Abstract
Purpose
To evaluate the efficacy of navigated focal laser photocoagulation in patients with chronic central serous chorioretinopathy (CSCR) and active leakage on fluorescein angiography (FA).
Methods
Thirty-two eyes of 32 patients (age 48 ± 11, m/f = 24/8) with persistent or recurrent CSCR (> 3 months) who received navigated laser photocoagulation (Navilas®) of leaking point(s) between June 2013 and 2016 were included in this retrospective case series. Outcome parameters after 4 weeks and 3 months were the number of patients presenting with complete resolution of subretinal fluid, the volume of subretinal fluid measured on SD-OCT (Spectralis Heidelberg Engineering©), and best corrected visual acuity (BCVA/ (Snellen equivalent).
Results
Complete resolution of subretinal fluid was achieved in 17 eyes (50%) after 4 weeks and in 24 eyes (75%) after 3 months with an average number of 1.3 laser procedures (range 1–3). Five eyes displayed a nearly complete resolution with a reduction of over 80% of the subretinal fluid compared to baseline. Three eyes showed no reduction in subretinal fluid. BCVA improved from median 0.58 (range 0.16–1.25) to 0.66 (0.16–1.0) (p = 0.001). The seven patients who had been treated within the central 1 mm of the ETDRS-OCT Grid but outside the avascular foveal zone showed an improvement of BCVA from median 0.6 (range 0.2–1.0) to 0.8 (0.2–1.0). No patient experienced a treatment-induced visual loss.
Conclusions
Laser treatment with Navilas® using eye tracking and FA-based planning is a safe and effective alternative therapy in patients with chronic CSCR.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Central serous chorioretinopathy (CSCR) is a common disorder of the posterior pole with a circumscribed imbalance of the outer retina-blood barrier, the retinal pigment epithelium. It is characterized by accumulation of subretinal fluid from one or more focal leaking points demonstrating in fluorescein angiography.
Single acute CSCR with spontaneous regression has a good prognosis with respect to visual acuity. Chronic and recurrent CSCR, however, is one of the most common causes of permanent loss of visual acuity in middle-age adults, due to photoreceptor and retinal pigment epithelial atrophy [1].
The incidence is reported to be at 9.9 per 100,000 in men and 1.9 in women [2]. Although the pathogenesis of CSCR still remains to be determined, the dysregulation of choroidal blood flow has been associated to elevated serum glucocorticoid and mineralocorticoid levels, local and systemic steroid exposure, and psychopathological/emotional distress [2,3,4,5,6,7].
In patients with non-resolving, persistent, and recurrent CSCR, treatment options are sparse and remain ineffective or associated with side effects. Administration of carboanhydrase inhibitors or recently mineralocorticoid receptor antagonists is usually the first option, followed in case of ineffectiveness by focal laser photocoagulation of the leaking point(s) on RPE or photodynamic therapy [8,9,10,11]. Laser photocoagulation bears the risk of scar and CNV induction, when areas of leakage are treated immediately after fluorescein injection using the blue light filter of the laser slit lamp. To improve safety and predictability especially in the treatment of peri- and juxtafoveal lesions, the NAVILAS® laser system provides the option to plan the treatment beforehand directly on the early FA angiogram. The latter is imported to the laser device, superimposed and continuously aligned with the live image using an eye tracking system. In this retrospective case series, we examined the effect of navigated laser treatment with respect to different distances of focal leakage from the foveal center.
Methods
Patients characteristics
Thirty-two eyes of 32 patients (age 48 ± 11, m/f = 16/7) with chronic CSCR who received navigated laser photocoagulation of the leaking point(s) between June 2013 and 2016 were included in this retrospective study.
Definition of chronic CSCR
Chronic CSCR was defined as persistent or recurrent subretinal fluid involving the macula with an active leakage as demonstrated on fluorescein angiography; duration of more than 3 months; and loss of visual acuity, metamorphopsia, or reduced contrast sensitivity.
Patients with subfoveal (within the avascular zone) or diffuse angiographic leakage were excluded from this study. All patients had received a treatment with oral carboanhydrase inhibitors (acetacolamid 125 to 250 mg three times daily) and/or mineralocorticoid receptor antagonist (spironolactone 50 mg daily) for a minimum of 12 weeks without an effect on exudation during the current episode prior to laser treatment (Table 1).
Treatment
All patients received a comprehensive ophthalmic exam including measurement of best corrected visual acuity (BCVA), Goldmann tonometry, biomicroscopy of the anterior and posterior segment, Spectral Domain Ocular Coherence Tomography (SD-OCT) of the posterior pole, and fluorescein angiography (FA) (Heidelberg Spectralis, Heidelberg Engineering GmbH, Germany) (2,5 ml Na-Fluorescein 10% Alcon, Alcon Pharma GmbH, Freiburg, Germany). Well-defined leaking points were identified on the earliest images of the FA series.
Prior to laser photocoagulation using the NAVILAS® Laser system (532 nm double-pulsed YAG Laser, OD-OS GMBH, Teltow, Germany), an image of the posterior pole was captured. The selected FA image demonstrating the focal leakage was imported and automatically or manually superimposed on the actual image captured.
Two to five partially overlapping laser spots of 50–100 μm per leaking point were marked together with a protection shield for the fovea at the superimposed image. After adjusting the appropriate energy (50–70 mW/80–100 ms per spot) required to create a barely visible laser effect on fundus image by 1–2 test spots outside the macula, the intended laser spots were applied. The laser software provides an additional feedback window which displays the immediate laser effect captured at the end of the pulse.
We defined treatment failure as focal leakage of the same lesion visible on FA after the first procedure. Treatment was then repeated. Patients who developed new leaking point(s) on FA distant from previous lesion(s) received also an additional laser treatment but were considered as a second treatment of the same eye.
Follow up and analysis
Patients were examined at least 4 weeks (± 1 week) after the last treatment and followed until complete resolution of subretinal fluid was achieved or a decision for other treatment options in case of persistence or recurrences was made.
Follow up visit included a complete eye exam, SD-OCT of the macula and FA, when appropriate.
The following parameters were collected from records of the patients in this study: age, sex, BCVA (decimal), treatment modalities prior to laser treatment, duration of history and of current CSCR-episode, OCT central foveal thickness (distance between the internal and external limiting membranes), central retinal thickness (distance between internal limiting membrane and retinal pigment epithelium, including subretinal fluid on 6 mm standard SD-OCT ETDRS Grid), volume of subretinal fluid, number of leaking points, number of laser spots, and average laser energy applied. Outcome parameters were BCVA, the rate of complete resolution of subretinal fluid, the volume of subretinal fluid based on the morphological evaluation of SD-OCT B-scans of the posterior pole. Therefore, the subretinal fluid of each individual OCT B-scan was outlined to obtain its area (Draw-Region-Function of the Heidelberg Spectralis Eye Explorer V. 1.9.10.0). Taken together with the number and distance between the OCT B-scans (from image information of the Eye Explorer), the numerical integral was calculated corresponding to the volume of the subretinal fluid (see equation):
with V = volume, n = number of OCT B-scans, d = distance between OCT B-scans, A = area of subretinal fluid within single OCT B-scan.
Statistics
Data are reported as average, medians, IQRs or as absolute numbers and percentages. Comparison between groups was done using the Wilcoxon and Mann-Whitney U-test as appropriate. Spearman rank correlation coefficient (RS)/Pearson’s correlation was calculated to investigate the relationship between BCVA and central retinal thickness, localization of leaking points, number of focal spots, number of applied laser spots, volume of subretinal fluid, and duration of CSCR history.
Statistical analysis was performed using IBM-SPSS Statistics Version 22. A p value less than 0.05 was considered statistically significant.
Results
Patients’ characteristics are shown in Table 1.
Thirty-two eyes of 32 patients with persistent/non-resolving or recurrent CSCR received 41 navigated focal laser photocoagulations and were followed up for a median period of 9.5 weeks (range 4–52).
A complete resolution of subretinal fluid (primary endpoint) was achieved in 17 eyes (50%) after 4 weeks and in 24 eyes (75%) after 3 months with an average number of 1.3 (range 1–3) laser procedures (Fig. 1; Table 2).
The remaining five eyes displayed a nearly complete resolution of more than 80% of the subretinal fluid compared to baseline. Three eyes showed no reduction in subretinal fluid at all.
Visual acuity improved significantly parallel to the reduction of subretinal fluid (Table 2). The seven patients who had been treated within the central 1 mm of the ETDRS-OCT Grid but outside the avascular foveal zone showed an improvement of BCVA from median 0.6 (range 0.2–1.0) to 0.8 (0.2–1.0) (Fig. 1) In seven eyes, nine laser treatments had to be repeated. In two eyes, spot size or energy applied was too low and required a repeated laser treatment which resulted in a complete resolution of subretinal fluid. Five eyes developed within the observational period additional focal leakage. In four of them, recurrence was detected at the margins of an area with pigment epithelial detachment but outside the previous treated area. Retreatment resulted in complete resolution of subretinal fluid in three eyes, incomplete in one and with no response in the remaining.
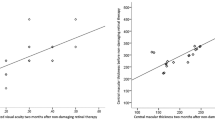
Preoperative central foveal thickness was correlated with an increase of visual acuity after successful treatment (0.555/p = 0.001). Localization and number of leaking spots, number of applied laser spots, volume of subretinal fluid, and overall and current duration of CSCR did not show any correlation to BCVA.
Examples of different treatment scenarios are shown in central focal leakage (Fig. 2), multiple focal points (Fig. 3), and management of recurrent focal leakage (Fig. 4).
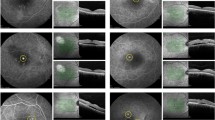
Case presentation of central lesion: female 53 years, CSCR for 48 months, active episode for 5 months, previous treatments with acetacolamide/spironolactone without effect. a Baseline SD-OCT/FA early/late phase with focal leakage within PED (BCVA 0.4). b Navilas laser photocoagulation: superimposed FA image, treatment/ parameters. c SD-OCT follow up 4 weeks (BCVA 0.5). d SD-OCT follow up 12 weeks with FA (BCVA 0.5)
Case presentation of multiple lesions: male 56 years, multifocal CSCR for 36 months, active episode for 5 months, previous treatment with acetacolamide without effect. a Baseline SD-OCT/FA early/late phase with multifocal leakage (BCVA 0.6). b Navilas laser photocoagulation: superimposed FA image, treatment/ parameters. c SD-OCT follow up 4 weeks (BCVA 0.8). d SD-OCT follow up 12 weeks with FA (BCVA 0.8)
Case presentation with recurrent leakage and multiple treatments: male 44 years, active CSCR for 3 months, previous treatment with acetacolamide without effect. a Baseline SD-OCT/FA early/late phase with focal leakage and serous pigmentepithelial detachment (sPED) (BCVA 0.5). b Navilas laser photocoagulation: superimposed FA image, treatment/parameters. c SD-OCT follow up 4 weeks (BCVA 0.7). d SD-OCT follow up 12 weeks with FA new focal leakage below previous spots (BCVA 0.5), retreatment with Navilas laser photocoagulation: superimposed FA image, treatment/parameters. e SD-OCT follow up 8 weeks with FA (BCVA 0.5) recurrence on previous focal spot, third retreatment with Navilas laser photocoagulation: superimposed FA image, treatment/parameters. f SD-OCT follow up 4 weeks complete resolution of subretinal fluid (BCVA 1.0)
Discussion
Recurrent or persistent subretinal fluid in CSCR leads to photoreceptor and retinal pigment epithelial atrophy with consecutive loss of contrast sensitivity and visual acuity. Focal leakage is often self-limiting and spontaneous resolution occurs within a few weeks. In persistent cases, focal laser treatment is one option to seal the leaking spot(s) [12,13,14].
Conventionally, the laser procedure is either done by memorizing the focal spot from FA images or immediately after injecting fluorescein to visualize the focal leak using the blue light filter of the laser slit lamp. Fluorescence and the commonly used 514 Argon-/532 nm Nd-YAG laser may interfere making the localization and energy distribution especially in high flow spots within the posterior pole less reliable [12, 15]. With the introduction of computer-assisted image recognition, eye alignment, and tracking software into new laser devices, we would expect to improve accuracy and reproducibility in the treatment of retinal and macular disease as, e.g., in focal spots in CSCR particularly regarding central lesions. The Navilas laser enables to plan the treatment on imported FA images beforehand, guide and monitor the laser application. In focal treatment of diabetic macular edema, 76% of the applied laser spots were visible 1 month postoperatively and 96% of these were within the intended area of 100 μm spot diameter [16].
Subretinal fluid resolution after laser photocoagulation in general lasts from a few weeks to 6 months [13, 15]. Rate of success in terms of complete subretinal fluid resolution differs. Chhalabani et al. reported a complete resolution after 2 months in 15 of 16 eyes with acute multifocal CSCR after applying a single shot per spot (70–100 μm, 30 ms with a barely visible grayish burn) using the Navilas laser [17]. Similar results were reported by Mastropasqua et al. in 32 previously untreated eyes after 6 months with an average of 3.2 burns per spot with the Navilas laser [18]. We used a similar approach in this case series to cover the leaking spot with an average of 3.5 laser spots being more restrictive in central lesions in terms of number, diameter, and energy per laser spot applied (Table 1). An unintended treatment other than the focal spot had not been observed.
The adjustment of fluence to a barely visible test spot of an uninvolved area outside the macula prior to treatment may have led to an underestimation of the laser energy/subthreshold effect that could explain the treatment failure in two cases of this series who required a second successful treatment. Chronic CSCR often displays with pigmentary changes of the RPE where single and potentially subthreshold spots are difficult to distinguish afterwards.
Five eyes developed during the observational period a recurrence close to the previously treated area. OCT analysis disclosed an elevated irregular RPE layer or serous pigment epithelial detachment in this region. Whether laser photocoagulation and the consecutive tissue response has provoked the new leakage remains unclear. On the other hand, similar RPE elevation in the leaking area was also detected in about half of all the treated cases in this series which is considered a pathological feature of CSCR.
Direct thermal photocoagulation of the leaking spot or area of RPE dysfunction creates a response of the neighbored RPE cells that cover the coagulated area [12, 19]. However, outer retinal segment damage and the risk of CNV development by interrupting Bruch’s membrane limit the application of threshold laser spots in foveal or juxtafoveal lesions. In contrast, subthreshold laser application confines the heating distribution within the RPE layer. Multimodal imaging and histopathological and metabolic studies in animals forms the basis of the current understanding of dose-depending subthreshold damaging laser (where the RPE cell is selectively destroyed) and subthreshold non-damaging laser (where intracellular regenerative processes are induced) [20,21,22]. As a result, the Endpoint Management algorithm (EpM) for 577 nm laser was developed using the Pascal Streamline Laser (Topcon Medical Laser Systems, Santa Clara CA, USA), setting the energy of a barely visible laser spot outside the treatment area to 100% and enabling a titration to subthreshold/but damaging (50%) and to non-damaging (30%) laser effects [20,21,22].
Wood et al. summarized recently available data in 398 patients of 16 studies on subthreshold retinal laser therapy on CSCR using various laser modalities from 532 nm continuous wave, 577 nm cw and micropulse to 810 nm micropulse lasers and found that non-damaging retinal laser efficiently reduces central macular thickness and improves visual acuity [14].
The main goal of laser treatment in CSCR is a complete resolution of subretinal fluid. This was achieved in 75% of patients with an average of 1.3 (1–3) treatment sessions in 3 months.
In the literature, treatment results regarding subthreshold laser are somewhat heterogeneous in terms of laser and outcome parameters, number of patients, and follow up time. Rates of complete remission and re-treatments have been reported for subthreshold laser (577 nm/810 nm micropulse) ranging from 40 to 87% requiring multiple treatment sessions (average 2, range 1–4) [21, 23,24,25,26].
Compared to threshold laser, the major advantage of non-damaging laser is enhanced cellular regeneration and the option to repeat the treatment within the same treatment area in case of initial ineffectiveness or recurrences compared. Further standardization and randomized prospective trails are therefore required.
The limitation of this series is its retrospective design and a lack of a control group. Although the immediate laser effect on subretinal fluid resolution was detectable with OCT, the direct coagulation effect was not reliably evaluable with the used OCT imaging technique. To study the impact of laser photocoagulation in central lesions would require further microperimetric studies with a longer follow up.
Furthermore, navigated laser therapy has to be investigated against low-dose photodynamic therapy, which has been recently shown to be equally effective compared to subthreshold/graded infrared laser treatment in longer-term follow up of 6 months but with a faster rate of regression [27, 28].
In conclusion, navigated laser photocoagulation in patients with chronic non-resolving CSCR and well-defined active leakage on FA represents a reliable treatment option to seal the source of exudation and permit the resolution of subretinal fluid in 75% of the cases. Particularly, patients with lesions of the foveal center but outside of the avascular zone who were resistant to previous conservative treatments could benefit from navigated laser treatment. In cases with leaking points within areas of pigment epithelial detachment, recurrences may occur potentially requiring multiple treatment sessions. Further studies in larger numbers of patients and with longer follow up are necessary to investigate efficacy and side effects of threshold/subthreshold laser treatments in combination with navigated laser spot application in this heterogeneous group of patients to improve effectivity, safety, and reproducibility.
References
Daruich A, Matet A, Dirani A, Bousquet E, Zhao M, Farman N, Jaisser F, Behar-Cohen F (2015) Central serous chorioretinopathy: recent findings and new physiopathology hypothesis. Prog Retin Eye Res 48:82–118. https://doi.org/10.1016/j.preteyeres.2015.05.003
Kitzmann AS, Pulido JS, Diehl NN, Hodge DO, Burke JP (2008) The incidence of central serous chorioretinopathy in Olmsted County, Minnesota, 1980-2002. Ophthalmology 115:169–173. https://doi.org/10.1016/j.ophtha.2007.02.032
Tewari HK, Gadia R, Kumar D, Venkatesh P, Garg SP (2006) Sympathetic-parasympathetic activity and reactivity in central serous chorioretinopathy: a case-control study. Invest Ophthalmol Vis Sci 47:3474–3478. https://doi.org/10.1167/iovs.05-1246
Conrad R, Geiser F, Kleiman A, Zur B, Karpawitz-Godt A (2014) Temperament and character personality profile and illness-related stress in central serous chorioretinopathy. TheScientificWorldJournal 2014:631687. https://doi.org/10.1155/2014/631687
Zakir SM, Shukla M, Simi ZU, Ahmad J, Sajid M (2009) Serum cortisol and testosterone levels in idiopathic central serous chorioretinopathy. Indian J Ophthalmol 57:419–422. https://doi.org/10.4103/0301-4738.57143
Haimovici R, Rumelt S, Melby J (2003) Endocrine abnormalities in patients with central serous chorioretinopathy. Ophthalmology 110:698–703. https://doi.org/10.1016/S0161-6420(02)01975-9
Khairallah M, Kahloun R, Tugal-Tutkun I (2012) Central serous chorioretinopathy, corticosteroids, and uveitis. Ocul Immunol Inflamm 20:76–85. https://doi.org/10.3109/09273948.2011.650776
Nicolo M, Eandi CM, Alovisi C, Grignolo FM, Traverso CE, Musetti D, Cardillo Piccolino F (2014) Half-fluence versus half-dose photodynamic therapy in chronic central serous chorioretinopathy. Am J Ophthalmol 157:1033–1037. https://doi.org/10.1016/j.ajo.2014.01.022
Bousquet E, Beydoun T, Rothschild PR, Bergin C, Zhao M, Batista R, Brandely ML, Couraud B, Farman N, Gaudric A, Chast F, Behar-Cohen F (2015) Spironolactone for nonresolving central serous chorioretinopathy: a randomized controlled crossover study. Retina 35:2505–2515. https://doi.org/10.1097/IAE.0000000000000614
Nicholson B, Noble J, Forooghian F, Meyerle C (2013) Central serous chorioretinopathy: update on pathophysiology and treatment. Surv Ophthalmol 58:103–126. https://doi.org/10.1016/j.survophthal.2012.07.004
Rubsam A, Thieme CE, Schlomberg J, Winterhalter S, Muller B, Joussen AM, Stubiger N (2017) Therapy rationale for mineralocorticoid-receptor antagonists, acetazolamide and a switch of therapy in nonresponders in central serous chorioretinopathy. J Ocul Pharmacol Ther 33:141–148. https://doi.org/10.1089/jop.2016.0068
Robertson DM, Ilstrup D (1983) Direct, indirect, and sham laser photocoagulation in the management of central serous chorioretinopathy. Am J Ophthalmol 95:457–466
Lim JW, Kang SW, Kim YT, Chung SE, Lee SW (2011) Comparative study of patients with central serous chorioretinopathy undergoing focal laser photocoagulation or photodynamic therapy. Br J Ophthalmol 95:514–517. https://doi.org/10.1136/bjo.2010.182121
Wood EH, Karth PA, Sanislo SR, Moshfeghi DM, Palanker DV (2017) Nondamaging retinal laser therapy for treatment of central serous chorioretinopathy: what is the evidence? Retina 37:1021–1033. https://doi.org/10.1097/iae.0000000000001386
Verma L, Sinha R, Venkatesh P, Tewari HK (2004) Comparative evaluation of diode laser versus argon laser photocoagulation in patients with central serous retinopathy: a pilot, randomized controlled trial [ISRCTN84128484]. BMC Ophthalmol 4:15. https://doi.org/10.1186/1471-2415-4-15
Kernt M, Cheuteu R, Liegl RG, Seidensticker F, Cserhati S, Hirneiss C, Haritoglou C, Kampik A, Ulbig M, Neubauer AS (2012) Navigated focal retinal laser therapy using the NAVILAS(R) system for diabetic macula edema. Ophthalmologe 109:692–698. https://doi.org/10.1007/s00347-012-2559-2
Chhablani J, Rani PK, Mathai A, Jalali S, Kozak I (2014) Navigated focal laser photocoagulation for central serous chorioretinopathy. Clin Ophthalmol (Auckland, NZ) 8:1543–1547. https://doi.org/10.2147/opth.s67025
Mastropasqua L, Di Antonio L, Toto L, Mastropasqua A, Di Iorio A, Carpineto P (2015) Central serous chorioretinopathy treated with navigated retinal laser photocoagulation: visual acuity and retinal sensitivity. Ophthalmic Surg Lasers Imaging 46:349–354. https://doi.org/10.3928/23258160-20150323-09
Sramek C, Mackanos M, Spitler R, Leung LS, Nomoto H, Contag CH, Palanker D (2011) Non-damaging retinal phototherapy: dynamic range of heat shock protein expression. Invest Ophthalmol Vis Sci 52:1780–1787. https://doi.org/10.1167/iovs.10-5917
Schuele G, Rumohr M, Huettmann G, Brinkmann R (2005) RPE damage thresholds and mechanisms for laser exposure in the microsecond-to-millisecond time regimen. Invest Ophthalmol Vis Sci 46:714–719. https://doi.org/10.1167/iovs.04-0136
Lavinsky D, Wang J, Huie P, Dalal R, Lee SJ, Lee DY, Palanker D (2016) Nondamaging retinal laser therapy: rationale and applications to the macula. Invest Ophthalmol Vis Sci 57:2488–2500. https://doi.org/10.1167/iovs.15-18981
Lavinsky D, Sramek C, Wang J, Huie P, Dalal R, Mandel Y, Palanker D (2014) Subvisible retinal laser therapy: titration algorithm and tissue response. Retina 34:87–97. https://doi.org/10.1097/IAE.0b013e3182993edc
Chen SN, Hwang JF, Tseng LF, Lin CJ (2008) Subthreshold diode micropulse photocoagulation for the treatment of chronic central serous chorioretinopathy with juxtafoveal leakage. Ophthalmology 115:2229–2234. https://doi.org/10.1016/j.ophtha.2008.08.026
Koss MJ, Beger I, Koch FH (2012) Subthreshold diode laser micropulse photocoagulation versus intravitreal injections of bevacizumab in the treatment of central serous chorioretinopathy. Eye (London, England) 26:307–314. https://doi.org/10.1038/eye.2011.282
Lanzetta P, Furlan F, Morgante L, Veritti D, Bandello F (2008) Nonvisible subthreshold micropulse diode laser (810 nm) treatment of central serous chorioretinopathy. A pilot study. Eur J Ophthalmol 18:934–940
Yadav NK, Jayadev C, Mohan A, Vijayan P, Battu R, Dabir S, Shetty B, Shetty R (2015) Subthreshold micropulse yellow laser (577 nm) in chronic central serous chorioretinopathy: safety profile and treatment outcome. Eye (London, England) 29:258–264; quiz 265. https://doi.org/10.1038/eye.2014.315
Russo A, Turano R, Morescalchi F, Gambicorti E, Cancarini A, Duse S, Costagliola C, Semeraro F (2017) Comparison of half-dose photodynamic therapy and 689 nm laser treatment in eyes with chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol 255:1141–1148. https://doi.org/10.1007/s00417-017-3626-9
Manayath GJ, Karandikar SS, Narendran S, Kumarswamy KA, Saravanan VR, Morris RJ, Venkatapathy N (2017) Low fluence photodynamic therapy versus graded subthreshold transpupillary thermotherapy for chronic central serous chorioretinopathy: results from a prospective study. Ophthalmic Surg Lasers Imaging 48:334–338. https://doi.org/10.3928/23258160-20170329-08
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Müller, B., Tatsios, J., Klonner, J. et al. Navigated laser photocoagulation in patients with non-resolving and chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol 256, 1581–1588 (2018). https://doi.org/10.1007/s00417-018-4031-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-018-4031-8