Abstract
Background
The pathogenesis of polypoidal choroidal vasculopathy (PCV), and even its clinical features, are controversial. Previous histopathological studies have identified different features; either dilated choroidal vessels or intra-Bruch’s neovascularization. These differences might be partly attributable to the influence of the disease stage. We therefore evaluated the clinical features of early and late stage PCV.
Methods
The medical records of 110 eyes of 97 PCV patients were retrospectively reviewed. The time between the subjective onset of visual abnormality and examination at our clinic and the greatest linear dimension of the total lesion at the first examination were investigated. The period of disturbed vision and lesion size data were placed in ascending order to determine the first quartile point. Eyes with both values at or below the first quartile point were classified as ‘small–short’ (early stage). Eyes with both values equal to at least the third quartile point were classified as ‘large–long’ (late stage). Fundus photography, indocyanine green and fluorescein angiography, visual acuity, and clinical course were compared.
Results
Twelve eyes from 12 patients were small-short cases (period of disturbed vision of 1 month or less, lesion size 2.0 disc diameters or less). Eleven eyes from ten patients were large-long cases (period of disturbed vision 36 months or more, lesion size at least 5.0 disc diameters). The large-long eyes were characterized by occult choroidal neovascular membrane or scar tissue secondary to exudative age-related macular degeneration. Noticeable in the small-short eyes were atrophic changes in the retinal pigment epithelium, choroidal vessel hyperpermeability and pulsation. The visual prognosis and clinical course were different between the groups.
Conclusions
The difference of clinical features between the groups might reflect different disease stages, although not all of the features observed in the small-short group appeared to represent the early stages of those recorded in the large-long group. Thus, the variation in histopathologic features among previous reports might be partly attributable to differences in disease stage.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Polypoidal choroidal vasculopathy (PCV) is characterized by branching of the choroidal vessels, with polyp-like aneurysmal dilations visible as reddish-orange structures in the peripapillary and/or macular areas. PCV shows recurrences and spontaneous resolutions of leakage or haemorrhage, resulting in macular degeneration and visual loss [1–4]. In Asia, PCV is the most common form of macular disease in middle and late life [5–9].
Vascular abnormalities in PCV occur within the inner choroid. Indocyanine green angiography (ICG-A) has identified two elements of such abnormalities: polypoidal structures projecting from the inner choroid towards the outer retina, and a branching vascular network [9–11]. However, the pathogenesis of PCV, and even its clinical features, remain controversial. Some histopathological studies suggest that the polypoidal structures represent choroidal neovascularization (CNV) [12–16], whereas others indicate an abnormality of the inner choroidal vasculature [17–19]. However, since most of these studies examined eyes indicating rather end-stage disease [12–16] and some dealt with cases after treatments [12–19], this diversity in clinical features might be partly attributable to differences reflecting the stage of the disease. Understanding could be improved by evaluating the clinical and histopathologic features of the various disease stages, particularly at the early stage. We therefore evaluated the clinical features of PCV during earlier and late disease stages, in terms of lesion size and disease duration.
Methods
Medical records of patients diagnosed with PCV between July 1998 and December 2005 at Kagoshima University Hospital, Japan, were retrospectively reviewed. Clinical diagnosis of PCV was based on ICG-A observation of polypoidal vessels corresponding to reddish-orange lesions that were visible on fundus examination. Patients were included whose medical records indicated when subjective visual impairment first developed, and who had undergone ICG-A. Patients were excluded who had not undergone ICG-A, together with those in whom accompanying eye diseases were causing the central visual impairment, thus obscuring the ability to judge whether the onset of subjective visual impairment was due to PCV. This study adhered to the tenets of the Declaration of Helsinki, and was approved by the Institutional Review Board of Kagoshima University Hospital.
Definitions of earlier-stage and later-stage PCV and subject selection
Earlier-stage cases were defined as having small lesions and a short period of disturbed vision (‘small-short’). Later-stage cases were defined as having large lesions and a long period of disturbed vision (‘large-long’). Patient selection was based on medical records from the time of the subjective onset of visual abnormality until the time of initial examination at our clinic. The period of disturbed vision was expressed in months (rounded up to the nearest month). The greatest linear dimension of the total lesion (reddish-orange lesion or polypoidal structures, serous/haemorrhagic detachment of the retinal pigment epithelium [RPE], thick contiguous blood, or atrophic change of the RPE) was measured using colour fundus photographs and confirmed by fluorescein angiography. The lesion size was expressed as the number of disc diameters (DD) of the maximum diameter (rounded up to the nearest decimal point). The period of disturbed vision and lesion diameter data were placed in ascending order, and the value at the quartile point from the smallest (the first quartile point) was determined. Eyes with a period of disturbed vision and lesion diameter at the initial examination below the first quartile point were designated as small-short. Eyes equal or greater to the third quartile point were designated as large-long.
Items investigated
We investigated several items retrospectively from medical records: (1) previous treatment of fundus lesions in the affected eye, (2) associated fundus findings, (3) ICG-A (Topcon, Tokyo, Japan) and fluorescein angiography (FA) findings, (4) best corrected visual acuity (BCVA) at the first examination, (5) BCVA at the last follow-up visit, and (6) clinical course. The first four items were determined from the initial examination by at least two observers (AO and MH).
The logarithmic value of the minimal angle of resolution (logMAR) BCVA values at the initial and the final examinations were statistically analysed using a Wilcoxon signed-ranks test. ‘Counting fingers’ visual acuity was defined as a BCVA of 0.001 or a logMAR VA of 3.0.
Results
Selection of small-short and long-large cases
In total, 102 eyes from 94 Japanese patients (26 women and 68 men) met the criteria. The average age at presentation was 69 years (range = 47–91 years). In addition to the 102 eyes, there were five asymptomatic patients with PCV that was detected during a health check or while examining disease in the other eye. Although the lesion size was small among patients in this group, suggesting early stage disease, it is difficult for such patients to determine the period of disturbed vision, and therefore their clinical findings were evaluated as a special category. The time from the subjective onset of visual deterioration in the affected eye until the examination at our clinic is shown in Fig. 1. For the period of disturbed vision, the first quartile point was 1 month and the third quartile point was 36 months. Fig. 2 shows the number of affected eyes for each lesion size; the most common size was 2.0 DD. For the lesion diameter, the first quartile point was 2.0 DD and the third quartile point was 5.0 DD. The relationship between disease period and lesion size is shown in Fig. 3.
Consequently, 12 eyes from 12 patients were designated as small-short (disease period 1 month or less, lesion size 2.0 DD or less). These patients had the following characteristics: mean age, 69.8 years (range = 56–82 years); mean disease period, 1 month; mean lesion size, 1.8 DD; mean follow-up time after PCV diagnosis, 32 months (range = 19–53 months). Eleven eyes from 10 patients were classified as large-long (disease period 36 months or more, lesion size at least 5.0 DD). These patients had the following characteristics: mean age, 72.2 years (range = 69–79 years); mean disease period, 76.4 months; mean lesion size, 5.9 DD; mean follow-up time after PCV diagnosis, 48 months (range = 7–107 months).
Clinical findings
Previous history of fundus lesion treatment in affected eye
None of the small–short eyes had received previous treatment for macular disease. Four of the large-long eyes had a history of treatment with laser photocoagulation (two cases for age-related macular degeneration (AMD), one for PCV, one for macular disease of an unknown type), and one eye had previously been treated by vitrectomy for vitreous haemorrhage.
Associated fundus findings
Associated fundus findings included serous/haemorrhagic detachment of the RPE and neurosensory retina, subretinal haemorrhage, hard exudates, cystoid macular edema, visible large vascular trunks, fibrotic scar tissue, atrophic scarring, and subretinal CNV (Table 1). The first four findings were observed less frequently in the small-short group than in the large-long group. Polypoidal vessels alone, with or without mild atrophic changes in the RPE, were observed in two small-short eyes (Fig. 4a) but not in any large-long eyes. Large radial vascular trunks or a large vascular network were visible as red vessels superimposed over the partly yellow-coloured larger choroidal vessels in seven large-long eyes (Fig. 5a). Some large-long eyes, but no small-short eyes, had fibrotic scar tissue, atrophic scarring, and/or subretinal CNV.
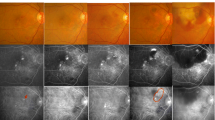
a Fundus photograph of the right eye of a 64-year-old-man with 25-day history of diminished right visual acuity showing an orange-reddish structure (arrow) and slight atrophic change of the RPE (arrowheads). b Fluorescein angiogram showing window defects (arrowheads) in the RPE. c ICG angiogram (392 seconds after dye injection) revealed a polypoidal structure (arrow) and hyperpermeability of choroidal veins within the areas indicated by arrowheads in Figure 4b
a Colour fundus photograph of a 67-year-old-woman with orange-reddish structures (arrowheads) accompanied with serous/haemorrhagic detachment of the neurosensory retina and RPE, hard exudates, subretinal haemorrhage, and cystoid macular edema. She noticed visual loss in this eye 5 years ago, and has been observed as retinal macroaneurysm in a clinic. b Fluorescein angiography (79 seconds after dye injection) shows a small classic area of choroidal neovascularization supranasally (arrows) and a large area of elevated stippled hyperfluorescence of the RPE with serous/haemorrhagic detachment of RPE (arrowhead) and blocked fluorescence due to haemorrhage. c Indocyanine green angiogram (414 seconds after dye injection) reveal polypoidal structures (arrowheads) and large vascular network (arrow)
ICG-A and FA findings
Of the 12 small-short eyes assessed by ICG-A, four showed no vascular network, and three showed a vascular network. In the remaining five eyes, thick haemorrhage—including haemorrhagic detachments of the RPE or neurosensory retina—prevented clear confirmatory identification of a vascular network. The regions of vascular network showed window defects (one eye), oozing (one eye), or fibrovascular RPE detachment (one eye) by FA. Three of the four eyes without a vascular network showed window defects (Fig. 4b) corresponding to various degrees of atrophic changes of the RPE, and the affected areas were revealed by ICG-A to be peripheral to polypoidal structures in the three eyes. Choroidal vessels (probably veins) within these areas showed hyperpermeability or focal dilation (Fig. 4b). In ten of the 11 large-long eyes, a vascular network was detected by ICG-A (Fig. 5c), and FA showed staining due to fibrotic scar or atrophic scarring, or fibrovascular RPE detachment (Fig. 5b). Overall, the small-short group had between one and four polypoidal structures, while the large-long group had numerous structures. Pulsation was seen in the polypoidal structures and/or vascular networks in 16.7% of the small-short group, but was not observed in the large-long group. FA and ICG-A examination of the polypoidal structures revealed dye leakage to varying degrees in both groups. Examination of the positional relationship between the total lesion and the fovea by colour fundoscopic images and FA revealed that the sub-juxtafovea area was affected in 83.3% and 90.9% of the small-short group and the large-long group respectively.
Visual acuity at first examination and final review
The relationship between visual acuity at the initial examination and the final review is shown in Fig. 6. In the small-short eyes, the initial logMAR BCVA values were −0.2 to 1.25 (mean = 0.39); the median logMAR BCVA value on presentation was 0.2. At the final examination, the logMAR BCVA values were −0.2 to 1.3 (mean = 0.21); the median logMAR BCVA value was −0.1. The change in logMAR BCVA values at the final examination was not statistically significant (Wilcoxon signed-ranks test, p = 0.331). In the large-long eyes, the initial logMAR BCVA values were 0.2 to 2.0 (mean = 1.38); the median logMAR BCVA value on presentation was 1.5. At the final examination, the logMAR BCVA values were 0.4 to 3.0 (mean = 1.51); the median logMAR BCVA value was 2.0. The change in logMAR BCVA values at the final examination was statistically significant (Wilcoxon signed-ranks test, p = 0.028). A rate of deterioration in logMAR BCVA of at least 0.2 was seen in 25.0% of the small-short eyes and 63.6% of the large-long eyes. When untreated eyes were analyzed, no statistical significant changes in logMAR BCVA values were recorded at the final examination in either the small-short or large-long eyes (Wilcoxon signed-ranks test, p = 0.498 and p = 0.144 respectively).
Clinical course
Five small-short eyes were followed without treatment and seven were treated by conventional laser photocoagulation (one eye), photodynamic therapy (PDT) (one eye), sub-Tenon’s injection of triamcinolone acetonide (four eyes), and surgical excision of subretinal tissue (one eye). One untreated eye that had manifested polypoidal vessels with mild atrophic change in the RPE retained stable fundus during follow-up. In the remaining four untreated eyes, exudative changes disappeared and polypoidal structures changed from reddish-orange to white, with or without disappearance of the corresponding polypoidal structures when examined using ICG-A. In the treated eyes, exudative changes disappeared except for one eye which had increased cystoid macular edema and hard exudates 5 months after a PDT session. With the exception of this eye, the total lesion size in the small-short eyes did not enlarge beyond two disc diameters, although it is difficult to compare the efficacy among the treatment options because of the small sample size. Six large-long eyes were followed without treatment because visual acuity was so poor that no treatment was deemed suitable, whereas the remaining five were treated by conventional laser photocoagulation (two eyes), sub-Tenon’s or vitreal injection of triamcinolone acetonide (two eyes), and/or vitreal injection of bevacizumab (two eyes). At the final examination of the five treated eyes, lesions were replaced by fibrotic scarring in one eye, exudative changes remained in three eyes, and disappeared in one eye (treated by conventional laser photocoagulation). At the final examination of the six untreated eyes, the lesion had been replaced by a fibrotic scar (one eye), the exudative changes had remained (four eyes), or serous detachment of the neurosensory retina had disappeared leaving atrophic scarring (one eye).
Clinical features of the special category
The five eyes in the special category showed similar clinical features to the small-short eyes, such as polypoidal vessels alone with or without mild RPE atrophy in fundus findings (two eyes), pulsation of polypoidal vessels assessed by ICG-A (two eyes), and window defects assessed by FA (four eyes). However, they did not show characteristic features seen in the large-long eyes, such as visible large radial vascular trunks, fibrotic scar, and atrophic scarring (Table 1).
Discussion
We have evaluated the clinical features of early and late stage PCV. Small lesions are likely to occur at an earlier stage and large lesions at a later stage. Throughout our evaluation, we considered the period of disturbed vision up until the initial examination. The main limitation of this is that the period of disturbed vision was defined as beginning when the patient subjectively felt visual impairment, which did not necessarily represent the time of the onset of the disease. For example, cases in which the lesion initially occurred in the extrafovea and subsequently involved the fovea might have had a delayed onset of subjective symptoms. However, although a patient who initially presented with these symptoms at the clinic might have been classified with short disease duration, they would have been excluded from the small lesion category because the lesion was enlarged. We therefore selected small-short cases as representative of early stage PCV, and large-long cases as representative of later stage PCV.
There were noticeable differences between both groups, but there were also common features. Since none of the small-short cases had a history of treatment for macular disease, the features of these cases can be regarded as representing the natural pathological course. Serous/haemorrhagic detachment of the RPE was most common in both groups, suggesting that PCV is a disease that underlies the RPE. Polypoidal vessels alone, with or without mild RPE atrophic changes in the RPE which corresponded to the ‘window defects’ in the RPE adjacent to the polypoidal structures assessed using FA, were found in the small-short group. Together with ICG-A findings of hyperpermeability or dilation in the choroidal vessels within the region, this suggests that the congestion and/or hyperpermeability of choroidal vessels causes, or is linked to, impairment of the overlying RPE in the area adjacent to polypoidal structures. Large radial vascular trunks or a large vascular network, fibrotic scar tissue, and atrophic scarring were characteristic of the large-long cases. These findings are known to be associated with the cicatricial stage of exudative AMD [20]. Thus, the large-long eyes resemble occult CNV or scar tissue secondary to exudative AMD, whilst atrophic changes in the RPE and choroidal vessel hyperpermeability are characteristic of at least some small-short eyes. The five eyes in the special category might have belonged to the small-short eyes category, as their clinical features were similar. Pulsation was evident in the polypoidal structures and vascular network in 16.7% of the small-short cases, but not in the large-long cases. This suggests that pulsatile polypoidal vessels in PCV might be representative of an early stage of the disease. Pulsatile vessels seem to be related to the choroidal vasculature [21]. The pathogenesis of PCV in the small-short group might therefore result from certain morphological characteristics of the choroidal vasculature: in the choroid, the arterioles and venules frequently intersect, particularly within the macular and peripapillary areas, and the choroidal vessels in these areas are tortuous.
The small-short group showed a lower rate of decline in visual acuity during observation than the large-long group. In most of the cases the exudative changes disappeared except for one eye, which progressed. This suggests that the lesions in some small-short cases might remain at a similar size for a relatively long period, as suggested by the results in Fig. 3, which shows cases with small lesions and a long disease period. However, the large-long group showed a higher rate of decline in visual acuity. Moreover, the exudative changes were either prolonged or developed into a fibrotic scar. Uyama et al. [5] conducted a retrospective evaluation of PCV patients, and reported that most of the patients had a favorable course, although some experienced recurrence and a few eyes developed disciform scarring. Their findings suggest the existence of subgroups with respect to visual prognosis. In the present study, it is difficult to compare the efficacy of treatment between the two groups: we did not carry out treatments for some large-long cases because visual acuity was so poor that no treatment was deemed suitable, whilst we observed some small-short cases without treatment because they did not manifest severe exudative changes. Although treatments for the small-short cases seem effective in keeping vision relatively good, it is also difficult to compare the efficacy among the treatment options because of the small sample size in the both groups.
Although we defined small-short cases as early stage and large-long cases as later stage, it is unclear whether all of the former represented the early stage of the disease in the latter group; that is, whether both groups had the same pathogenesis. Some small-short cases seemed to develop into large-long cases; one eye revealed cystoid macular edema and fibrovascular RPE detachment that did not disappear and enlarged after PDT with decreased visual acuity. Unexpectedly, however, many small-short cases did not vary over time. This slow rate of progression, together with the lower rate of decline in visual acuity, seems to be different from the findings relating to CNV secondary to the typical exudative AMD, most of which show a lesion enlargement and decrease of vision with time. This suggests that some small-short eyes might have pathological origins other than CNV. Candidates include the abnormality of the inner choroidal vasculature reported in previous studies [17–19]. Long-term observation of the natural course of small-short cases could resolve this uncertainty.
In conclusion, we studied clinical features of early and late PCV cases, identified according to lesion size and disease duration. The variation in clinical features between the groups may reflect different disease stages to some extent. However, not all of the features observed in the small-short group appear to represent the early stages of the large-long group. Thus, the different histopathological features as reported by previous studies [12–19] are likely to reflect differences in both disease stage and pathology.
References
Stern RM, Zakov ZN, Zegarra H, Gutman FA (1985) Multiple recurrent serosanguineous retinal pigment epithelial detachments in black women. Am J Ophthalmol 100:560–569
Yannuzzi LA, Sorenson J, Spaide RF, Lipson B (1990) Idiopathic choroidal vasculopathy (IPCV). Retina 10:1–8
Kleiner RC, Brucker AJ, Johnston RL (1990) The posterior uveal bleeding syndrome. Retina 10:9–17
Perkovich BT, Zakov ZN, Berlin LA, Weidenthal D, Avins LR (1990) An update on multiple recurrent serosanguineous retinal pigment epithelial detachments in black women. Retina 10:18–26
Uyama M, Matsubara T, Fukushima I, Matsunaga H, Iwashita K, Nagai Y, Takahashi K (1999) Idiopathic polypoidal choroidal vasculopathy in Japanese patients. Arch Ophthalmol 117:1035–1042
Kwok AK, Lai TY, Chan CW, Neoh EL, Lam DS (2002) Polypoidal choroidal vasculopathy in Chinese patients. Br J Ophthalmol 86:892–897
Sho K, Takahashi K, Yamada H, Wada M, Nagai Y, Otsuji T, Nishikawa M, Mitsuma Y, Yamazaki Y, Matsumura M, Uyama M (2003) Polypoidal choroidal vasculopathy: incidence, demographic features, and clinical characteristics. Arch Ophthalmol 121:1392–1396
Wen F, Chen C, Wu D, Li H (2004) Polypoidal choroidal vasculopathy in elderly Chinese patients. Graefe’s Arch Clin Exp Ophthalmol 242:625–629
Yannuzzi LA, Ciardella A, Spaide RF, Rabb M, Orlock DA (1997) The expanding clinical spectrum of idiopathic polypoidal choroidal vasculopathy. Arch Ophthalmol 115:478–485
Spaide RF, Yannuzzi LA, Slakter JS, Sorenson J, Orlach DA (1995) Indocyanine green videoangiography of idiopathic polypoidal choroidal vasculopathy. Retina 15:100–110
Yuzawa M, Mori R, Kawamura A. (2005) The origins of polypoidal choroidal vasculopathy. Br J Ophthalmol 89:602–607
MacCumber MW, Dastgheib K, Bressler NM, Chan CC, Harris M, Fine S, Green WR (1994) Clinicopathologic correlation of the multiple recurrent serosanguineous retinal pigment epithelial detachments syndrome. Retina 14:143–152
Spraul CW, Grossniklaus HE, Lang GK (1977) Idiopathische polypose choroidale Vaskulopathie (IPCV). Klin Monatsbl Augenheilkdd 210:405–406
Lafaut BA, Aisenbrey S, Van den Broecke C, Bartz-Schmidt KU, Heimann K (2000) Polypoidal choroidal vasculopathy pattern in age-related macular degeneration. A clinicopathologic correlation. Retina 20:650–654
Terasaki H, Miyake Y, Suzuki T, Nakamura M, Nagasaka T (2002) Polypoidal choroidal vasculopathy treated with macular translocation: clinical pathological correlation. Br J Ophthalmol 86:321–327
Rosa RH Jr, Davis JL, Eifrig CW (2002) Clinicopathologic correlation of idiopathic polypoidal choroidal vasculopathy. Arch Ophthalmol 120:502–508
Kuroiwa S, Tateiwa H, Hisatomi T, Ishibashi T, Yoshimura N (2004) Pathologic features of surgically excised polypoidal choroidal vasculopathy membranes. Clin Exp Ophthalmol 32:292–302
Okubo A, Sameshima M, Uemura A, Kanda S, Ohba N (2002) Clinicopathological correlation of polypoidal choroidal vasculopathy revealed by ultrastructural study. Br J Ophthalmol 86:1093–1098
Nakajima M, Yuzawa M, Shimada H, Mori R (2004) Correlation between indocyanine green angiographic findings and histopathology of polypoidal choroidal vasculopathy. Jpn J Ophthalmol 48:249–255
Green WR, Enger C (1993) Age-related macular degeneration histopathology studies. The 1992 Lorenz E. Zimmerman Lecture. Ophthalmology 100:1519–1535
Okubo A, Ito M, Sameshima M, Uemura A, Sakamoto T (2005) Pulsatile blood flow in the polypoidal choroidal vasculopathy. Ophthalmology 112:1436–1441
Author information
Authors and Affiliations
Corresponding author
Additional information
Grant information: This work was supported by research grant number 19592028 from the Japanese Ministry of Education, Science, Sports, and Culture.
Disclosures: The authors report no conflicting interests.
Rights and permissions
About this article
Cite this article
Okubo, A., Hirakawa, M., Ito, M. et al. Clinical features of early and late stage polypoidal choroidal vasculopathy characterized by lesion size and disease duration. Graefes Arch Clin Exp Ophthalmol 246, 491–499 (2008). https://doi.org/10.1007/s00417-007-0680-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-007-0680-8