Abstract
Background
At the time of posterior subtenon infusion of triamcinolone acetonide (STI-TA) in patients with diabetic macular edema (DME), drug reflux of TA has sometimes been observed from the conjunctival incision site. We investigated the influence of this reflux on regression of DME and postoperative intraocular pressure (IOP).
Methods
STI-TA was performed on one hundred and twenty-four eyes of 88 consecutive patients with DME. Eligible eyes were divided into two groups: those with observed drug reflux of TA and those without observed drug reflux of TA. Visual acuity (VA), foveal thickness (FT) and IOP were monitored in each eye for up to 12 weeks after STI-TA.
Results
STI-TA with drug reflux was observed in ten individual eyes of seven patients. These patients were significantly younger than those patients without observed drug reflux. After STI-TA, both improvement of VA and regression of FT in reflux(+) eyes were less than in reflux(-) eyes. Postoperative IOP elevation in reflux(+) eyes was much higher, and four of the ten eyes needed anti-glaucoma therapy. This was in contrast to three of the 118 eyes without drug reflux that required anti-glaucoma therapy.
Conclusions
At the time of STI-TA in DME, drug reflux of TA is a risk factor not only for insufficient reduction of edema, but also for postoperative IOP elevation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Diabetic macular edema (DME) is the most common cause of vision loss in diabetic patients [1]. Recently, the intravitreal administration of triamcinolone acetonide (IV-TA) has provided promising results for the treatment of DME [2, 3]. However, intravitreal administration is associated with many ocular complications, such as intraocular pressure (IOP) elevation [4], endophthalmitis [5], intraocular hemorrhages, or retinal detachment [6]. Posterior subtenon infusion of triamcinolone acetonide (STI-TA) is an alternative to intravitreal injection and has been reported to be an equally effective and safer treatment for DME [7, 8]. In contrast, it has also been reported that STI-TA may be less effective than IV-TA for the management of DME [9]. Compared with IV-TA, the injection technique of STI-TA is more complicated because: (1) Tenon’s capsule is sometimes too thick to make a clear incision, and (2) the tip of the infusion needle is inserted thorough the back of the eye, and thus drug delivery is hard to confirm. Even with a carefully performed procedure, back flow (reflux) of triamcinolone acetonide (TA) through the subtenon incision is sometimes observed during STI, resulting in TA being incorrectly infused. Therefore, the effectiveness of STI-TA in reducing DME appears to be more case-sensitive than that of IV-TA.
In this prospective study, we investigated whether drug reflux of TA during STI affects the reduction of DME and IOP elevation.
Materials and methods
After informing the patients of the purpose of this study and the possible outcomes, informed consent was obtained from all patients prior to the intervention. This study was also approved by the local institutional review board of NTT East Japan Tohoku Hospital Clinical Research Ethics Committee. The procedures conformed to the tenets of the Declaration of Helsinki.
Patient eligibility
In this prospective study, patients with DME whose foveal thickness was more than 400 μm, and who had visual acuity less than logMAR 0.3, were recruited. DME was defined as retinal thickening of two or more disk areas involving some portion of the foveal avascular zone [10]. Indirect ophthalmoscopy and slit-lamp biomicroscopy of the posterior segment with a Volk Superfield contact lens [Volk, Mentor, OH, USA] were performed by at least two independent clinicians to establish the presence of DME. Exclusion criteria were: (1) a history of previous subtenon injection of any drugs, (2) history of glaucoma or ocular hypertension, (3) history of vitreous surgery, (4) loss of vision as a result of other causes, (5) HbA1c greater than 9.0%, (6) systemic or local corticosteroid therapy, (7) severe systemic disease, or (8) any condition affecting follow-up or documentation.
During the inclusion period of the study, DME was identified in 128 eyes of 88 consecutive patients (40 patients had bilateral DME) based on clinical and angiographic evaluation of diffuse fluorescein leakage involving the foveal center and most of the macular area. All of the patients had pan-retinal photocoagulation at least 12 months prior to entry into the study, and DME had been identified within the previous year. Of the 128 eyes, 86 had a history of cataract surgery with no complications at least 6 months prior to the study.
All patients received a comprehensive ocular examination before and after treatment. The best-corrected visual acuity (BCVA) with the logMAR chart (5 m) (NEITZ LVC-10, Tokyo, Japan) and retinal thickness by optical coherence tomography (OCT) (OCT-3000: Zeiss-Humphrey, Dublin, CA, USA) were measured during the follow-up examinations. A macular thickness map was made from six radial scans that intersected at the fovea using the OCT retinal-mapping program [Ver 6.2]. This program calculates mean thickness in nine regions: the 1000-μm central area, and the four quadrants of the inner and outer rings. The diameters of the inner and outer rings were 1000 to 3000 and 3000 to 6000 μm, respectively [11]. In this study, foveal thickness (FT) was defined as the value of a 1000-μm central area. IOP was measured by applanation tonometry during the clinical course.
Experimental design
After the initial ocular and general examinations, STI-TA was performed in all eyes by the same retinal specialist (MS) using topical anesthesia under appropriate sterile conditions. A conjunctival incision was made at the superotemporal quadrant, then Tenon’s incision was made midway between the superior and lateral rectus muscles, 7–9 mm posterior to the limbus, and the surface of the sclera was confirmed. A curved blunt cannula (21-gauge tri-port subtenon cannula: Eagle Laboratories, Rancho Cucamonga, CA, USA) was inserted through the subtenon space reaching the posterior pole [12]. Next, 20 mg (0.5 ml) of triamcinolone acetonide [Kenacort™ 40 mg/ml] was injected. At the time of injection, a back flow of the white, particulate fluid of triamcinolone acetonide from Tenon’s incision was observed in some cases, and this defined drug reflux. All of the STI-TA procedures were recorded on a video disc recorder (VDR). In each case, drug reflux was confirmed from the VDR recording by two observers (TN and KY), and the eyes were then classified as being in the reflux(+) group or the reflux(-) group.
In all eyes, BCVA, FT and IOP were measured at 1, 2, 4, 8, and 12 weeks after the STI-TA injection. If IOP was increased to more than 25 mmHg during the 12-week clinical course, the corresponding patients were treated with anti-glaucoma therapy as appropriate. δIOP was defined as the subtraction of IOP at each point in time from the baseline.
Statistical analyses
The data are presented as mean ± standard deviation. Statistical differences between pre- and post-treatment data were assessed using the Wilcoxon signed-rank test, and differences between reflux(+) and reflux(-) eyes were assessed using the Mann Whitney U test (SPSS, Chicago, IL, USA). A p value of less than 0.05 was considered to be statistically significant.
Results
Drug reflux of TA during STI was observed in 10 individual eyes of seven patients out of a total of 128 eyes of 88 patients. Four of the seven patients had only one of their eyes injected, as their contra-lateral eye was not eligible for this study.
Alterations of FT and VA
Baseline characteristics of the reflux(+) group and the reflux(-) group are shown in the Table 1. The mean age of the reflux(+) group was a statistically significant lower age than that of the normal group (p = 0.011). In contrast, there was no significant difference in gender (p = 0.579), duration of diabetes (p = 0.260), HbA1c (p = 0.649), total cholesterol (p = 0.865) or history of cataract surgery (p = 0.473) between the groups.
Before starting this study, FT was 624.9 ± 102.8 μm in the reflux(+) group, and 653.2 ± 119.4 μm in the reflux(-) group, which is not significantly different (p = 0.469). After the STI-TA, FT in the reflux(-) group showed a significant reduction at each time point; in contrast, FT in the reflux(+) group showed a significant reduction from baseline only at 2 and 4 weeks. As depicted in Fig. 1a, at all time points after STI-TA, FT in the reflux(+) group was significantly higher than that in the reflux(-) group. Maximum reduction of edema after STI-TA in the reflux(+) group was 87.3 ± 11.0%, which is significantly less effective than that in the reflux(-) group of 64.2 ± 17.9% (p = 0.0004).
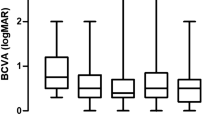
a Alteration of foveal thickness (FT) after subtenon infusion of triamcinolone acetonide (STI-TA) with (open circle) and without (closed circle) drug reflux. At the time of STI (0 wk), there is no significant difference between the two groups. After STI, FT in the group of eyes without drug reflux showed significant decrease compared with the group with drug reflux at each time point (1, 2, 4, 8 and 12 wks). b Alteration of best corrected logMAR visual acuity (BCVA) after STI-TA with (open circle) and without (closed circle) drug reflux. At the time of STI-TA (0 wk), and at 1 week, there is no significant difference between the two groups. Two weeks after STI-TA, BCVA in eyes without drug reflux showed significant decrease compared with the group with drug reflux at each time point (2, 4, 8 and 12 wks). Each point and vertical bar indicates the mean ± standard deviation. The asterisk (*) indicates a statistically significant difference between the groups of eyes with and without drug reflux at each time point (p < 0.05)
As depicted in Fig. 1b, initial logMAR BCVA was 0.62 ± 0.22 in the reflux(+) group, and 0.63 ± 0.20 in the reflux(-) group, which indicates no significant difference between the two groups (p = 0.873). After the STI, logMAR BCVA in both groups was slightly decreased (improved); however, alteration of logMAR BCVA in the reflux(+) group was less dramatically improved than that in the reflux(-) group. There is a significant difference of logMAR BCVA between the two groups at every time point after 2 weeks.
Alteration of IOP
At the initial examination, IOP was 17.9 ± 2.1 mmHg in the reflux(+) group, and 16.9 ± 2.5 mmHg in the reflux(-) group, which shows no significant difference between the two groups (p = 0.221). After the STI-TA, IOP of more than 25 mmHg (high IOP eye) was observed in four of the ten eyes (40%) in the reflux(+) group. Anti-glaucoma eye drops were started at the 2-week point in two of these eyes, and at the 8-week point in the other two eyes. In contrast, high IOP was observed in three of the 118 eyes (2.5%) in the reflux(-) group, and anti-glaucoma eye drops were started at the 12-week point in these three eyes (Fig. 2a). During the clinical course, maximum elevation of δIOP in the reflux(+) group was 5.7 ± 2.9 mmHg, which is significantly higher than that in the reflux(-) group, which was 2.6 ± 2.2 mmHg (p < 0.0001).
a After the subtenon infusion of triamcinolone acetonide (STI-TA), IOP of more than 25 mmHg (high IOP eye) was observed in four out of ten (40%) eyes in the reflux(+) group. In contrast, high IOP eye was observed in three out of 118 (2.5%) eyes in the reflux(-) group. The vertical bar indicates the ratio of high IOP eye (white bar) and normal IOP eye (black bar) in eyes with (upper bar) and without (lower bar) drug reflux at STI-TA. b Alteration of δIOP after STI-TA with (open circle) and without (closed circle) drug reflux. Although δIOP in both groups gradually increased from baseline, δIOP in the group of eyes with drug reflux showed significant increase compared with the group without drug reflux at each time point after 2 weeks. Note that the data of high IOP eye in both groups [four eyes in the reflux(+) group, three eyes in the reflux(-) group] were excluded. Each point and vertical bar indicates the mean ± standard deviation. The asterisk (*) indicates a statistically significant difference between the groups of eyes with and without drug reflux at each time point (p < 0.05)
Alterations of δIOP after treatment are depicted in Fig. 2b. Note that the data for high IOP eye in both groups (four eyes in the reflux(+) group, three eyes in the reflux(-) group) are excluded. Although δIOP in both groups gradually increases from the baseline, δIOP in the eyes in the reflux(+) group shows a significant increase compared with those eyes in the reflux(-) group at each time point after 2 weeks.
Discussion
Three main conclusions were reached from the results of this prospective clinical study: (1) STI-TA with drug reflux is less effective for reduction of DME than STI-TA without drug reflux, (2) STI-TA with drug reflux also causes more elevation of IOP up to 12 weeks post-injection, and (3) STI-TA with drug reflux was more often observed in DME patients with a younger average age.
The pharmacokinetics of STI-TA is that TA diffuses through the sclera to reach the retina [13], and the effect of STI-TA is dose-dependent [14]. Although the amount of STI-TA used in this clinical study was 20 mg, when STI with drug reflux occurs the actual amount of TA is most likely less than an effective dose. A previous study indicated that STI-TA using 5 mg or less of TA resulted in no detectable vitreous drug concentration [14]. Even with 40 mg of STI-TA, vitreous drug concentration was reported to vary considerably, from 0 to 4.93 μg/ml [15]. Thus, it seems to be difficult for STI-TA to be injected in the posterior subtenon space in a fixed concentration, and therefore it is not surprising that STI-TA with drug reflux is less effective at reducing macular edema.
It is surprising that STI-TA with drug reflux more often resulted in IOP elevation. The incidence of IOP elevation after STI-TA was reported to be about 20% [16, 17]. In this study, a high-risk increase in IOP (>24 mmHg) occurred after STI-TA in 40% of the eyes with drug reflux. In contrast, this increase in IOP was observed in only 2% of the eyes without drug reflux. Although the pathogenesis of steroid-induced IOP elevation remains unclear, corticosteroids alter the gene expression of actins and their associated proteins [18], and change the formation of cross-linked actin networks in trabecular meshwork cells [19]. It is also reported that corticosteroids alter the activity of matrix metalloproteinase, which interferes with the ocular fluid drainage at the trabecular meshwork [20]. Another researcher disclosed that corticosteroids alter glaucoma-associated gene expression in the ciliary body [21]. These previous investigations indicate that functional alterations in the trabecular meshwork and/or the ciliary body are responsible for steroid-induced ocular hypertension. Thus, it is possible that STI-TA with drug reflux results in accumulation of TA into the anterior subtenon space, and juxtascleral infusion into the ciliary body and trabecular meshwork, affecting the production or discharge of aqueous humor. To prevent STI-TA-induced IOP elevation, it is important to place the triamcinolone in the back of the eye, not in the anterior (subconjunctival) space, so that STI will occur without drug reflux. To minimize drug reflux at the time of STI-TA delivery, if back pressure is encountered, momentarily stop the delivery and then slow the delivery rate to allow for expansion of subtenon space, or then reassess the position of the cannula on the sclera, or withdraw the cannula and reinsert if necessary [22]. Another technique using a counter-pressure device is also effective, which is the application of pressure with a cotton tip over the cannula during retraction of the cannula [22].
It has previously been reported that a younger age was associated with an increased incidence of triamcinolone-induced ocular hypertension [16, 23], but the reason why a younger age is a risk factor for STI-TA-induced IOP elevation remains unclear. At a younger age, Tenon's capsule is composed mainly of round and smooth collagen fibrils and contains more elastic fibers [24], which makes the connective tissue thicker. This thicker subtenon connective tissue may make smooth insertion of the needle in STI-TA more difficult. In this study, in three of the 20 patients with bilateral eligible eyes, STI-TA with drug reflux occurred in both eyes, even though the procedure was carefully performed, while in the other 17 bilateral eligible eyes, STI-TA was easily performed without drug reflux in either eye, indicating that STI-TA with drug reflux was patient-dependent. Thus STI-TA in younger patients, with a thicker subtenon space, may result in a reflux of triamcinolone acetonide which induces IOP elevation.
In this study, we conclude that STI-TA with drug reflux results not only in an insufficient reduction of DME, but also in a higher risk of IOP elevation, especially in younger patients, as well as in patients with DME, who need a careful ophthalmologic check on drug reflux of TA at the time of STI-TA.
References
Moss SE, Klein R, Klein BEK (1988) The incidence of visual loss in a diabetic population. Ophthalmology 95:1340–1348
Jonas JB, Kreissig I, Sofker A, Degenring RF (2003) Intravitreal injection of triamcinolone for diffuse diabetic macular edema. Arch Ophthalmol 121:57–61, doi:10.1001/archopht.121.5.729
Massin P, Andren F, Haouchine B, Erginay A, Bergmann JF, Benosman R, Caulin C, Gaudric A (2004) Intravitreal triamcinolone acetonide for diabetic macular edema-Preliminary results of a prospective controlled trial-. Ophthalmology 111:218–225, doi:10.1016/j.ophtha.2003.05.037
Jonas JB, Degenring RF, Kreissig I, Akkoyun I, Kamppeter BA (2005) Intraocular pressure elevation after intravitreal triamcinolone acetonide injection. Ophthalmology 112:593–598, doi:10.1016/j.ophtha.2004.10.042
Moshfeghi DM, Kaiser PK, Scott IU, Sears JE, Benz M, Sinesterra JP, Kaiser RS, Bakri SJ, Maturi RK, Belmont J, Beer PM, Murray TG, Quiroz-Mercado H, Mieler WF (2003) Acute endophthalmitis following intravitreal triamcinolone acetonide injection. Am J Ophthalmol 136:791–796, doi:10.1016/S0002-9394(03)00483-5
Jonas JB, Spandau UH, Schlichtenbrede F (2008) Short-term complication of intravitreal injections of triamcinolone and bevacizumab. Eye 22:590–591, doi:10.1038/eye.2008.10
Cellini M, Pazzaglia A, Zamparini E, Leonetti P, Campos EC (2008) Intravitreal vs. subtenon triamcinolone acetonide for the treatment of diabetic cystoid macular edema. BMC Ophthalmol 8(5)
Choi YJ, Oh IK, Oh JR, Huh K (2006) Intravitreal versus posterior subtenon injection of triamciolone acetonide for diabetic macular edema. Korean J Ophthalmol 20:205–209
Bonini-Filho MA, Jorge R, Barbosa JC, Calucci D, Cardillo JA, Costa RA (2005) Intravitreal injection versus sub-tenon's infusion of triamcinolone acetonide for refractory diabetic macular edema: A randomized clinical trial. Invest Ophthalmol Vis Sci 46:3845–3849, doi:10.1167/iovs.05-0297
Lee CM, Olk J, Akduman L (2000) Combined modified grid and panretinal photocoagulation for diffuse diabetic macular edema and proliferative diabetic retinopathy. Ophthalmic Surg Lasers 31:292–300
Shimura M, Nakazawa T, Yasuda K, Shiono T, Nishida K (2007) Pretreatment of posterior subtenon injection of triamcinolone acetonide has beneficial effects for grid pattern photocoagulation against diffuse diabetic macular edema. Br J Ophthalmol 91:449–454, doi:10.1136/bjo.2006.106336
Shimura M, Yasuda K, Shiono T (2006) Posterior subtenon injection of triamcinolone acetonide prevents pan-retinal photocoagulation-induced visual dysfunction in patients with severe diabetic retinopathy and good vision. Ophthalmology 113:381–387, doi:10.1016/j.ophtha.2005.10.035
Lee SJ, Kim ES, Geroski DH, McCarey BE, Edelbauser HF (2008) Pharmacokinetics of intraocular drug delivery of oregon green 488-labeled triamcinolone by subtenon injection using ocular fluorophotometry in rabbit eyes. Invest Ophthalmol Vis Sci 49:4506–4514, doi:10.1167/iovs.08-1989
Inoue M, Takeda K, Morita K, Yamada M, Tanigawa Y, Oguchi Y (2004) Vitreous concentration of triamcinolone acetonide in human eyes after intravitreal or subtenon injection. Am J Ophthalmol 138:1046–1048, doi:10.1016/j.ajo.2004.05.028
Thomas ER, Wang J, Ege E, Mdsen R, Haisworth DP (2006) Intravitreal triamcinolone acetonide concentration after subtenon injection. Am J Ophthalmol 142:860–861, doi:10.1016/j.ajo.2006.05.023
Iwao K, Inatani M, Kawaji T, Koga T, Mawatari Y, Tanihara H (2007) Frequency and risk factors for intraocular pressure elevation after posterior sub-Tenon capsule triamcinolone acetonide injection. J Glaucoma 16:251–256, doi:10.1097/IJG.0b013e31802d696f
Hirooka K, Shiraga F, Tanaka S, Baba T, Mandai H (2006) Risk factors for elevated intraocular pressure after trans-tenon retrobulbar injection of triamcinolone. Jpn J Ophthalmol 50:235–238, doi:10.1007/s10384-005-0306-9
Rozsa FW, Reed DM, Scott KM, Pawar H, Moroi SE, Kijek TG, Krafchak CM, Othman MI, Vollrath D, Elner VM, Richards JE (2006) Gene expression profile of human trabecular meshwork cells in response to long-term dexamethasone exposure. Mol Vis 12:125–141
Clark AF, Wilson K, McCartney MD, Miggans ST, Kunkle M, Howe W (1994) Glucocorticoid-induced formation of cross-linked actin networks in cultured human trabecular meshwork cells. Invest Ophthalmol Vis Sci 35:281–194
Saadat F, Raji A, Zomorodian K, Eslami MB, Pezeahiki M, Khorramizadeh MR, Aalizadeh N (2003) Alteration in matrixmetalloproteinases (MMPS) activity in fibroblast cell line by dexamethasone: A possible mechanism in corticosteroid-induced glaucoma. Iran J Allergy Asthma Immunol 2:145–148
Coca-Prados M, Escribano J (2007) New perspectives in aqueous humor secretion and in glaucoma: the ciliary body as a multifunctional neuroendocrine gland. Prog Retin Eye Res 26:239–262, doi:10.1016/j.preteyeres.2007.01.002
Kaiser PK, Goldberg MF, Davis AA, Group. TAACS (2007) Posterior juxtascleral depot administration of anecortave acetate. Surv Ophthalmol 52:S62–S69, doi:10.1016/j.survophthal.2006.10.015
Inatani M, Iwao K, Kawaji T, Hirano Y, Ogura Y, Hirooka K, Shiraga F, Nakanishi Y, Yamamoto H, Negi A, Shimonagano Y, Sakamoto T, Shima C, Matsumura M, Tanihara H (2008) Intraocular pressure elevation after injection of triamcinolone acetonide: A multicenter retrospective case-control study. Am J Ophthalmol 145:676–681, doi:10.1016/j.ajo.2007.12.010
Meyer E, Ludatscher RM, Miller B, Lichtig C (1992) Connective tissue of the orbital cavity in retinal detachment: an ultrastructural study. Ophthalmic Res 24:365–371
Acknowledgements
The authors thank Dr. Norio Sugimoto in Theranostic Instruments Research Laboratories for technical support of statistical analyses, and also thank Dr. Dee Worman for English editing and useful comments. This study was supported by 2006-2008 scientific grants in aid for clinical research by NTT East Japan Tohoku Hospital
Author information
Authors and Affiliations
Corresponding author
Additional information
Proprietary Interest None.
Rights and permissions
About this article
Cite this article
Shimura, M., Yasuda, K., Nakazawa, T. et al. Drug reflux during posterior subtenon infusion of triamcinolone acetonide in diffuse diabetic macular edema not only brings insufficient reduction but also causes elevation of intraocular pressure. Graefes Arch Clin Exp Ophthalmol 247, 907–912 (2009). https://doi.org/10.1007/s00417-009-1074-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-009-1074-x