Abstract
Background
To assess the role of Spectral Domain Optical Coherence Tomography (SD-OCT) measurements as prognostic factors in myopic macular hole (MMH) surgery.
Methods
In a retrospective cohort study, we evaluated 42 eyes of 42 patients (Spherical equivalent > −6.00 D) who underwent pars plana vitrectomy with internal limiting membrane peeling for MMH without foveoschisis. Statistical analysis was performed to correlate postoperative best corrected visual acuity (BCVA) with preoperative BCVA, age, degree of myopia and seven preoperative OCT measurements: macular hole (MH) base, MH minimum diameter, MH height, Hole Form Factor (HFF), Macular Hole Index (MHI), Diameter Hole Index (DHI) and Tractional Hole Index (THI).
Results
Primary and final anatomical success rate were 83.3 % (35/42) and 90.5 % (38/42), respectively. Four patients deferred reoperation and three underwent a second surgical approach to achieve MH closure. A posterior staphyloma was observed in 27 of 42 patients, and in three of them the MH was located in the apex of the staphyloma. Two of these three cases showed an open MH after the first surgery. Postoperative visual acuity improved in 22/42 (52.4 %) patients, worsened in 7/42 (16.7 %) and remained unchanged in 13/42 (30.9 %). Only MH minimum diameter (P = 0.03) and HFF (P = 0.02) correlated significantly with postoperative BCVA.
Conclusions
Minimum diameter and HFF are strongly correlated with postoperative visual outcomes in cases of MMH. Since analyzing MH configuration seems to improve the anatomical success rate after vitreous surgery in highly myopic patients, these parameters should be preoperatively evaluated by SD-OCT.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Macular hole (MH) was first described by Knapp in 1869 in a patient who sustained an ocular blunt trauma [1]. However, it has increasingly being recognized that this condition is more frequently idiopathic, although it can be also considered a common complication in highly myopic eyes. Due to an excessive axial elongation [2–4] , these patients can develop posterior ectasia of the globe known as posterior staphyloma (PS) [5], which can be associated with a full thickness MH with or without foveoschisis and retinal detachment (RD) in some cases [6–8]. Several surgical procedures are available for MH repair with or without RD in high myopia, including pars plana vitrectomy (PPV) with gas or silicone oil tamponade, macular buckling (MB), and scleral shortening technique [9–12]. Moreover, although spontaneous closure of a recurrent myopic macular hole (MMH) can occur [13], some patients require multiple interventions to achieve MH closure [14–16].
However, due to some degree of posterior chorioretinal atrophy, which causes a lack of contrast between the MH and the retinal pigment epithelium (RPE), biomicroscopic diagnosis of MH can be difficult. For this reason, since its first use in 1991 [17], Optical Coherence Tomography (OCT), and even more Spectral Domain (SD)-OCT [18–20], is considered a useful technique to evaluate the status of the macula, since it can reveal myopic foveoschisis and even full-thickness macular holes that are not apparent on fundus biomicroscopy. [21–23]. Moreover, it may play an important role in the follow-up of patients who have undergone vitreoretinal surgery.
Contrary to idiopathic MH, little is found in literature regarding the preoperative MH configuration in highly myopic eyes, and the relationship between the preoperative MH sizes and postoperative visual outcome. Therefore, the aim of our study was to evaluate the role of preoperative OCT measurements, as possible prognostic factors in MMH surgery.
Methods
In a retrospective cohort study, we evaluated 42 eyes of 42 consecutive patients who underwent standard three-port pars plana vitrectomy for MMH without foveoschisis, between 2007 and 2011. All patients had been operated and followed up at the IMO—Instituto de Microcirugía Ocular (Barcelona, Spain). Surgery was performed by the same surgeon (CM). All examinations and investigations adhered to the tenets of the Declaration of Helsinki. All patients had given written informed consent prior to the surgery and all the procedures were approved by the institutional ethics committee of our hospital.
During their visit, all patients underwent a complete ophthalmic evaluation, including refraction, measurement of best corrected visual acuity (BCVA, logMAR) by means of an ETDRS chart, biomicroscopy of the anterior segment and of the retina, color fundus photographs with a mydriatic fundus camera (TRC-50DX type IA; Topcon, Tokyo, Japan) and SD-OCT scans (Cirrus HD-OCT 4000, version 5.0; Carl Zeiss Meditec, Dublin, California, USA). Visual improvement was arbitrarily defined as a gain in the BCVA of 0.20 log units, equivalent to one to two lines on a Snellen chart. Inclusion criteria were a spherical equivalent of at least −6.00 D and a MH base diameter of more than 400 μm (stage 3 or 4 according to the Gass Classification [24, 25]). Exclusion criteria were a refractive error lower than −6.00 D, significant media opacities or other macular conditions such as foveoschisis, posterior retinal detachment, traumatic MH, macular RPE atrophy, choroidal neovascularization or history of past ocular surgery except phacoemulsification.
Surgery
All 42 patients underwent standard 20G or 23G three-port PPV with induced Triamcinolone Acetonide (TA)-assisted posterior vitreous detachment (PVD), if not already present. Epiretinal membrane (ERM) peeling, if present, and internal limiting membrane (ILM) removal were performed using 0.2 mL of dyes, either trypan blue (Membran-Blue; DORC, Zuidland, The Netherlands), brilliant blue G (BrilliantPeel; Geuder, Heidelberg, Germany) or both.
Because of retinal thinning, poor visualization of the ILM, fragility of the same, and in order to avoid the tendency of the eyewall to collapse due to the thinner sclera in highly myopic patients, ILM was peeled under perfluorocarbon (PFC) liquid. Morevoer, its removal was considered complete when the ILM was removed with the end-gripping forceps to the vascular arcades, 360° all around the hole within a minimal extension of approximately 2 or 3 disc diameters centred on the centre of the hole. Finally, a PFC/air exchange and subsequent injection of 20 % of sulfur hexafluoride (SF6) for tamponade were performed. Patients were asked to maintain a prone position for 5 days postoperatively. Phacoemulsification with intraocular lens implants was performed in the follow-up period only in cases that developed post-vitrectomy cataract. Postoperative anatomical success was defined as the complete closure of the MH and absence of subretinal fluid on OCT.
Optical Coherence Tomography (OCT) measurements
Based on what has been reported in previous studies [26–31], seven OCT parameters (Figs. 1 and 2) that defined the geometry of the MMH were analyzed by two observers (MA and LP): MH base diameter, MH minimum diameter, MH height, Hole Form Factor (HFF) [28–31], Macular Hole Index (MHI) [26], Diameter Hole Index (DHI) and Tractional Hole Index (THI) [27]. For each patient, after detecting the area, which included the MH using the Macular Cube 512x128 acquisition protocol, horizontal and vertical 5 Line Raster analyses were performed to select the foveal scan with the greatest MH dimension. Finally, the corresponding single high-definition line scan image was acquired using the HD 5 Line Raster for the subsequent parameters determination.
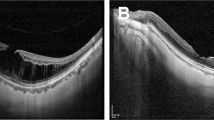
Preoperative Hole Form Factor (HFF) determination by OCT measurements in a stage 3 MMH. Base diameter (B) was measured as the greater distance between the MH margins at the level of the retinal pigment epithelium (RPE). Minimum diameter (M) was considered as the shorter distance across the MH at the level of the inner retinal layers. Left (L) and right (R) arms corresponded to the distance between the left and right edge of the base and minimum diameter. The HFF was finally calculated as shown. A posterior staphyloma involving the macula and a hyperreflective band at the vitreoretinal interface (arrow heads), which possibly represents the posterior hyaloids rather than an epiretinal membrane, are evident in preoperative B-scan. A complete MMH closure was achieved postoperatively, showing fine dimples in the retinal nerve fiber layer [32] maybe due to the internal limiting membrane (ILM) peeling (white arrows). Notes the mirror artifacts which often occurs on spectral domain-OCT images in highly myopic eyes (white asterisks) [33]
Preoperative Macular Hole Index (MHI), Diameter Hole Index (DHI), Tractional Hole Index (THI) measurements on OCT scans in a stage 4 MMH. The MH height (H) was considered as the greater distance between the RPE and the ILM on the highest side of the MH. MHI was the ratio of MH height (H) to the base diameter (B) [26]. DHI and THI were calculated as the ratio of the minimum diameter (M) to the base (B), and the ratio of the height (H) to the minimum diameter (M) of the MH, respectively [27]. A mirror artifact is present (white asterisk) [33]. Bottom left B-scan indicates a complete MMH closure 1 year after surgery with the evidence of shallow dimples in the optic nerve fiber layer (white arrows) [32]
Statistical analysis
Best corrected postoperative visual acuity (LogMAR) achieved by each patient at the end of follow-up was used for statistical analyses (MedCalc Software version 9.3.1.0, Mariakerke, Belgium). To assess if functional outcome (postoperative BCVA) might be influenced by cataract surgery, a two-tailed t-test was performed between patients who underwent phacoemulsification with intraocular lens implants and patients who did not. Spearman rank coefficient was calculated to assess correlation between the postoperative BCVA (LogMAR) and preoperative variables such as age, preoperative BCVA (LogMAR), degree of myopia and all seven MMH indexes. A P value < 0.05 was considered statistically significant, and the interobserver variability (i.e. repeatability) for the OCT measurements was calculated using the intraclass correlation coefficient (ICC) before performing any statistical analysis.
Results
Table 1 summarizes the demographic and ophthalmic data of the 42 study participants.
Six of 20 phakic patients (14 %) developed cataract during the follow-up period and underwent phacoemulsification with intraocular lens implants. There was no statistically significant difference (P > 0.05, t-test) concerning postoperative BCVA (LogMAR) between patients who underwent cataract extraction and patients who did not.
Mean preoperative BCVA (LogMAR) was 0.82 ± 0.37 SD and improved to 0.71 ± 0.44 SD postoperatively. Nevertheless, this difference was not statistically significant (P = 0.19, t-test). In 22/42 patients (52.4 %), BCVA was improved after surgery. Mean gain in visual acuity at the end of follow-up was 0.12 ± 0.41 SD (ΔBCVA, LogMAR). Visual acuity worsened or remained unchanged in 7/42 (16.7 %) and 13/42 (30.9 %) eyes, respectively. Among the group of patients who experimented a decrease in their visual acuity (7/42), three had undergone a second surgery, two had an open MH and two were affected by glaucoma.
Mean preoperative MH measurements were calculated as showed in Table 1.
For our series of measurements agreement, the ICC for the OCT parameters ranged from 0.70 to 0.89.
Primary and final anatomical success rate were 83.3 % (35/42) and 90.5 % (38/42) respectively. Four patients deferred reoperation and three patients underwent a second surgical approach to achieve MH closure. In these two subgroups, mean preoperative refractive errors (Spherical Equivalent, SE) were −17.00 D ± 3.16 SD (range −14.00 to −21.00) and −16.83 D ± 9.17 SD (range −6.50 to −24.00) respectively. No reopened MH and no posterior RD resulting from a persistent MH were observed during the follow-up.
As indicated in Table 2, MH minimum diameter (P = 0.03) and HFF (P = 0.02) showed a statistically significant correlation with postoperative BCVA (LogMAR), while no significant correlation was found regarding the other five OCT parameters. HFF had a higher correlation with the postoperative BCVA (LogMAR) than any other OCT variable evaluated. Among patients with primary successful MH closure (35/42) HFF ranged from 0.58 to 1.11 (mean value 0.82 ± 0.13) and mean MH minimum diameter was 354.40 μm ± 174.52 SD. In MHs that were closed after a second surgical approach (3/42), mean preoperative HFF and MH minimum diameter were 0.78 ± 0.14 SD and 397 μm ± 90.44 SD, respectively. Concerning MMH which remained open after one surgical procedure (7/42), these two OCT parameters measured 0.68 ± 0.13 SD and 436.71 ± 150.37 SD respectively. In addition, no statistically significant correlation was found between any OCT parameter and postoperative BCVA in this subgroup, probably due to the low sample size.
Regarding the other preoperative variables, both MH minimum diameter and HFF had a statistically significant correlation with preoperative BCVA (P = 0.005 and P = 0.03, respectively), but not with the degree of myopia (P = 0.54 and P = 0.37, respectively).
No intraoperative or early postoperative complications were observed. Cataract progression in some cases and refractary glaucoma in two cases that required filtration surgery, were observed as the later complications during the follow-up.
Discussion
Individuals with high myopia are subject to various posterior retinal diseases, including MH which can also be associated with foveoschisis or RD if a posterior staphyloma is present. In these patients, SD-OCT appears to be a very useful and practical tool to avoid delayed diagnosis of some important and progressive retinal pathologies, such as MH. Several authors tried to correlate the preoperative features of idiopathic MH determined by OCT with the anatomical and functional results after surgery [26–31, 34–37]. Until now, little attention has been given to the preoperative MH configuration in highly myopic eyes [38–43] and, to our knowledge, this is the first study with the largest sample of MMH without foveoschisis that analyzes and correlates postoperative functional outcomes with MMH sizes measured by SD-OCT, which is an accurate tool for a quantitative evaluation of the preoperative MH deformation [Shih CY, et al. IOVS. 2002;43:ARVO E-Abstract 4405].
Since we supposed that the pathogenetic mechanism of MMH, with or without posterior staphyloma, could be similar in some cases to that in emmetropic eyes, we evaluated the MMH anatomical configuration using the same measurements as described in previous studies for idiopathic MH [26–31, 34–37]. In our study, MH minimum diameter and Hole Form Factor (HFF) were the only two OCT parameters that showed a statistically significant correlation with postoperative BCVA , while no significant correlation was found regarding the other five OCT measurements.
In the series of Puliafito et al. regarding idiopathic MH [28, 34, 35] , HFF was considered to be of greater influence on the anatomical success rate than the base diameter alone. They found an 80 % anatomical success rate in patients with HFF > 0.9 and an anatomical success rate less than 25 % in patients with HFF < 0.5. Thus, 0.9 and 0.5 were considered the HFF cut-off values for a good or unfavorable anatomical outcome. In our series, primary and final MH closure rate were 83.3 % (35/42) and 90.5 % (38/42) with a mean HFF of 0.80 and 0.82, respectively. Furthermore, in 11 of 12 patients with HFF > 0.9, the MH was closed following one operation. Among patients who underwent a second surgical procedure and in which MH was closed (3/42), mean preoperative HFF was similar (0.78 ± 0.14) to primarily closed MMH, suggesting that other factors might influence anatomical success in these cases. In addition, according to previous studies [28, 34, 35], in eyes without anatomical MH closure (4/42) HFF has a lower value, ranging from 0.54 to 0.63 (mean HFF 0.60 ± 0.04). This result seemed to suggest that a greater HFF was more likely associated with a higher anatomical success rate , while a MH closure failure might occur in case of a low HFF value.
Concerning linear horizontal and vertical dimensions, MH minimum diameter showed a statistically significant correlation with postoperative BCVA (P = 0.03), while no significant correlation was found with MH base diameter (P = 0.40) and MH height (P = 0.58). Thus, it might be supposed that MH minimum diameter, which is usually the inner one, rather than base diameter, is mostly implicated in the process of the MH closure.
In 7/42 patients, there was a persistent MH after one surgical approach. Postoperatively, mean MH base and minimum diameter were increased in this group. Although it is well known for idiopathic MH, this confirmed that also in highly myopic eyes, there is a tendency of persistent MHs to enlarge after vitrectomy [15].
Our study had several limitations, mostly due to its retrospective nature. We did not evaluate any correlation between preoperative variables such as axial length and duration of symptoms.
Firstly, the use of the refractive error (spherical equivalent) as an index could not be suggestive of the real degree of myopia and, therefore, of the effective anteroposterior traction produced by posterior staphyloma, if present. [44, 45] This might explain why no correlation was found between any preoperative OCT parameter and refractive error. Nevertheless, PS involving the macula was not observed in 15 of 42 patients in our series, and only three of 27 patients with PS had the MH located just at the apex of PS, where anteroposterior tractional forces might be increased. Interestingly, in two of these three cases in which the HFF had lower values than in all other patients (0.54 and 0.56 respectively), the MH remained open. However, the size and shape of posterior stahpyloma, as well as the axial length, was not measured in this study, and it is difficult to make conclusions regarding these two aspects of high myopia.
Secondly, we did not consider the duration of symptoms for our statistical analysis because this criterion could be conditioned by the subjective patients’ estimation. In addition, it has been demonstrated that a MH in highly myopic eyes may be totally asymptomatic for a long time before being detected [21].
In conclusion, we retrospectively evaluated 42 eyes with MMH without foveoschisis or posterior retinal detachment, reporting a final anatomical MH closure rate of 90.5 % after vitreous surgery.
However, our study was not designed to assess the value of vitreous surgery in MMH for its potential visual success; nevertheless, we achieved a high anatomical success rate in a large cohort of patients affected by MMH without foveoschisis. We believe that further studies are necessary to confirm which seems to be the best surgical approach in these group of patients.
In our series, myopic MH without foveoschisis seem to have less favorable visual results than non-myopic MH. Regarding the seven MMH geometric parameters, HFF and MH minimum diameter were the only OCT measurements that correlated with postoperative visual acuity. However, preoperative BCVA still remained the best predictor of postoperative functional outcomes of all parameters assessed.
Nevertheless, to our knowledge, this is the first study that analyzes the OCT measurements of MH in high myopia, and this does not allow a comparison between our series and previous data regarding the prognostic value of the MMH parameters.
References
Knapp H (1869) Ueber isolirte zerreissungen der aderhaut in folge von traumen auf dem augapfel. Arch Augenheilkd 1:6–29
Coppé AM, Ripandelli G (2003) Optical coherence tomography in the evaluation of vitreoretinal disorders of the macula in highly myopic eyes. Semin Ophthalmol 18(2):85–88
Faghihi H, Hajizadeh, Riazi-Esfahani M (2010) Optical coherence tomographic findings in highly myopic eyes. J Ophthalmic Vis Res 5(2):110–121
Tano Y (2002) Pathologic myopia: where are we now? Am J Ophthalmol 134(5):645–660
Curtin BJ (1977) The posterior staphyloma of pathologic myopia. Trans Am Ophthalmol Soc 75:67–86
Oie Y, Ikuno Y, Fujikado T, Tano Y (2005) Relation of posterior staphyloma in highly myopic eyes with macular hole and retinal detachment. Jpn J Ophthalmol 49(6):530–532
Matsumura N, Ikuno Y, Tano Y (2004) Posterior vitreous detachment and macular hole formation in myopic foveoschisis. Am J Ophthalmol 138(6):1071–1073
Sayanagi K, Ikuno Y, Tano Y (2006) Reoperation for persistent myopic foveoschisis after primary vitrectomy. Am J Ophthalmol 141(2):414–417
Kono T, Takesue Y, Shiga S (2006) Scleral resection technique combined with vitrectomy for a macular hole retinal detachment in highly myopic eyes. Ophthalmologica 220(3):159–163
Nakagawa N, Parel JM, Murray TG, Oshima K (2000) Effect of scleral shortening on axial length. Arch Ophthalmol 118(7):965–968
Tanaka T, Ando F, Usui M (2005) Episcleral macular buckling by semirigid shaped-rod exoplant for recurrent retinal detachment with macular hole in highly myopic eyes. Retina 25:147–151
Qian J, Jiang YR (2010) Anatomic evaluation of macular holes with silicone oil tamponades in highly myopic eyes using optical coherence tomography. Eur J Ophthalmol 20(5):938–944
Bonnet M, Semiglia R (1993) Late recurrences after successful surgery for retinal detachment with macular hole. Graefes Arch Clin Exp Ophthalmol 231:347–350
Seike C, Kusaka S, Sakagami K, Ohashi Y (1997) Reopening of macular holes in highly myopic eyes with retinal detachments. Retina 17(1):2–6
Sayanagi K, Ikuno Y, Tano Y (2005) Macular hole diameter after vitrectomy for macular hole and retinal detachment. Retina 25(5):608–611
Yonekawa Y, Hirakata A, Inoue M, Okada AA (2010) Spontaneous closure of a recurrent myopic macular hole previously repaired by pars plana vitrectomy. Acta Ophthalmol 89(6):e536–e537
Huang D, Swanson EA, Lin CP, Schuman JS, Stinson WG, Chang W, Hee MR, Flotte T, Gregory K, Puliafito CA (1991) Optical coherence tomography. Science 254(5035):1178–1181
Van Velthoven ME, Faber DJ, Verbraak FD, Van Leeuwen TG, De Smet MD (2007) Recent developments in optical coherence tomography for imaging the retina. Prog Retin Eye Res 26:57–77
Sakata LM, Deleon-Ortega J, Sakata V, Girkin CA (2009) Optical coherence tomography of the retina and optic nerve—a review. Clin Exp Ophthalmol 37(1):90–99
Jaffe GJ, Caprioli J (2004) Optical coherence tomography to detect and manage retinal disease and glaucoma. Am J Ophthalmol 137:156–169
Coppé AM, Ripandelli G, Parisi V, Varano M, Stirpe M (2005) Prevalence of asymptomatic macular holes in highly myopic eyes. Ophthalmology 112(12):2103–2109
Gaucher D, Erginay A, Lecleire-Collet A, Haouchine B, Puech M, Cohen SY, Massin P, Gaudric A (2008) Dome-shaped macula in eyes with myopic posterior staphyloma. Am J Ophthalmol 145(5):909–914
Panozzo G, Mercanti M (2004) Optical coherence tomography findings in myopic traction maculopathy. Arch Ophthalmol 122:1455–1460
Gass JDM (1988) Idiopathic senile macular holes: its early stages and pathogenesis. Arch Ophthalmol 106:629–639
Johnson RN, Gass JDM (1998) Idiopathic macular holes: observations, stages of formation and implications of surgical intervention. Ophthalmology 95:917–924
Kusuhara S, Teraoka Escaño MF, Fujii S, Nakanishi Y, Tamura Y, Nagai A, Yamamoto H, Tsukahara Y, Negi A (2004) Prediction of postoperative visual outcome based on hole configuration by optical coherence tomography in eyes with idiopathic macular holes. Am J Ophthalmol 138(5):709–716
Ruiz-Moreno JM, Staicu C, Piñero DP, Montero J, Lugo F, Amat P (2008) Optical coherence tomography predictive factors for macular hole surgery outcome. Br J Ophthalmol 92(5):640–644
Ip MS, Baker BJ, Duker JS, Reichel E, Baumal CR, Gangnom R, Puliafito CA (2002) Anatomical outcomes of surgery for idiopathic macular hole as determined by optical coherence tomography. Arch Ophthalmol 120:29–35
Benson SE, Schlottmann PG, Bunce C, Charteris DG (2008) Comparison of macular hole size measured by optical coherence tomography, digital photography, and clinical examination. Eye (Lond) 22(1):87–90
Uemoto R, Yamamoto S, Aoki T, Tsukahara I, Yamamoto T, Takeuchi S (2002) Macular configuration determined by optical coherence tomography after idiopathic macular hole surgery with or without internal limiting membrane peeling. Br J Ophthalmol 86(11):1240–1242
Hoerauf H (2007) Predictive values in macular hole repair. Br J Ophthalmol 91(11):1415–1416
Alkabes M, Salinas C, Vitale L, Burès A, Nucci P, Mateo C (2011) En face optical coherence tomography of inner retinal defects after internal limiting membrane peeling for idiopathic macular hole. Invest Ophthalmol Vis Sci 52(11):8349–8355
Song Y, Lee BR, Shin YW, Lee YJ (2012) Overcoming segmentation errors in measurements of macular thickness made by spectral-domain optical coherence tomography. Retina 32(3):569–580
Hee MR, Puliafito CA, Wong C, Duker JS, Reichel E, Schuman JS, Swanson EA, Fujimoto JG (1995) Optical coherence tomography of macular hole. Ophthalmology 102:748–756
Desai VN, Hee MR, Puliafito CA (1999) Optical coherence tomography of macular holes. In: Madreperla SA, McCuen BW (eds) Macular hole: Pathogenesis, diagnosis and treatment. Butterworth-Heinemann, Oxford, pp 37–47
Haritoglou C, Neubauer AS, Reiniger IW, Priglinger SG, Gass CA, Kampik A (2007) Long-term functional outcome of macular hole surgery correlated to optical coherence tomography measurements. Clin Exp Ophthalmol 35(3):208–213
Ullrich S, Haritoglou C, Gass C, Schaumberger M, Ulbig MW, Kampik A (2002) Macular hole size as a prognostic factor in macular hole surgery. Br J Ophthalmol 86(4):390–393
Salman AG (2006) Healing pattern of myopic macular hole based on optical coherence tomography (OCT). Saudi J Ophth 20(2):124–130
Georgalas I, Ezra E (2007) Delayed closure after surgery for a full-thickness macular hole in a highly myopic eye. Can J Ophthalmol 42(2):341–342
Ikuno Y, Tano Y (2003) Early macular holes with retinoschisis in highly myopic eyes. Am J Ophthalmol 136(4):741–744
Ikuno Y, Sayanagi K, Oshima T, Gomi F, Kusaka S, Kamei M, Ohji M, Fujikado T, Tano Y (2003) Optical coherence tomography findings of macular hole and retinal detachment after vitrectomy in highly myopic eyes. Am J Ophthalmol 136:477–481
Kokame GT (1995) Early stage of macular hole in a severely myopic eye. Am J Ophthalmol 119(2):240–242
Sun CB, Liu Z, Xue AQ, Yao K (2010) Natural evolution from macular retinoschisis to full-thickness macular hole in highly myopic eyes. Eye (Lond) 24(12):1787–1791
Saka N, Ohno-Matsui K, Shimada N, Sueyoshi S, Nagaoka N, Hayashi W, Hayashi K, Moriyama M, Kojima A, Yasuzumi K, Yoshida T, Tokoro T, Mochizuki M (2010) Long-term changes in axial length in adult eyes with pathologic myopia. Am J Ophthalmol 150(4):562–568
Hsiang HW, Ohno-Matsui K, Shimada N, Hayashi K, Moriyama M, Yoshida T, Tokoro T, Mochizuki M (2008) Clinical characteristics of posterior staphyloma in eyes with pathologic myopia. Am J Ophthalmol 146(1):102–110
Acknowledgments
We are greatly indebted to Giovanni Taibbi, Davide Ausili and Daniela Gilardi for their precious help in the statistical analysis.
Financial disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alkabes, M., Padilla, L., Salinas, C. et al. Assessment of OCT measurements as prognostic factors in myopic macular hole surgery without foveoschisis. Graefes Arch Clin Exp Ophthalmol 251, 2521–2527 (2013). https://doi.org/10.1007/s00417-013-2347-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-013-2347-y