and Le Rossignol 1998; Bishop et al. 2003; Bishop et al. 2004b) and that better sprinters utilise more of their available PCr stores than sprinters of lesser ability (Hirvonen et al. 1987). Furthermore, subjects with a greater initial power output have previously been reported to have a greater glycogenolytic rate and subsequent H+ accumulation (Gaitanos et al. 1993). It has also been reported that subjects with a higher endurance fitness have a lower initial sprint performance (Bogdanis et al. 1996). Therefore, the lower sprint decrement previously reported in subjects with a high \(\dot{V}\hbox{O}_{2\max}\) may have been due, in part, to their lower initial sprint performance, and subsequent smaller metabolic disturbances, rather than due solely to their greater \(\dot{V}\hbox{O}_{2\max}\).
Matching subjects for initial sprint performance allowed a different examination of the relationship between \(\dot{V}\hbox{O}_{2\max}\) and RSA. Consistent with previous reports, the group with the higher \(\dot{V}\hbox{O}_{2\max}\) was better able to maintain performance over repeated sprints (Hamilton et al. 1991; Tomlin and Wenger 2002). While similar results were noted by Hamilton et al. (1991), the high \(\dot{V}\hbox{O}_{2\max}\) group consisted of endurance athletes, while the low \(\dot{V}\hbox{O}_{2\max}\) group consisted of team-sport athletes. It has previously been reported that even when matched for \(\dot{V}\hbox{O}_{2\max}\), endurance-trained athletes have a smaller sprint decrement than team-sport athletes (Bishop and Spencer 2004). Hamilton et al. (1991) also suggested that the smaller decrement reported in their high \(\dot{V}\hbox{O}_{2\max}\) group (endurance athletes) may have been associated with the smaller amounts of work performed during the initial sprints. In addition, neither of these two previous studies measured muscle buffer capacity which has been shown to differ according to training status (Edge et al. 2006) and to be related to RSA in this and a previous study (Bishop et al. 2004b). It is therefore possible that factors other than an elevated \(\dot{V}\hbox{O}_{2\max}\) were responsible, in part, for previous reports of greater RSA in subjects with a higher \(\dot{V}\hbox{O}_{2\max}\).
It has been reported that MT subjects consume significantly more O2 than UT subjects throughout sprint-recovery bouts (Tomlin and Wenger 2002). This supports previous findings that the aerobic contribution to both a sprint (r = 0.79; Bogdanis et al. 1996) and to sprint-recovery intervals (r = 0.83; Hamilton et al. 1991) is related to \(\dot{V}\hbox{O}_{2\max}.\) In addition, following EPO administration (that increased \(\dot{V}\hbox{O}_{2\max}\)), individuals performing 15×6-s treadmill sprints showed decreased accumulation of blood lactate, despite performing the same amount of work as compared to the control condition (Balsom et al. 1994a). This suggests that consuming more O2 during repeated sprints will result in less reliance on anaerobic metabolism. RSA has previously been negatively related to H+ accumulation (Gaitanos et al. 1993; Bishop et al. 2004b). Therefore an increased aerobic contribution during the RSA test, in our MT subjects with a greater \(\dot{V}\hbox{O}_{2\max},\) may have allowed them to produce more work in the latter sprints (smaller W dec) without a greater anaerobic contribution (as indicated by the similar La− and pH values between groups).
Individuals with a higher \(\dot{V}\hbox{O}_{2\max}\) are also likely to attain a higher \(\dot{V}\hbox{O}_{2}\) during sprinting and at the start of recovery. This will result in the provision of more aerobically-derived energy during recovery, despite endurance-trained athletes also having a more rapid recovery of excess post-exercise O2 consumption (Short and Sedlock 1997). As glycolysis ceases at the termination of exercise (Crowther et al. 2002) and the recovery of PCr is almost exclusively O2 dependent (Harris et al. 1976), greater O2 delivery to muscle post exercise should accelerate the rate of PCr resynthesis. This is supported by most (McCully et al. 1989; McCully et al. 1992; Takahashi et al. 1995), but not all (Petersen and Cooke 1994) studies that have reported increased PCr resynthesis in endurance versus UT individuals. In addition, \(\dot{V}\hbox{O}_{2\max}\) has been reported to be significantly correlated with the rate of PCr recovery in the quadriceps muscle following exhaustive exercise (Takahashi et al. 1995). Thus, a higher \(\dot{V}\hbox{O}_{2\max}\) appears to be associated with a greater ability to resynthesise PCr.
Restoration of PCr levels during recovery has been suggested to be critical for maintenance of performance during repeated 6-s sprints (Gaitanos et al. 1993). The importance of PCr availability for repeated-sprint performance is supported by the observation that power recovery for repeated cycle sprints and PCr resynthesis occur in parallel (Bogdanis et al. 1995). Furthermore, a strong relationship has been reported between power recovery in the first 10 s of a second 30-s sprint and the resynthesis of PCr (r = 0.84; Bogdanis et al. 1996). Thus, greater PCr resynthesis (as a result of greater post-exercise O2 consumption) may at least partially explain why MT subjects had a smaller W dec, even when matched for initial sprint performance.
While the two groups in the present study were significantly different for \(\dot{V}\hbox{O}_{2\max}\), it is also likely that they differed in a number of other physiological variables. Individuals with a high \(\dot{V}\hbox{O}_{2\max}\) typically have a greater percentage of slow-twitch (ST) fibres, increased concentrations of aerobic enzymes and increased mitochondrial size and number (Costill et al. 1976; Holloszy and Coyle 1984). Aerobic training is also associated with an increased muscle capillary density (Andersen and Henricksson 1977). It has been reported that PCr resynthesis is faster in ST fibres (Tesch et al. 1989) and that there is a significant relationship between citrate synthase activity and the time course of PCr recovery (Paganini et al. 1997). Furthermore, increased capillary density provides a decreased diffusion distance enhancing the removal of H+ from muscle (Holloszy and Coyle 1984). As both PCr resynthesis (Bogdanis et al. 1996) and H+ accumulation (Bishop et al. 2004b) have been associated with RSA, it may be that adaptations associated with a high \(\dot{V}\hbox{O}_{2\max}\) may facilitate greater recovery and improve RSA, rather than a high \(\dot{V}\hbox{O}_{2\max}\) per se.
The many other adaptations associated with a moderate \(\dot{V}\hbox{O}_{2\max}\) may explain why there was a low correlation between \(\dot{V}\hbox{O}_{2\max}\) and W dec in the present study (r = −0.41 to −0.50). This is consistent with the low correlations that have previously been reported between \(\dot{V}\hbox{O}_{2\max}\) and performance decrement (−0.42 < r < −0.65; Wadley and Le Rossignol 1998; McMahon and Wenger 1998; Tomlin and Wenger 2002; Bishop et al. 2003). The relationship between \(\dot{V}\hbox{O}_{2\max}\) and W dec was improved slightly when a semi-partial correlation was performed to account for the contribution of the initial sprint to performance decrement. However, \(\dot{V}\hbox{O}_{2\max}\) still only explained 25% of the variance in W dec in the present study.
Summary
The results of the present study have shown that subjects with a higher \(\dot{V}\hbox{O}_{2\max},\) but similar initial sprint performance, have a smaller sprint decrement (W dec). This suggests that \(\dot{V}\hbox{O}_{2\max}\) does contribute to the maintenance of performance during repeated-sprint efforts. However, the small variance in W dec explained by \(\dot{V}\hbox{O}_{2\max}\) suggests that other factors also contribute. As the \(\dot{V}\hbox{O}_{2\max}\) of subjects in this study was low to moderate, caution should be used in extrapolating these results to elite, team-sport athletes.
References
Andersen P, Henricksson J (1977) Capillary supply of the quadriceps femoris muscle of man. Adaptive response to exercise. J Physiol (Lond) 270:677–690
Balsom PD, Seger JY, Sjodin B, Ekblom B (1992) Maximal-intensity intermittent exercise: effect of recovery duration. Int J Sports Med 13(7):528–533
Balsom P, Ekblom B, Sjodin B (1994a) Enhanced oxygen availablility during high intensity intermittent exercise decreases anaerobic metabolite concentration in blood. Acta Physiol Scand 150:455–456
Balsom PD, Gaitanos GC, Ekblom B, Sjodin B (1994b) Reduced oxygen availability during high intensity intermittent exercise impairs performance. Acta Physiol Scand 152:279–285
Bishop D, Spencer M (2004) Determinants of repeated sprint ability in well-trained team-sport and endurance-trained athletes. J Sports Med Phys Fit 44(1):1–6
Bishop D, Spencer M, Duffield R, Lawrence S (2001) The validity of a repeated sprint ability test. J Sci Med Sport 4(1):19–29
Bishop D, Lawrence S, Spencer M (2003) Predictors of repeated-sprint ability in elite female hockey players. J Sci Med Sport 6(2):199–209
Bishop D, Davis C, Edge J, Goodman C (2004a) Induced metabolic alkalosis affects muscle metabolism and repeated-sprint ability. Med Sci Sports Exerc 36:807–813
Bishop D, Edge J, Goodman C (2004b) The relationship between muscle buffer capacity and repeated-sprint ability in females. Eur J Appl Physiol 92:540–547
Bogdanis GC, Nevill ME, Boobis LH, Lakomy HKA, Nevill AM (1995) Recovery of power output and muscle metabolites following 30 s of maximal sprint cycling in man. J Physiol (Lond) 482:467–480
Bogdanis GC, Nevill ME, Boobis LH, Lakomy HKA (1996) Contribution of phosphocreatine and aerobic metabolism to energy supply during repeated sprint exercise. J Appl Physiol 80(3):876–884
Christensen EH, Hedman R, Saltin B (1960) Intermittent and continuous running. Acta Physiol Scand 50:269–286
Costill DL, Daniels D, Evans W, Fink W, Krahenbuhl G, Saltin B (1976) Skeletal muscle enzymes and fibre composition in male and female track and field athletes. J Appl Physiol 40:149–154
Crowther GJ, Kemper WF, Carey MF, Conley KE (2002) Control of glycolysis in contracting muscle. 1. Turning it off. Am J Physiol 282:E74–E79
Edge J, Bishop D, Hill-Haas S, Goodman C (2006) The influence of training status on muscle buffer capacity. Eur J Appl Physiol 96:225–234
FitzSimons M, Dawson B, Ward D, Wilkinson A (1993) Cycling and running tests of repeated sprint ability. Aust J Sci Med Sport 25(4):82–87
Gaitanos GC, Williams C, Boobis LH, Brooks S (1993) Human muscle metabolism during intermittent maximal exercise. J Appl Physiol 75(2):712–719
Hamilton AL, Nevill ME, Brooks S, Williams C (1991) Physiological responses to maximal intermittent exercise: differences between endurance-trained runners and games players. J Sports Sci 9:371–382
Harris RC, Hultman E, Nordesjo LO (1974) Glycogen, glycolytic intermediates and high-energy phosphates determined in biopsy samples of musculus quadriceps femoris of man at rest: methods and variance of values. Scand J Clin Lab Invest 33:109–120
Harris C, Edwards RHT, Hultman E, Nordesjo LO, Nylind B (1976) The time course of phosphorylcreatine resynthesis during recovery of the quadriceps muscle in man. Pflugers Archiv 367:137–142
Hirvonen J, Rehunen S, Rusko H, Harkonen M (1987) Breakdown of high-energy phosphate compounds and lactate accumulation during short supramaximal exercise. Eur J Appl Physiol 56:253–259
Holloszy JO, Coyle EF (1984) Adaptations of skeletal muscle to endurance exercise and their metabolic consequences. J Appl Physiol 56:831–838
Mannion AF, Jakeman PM, Willan PLT (1993) Determination of human skeletal muscle buffer value by homogenate technique: methods of measurement. J Appl Physiol 75(3):1412–1418
McCully KK, Boden BP, Tuchler M, Fountain MR, Chance B (1989) Wrist flexor muscles of elite rowers measured with magnetic resonance spectroscopy. J Appl Physiol 67(3):926–932
McCully KK, Vandenborne K, DeMeirleir K, Posner JD, Leigh JS (1992) Muscle metabolism in track athletes, using 31P magnetic resonance spectroscopy. Can J Physiol Pharm 70:1353–1359
McMahon S, Wenger HA (1998) The relationship between aerobic fitness and both power output and subsequent recovery during maximal intermittent exercise. J Sci Med Sport 1(4):219–227
Paganini AT, Foley JM, Meyer RA (1997) Linear dependence of muscle phosphocreatine kinetics on oxidative capacity. Am J Physiol 272(41):C501–C510
Petersen SR, Cooke SR (1994) Effects of endurance training on recovery from high-intensity exercise. 10th Commonwealth & International Scientific Congress Proceedings Victoria, British Columbia, Canada, 227–236
Sahlin K, Harris RC, Nylind B, Hultman E (1976) Lactate content and pH in muscle samples obtained after dynamic exercise. Pflugers Archiv 367:143–149
Short KR, Sedlock SR (1997) Excess postexercise oxygen consumption and recovery rate in trained and untrained subjects. J Appl Physiol 83(1):153–159
Spencer M, Lawrence S, Rechichi C, Bishop D, Dawson B, Goodman C (2004) Time-motion analysis of elite field-hockey: special reference to repeated-sprint activity. J Sports Sci 22:843–850
Takahashi H, Inaki M, Fujimoto K, Katsuta S, Anno I, Niitsu M, et al (1995) Control of the rate of phosphocreatine resynthesis after exercise in trained and untrained human quadriceps muscles. Eur J Appl Physiol 71:396–404
Tesch PA, Thorsson A, Fujitsuka N (1989) Creatine phosphate in fiber types of skeletal muscle before and after exhaustive exercise. J Appl Physiol 66(4):1756–1759
Thomas JR, Nelson JK (1990) Research methods in physical activity. Champaign, IL: Human Kinetics pp
Tomlin DL, Wenger HA (2002) The relationship between aerobic fitness, power maintenance and oxygen consumption during intense intermittent exercise. J Sci Med Sport 5(3):194–203
Wadley G, Le Rossignol P (1998) The relationship between repeated sprint ability and the aerobic and anaerobic energy systems. J Sci Med Sport 1(2):100–110
Weston AR, Wilson GR, Noakes TD, Myburgh KH (1996) Skeletal muscle buffering capacity is higher in the superficial vastus than in the sloeus of spontaneously running rats. Acta Physiol Scand 157:211–216
Acknowledgments
The authors wish to express their sincere gratitude to all the participants for their maximal effort and cooperation. We would also like to thanks Drs Daelyn Cullen, Carmel Goodman and Peter Steele for performing the muscle biopsies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bishop, D., Edge, J. Determinants of repeated-sprint ability in females matched for single-sprint performance. Eur J Appl Physiol 97, 373–379 (2006). https://doi.org/10.1007/s00421-006-0182-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-006-0182-0