Abstract
Purpose
To analyze the influence of spectral-domain optical coherence tomography (SD-OCT) features on visual acuity changes in patients with idiopathic epiretinal membranes (ERMs).
Methods
Seventy-nine eyes of 71 patients were included in this study. SD-OCT was performed for all patients; data were collected upon ERM diagnosis and at the final visit. The patients were divided into subgroups based on their SD-OCT features. The initial best corrected visual acuity (BCVA) and changes in BCVA for each subgroup were compared. A multivariate analysis was performed to assess the factors associated with changes in BCVA.
Results
During a mean follow-up period of 20.78 months, the mean change in logMAR visual acuity was 0.052 ± 0.089. Eyes with inner segment/outer segment (IS/OS) junction disruption and cystoid macular edema (CME) had a significantly lower mean initial BCVA than those without disruption and CME (P = 0.036 and P = 0.012, respectively). However, only eyes with CME had significant changes in BCVA (P = .034). Multivariate analysis revealed the presence of CME as the only factor that had a significant correlation with VA changes.
Conclusions
In patients with idiopathic ERMs, the presence of CME and IS/OS disruption detected by OCT correlated with a poorer initial BCVA. Most patients’ visual acuity remained stable during follow-up. The presence of CME with OCT represented a predictor of the progression of visual acuity. These results may provide valuable clinical information regarding the management of patients with idiopathic ERMs.
Specific note on abstract
We demonstrated that the presence of CME and IS/OS disruption detected with OCT correlated with a poorer BCVA in idiopathic ERMs. The visual acuity of most patients was stable during the follow-up period. The presence of CME in OCT represented a predictor of vision deterioration for patients with idiopathic ERMs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
An epiretinal membrane (ERM) is a retinal disease in which the vitreomacular interface induces a tangential tractional force on the retina, leading to deformation of the retinal architecture [1]. Epidemiological studies showed that ERM is a common retinal disease, with a prevalence rate ranging from 2.2 to 18.5 % among different ethnic groups [2–5]. It has been associated with a variety of ocular diseases such as diabetic retinopathy and retinal vein occlusion, or secondary to inflammation and retinal surgery [6–8]. However, most cases without antecedent ocular pathology are termed idiopathic ERMs.
The clinical course of an ERM may be stable or progress slowly. The initial presentation of idiopathic ERM formation does not usually cause any clinically important reduction in vision; however, in some cases, this disease will progress, leading to a significant reduction in visual acuity, necessitating surgical intervention [9, 10]. Previous studies have reported the longitudinal analysis of visual acuity and OCT features in ERM patients with uveitis [11, 12]. However, to our knowledge, no data have evaluated the natural course of idiopathic ERMs and whether morphological OCT findings have influence on the visual acuity changes in idiopathic ERMs.
Spectral-domain OCT (SD-OCT) is a non-invasive imaging technique that provides high-resolution cross-section images of the retinal structure, and is particularly useful in examining the macula [13]. For ERMs, SD-OCT facilitates the visualization of not only retinal thickness, but also the pathologic features of the ERM and associated retinal changes [14–16]. Furthermore, SD-OCT is a sensitive approach for the detection of an ERM and has become an integral part of the assessment and follow-up for ERMs [15, 17, 18].
In this study, we first analyzed the influence of the morphological OCT findings and central subfoveal thickness (CST) on visual acuity. Furthermore, we evaluated the influence of the morphological OCT findings on visual acuity changes during follow-up in patients with idiopathic ERMs.
Materials and methods
We conducted a retrospective chart review of 133 consecutive patients who were diagnosed with idiopathic ERM using SD-OCT from Jan 2, 2011 to June 30, 2014 in the Department of Ophthalmology at Taipei City Hospital Zhongxiao Branch. Patients were excluded from this study for the following: eyes with secondary ERM (diabetic retinopathy, venous occlusion, retinal tear, retinal detachment, uveitis, trauma, etc.); eyes with myopia of >6 diopters; and eyes with other ocular pathologic features that could have interfered with functional results, such as glaucoma, visually significant cataract, or age-related macular degeneration. In addition, patients with follow-up periods of <6 months were also excluded from this study.
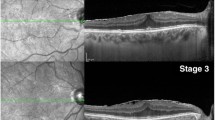
All patients underwent thorough ophthalmologic examinations including best-corrected visual acuity (BCVA; Snellen visual acuity chart), biomicroscopy of the anterior and posterior segments, indirect ophthalmoscopy, fundus photography, and SD-OCT (RTVue, Optovue Inc, Fremont, USA) that were performed at diagnosis and at each follow-up visit. Patients with poor SD-OCT images that prevented the evaluation and quantification of the SD-OCT data were excluded. The eyes were divided into OCT morphological subgroups according to whether the photoreceptor IS/OS junction was disrupted or intact (Fig. 1a and b), presence or absence of macular edema (Fig. 1c and d), presence or loss of foveal concavity (Fig. 1e and f), or whether the external limiting membranes (Fig. 1g and h) were disrupted or intact [19]. The OCT patterns were interpreted by two independent retinal specialists (F.I.M. and H.C.C.). To analyze central retinal thickness, an “MM5” grid-scanning mode was used. In this mode, 17 horizontal line scans and 17 vertical line scans were performed with each set of scans including 11 lines with a 5-mm scan length and 6 lines with a 3-mm scan length. After scanning in MM5 mode, the RTVue-100 device automatically calculates the mean retinal thickness at the diameter (1, 3, or 5 mm) of the macular area. Central subfoveal thickness (CST) is the mean retinal thickness in the 1-mm diameter circle centered on the fovea.
Spectral-domain optical coherence tomography (SD-OCT) features of patients with an epiretinal membrane (ERM). a Disruption of the inner segment and outer segment (IS/OS) junction (arrow); b integrity of the IS/OS junction; c Presence of cystoid macular edema (CME); d absence of CME; e Disruption of the external limiting membrane (ELM); f integrity of the ELM; g Loss of foveal concavity; h presence of foveal concavity
The initial BCVA, final BCVA, BCVA changes, initial CST, final CST, CST changes, and follow-up periods for each OCT morphological subgroup were compared. A multivariate analysis was used to evaluate the relationship between initial BCVA or BCVA changes and other parameters including sex, age, and OCT morphology.
Statistical analysis
Descriptive and comparative statistics were performed using SPSS version 10.0 (SPSS Inc., Chicago, IL, USA). Visual acuity measurements with Snellen charts were converted to a logarithm of the minimal angle of resolution (log MAR) for statistical analysis. Continuous variables were compared using a student’s t test. Categorical variables were compared using Pearson’s chi-square test. P values < 0.05 were considered statistically significant.
Results
In this study, 79 eyes of 71 consecutive patients with an SD-OCT–supported ERM diagnosis were included. The average patient age at the time of data collection was 65.1 ± 9.66 years (range, 42 to 84 years); 33 patients (46.48 %) were female. For 8 patients, both eyes had a documented ERM and were included in the study. The OCT morphological subgroup had the following: 73 eyes were intact and 6 eyes had a disrupted IS/OS junction; 23 eyes had CME presence and 56 had CME absence; 74 eyes had an intact ELM and 5 eyes had a disrupted ELM; and 25 eyes had a normal foveal contour and 54 had a decreased foveal contour. The demographic data and clinical characteristics for all patients and each subgroup are shown in Tables 1 and 2. The mean follow-up period was 20.78 months (range, 7 to 46 months), and the mean initial visual acuity was 0.39 ± 0.32. The mean change in visual acuity was 0.052 ± 0.089. Scatter plots of the initial and final visual acuities for all patients and each subgroup are shown in Fig. 2.
Scatter plots of initial visual acuity with final visual acuity in: a all patients; b subgroup of patients with a disrupted or intact photoreceptor IS/OS junction; c subgroup with the presence or absence of macular edema; d subgroup with a disrupted or intact external limiting membrane; and e subgroup with the loss or presence of a foveal concavity
The visual acuity and CST during follow-up for each subgroup are shown in Table 3. The mean initial visual acuity was 0.54 ± 0.38 for patients with CME, whereas the mean initial visual acuity was 0.31 ± 0.27 for patients without CME. ERM patients with CME had statistically significant poorer visual acuity upon diagnosis than those without CME (P = 0.034). No significant difference in mean follow-up period was noted between the 2 subgroups (21.09 ± 10.11 for patients with CME versus 20.82 ± 9.65 for patients without CME; P = 0.91). The mean final visual acuity was significantly different between patients with and without CME (0.61 ± 0.38 for patients with CME versus 0.33 ± 0.27 for patients without CME; P = 0.036). Similarly, we also found statistically significant differences in the mean BCVA change between the 2 subgroups during follow-up (0.068 ± 0.011 for patients with CME versus 0.017 ± 0.089 for patients without CME; P = 0.034).
The mean initial visual acuity was 0.64 ± 0.21 for patients with IS/OS disruption and 0.36 ± 0.32 in patients with an intact IS/OS junction. There was a significant difference in mean initial visual acuity between patients with and without IS/OS disruption (P = 0.036). The mean follow-up period was not significantly different between the 2 subgroups (20.58 ± 9.46 for patients with an intact IS/OS junction versus 24.83 ± 12.89 for patients with IS/OS disruption; P = 0.31). The mean final visual acuity was significantly different between patients with an intact and disrupted IS/OS junction (0.39 ± 0.33 for patients with an intact IS/OS junction versus 0.69 ± 0.17 for patients with IS/OS disruption; P = 0.028). However, there were no significant differences in the mean BCVA change between the 2 subgroups during follow-up (0.03 ± 0.097 for patients with an intact IS/OS junction versus 0.05 ± 0.12 for patients with IS/OS disruption; P = 0.63).
As for an intact or disrupted ELM, in the foveal subgroups, there were no significant differences in the mean initial BCVA, final BCVA, change in BCVA, or follow-up periods between the two subgroups. Similarity, there were no significant differences in mean initial BCVA, final BCVA, change in BCVA, or follow-up periods between the normal and decreased foveal contour subgroups.
The mean initial CST was 401.70 ± 84.65 for patients with CME and 332.85 ± 66.34 for patients without CME. There was a significant difference in the mean initial CST between patients with and without CME (P = 0.001). The mean final CST was significantly different between patients with and without CME (405.91 ± 80.86 for patients with CME versus 338.95 ± 68.63 for patients without CME; P = 0.001). However, there were no significant differences in the mean CST change between the 2 subgroups during follow-up (4.22 ± 14.66 for patients with CME versus 6.05 ± 22.28 for patients without CME; P = 0.72).
The mean initial CST was 376.09 ± 75.98 for patients with a decreased foveal contour, whereas the mean initial CST was 302.88 ± 57.48 for patients with a normal foveal contour. The ERM patients with a decreased foveal contour had a statistically significant increase in CST upon diagnosis, as compared with those patients who had a normal foveal contour (P = 0.001). The mean final CST was significantly different between patients with decreased and normal foveal contours (383.39 ± 73.36 for patients with decreased foveal contours versus 304.56 ± 59.35 for patients with normal foveal contours; P = 0.001). However, there were no significant differences in the mean CST change between the 2 subgroups during follow-up (7.30 ± 22.49 for patients with decreased foveal contours versus 1.68 ± 14.05 for patients with normal foveal contours; P = 0.26).
There were no significant differences in the mean initial CST, final CST, change in CST, and the follow-up periods between subgroups with an intact ELM and a disrupted ELM. Further, there were no significant differences in mean initial CST, final CST, and change in CST between the normal and decreased foveal contour subgroups.
A multivariate analysis using a backward stepwise selection considering all variables revealed the presence of CME as the only factor with a significant correlation with a visual acuity change during the follow-up period (Table 4).
Discussion
In this study, we demonstrated that the visual acuity of ERM patients is relatively stable with a mean change of 0.034 during a mean follow-up of 20.78 months. In addition, disruption of the IS/OS junction and the presence of CME are factors associated with poorer initial vision for patients with idiopathic ERMs. However, the presence of CME was the only factor that had a significant correlation with a change in visual acuity during the follow-up period. As progressive deterioration in visual acuity is the principle indication and as the preoperative visual acuity is a significant prognostic factor for ERM surgery, our findings implied that early surgery should be considered for ERM patients with the presence of CME detected by OCT.
The influence of the central retinal thickness on visual acuity for patients with ERM is still controversial [9, 20–22]. Michalewski et al. found a negative correlation between foveal thickness and visual acuity [23]. In contrast, Suzuki et al. found no relationship between foveal thickness and visual acuity for patients with an ERM and pseudoholes [24]. In this study, we showed that eyes with a disrupted IS/OS junction tended to have a poorer initial visual acuity than those without disruption. However, the central foveal thickness was not significant between the two groups. Similarity, although central foveal thickness was significantly thicker in eyes with a decreased foveal contour than those with a normal one, no difference was noted between the two groups. Considering these ideas, our findings indicated that OCT morphological features are more important than central retinal thickness regarding the influence of visual acuity. Several mechanisms have been proposed to account for ERM-induced retinal damage such as direct damage to the neurons in the inner retinal layers, reduced perfusion of the macular capillaries, and photoreceptor impairment [25–27]. Therefore, it is reasonable that visual acuity could not be only attributable to central macular thickness for patients with idiopathic ERMs.
The natural course of an ERM is generally good with the preservation of visual acuity. Wise demonstrated that the majority of ERM patients have a benign natural course that leads to a visual acuity of ≤0.1 in <5 % of patients [28]. Nazari et al. reported that the visual acuity of eyes with ERM and uveitis remains stable over 1 to 2 years of follow-up [11]. In this study, we also showed that the visual acuity in idiopathic ERMs was relative stable, with a mean change in logMAR acuity of 0.052 ± 0.089 during a mean follow-up of 20.78 months. However, subgroup and multivariate analyses showed that the presence of CME detected by OCT is a predictor of visual acuity progression during the follow-up period. Our finding is similar to Poliner et al. who demonstrated that the presence of CME was significantly related to a poor visual outcome for ERM patients receiving pars plana vitrectomy [29]. Several studies indicated that the presence of CME in OCT was found to be associated with the deterioration of visual acuity in several eye diseases such as diabetic retinopathy and uveitis [30, 31]. Kim et al. showed that the mere presence of CME corresponded to a mean reduction in logMAR acuity of 0.40, as compared with 0.16 for other patients with diabetic retinopathy [32]. Tanikawa et al. demonstrated that the focal macular electroretinograms for eyes with ERM were similar to those of eyes with CME [33]. They suggested that the ERM probably caused damage to the neurons in the inner retinal layers and that dysfunction of the retinal neurons played an important role in reducing vision. A recent study analyzing the impact of OCT on surgical decision-making for ERMs demonstrated that the presence of macular edema influenced surgeons to recommend patients with ERMs to undergo surgical intervention [34]. Our results supported this notion and provided evidence by showing that the presence of CME detected by OCT is a predictor of visual deterioration in ERMs.
There were several limitations in this study. Because of the retrospective study design, the follow-up periods were variable, which may influence the final visual acuity and change in visual acuity. However, no statistically significant differences in mean follow-up period were noted among all the subgroups, which may have reduced the confounding of this factor. Second, as idiopathic ERM is a slow progressive disease, the follow-up period in this study was too short. The patients were followed from 7 to 46 months, with a mean of 20.78 months. Third, the sample size was relatively small. In this study, 73 eyes were intact while only 6 eyes had a disrupted IS/OS junction. Results should be interpreted with caution due to the limited number of patients with IS/OS disruption. Further prospective study with regular and longer-term follow-up periods and large sample sizes are necessary to clarify the issue.
In conclusion, our results showed that the presence of CME and IS/OS disruption detected with OCT correlated with a poorer BCVA in idiopathic ERMs. The visual acuity of most patients was relative stable during the follow-up period. The presence of CME in OCT represented a predictor of vision deterioration for patients with idiopathic ERMs. These results may provide valuable clinical information for the management of patients with idiopathic ERMs.
References
Snead DR, James S, Snead MP (2008) Pathological changes in the vitreoretinal junction I: epiretinal membrane formation. Eye 22:1310–1317
Zhu XF, Peng JJ, Zou HD et al (2012) Prevalence and risk factors of idiopathic epiretinal membranes in Beixinjing blocks, Shanghai, China. PLoS One 7(12), e51445
McCarty DJ, Mukesh BN, Chikani V et al (2005) Prevalence and associations of epiretinal membranes in the visual impairment project. Am J Opthalmol 140:288–294
Fraser-Bell S, Ying-Lai M, Klein R, Varma R, Los Angeles Latino Eye Study (2004) Prevalence and associations of epiretinal membranes in Latinos: the Los Angeles Latino Eye Study. Invest Ophthalmol Vis Sci 45:1732–1736
Ng CH, Cheung N, Wang JJ et al (2011) Prevalence and risk factors for epiretinal membranes in a multiethnic United States population. Ophthalmology 118(4):694–699
Appiah AP, Hirose T (1989) Secondary causes of premacular fibrosis. Ophthalmology 96:389–392
Kraushar MF, Morse PH (1988) The relationship between retina surgery and preretinal macular fibrosis. Ophthalmic Surg 19:843–848
Jahn CE, Minich V, Moldaschel S et al (2001) Epiretinal membranes after extracapsular cataract surgery. J Cataract Refract Surg 27:753–760
Mori K, Gehlbach PL, Sano A et al (2004) Comparison of epiretinal membranes of differing pathogenesis using optical coherence tomography. Retina 24:57–62
Falkner-Radler CI, Glittenberg C, Hagen S et al (2010) Spectral-domain optical coherence tomography for monitoring epiretinal membrane surgery. Ophthalmology 117(4):798–805
Nazari H, Dustin L, Heussen FM et al (2012) Morphometric spectal-domain optical coherent tomography features of epiretinal membrane correlated with visual acuity in patients with uveitis. Am J Ophthalmol 154:78–86
Nazari H, Rao N (2013) Longitudinal morphometric analysis of epiretinal membrane in patients with uveitis. Ocul Immunol Inflamm 21:2–7
Chen TC, Cense B, Pierce MC et al (2005) Spectral domain optical coherence tomography: ultra-high speed, ultra-high resolutionophthalmic imaging. Arch Ophthalmol 123:1715–1720
Niwa T, Terasaki H, Kondo M et al (2003) Function and morphology of macula before and after removal of idiopathic epiretinal membrane. Invest Ophthalmol Vis Sci 44:1652–1656
Wilkins JR, Puliafito CA, Hee MR et al (1996) Characterization of epiretinal membranes using optical coherence tomography. Ophthalmology 103:2142–2151
Nigam N, Bartsch DU, Cheng L et al (2010) Spectral domain optical coherence tomography for imaging ERM, retinal edema, and vitreomacular interface. Retina 30(2):246–253
Gupta P, Sadun AA, Sebag J (2008) Multifocal retinal contraction in macular pucker analyzed by combined optical coherence tomography/scanning laser ophthalmoscopy. Retina 28:447–452
Pilli S, Lim P, Zawadzki RJ et al (2011) Fourier-domain optical coherence tomography of eyes with idiopathic epiretinal membrane: correlation between macular morphology and visual function. Eye 25:775–783
Iannetti L, Tortorella P, D’Ambrosio E et al (2013) Epiretinal membranes in patients with uveitis: morphological and functional analysis with spectral domain optical coherence tomography. Biomed Res Int 2013:284821
Azzolini C, Patelli F, Codenotti M et al (1999) Optical coherence tomography in idiopathic epiretinal macular membrane surgery. Eur J Ophthalmol 9:206–211
Massin P, Allouch C, Haouchine B et al (2000) Optical coherence tomography of idiopathic macular epiretinal membranes before and after surgery. Am J Ophthalmol 130:732–739
Oster SF, Mojana F, Brar M et al (2010) Disruption of the photoreceptor inner segment/outer segment layer on spectral domain-optical coherence tomography is a predictor of poor visual acuity in patients with epiretinal membranes. Retina 30(5):713–718
Michalewski J, Michalewska Z, Cisiecki S, Nawrocki J (2007) Morphologically functional correlations of macular pathology connected with epiretinal membrane formation in spectral optical coherence tomography (SOCT). Graefes Arch Clin Exp Ophthalmol 245:1623–1631
Suzuki T, Terasaki H, Niwa T et al (2003) Optical coherence tomography and focal macular electroretinogram in eyes with epiretinal membrane and macular pseudohole. Am J Ophthalmol 136(1):62–67
Mitamura Y, Hirano K, Baba T, Yamamoto S (2009) Correlation of visual recovery to presence of photoreceptor inner/outer segment junction in optical coherence images after epiretinal membrane surgery. Br J Ophthalmol 93:171–175
Suh MH, Seo JM, Park KH, Yu HG (2009) Associations between macular findings by optical coherence tomography and visual outcomes after epiretinal membrane removal. Am J Ophthalmol 147:473–480
Kadonosono K, Itoh N, Nomura E, Ohno S (1999) Perifoveal microcirculation in eyes with epiretinal membranes. Br J Opthalmol 83:1329–1331
Wise GN (1975) Clinical features of idiopathic preretinal macular fibrosis. Am J Ophthalmol 79:349–357
Poliner LS, Olk RJ, Grand MG et al (1988) Surgical management of premacular fibroplasias. Arch Ophthalmol 106:761–764
Yamamoto S, Yamamoto T, Hayashi M (2001) Morphological and functional analyses of diabetic macular edema by optical coherence tomography and multifocal electroretinograms. Graefes Arch Clin Exp Ophthalmol 239:96–101
Hassenstein A, Bialasiewicz AA, Richard G (2000) Optical coherence tomography in uveitis patients. Am J Ophthalmol 130:669–670
Kim BY, Smith SD, Kaiser PK (2006) Optical coherence tomographic patterns of diabetic macular edema. Am J Ophthalmol 142:405–412
Tanikawa A, Horiguchi M, Kondo M et al (1999) Abnormal focal macular electroretinograms in eyes with idiopathic epimacular membrane. Am J Ophthalmol 127:559–564
Do DV, Cho M, Nguyen QD et al (2006) The impact of optical coherence tomography on surgical decision making in epiretinal membrane and vitreomacular traction. Trans Am Ophthalmol Soc 104:161–166
Conflict of interest statement
All authors certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fang, IM., Hsu, CC. & Chen, LL. Correlation between visual acuity changes and optical coherence tomography morphological findings in idiopathic epiretinal membranes. Graefes Arch Clin Exp Ophthalmol 254, 437–444 (2016). https://doi.org/10.1007/s00417-015-3069-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-3069-0