Abstract
Mutations in polycystin-1 (PC-1) are responsible for autosomal dominant polycystic kidney disease (ADPKD), characterized by formation of fluid-filled tubular cysts. The PC-1 is a multifunctional protein essential for tubular differentiation and maturation found in desmosomal junctions of epithelial cells where its primary function is to mediate cell–cell adhesion. To address the impact of mutated PC-1 on intercellular adhesion, we have analyzed the structure/function of desmosomal junctions in primary cells derived from ADPKD cysts. Primary epithelial cells from normal kidney showed co-localization of PC-1 and desmosomal proteins at cell–cell contacts. A striking difference was seen in ADPKD cells, where PC-1 and desmosomal proteins were lost from the intercellular junction membrane, despite unchanged protein expression levels. Instead, punctate intracellular expression for PC-1 and desmosomal proteins was detected. The N-cadherin, but not E-cadherin was expressed in adherens junctions of ADPKD cells. These data together with co-sedimentation analysis demonstrate that, in the absence of functional PC-1, desmosomal junctions cannot be properly assembled and remain sequestered in cytoplasmic compartments. Taken together, our results demonstrate that PC-1 is crucial for formation of intercellular contacts. We propose that abnormal expression of PC-1 causes disregulation of cellular adhesion complexes leading to increased proliferation, loss of polarity and, ultimately, cystogenesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Autosomal dominant polycystic kidney disease (ADPKD) is a common genetic disease with an incidence of 1/1000. The disease is characterized by progressive growth of focal cysts in the kidney as well as other organs such as liver, pancreas, and spleen (Calvet and Grantham 2001; Grantham 2003; Cowley 2004; Wilson 2004a). Cysts can arise from multiple segments of the nephron. Mutations in the PKD1 gene, encoding polycystin-1 (PC-1), account for ~85% of ADPKD cases (Burn et al. 1995; Hughes et al. 1995). The rest of the ADPKD cases can be attributed to a mutation in the PKD2 gene, encoding polycystin-2 (PC-2) (Mochizuki et al. 1996). Cystogenesis is initiated in mutant tubular epithelial cells and progresses with increased proliferation and apoptosis, fluid secretion into the cystic lumen, changes in cellular polarity, and extracellular matrix remodelling (Bacallao and Carone 1997; Grantham 1996; Wilson 2004a). The role of polycystins in these cellular processes has been extensively studied over the last few years.
The PC-1 is a large integral membrane glycoprotein with multiple extracellular adhesive domains that functions at multiple subcellular locations, including intercellular junctions, cell–matrix junctions, and cilia (Ong and Wheatley 2003). A number of studies have revealed the diversity of the functions of PC-1, implicating it in signaling pathways together with PC-2. It has been shown that PC-1 is involved in the regulation of the cell cycle through the JAK-STAT pathway, Wnt signaling, G-protein coupled signaling and, most importantly, in mediating cation channel activity (Babich et al. 2004; Bhunia et al. 2002; Ikeda and Guggino et al. 2002; Kim et al. 1999; Parnell et al. 1998).
Experimental evidence from several groups established an important role for PC-1 in epithelial cell morphogenesis, including differentiation and maturation in vivo (Kim et al. 2000; Lu et al. 1997). Recently, we have demonstrated that functional PC-1 expression at desmosomal cell–cell junctions at controlled levels is critical for proper tubular differentiation and maturation in vitro (Bukanov et al. 2002). We have also shown that PC-1 functions in cell–cell adhesion through homophilic interactions of its PKD (Ig-like) domains (Ibraghimov-Beskrovnaya et al. 2000).
Investigations using diverse experimental systems have also shown localization of PC-1 to desmosomes of epithelial cells, where it can modulate intercellular adhesion (Scheffers et al. 2000; Streets et al. 2003; Xu et al. 2001). Alterations in PC-1 mediated adhesion may explain the abnormal epithelial cellular phenotype observed in ADPKD cells, including dedifferentiation, loss of epithelial polarity, and increased proliferation. Therefore, we have set out to perform the structure–function analysis of cell–cell adhesion junctions in primary cells derived from ADPKD kidneys.
In this study, we report abnormal expression of proteins, comprising desmosomal junctions in ADPKD primary cystic epithelial cells. Desmosomal cadherins and associated proteins are no longer localized at intracellular junctions, but instead are sequestered to the cytoplasmic pools with mutant PC-1. In addition, adherens junctions appear disrupted, with great reduction of E-cadherin complexes, which is in agreement with Roitbak et al. (2004). Collectively, our data demonstrate that mutations in PC-1 lead to perturbation of intercellular adhesion complexes, which in turn contributes to a loss of epithelial polarity and differentiation, resulting in cystogenesis.
Materials and methods
Primary cell cultures and immunofluorescence
Primary tubular epithelial cells were isolated from two normal and three ADPKD kidneys. Normal primary kidney epithelial cells were obtained from normal portions of kidney containing localized adenocarcinoma. The ADPKD samples were selected based on clinical observations and family history consistent with mutation in the PKD1 gene. All primary ADPKD cells were derived from a pool of cysts dissected within a kidney and were not clonal. Briefly, multiple ADPKD cysts were cleaned of fibrous and adipose tissue and the cyst domes were excised. The cyst domes were digested in Dulbecco’s modified Eagle’s medium (DMEM) containing 1 mg/ml collagenase (Sigma–Aldrich, St Louis, MO, USA) to loosen the epithelial cells from basal membrane. The cell suspension was washed to remove cell debris and collagenase (Yamaguchi et al. 2000). Cells were grown in DMEM containing 1% fetal bovine serum, 5 μg/ml insulin, 10 μg/ml transferrin, 5 ng/ml sodium selenite, 6.5 ng/ml triiodothyronin, 10 ng/ml EGF, 500 ng/ml hydrocortisone, and 10 mM HEPES (Invitrogen, Carlsbad, CA, USA) on collagen I coated vessels (Beckton Dickinson, Bedford, MA, USA). All primary cultures were used between passages two and five. Immunofluorescence was performed as follows: normal or ADPKD cells grown on collagen I coated eight well culture slides (Becton Dickinson, San Diego, CA, USA) were fixed in 10% acetic acid/50% ethanol (vol/vol) for 30 min on ice, washed three times for 5 min with phosphate buffered saline (PBS) and blocked with PBS/3% bovine serum albumin (PBS–BSA) (Bukanov et al. 2002; Ibraghimov-Beskrovnaya et al. 2000). Primary antibodies directed against desmocollin-2/-3 (Zymed Laboratories Inc., San Francisco, CA, USA), desmoplakin-1/-2 (RDI, Flanders, NJ, USA), desmoglein-1 (BD Transduction Laboratories, Lexington, KY, USA), E-cadherin (BD Transduction Laboratories), or pan-cytokeratin (Sigma–Aldrich, St Louis, MO, USA) were used as recommended by the manufacturers. Anti-PC-1 antibody (anti-LRR) was used at 1:100 dilution. Primary antibodies were incubated for 1 h at 37°C followed by incubation with FITC-labeled or Cy3-labeled secondary antibodies at 1:100 dilution. The staining specificity was determined using secondary antibody alone. Staining was visualized on an Olympus IX70 microscope with 20 times objectives. Images were captured with QED Camera Plus-In imaging system (QED Imaging Inc., Pittsburgh, PA, USA).
Immunohistochemistry
Immunohistochemical staining was performed using an indirect immunoperoxidase protocol as described previously (Joly et al. 2003). Acetone-treated cryostat sections (5 μm) were fixed in chloroform and blocked in PBS containing 5% goat serum and 1% BSA, and incubated for 1 h with primary antibodies: E-cadherin (clone 4 A2C7, Zymed Laboratories, Inc. South San Francisco, USA), N-cadherin (clone3B9, Zymed), Desmoglein-1/-2 (clone DG.3.10, Progen, Montigny le Bretonneux, France) or Desmoplakin-1 (clone DP-2.17, Progen) and treated with LSAB2-horseradish peroxydase system according to the manufacturer’s recommendations (Dako, Carpinteria, CA, USA). Peroxidase staining was examined with an ortho LEICA microscope coupled with a CDD camera (Olympus).
Subcellular fractionation by continuous sucrose gradient
Subcellular fractionations through a continuous sucrose gradient were performed as previously described (Scheffers et al. 2002). Briefly, 3 days post confluence cells were homogenized in 10 mM Tris–HCl (pH 7.4), 179 mM sucrose, 750 mM KCl, 19.2 mM NaCl, and complete protease inhibitor cocktail (Roche Diagnostics, Mannheim, Germany) using a dounce homogenizer on ice. Homogenates were centrifuged at 1000 g to remove cellular debris. The supernatant was layered on 5–40% continuous sucrose gradients. Gradients were centrifuged at 40,000 g overnight at 4°C. Fractions of 0.5 ml were collected from the bottom of the gradients and numbered. Proteins (100 μl) were then separated by SDS-PAGE and immunoblotted.
SDS-PAGE and immunoblotting
For total protein western blotting analysis, cells were harvested in lysis buffer (50 mM Tris/HCl pH 7.4, 150 mM NaCl, 5 mM EDTA, and 1% Nonidet P40 in the presence of protease inhibitor cocktail) using cell scrapers. Homogenates were centrifuged at 10,000 g for 10 min at 4°C and cellular proteins were normalized using a BCA protein assay (Pierce, Rockford, IL, USA). Proteins (200 μg) were resolved by SDS-PAGE (3–12% gradients) and transferred onto Immobilon P membranes (Millipore, Bedford, MA, USA) as previously described (Ibraghimov-Beskrovnaya et al. 2000). Antibodies against GS28 (BD Transduction Laboratories, Lexington, KY) and Na+/K+ ATPase (BD Transduction Laboratories) were used at a dilution of 1:1000, and anti-polycystin antibody (LRR) was used at 1:100 followed by incubation with HRP-labeled secondary antibodies at 1:10,000. Immunoreactive proteins were detected using enhanced chemi-luminescence (Amersham Pharmacia Biotech, Little Chalfont Buckinghamshire, UK).
Results
Loss of polycystin-1 and desmosomal proteins from intercellular junctions of ADPKD cells
The PC-1 has recently been shown to play an essential role in intercellular adhesion (Charron et al. 2000; Ibraghimov-Beskrovnaya et al. 2000; Scheffers et al. 2000). We and others have determined that PC-1 is a novel component of desmosomal junctions of immortalized canine and human epithelial cells (Bukanov et al. 2002; Scheffers et al. 2000). In order to address the role of PC-1 and desmosomal components in the normal and cystic states, we have chosen human normal and ADPKD-derived primary epithelial cells for further analysis. Both normal and cystic cells were positive for pan-cytokeratin staining, confirming their epithelial origin (not shown).
First, utilizing primary normal human renal epithelial cells, we examined colocalization of PC-1 with several components of desmosomal junction by performing dual color immunofluorescence staining experiments. We have used anti-human PC-1 antibody (anti-LRR), which has been extensively characterized for its specificity. Thus, we and others have previously shown that anti-LRR antibody specifically recognizes the full length in vitro translated PC-1 as well as endogenous PC-1 in multiple tissues and cell lines (Foggensteiner et al. 2000; Ibraghimov-Beskrovnaya et al. 1997; Streets et al. 2003). As shown in Fig. 1, a concordant punctate pattern of staining was produced for PC-1 and desmoplakin, desmoglein, and desmocollin, resulting in a completely overlapping merged pattern. This analysis confirms that PC-1 is a component of desmosomal junction in primary human epithelial cells. Culture conditions used in these experiments were optimal for detection of PC-1 at intercellular contacts and therefore expression of PC-1 in the cilia was not analyzed. Specificity of staining in each experiment was controlled by using secondary antibody only.
Localization of polycystin-1 (PC-1) to desmosomal junctions in normal human primary renal tubular epithelial cells. The PC-1 staining (red) and desmoplakin, desmoglein, and desmocollin staining (green) are shown. The merged image shows complete colocalization of PC-1 and desmosomal markers, producing yellow overlapping pattern. Scale bar equals to 10 μm as indicated
As PC-1 is mutated in ADPKD, we set out to evaluate the status of desmosomal junctions in primary ADPKD cyst-derived epithelial cells. Figure 2a shows immunostaining of normal and ADPKD cells with antibodies directed against PC-1, desmoplakin, and two desmosomal cadherins, desmoglein and desmocollin. Immunostaining of normal cells (Fig. 2a, top panel) makes evident the typical localization patterns of PC-1 and desmosomal proteins at cell–cell contacts as expected. In ADPKD cells, however, the intercellular membrane localization of PC-1 as well as each desmosomal junction marker is lost (Fig. 2a, bottom panel). Instead, punctate intracellular expression for PC-1 and desmosomal proteins is seen (Fig. 2a). Similar results were obtained using three independent primary cultures derived from separate ADPKD donors (not shown). These data suggest that desmosomal junctions are disrupted in ADPKD primary epithelial cells.
a Abnormal expression of desmosomal junctional proteins in ADPKD cells. The top row shows normal human renal epithelial cells and the bottom row shows ADPKD primary cystic epithelial cells. Cells were immunostained with antibodies against markers of desmosomal cell–cell junction as indicated on the top of each image. Note the membrane localization of PC-1, desmoplakin, desmoglein, and desmocollin in the normal state (top row) and loss of membrane association for all these proteins in cystic state (bottom row). In ADPKD cells, an intracellular punctate pattern of staining for PC-1 and desmosomal proteins is detected. b Expression of adherens junctions (AJ) in normal and ADPKD cells. Membrane localization of E-cadherin is detected only in normal cells. Note unexpected membrane localization of N-cadherin in ADPKD cells, but not in normal cells. Scale bar equal to 10 μm as indicated
The structure of adherens junctions in ADPKD cells
We have tested our primary normal and ADPKD epithelial cells for integrity of adherens junctions (Fig. 2b). Antibodies against E-cadherin were used to determine the expression of adherens junctions by immunostaining. The E-cadherin expression in normal primary cells showed characteristic intercellular membrane pattern of staining (Fig. 2b, top panel). In contrast, E-cadherin staining was not detectable in the lateral membranes of ADPKD cells (Fig. 2b, bottom panel). The observed loss of E-cadherin expression in ADPKD cells was further confirmed by western blot analysis. Interestingly, N-cadherin showed no expression at the membranes of normal primary cells; however, it was expressed at the cell–cell junctional membranes of ADPKD cells (Fig. 2b, bottom panel). Thus, the loss of PC-1 from intercellular contacts of ADPKD cells leads to a loss of E-cadherin expression from adherens junctions. Instead, N-cadherin is expressed at adherens junction of ADPKD cells.
Differential regulation of expression for desmosomal and adherens junction proteins in ADPKD cells
The loss of PC-1 along with junctional proteins from intercellular contacts may be a result of their redistribution from lateral membranes in the normal state to the cytoplasmic pools in ADPKD. It is also possible that depletion of membrane-associated proteins may be due to down-regulation of their expression. To address this question, we have compared protein expression levels for several components of desmosomal and adherens junctions by western blot analysis.
Figure 3 presents a comparison of PC-1 and junctional marker protein expression between normal and ADPKD primary cells by western blot. The expression of E-cadherin is not detectable in ADPKD cells, suggesting that loss of E-cadherin from adherens junctions is a result of down-regulation of its expression or posttranslational degradation. The N-cadherin was found to be expressed at a higher level in ADPKD cells as compared to normal cells. In combination with our immunofluorescent results (Fig. 2) showing membrane localization of N-cadherin in cystic cells but not in normal cells, our data suggest possible compensatory expression of N-cadherin in place of E-cadherin in cystic epithelia. In contrast, the desmosomal junction components desmoglein and desmocollin appear to be similarly expressed in normal and ADPKD cells (Fig. 3). This suggests that desmosomal proteins are still expressed at a normal level in ADPKD, but are not able to properly localize to cell–cell contacts. Similar to the expression of desmosomal components, PC-1 is expressed at wild-type levels in ADPKD cells (Fig. 3), but is not targeted to the membrane (Fig. 2a). Interestingly, we observed continuous expression of PC-1 in all three cystic ADPKD epithelia cultures examined (not shown). Similar results were reported by others showing that ADPKD cyst-derived epithelia continue to express PC-1 (Ong et al. 1999; Roitbak et al. 2004; Ward et al. 1996).
Expression levels of intercellular junctional proteins in normal and ADPKD cellular lysates. Equal amounts of total protein lysates isolated from normal and ADPKD cells were blotted with antibodies against PC-1 and junctional markers as indicated on top of each panel and identified by arrowheads E-cadherin (E-cad), N-cadherin (N-cad), Desmoglein-1 (Desmgl-1), Desmocolin-2/ Desmocolin-3 (Desmocol-2/3), and polycystin-1 (PC-1). Note the down-regulation of E-cadherin and up-regulation of N-cadherin expression levels in ADPKD cells. Molecular weight standards (×10−3) are indicated on the left of each panel
Co-sedimentation of polycystin-1 with desmosomal proteins in normal and ADPKD cells
To investigate whether intracellular redistribution for PC-1 and desmosomal proteins are concordant in ADPKD cells and to confirm the importance of PC-1 for desmosomal junctions, we have performed sucrose gradient co-sedimentation experiments. We aimed to examine which cell–cell junctional markers co-sediment with PC-1 on sucrose density gradients in normal and ADPKD cells.
Normal and ADPKD cell homogenates were centrifuged through continuous sucrose gradients and fractions were subjected to western blot analysis. A representative experiment is shown in Fig. 4. In normal human primary epithelial cells, PC-1 was found in fractions 14–19 as shown in Fig. 4. As expected, the desmosomal proteins desmoplakin and desmoglein were copurifying in these fractions. Desmoglein distributed more broadly than desmoplakin over several fractions, however. The presence of E-cadherin and Na+/K+ ATPase in these fractions confirms the membrane localization for PC-1 and desmosomal markers as seen by immunofluorescence in Fig. 1. In ADPKD cells, however, PC-1 was no longer seen in fractions 16–19, but was found in intracellular vesicles partially overlapping with the Golgi marker. Desmosomal markers in ADPKD cells exhibited the same sedimentation peak as PC-1 in fractions 12–15 (Fig. 4). An additional pool of desmoglein immunoreactive bands was detected in fractions 3–5, where no PC-1 was found. The significance of this pool needs further investigation. Thus, it appears that PC-1 co-sediments with desmosomal proteins in both normal and ADPKD cells: PC-1 and desmosomal proteins are localized to the cellular membranes in the normal state and to the intracellular compartments in the cystic state. Control proteins Na+/K+-ATPase and GS 28 exhibited overlapping patterns of sedimentation in both normal and ADPKD cell lines. The E-cadherin expression in ADPKD cells was almost undetectable in sucrose gradient fractions, corroborating down-regulation of its expression seen in western blot analysis (Fig. 3). A similar pattern of E-cadherin expression in the cystic state was also reported in primary cultures of independent origin (Roitbak et al. 2004). Therefore, our data demonstrate perturbation of desmosomal cell–cell junctions in ADPKD cells.
Cosedimentation of PC-1 and desmosomal proteins from normal and ADPKD cells through 5–40% sucrose gradients. Shown are PVDF membrane transfers of sucrose gradient fractions 1–19 from either normal or ADPKD cells (as indicated on the left of each panel) separated by SDS-PAGE and stained with different antibodies (as indicated on the right). Co-sedimentation of PC-1 with desmoplakin and desmoglein in the membrane fraction of normal epithelial cells (fractions 14–19) is seen. Note the shift of co-sedimentating fractions of PC-1 and desmosomal markers to the pool of intracellular vesicles in ADPKD cells. As controls for the sucrose gradients, Na+/K+ATPase and GS 28 were analyzed. Note the overlapping patterns in both normal and ADPKD cells
Expression of desmosomal proteins in normal and ADPKD kidneys in vivo
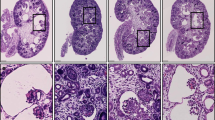
Analysis of immunoperoxidase staining of normal and ADPKD renal tissues was performed to address distribution and expression patterns of desmosomal junctional proteins. As shown in Fig. 5a, desmoplakin showed strong staining of tubular epithelia with membrane accentuation in normal kidney. In contrast, cytoplasmic expression of desmoplakin was mainly seen in cystic kidney tissue from an ADPKD patient (Fig. 5b). Desmoglein exhibited weaker expression in lateral membranes of normal kidney tubules with additional punctate cytoplasmic staining (Fig. 5c). Desmoglein was seen only in cytoplasmic pools of mature cyst-lining epithelial cells, but still could be found partially membrane-associated in early stages of cystic growth (Fig. 5d). Therefore, the observed expression of desmosomal junctional proteins in normal and cyst-derived primary epithelial cells is in concordance with expression observed in normal and ADPKD kidney tissue.
Distribution of intercellular junctional markers in normal and ADPKD kidney. Human normal and ADPKD kidneys were frozen and 7 μM sections were labeled with antibodies against desmosomal markers (a–d), E-cadherin, (e, f) and N-cadherin (g, h) as indicated on the right of each row. The cystic lumen is indicated (Cy)
We have also analyzed the expression of adherens junction proteins in normal and cystic kidney cortex. The E-cadherin, as expected, showed strong basolateral expression in the distal nephron of normal kidney, but no expression in the normal proximal tubule (Fig. 5e). Cystic samples demonstrated preferentially cytoplasmic staining with partial basolateral localization (Fig. 5f). A weak expression of N-cadherin is detected in proximal tubules of normal kidney, but increased expression is observed in cystic epithelia (Fig. 5g, h). Thus, the aberrant expression of junctional proteins in ADPKD cystic epithelia clearly shows that intercellular adhesion is compromised at both adherens and desmosomal junctions during cytogenesis.
Discussion
The role of PC-1 in epithelial cell differentiation and maturation has been directly established by Pkd1 knock-out studies (Boulter et al. 2001; Kim et al. 2000; Lu et al. 1997). We and others have previously demonstrated that PC-1 is an important dynamic player in normal tubular epithelial morphogenesis with a direct role in cell–cell adhesion (Bukanov et al. 2002; Huan and van Adelsberg 1999; Scheffers et al. 2000; Streets et al. 2003). Specifically, PC-1 was shown to be a novel component of desmosomes, interacting with intermediate filaments (Bukanov et al. 2002; Scheffers et al. 2000; Xu et al. 2001). Also, PC-1 is likely to employ homophilic interactions of its Ig-like domain in order to mediate intercellular adhesion (Ibraghimov-Beskrovnaya et al. 2000). The role of PC-1 in molecular cystogenesis, however, has not been fully characterized.
We reasoned that, because intercellular adhesion junctions are well known to rearrange during differentiation and in pathologic conditions, it is possible that mutations in PC-1 may result in perturbation of cell–cell adhesion in ADPKD epithelia. We have specifically focused on desmosomal junctions of primary cells derived from ADPKD cysts. Indeed, we have found profound abnormalities in desmosomal protein localization in cystic epithelia. First, using normal primary kidney tubular epithelial cells, we have confirmed PC-1 localization to the desmosomes, as previously demonstrated for immortalized cell lines (Bukanov et al. 2002). In contrast, we have seen no expression of the desmosomal proteins desmoglein, desmocollin, and desmoplakin at the lateral membranes of ADPKD cells. Interestingly, desmosomal proteins are still produced, as detected by western blotting, but are evidently unable to properly localize to the intercellular membranes and were only found in the cytoplasmic pools. Such an expression pattern for desmosomal proteins is concordant with the PC-1 expression pattern in ADPKD cells. The expression of PC-1 in ADPKD primary cells is also detected in cystic cultures of independent origins, suggesting that cyst formation is not accompanied by the loss of protein (Roitbak et al. 2004; Streets et al. 2003). We further confirmed by co-sedimentation experiments that intracellular vesicular redistribution for PC-1 and desmosomal proteins were concordant in ADPKD cells. Based on these data as well as previous observations made by us and others, we propose a model, showing engagement of PC-1 in formation of desmosomal junctions in normal and cystic epithelial cells (Fig. 6). Our data demonstrate that PC-1 is essential for normal desmosomal function and that desmosomal junctions cannot form properly in ADPKD. To understand the molecular events in the dynamic process of formation of cell–cell junctions and how mutated PC-1 leads to mislocalization of junctional proteins, it would be interesting to test a direct effect of down-regulation of PKD1 gene using RNAi technology.
Possible arrangement of polycystin-1 and cell–cell contacts of normal and ADPKD epithelial cells. In normal tubular epithelial cell, polycystin-1 co-localizes and co-sediments with desmosomal junctional (DJ) proteins (Bukanov et al. 2002; Scheffers et al. 2000; Xu et al. 2001). Shown are DJ components: desmoplakin (Dp), desmogleins (Dsg), desmocollins (Dsc), and plakoglobin (Pg). Polycystin-1 (PC-1) is likely to engage its extracellular PKD (Ig-like) domains for homotypic interactions from opposing cells (Ibraghimov-Beskrovnaya et al. 2000). DJ proteins link together intermediate filaments (IF) of epithelial cells (Gumbiner 1996). A major component of adherens junctions (AJ) is E-cadherin (E-cad), associated with α, β, and γ-catenins (catenins), providing linkage to the actin cytoskeleton (Gumbiner 1996). In ADPKD epithelial cells, disfunctional PC-1 (shown by crossed red lines) and desmosomal proteins are lost from cell–cell contacts and remain in intracellular vesicles. Expression of E-cadherin is greatly reduced, resulting in compensatory expression of N-cadherin (N-cad) (Roitbak et al. 2004)
Abnormalities were also detected in adherens junctions, where significant down-regulation of E-cadherin was seen, in agreement with previous reports (Roitbak et al. 2004; Streets et al. 2003). Interestingly, N-cadherin was expressed at intercellular membranes of ADPKD cells, but not in normal cells. In addition, we have found that N-cadherin was expressed at higher levels in ADPKD cysts in vivo, while only weak staining was seen in normal proximal nephron. We propose that it may be expressed in place of E-cadherin, possibly by a compensatory mechanism as described in similar studies (Streets et al. 2003). It is important to note that some cysts were negative for N-cadherin expression, suggesting possible compensatory expression of other as yet unidentified junctional proteins.
Interestingly, desmoglein and E-cadherin still exhibited partial membrane association (in addition to the cytoplasmic pools) in epithelia lining ADPKD cysts in the early stages of cystogenesis. In view of compensatory N-cadherin expression in ADPKD cysts, it is likely that in vivo cystic epithelial cells remain partially polarized and still maintain cell–cell/matrix adhesion. At later stages of cystogenesis, the cyst-lining cells dedifferentiate so that the loss of normal intercellular adhesion becomes more evident. In primary ADPKD cultures we do not detect any membrane-associated E-cadherin and only a small amount of protein can still be detected in the cytoplasm. Because cyst formation in ADPKD occurs over time and newly formed cysts arise from fully formed tubules, deregulation of cell–cell contacts and loss of normal epithelial phenotype become more prominent as cystic growth progresses. We suggest that since ADPKD cultures are derived from cysts where critical molecular mechanisms of dedifferentiation are already engaged, cell–cell contacts cannot form properly in culture resulting in mislocation of desmosomal proteins and down-regulation of E-cadherin.
Several other functions were recently ascribed to PC-1, particularly in primary cilia (Ong et al. 2003). Importantly, by using the same anti-polycystin antibody, we and others have demonstrated that PC-1 localizes to desmosomal junctions in the early stages of epithelial sheet formation, and at late stages can be detected in primary cilia as they are formed (Yoder et al. 2002). Also, PC-1 was shown to be important in cell/matrix interactions (Joly et al. 2003; Wilson 2004b). The inactivation of tensin and insertional mutation in laminin alpha five result in cystogenesis (Lo et al. 1997; Shannon and Miner et al. 2004). Therefore, it will be of great interest to determine other functional abnormalities of ADPKD epithelia.
In conclusion, we have shown abnormal expression of desmosomal proteins and perturbation of intercellular adhesion in ADPKD primary cells. The disruption of cell–cell interactions would lead to increased proliferation and dedifferentiation seen in ADPKD. Moreover, the extracellular region of PC-1 was shown to be critical for regulating ion currents—the downstream signals for intercellular interactions (Babich et al. 2004). Thus, altered intercellular adhesion causes changes in ionic currents and, together with other signaling pathways, culminates in cystic transformation.
References
Babich V, Zeng WZ, Yeh BI, Ibraghimov-Beskrovnaya O, Cai Y, Somlo S, Huang CL (2004) The N-terminal extracellular domain is required for polycystin-1-dependent channel activity. J Biol Chem 279:25582–25589
Bacallao RL, Carone FA (1997) Recent advances in the understanding of polycystic kidney disease. Curr Opin Nephrol Hypertens 6:377–383
Bhunia AK, Piontek K, Boletta A, Liu L, Qian F, Xu PN, Germino FJ, Germino GG (2002) PKD1 induces p21(waf1) and regulation of the cell cycle via direct activation of the JAK-STAT signaling pathway in a process requiring PKD2. Cell 109:157–168
Boulter C, Mulroy S, Webb S, Fleming S, Brindle K, Sandford R (2001) Cardiovascular, skeletal, and renal defects in mice with a targeted disruption of the Pkd1 gene. Proc Natl Acad Sci USA 98:12174–12179
Bukanov NO, Husson H, Dackowski WR, Lawrence BD, Clow PA, Roberts BL, Klinger KW, Ibraghimov-Beskrovnaya O (2002) Functional polycystin-1 expression is developmentally regulated during epithelial morphogenesis in vitro: downregulation and loss of membrane localization during cystogenesis. Hum Mol Genet 11:923–936
Burn TC, Connors TD, Dackowski WR, Petry LR, Van Raay TJ, Millholland JM, Venet M, Miller G, Hakim RM, Landes GM, et al (1995) Analysis of the genomic sequence for the autosomal dominant polycystic kidney disease (PKD1) gene predicts the presence of a leucine-rich repeat. The American PKD1 consortium (APKD1 consortium). Hum Mol Genet 4:575–582
Calvet JP, Grantham JJ (2001) The genetics and physiology of polycystic kidney disease. Semin Nephrol 21:107–123
Charron AJ, Nakamura S, Bacallao R,Wandinger-Ness A (2000) Compromised cytoarchitecture and polarized trafficking in autosomal dominant polycystic kidney disease cells. J Cell Biol 149:111–124
Cowley BD Jr (2004) Recent advances in understanding the pathogenesis of polycystic kidney disease: therapeutic implications. Drugs 64:1285–1294
Foggensteiner L, Bevan AP, Thomas R, Coleman N, Boulter C, Bradley J, Ibraghimov-Beskrovnaya O, Klinger K, Sandford R (2000) Cellular and subcellular distribution of (PC-2), the protein product of the PKD2 gene. J Am Soc Nephrol 11:814–827
Grantham JJ (1996) The etiology, pathogenesis, and treatment of autosomal dominant polycystic kidney disease: recent advances. Am J Kidney Dis 28:788–803
Grantham JJ (2003) Lillian jean kaplan international prize for advancement in the understanding of polycystic kidney disease. Understanding polycystic kidney disease: a systems biology approach. Kidney Int 64:1157–1162
Gumbiner BM (1996) Cell adhesion: the molecular basis of tissue architecture and morphogenesis. Cell 84:345–357
Huan Y, van Adelsberg J (1999) Polycystin-1, the PKD1 gene product, is in a complex containing E-cadherin and the catenins. J Clin Invest 104:1459–1468
Hughes J, Ward CJ, Peral B, Aspinwall R, Clark K, San Millan JL, Gamble V, Harris PC (1995) The polycystic kidney disease 1 (PKD1) gene encodes a novel protein with multiple cell recognition domains. Nat Genet 10:151–160
Ibraghimov-Beskrovnaya O, Bukanov NO, Donohue LC, Dackowski WR, Klinger KW, Landes GM (2000) Strong homophilic interactions of the Ig-like domains of polycystin-1, the protein product of an autosomal dominant polycystic kidney disease gene, PKD1. Hum Mol Genet 9:1641–1649
Ibraghimov-Beskrovnaya O, Dackowski WR, Foggensteiner L, Coleman N, Thiru S, Petry LR, Burn TC, Connors TD, Van Raay T, Bradley J, Qian F, Onuchic LF, Watnick TJ, Piontek K, Hakim RM, Landes GM, Germino GG, Sandford R, Klinger KW (1997) Polycystin: in vitro synthesis, in vivo tissue expression, and subcellular localization identifies a large membrane-associated protein. Proc Natl Acad Sci USA 94:6397–6402
Ikeda M, Guggino WB (2002) Do polycystins function as cation channels? Curr Opin Nephrol Hypertens 11:539–545
Joly D, Morel V, Hummel A, Ruello A, Nusbaum P, Patey N, Noel LH, Rousselle P, Knebelmann B (2003) Beta4 integrin and laminin 5 are aberrantly expressed in polycystic kidney disease: role in increased cell adhesion and migration. Am J Pathol 163:1791–1800
Kim E, Arnould T, Sellin LK, Benzing T, Fan MJ, Gruning W, Sokol SY, Drummond I, Walz G (1999) The polycystic kidney disease 1 gene product modulates Wnt signaling. J Biol Chem 274:4947–4953
Kim K, Drummond I, Ibraghimov-Beskrovnaya O, Klinger K, Arnaout MA (2000) Polycystin 1 is required for the structural integrity of blood vessels. Proc Natl Acad Sci USA 97:1731–1736
Lo SH, Yu QC, Degenstein L, Chen LB, Fuchs E (1997) Progressive kidney degeneration in mice lacking tensin. J Cell Biol 136:1349–1361
Lu W, Peissel B, Babakhanlou H, Pavlova A, Geng L, Fan X, Larson C, Brent G, Zhou J (1997) Perinatal lethality with kidney and pancreas defects in mice with a targetted Pkd1 mutation. Nat Genet 17:179–181
Mochizuki T, Wu G, Hayashi T, Xenophontos SL, Veldhuisen B, Saris JJ, Reynolds DM, Cai Y, Gabow PA, Pierides A, Kimberling WJ, Breuning MH, Deltas CC, Peters DJ, Somlo S (1996) PKD2, a gene for polycystic kidney disease that encodes an integral membrane protein. Science 272:1339–1342
Ong AC, Ward CJ, Butler RJ, Biddolph S, Bowker C, Torra R, Pei Y, Harris PC (1999) Coordinate expression of the autosomal dominant polycystic kidney disease proteins, polycystin-2 and polycystin-1, in normal and cystic tissue. Am J Pathol 154:1721–1729
Ong AC, Wheatley DN (2003) Polycystic kidney disease—the ciliary connection. Lancet 361:774–776
Parnell SC, Magenheimer BS, Maser RL, Rankin CA, Smine A, Okamoto T, Calvet JP (1998) The polycystic kidney disease-1 protein, polycystin-1, binds and activates heterotrimeric G-proteins in vitro. Biochem Biophys Res Commun 251:625–631
Roitbak T, Ward CJ, Harris PC, Bacallao R, Ness SA, Wandinger-Ness A (2004) A polycystin-1 multiprotein complex is disrupted in polycystic kidney disease cells. Mol Biol Cell 15:1334–1346
Scheffers MS, Le H, van der Bent P, Leonhard W, Prins F, Spruit L, Breuning MH, de Heer E, Peters DJ (2002) Distinct subcellular expression of endogenous polycystin-2 in the plasma membrane and Golgi apparatus of MDCK cells. Hum Mol Genet 11:59–67
Scheffers MS, van der Bent P, Prins F, Spruit L, Breuning MH, Litvinov SV, de Heer E, Peters DJ (2000) Polycystin-1, the product of the polycystic kidney disease 1 gene, co-localizes with desmosomes in MDCK cells. Hum Mol Genet 9:2743–2750
Shannon MB, Miner JH (2004) Insertional mutation in laminin alpha 5: a new mouse model for polycystic kidney disease. In: Couser WG (ed) American society of nephrology. LW&W, St. Louis, MO, USA, vol. 15, p 652A
Streets AJ, Newby LJ, O’Hare MJ, Bukanov NO, Ibraghimov-Beskrovnaya O, Ong AC (2003) Functional analysis of PKD1 transgenic lines reveals a direct role for polycystin-1 in mediating cell–cell adhesion. J Am Soc Nephrol 14:1804–1815
Ward CJ, Turley H, Ong AC, Comley M, Biddolph S, Chetty R, Ratcliffe PJ, Gattner K, Harris PC (1996) Polycystin, the polycystic kidney disease 1 protein, is expressed by epithelial cells in fetal, adult, and polycystic kidney. Proc Natl Acad Sci USA 93:1524–1528
Wilson PD (2004a) Polycystic kidney disease N Engl J Med 350:151–164
Wilson PD (2004b) Polycystic kidney disease: new understanding in the pathogenesis. Int J Biochem Cell Biol 36:1868–1873
Xu GM, Sikaneta T, Sullivan BM, Zhang Q, Andreucci M, Stehle T, Drummond I, Arnaout MA (2001) Polycystin-1 interacts with intermediate filaments. J Biol Chem 276:46544–46552
Yamaguchi T, Pelling JC, Ramaswamy NT, Eppler JW, Wallace DP, Nagao S, Rome LA, Sullivan LP, Grantham JJ (2000) cAMP stimulates the in vitro proliferation of renal cyst epithelial cells by activating the extracellular signal-regulated kinase pathway. Kidney Int 57:1460–1471
Yoder BK, Hou X, Guay-Woodford LM (2002) The polycystic kidney disease proteins, polycystin-1, polycystin-2, polaris, and cystin, are co-localized in renal cilia. J Am Soc Nephrol 13:2508–2516
Acknowledgements
We are grateful to Drs. Kathy Klinger and Thomas Natoli for helpful discussions and comments on this manuscript. We thank AIRG and the GIS-Maladies rares Institute for grant support to BK.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Russo, R.J., Husson, H., Joly, D. et al. Impaired formation of desmosomal junctions in ADPKD epithelia. Histochem Cell Biol 124, 487–497 (2005). https://doi.org/10.1007/s00418-005-0055-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-005-0055-3