Abstract
Background
To evaluate the effects of thalidomide treatment on the temporal course of TNF-α, VEGF production and the histopathological changes in ischemia/reperfusion (I/R) injured guinea pigs retina.
Methods
Control, ischemia, and thalidomide/ischemia groups including seven animals each were formed. Retinal ischemia was induced in male guinea pigs by cannulating anterior chambers and lifting the bottle to a height of 205 cm for 90 min in the ischemia and thalidomide/ischemia groups. The thalidomide/ischemia group received thalidomide (300 mg/kg/day) via nasogastric tube 24 h before ischemia and during 7 days of reperfusion. Guinea pigs were sacrificed for histopathological examination to evaluate the mean thickness of the inner plexiform layer (IPL), polymorphonuclear leukocyte (PMNL) infiltration, and biochemical analysis of retinal VEGF and TNF-α levels by ELISA.
Results
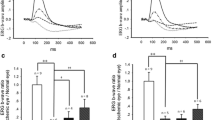
The mean retinal VEGF and TNF-α levels of the control, ischemia, and thalidomide/ischemia groups were 10.22 ± 2.58 and 270.41 ± 69.77 pg/ml; 35.80 ± 5.97 and 629.93 ± 146.41 pg/ml; 19.01 ± 3.01 and 340.93 ± 158.26 pg/ml, respectively. The retinal VEGF levels were significantly higher in I/R injured groups. The thalidomide/ischemia group retinal VEGF level was significantly lower versus the ischemia group. The retinal TNF-α levels were significantly elevated in the ischemia group, but no difference was observed between the thalidomide/ischemia and control groups. Also, the retinal TNF-α level was significantly lower in the thalidomide/ischemia group versus the ischemia group. The mean thickness of IPL and PMNL infiltration showed no difference between the control and thalidomide/ischemia groups. However, there was a significant difference between the control and ischemia groups.
Conclusion
Thalidomide treatment decreases PMNL infiltration, retinal edema, VEGF, and TNF-α synthesis following I/R injury to the guinea pig retina.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Retinal ischemia, which occurs as the consequence of a primary ocular disease or of a systemic disease, is a common cause of visual impairment and even irreversible blindness in the world. To counteract retinal ischemia, the tissue increases the production and release of angiogenic stimulators such as tumor necrosis factor-alpha (TNF-α) and vascular endothelial growth factor (VEGF) in order to promote angiogenesis that is the key event in pathogenesis [1, 2].
VEGF is a potent angiogenic and vasopermeability factor that has been initially identified in the bovine retina and later in the human retina [3, 4]. Intravitreal injections of recombinant human VEGF into healthy monkey eyes produce iris neovascularization and glaucoma [5]. Conversely, injection of VEGF antibodies to human vitreous causes regression of neovascularization [6].
TNF-α is a macrophage/monocyte-derived pluripotent polypeptide mediator that stimulates angiogenesis in the cornea and chorioallantoic membrane in vivo by its direct effect and also enhances the synthesis of VEGF, which is an angiogenic stimulator [7–10]. Additionally, TNF-α is a chemoattractant for leukocytes and causes breakdown of the blood-retinal barrier [11, 12].
In the 1950s, thalidomide [N(α).-phthalimidoglutarimide] (THD) was marketed as a sedative in Europe, but eventually withdrawn when it was found to be a potent teratogen. In 1961, shortly after its withdrawal, the anti-inflammatory properties were observed in erythema nodosum leprosum patients [13]. The anti-angiogenic effect of THD in a rabbit corneal neovascularization model [14] and inhibitor effect on TNF-α synthesis [15] were discovered in later years. To our knowledge the effect of THD in a model of retinal ischemia has not been studied. In our study we investigated the influence of THD treatment on TNF-α, VEGF production, and the histopathological changes in I/R injured guinea pig retinas.
Materials and methods
Experimental protocol
Twenty-one pigmented male guinea pigs weighing between 470 and 640 g were treated in accordance with the Association for Research in Vision and Ophthalmology in Experimental Research Center of our university upon the consent of the local ethics committee. Three groups (control, ischemia, and thalidomide/ischemia) including seven animals each were formed.
Induction of ischemia and reperfusion
Intramuscular ketamine HCl (50 mg/kg) and xylazine HCl (5 mg/kg) were used for the anesthesia of the animals. Topical anesthetic consisting of 0.005% proparacaine HCl was administered to both eyes of each animal. Pressure-induced retinal ischemia was induced by cannulating both anterior chambers with a 27-gauge needle connected to a bottle of normal saline and lifting the bottle rapidly to a height of 205 cm in order to raise the intraocular pressure to 150 mmHg. This lasted for 90 min, and reperfusion was established by lowering the saline bottle to eye level. Then the eyes were decanulated. In the control group, ischemia/reperfusion (I/R) was not induced, and no drug was administered. In the ischemia group, saline was administered orally via nasogastric tube with a dose of 2 ml/day along the experimental period. In the thalidomide/ischemia group, 300 mg/kg/day of THD was given in 2 ml saline via the same route 24 h before the ischemic insult and during the 7-day reperfusion period. At the 7th day of the experiment, the animals were reanesthetized, and both eyes of all the animals were rapidly enucleated; the animals were sacrificed by intracardiac thiopental sodium (50 mg/kg).
Biochemical assay
One eye of each animal was randomly selected for biochemical assays and the other for histopathological evaluation. The enucleated eyes for biochemical assays were dissected coronally through the pars plana. After removing the vitreous, the retinal tissue was gently peeled off from the choroidea and cut from the optic disc with fine forceps and scissors under an operating microscope and washed with phosphate buffered saline (PBS) (pH 7.4 molar 0.2). The retina was frozen, smashed, and homogenized in PBS at a dilution of 1/20. The supernatants were collected by centrifugation at 150 g for 10 min and stored at −80°C until the assay. Respectively, retinal VEGF and TNF-α levels were analyzed with RayBio Rat VEGF ELISA kit (cat. no.: ELR-VEGF-001c, USA), Biosource Rat TNF-α immunoassay kit (cat. no.: KRC3011, USA). The results were expressed as mean ± standard deviation.
Preparation of tissue samples
The eyes selected for histopathological examination were fixed in 10% formalin immediately after enucleation, and transverse sections passing through the optic nerve were obtained. The samples were embedded in paraffin wax, 5-μm-thick paraffin sections were prepared, and the specimens were stained with hematoxylin and eosin.
Histological changes
An Olympus BX50 light microscope was used for the histopathological evaluation of the tissue sections of three groups in a masked fashion. The quantification of the retinal ischemic damage was made by measuring the thickness of the inner plexiform layer (IPL) of the retina [16, 17]. The measurements were made with an ocular micrometer ×400 magnification within 0.5 mm from the optic nerve. Three measurements from adjacent locations in each nasal and temporal hemisphere were obtained, and a mean retinal thickness for each eye was obtained by averaging the six measurements. Polymorphonuclear leukocyte (PMNL) infiltration between the internal limiting membrane (ILM) and IPL was evaluated. The number of PMNLs was counted in ten high power fields (HPF)/slide. The results were expressed as mean ± standard deviation per HPF.
Statistical analysis
Statistical analyses were carried out by employing the Statistical Package for Social Sciences soft-ware 11.0 for Windows (SPSS, Inc., Chicago, IL). The Mann-Whitney U test was used, and a p value less than 0.05 was considered statistically significant.
Results
The mean retinal VEGF levels of the control, ischemia, and thalidomide/ischemia groups were 10.22 ± 2.58 pg/ml, 35.80 ± 5.97 pg/ml, and 19.01 ± 3.01 pg/ml, respectively (Fig. 1). The retinal VEGF levels were significantly elevated in the ischemia (p = 0.001) and thalidomide/ischemia (p = 0.001) groups compared with the control (Fig. 1). However, the retinal VEGF level was significantly lower in the thalidomide/ischemia group versus the ischemia group (p = 0.001) (Fig. 1).
The mean retinal TNF-α levels of the control, ischemia, and thalidomide/ischemia groups were 270.41 ± 69.77 pg/ml, 629.93 ± 146.41 pg/ml, and 340.93 ± 158.26 pg/ml, respectively (Fig. 2). The retinal TNF-α levels were significantly elevated in the ischemia group compared with the control (p = 0.001) (Fig. 2). There was no significant difference between the thalidomide/ischemia and the control group (p = 0.620) (Fig. 2). Also, the retinal TNF-α levels were significantly lower in the ischemia/thalidomide group versus the ischemia group (p = 0.017) (Fig. 2).
Figure 3 shows the histological sections of retina in non-ischemic control eye (Fig. 3a), in retina subjected to I/R injury without treatment (Fig. 3b), and in I/R injured retina treated with THD (Fig. 3c), respectively. The mean thickness of the IPL in the control, ischemia, and thalidomide/ischemia groups were 18.92 ± 3.18, 29.28 ± 7.31, and 21.07 ± 3.49 μm, respectively (Fig. 4). In the statistical analysis, there was no difference between the ischemia and the thalidomide/ischemia group (p = 0.07) or between the control and the thalidomide/ischemia group (p = 0.318). However there was significant difference between the control and the ischemia group (p = 0.001). The mean PMNL infiltrations in the control, ischemia, and thalidomide/ischemia groups were 0.28 ± 0.48, 3.28 ± 2.28, and 0.85 ± 0.89, respectively. There was no statistical difference between the control and the thalidomide/ischemia groups (p = 0.259). However, there was a statistically significant difference between the control and the ischemia groups (p = 0.011) and between the ischemia and the thalidomide/ischemia groups (p = 0.038).
Hematoxylin and eosin staining of the retina. a. Control group retina. GCL: ganglion cell layer; IPL: inner plexiform layer; INL: inner nuclear layer; OPL: outer plexiform layer; ONL: outer nuclear layer; PL: photoreceptor layer. b. PMNLs infiltration in the internal limiting membrane (vertical arrows) and inner plexiform (horizontal arrow) layers of retina in the ischemia group. c. Retina from guinea pigs treated with oral thalidomide
Discussion
Angiogenesis is tightly regulated by the action of both stimulatory and inhibitory factors that are in balance. Hypoxia shifts the balance in favor of angiogenesis. Many diseases such as diabetic retinopathy, occlusion of the central retinal artery, sickle cell anemia, and retinopathy of prematurity are driven by this unregulated angiogenesis.
VEGF is a potent angiogenic factor that regulates both physiological and pathological neovascularization [9, 10] secreted from pericytes, retinal endothelial, retinal pigment epithelial, and ganglion cells in response to hypoxia [2, 18, 19]. In our study, in the I/R injured groups we have also shown that the ischemia has increased the production of VEGF.
The antiangiogenic effects of THD initially became evident in the 1950s, but the mechanisms of its action were documented by subsequent studies. Blocking the action of basic fibroblast growth factor (bFGF) and reducing the release of matrix metalloproteinase-2 (MMP-2) are probable mechanisms of its action [14, 20]. In recent studies additional mechanisms were shown such as depletion of VEGF membrane receptors neuropilin-1, Flk-1 [21], and direct inhibition of VEGF secretion [22]. In our study we also demonstrated that THD decreased the VEGF synthesis.
Ischemia/reperfusion injury is a multistage process. Immediate hypoxia as well as return of oxygenated blood damages the tissue. Free radical generation and inflammatory-immunologic reactions are involved in the pathogenesis of I/R injury.
TNF-α increases nitric oxide (NO) production via induction of nitric oxide synthase (NOS)-2 in astrocytes [23]. NO interacts with \( \raise0.145em\hbox{${\scriptscriptstyle \bullet}$}O^{ - }_{2} \) and form peroxynitrite free radicals (·ONOO¯) that cause lipid peroxidation of cellular membranes in the mechanism of ischemic-reperfusion injury [24–26]. THD suppresses NO production via its direct effect and its indirect effect on TNF-α by increasing degradation of its mRNA [27, 28].
TNF-α leads to activation of PMNLs and upregulates cell adhesion molecules both on PMNLs and endothelium, followed by transmigration into the interstitium, release of toxic enzymes, and tissue damage, which is the inflammatory-immunologic reaction part of the I/R injury [29]. THD inhibits transmigration of PMNLs across human umbical vein endothelial cells [30]. The alternative way of its anti-inflammatory action is its inhibitor action on NF-κB (a multi-subunit transcription factor that takes part in the transcriptional regulation of many cytokines and regulation of inflammation mediator genes) activation induced by TNF [31, 32]. Our study demonstrates the first evidence that systemic THD reduces I/R injury in a guinea pig retina via reduction of TNF-α synthesis.
Thickness of the IPL has been selected for the quantification of ischemic retinal damage in the present study. IPL thickness has been associated with I/R injury in a number of studies [16, 17]. The IPL thicknesses have been observed to increase significantly in the ischemic group compared with the control. This result has been interpreted as the consequence of persisting retinal edema till the 7th day. Although statistically not significant, in the thalidomide/ischemia group compared with the ischemia group, the thickness of the IPL has been observed to be decreased. This insignificant decrement in the thickness of IPL in the thalidomide/ischemia groups may be interpreted as the consequence of the antiedematous effect of THD, which has been shown previously during early wallerian degeneration in neurons [33].
In conclusion, oral THD treatment was found to reduce PMNL infiltration, retinal edema, and VEGF and TNF-α synthesis of the retina in guinea pigs after ischemic insult. These effects of THD may have important therapeutic implications in diseases that are driven by ischemia compared to unregulated angiogenesis.
References
Hangai M, Yoshimura N, Honda Y (1996) Increased cytokine gene expression in rat retina following transient ischemia. Ophthalmic Res 28:248–254
Ogata N, Yamanaka R, Yamamoto C, Miyashiro M, Kimoto T, Takahashi K, Maruyama K, Uyama M (1998) Expression of vascular endothelial growth factor and its receptor, KDR, following retinal ischemia-reperfusion injury in the rat. Curr Eye Res 17:1087–1096
Chen CH, Chen SC (1987) Evidence of the presence of a specific vascular endothelial growth factor in fetal bovine retina. Exp Cell Res 169:287–295
Shima DT, Adamis AP, Ferrara N, Yeo KT, Yeo TK, Allende R, Folkman J, D’Amore PA (1995) Hypoxic induction of endothelial cell growth factors in retinal cells: identification and characterization of vascular endothelial growth factor (VEGF) as the mitogen. Mol Med 1:182–193
Hofman P, van Blijswijk BC, Gaillard PJ, Vrensen GF, Schlingemann RO (2001) Endothelial cell hypertrophy induced by vascular endothelial growth factor in the retina: new insights into the pathogenesis of capillary nonperfusion. Arch Ophthalmol 119:861–866
Avery RL (2006) Regression of retinal and iris neovascularization after Intravitreal bevacizumab (Avastin) treatment. Retina 26:352–354
Frater-Schroder M, Risau W, Hallmann R, Gautschi P, Bohlen P (1987) Tumor necrosis factor type alpha, a potent inhibitor of endothelial cell growth in vitro, is angiogenic in vivo. Proc Natl Acad Sci USA 84:5277–5281
Leibovich SJ, Polverini PJ, Shepard HM, Wiseman DM, Shively V, Nuseir N (1987) Macrophage-induced angiogenesis is mediated by tumour necrosis factor-alpha. Nature 329:630–632
Ferrara N, Davis-Smyth T (1997) The biology of vascular endothelial growth factor. Endocr Rev 18:4–25
Folkman J, Shing Y (1992) Angiogenesis. J Biol Chem 267:10931–10934
Majka S, McGuire PG, Das A (2002) Regulation of matrix metalloproteinase expression by tumor necrosis factor in a murine model of retinal neovascularization. Invest Ophthalmol Vis Sci 43:260–266
Derevjanik NL, Vinores SA, Xiao WH, Mori K, Turon T, Hudish T, Dong S, Campochiaro PA (2002) Quantitative assessment of the integrity of the blood-retinal barrier in mice. Invest Ophthalmol Vis Sci 43:2462–2467
Sheskin J (1965) Thalidomide in the treatment of lepra reactions. Clin Pharmacol Ther 6:303–306
D’Amato RJ, Loughnan MS, Flynn E, Folkman J (1994) Thalidomide is an inhibitor of angiogenesis. Proc Natl Acad Sci USA 91:4082–4085
George A, Marziniak M, Schafers M, Toyka KV, Sommer C (2000) Thalidomide treatment in chronic constrictive neuropathy decreases endoneurial tumor necrosis factor-alpha, increases interleukin-10 and has long-term effects on spinal cord dorsal horn met-enkephalin. Pain 88:267–275
Hughes WF (1991) Quantitation of ischemic damage in the rat retina. Exp Eye Res 53:573–582
Szabo ME, Droy-Lefaix MT, Doly M, Carre C, Braquet P (1991) Ischemia and reperfusion-induced histologic changes in the rat retina. Demonstration of a free radical mediated mechanism. Invest Ophthalmol Vis Sci 32:1471–1478
Shima DT, Adamis AP, Ferrara N, Yeo KT, Yeo TK, Allende R, Folkman J, D’Amore PA (1995) Hypoxic induction of endothelial cell growth factors in retinal cells: identification and characterization of vascular endothelial growth factor (VEGF) as the mitogen. Mol Med 1:182–193
Aiello LP, Northrup JM, Keyt BA, Takagi H, Iwamoto MA (1995) Hypoxic regulation of vascular endothelial growth factor in retinal cells. Arch Ophthalmol 113:1538–1544
Kenyon BM, Browne F, D’Amato RJ (1997) Effects of thalidomide and related metabolites in a mouse corneal model of neovascularization. Exp Eye Res 64:971–978
Yabu T, Tomimoto H, Taguchi Y, Yamaoka S, Igarashi Y, Okazaki T (2005) Thalidomide-induced antiangiogenic action is mediated by ceramide through depletion of VEGF receptors, and is antagonized by sphingosine-1-phosphate. Blood 106:125–134
Komorowski J, Jerczynska H, Siejka A, Baranska P, Lawnicka H, Pawlowska Z, Stepien H (2006) Effect of thalidomide affecting VEGF secretion, cell migration, adhesion and capillary tube formation of human endothelial EA.hy 926 cells. Life Sci 78:2558–2563
Yuan L, Neufeld AH (2000) Tumor necrosis factor-alpha: a potentially neurodestructive cytokine produced by glia in the human glaucomatous optic nerve head. Glia 32:42–50
Geyer O, Almog J, Lupu-Meiri M, Lazar M, Oron Y (1995) Nitric oxide synthase inhibitors protect rat retina against ischemic injury. FEBS Lett 374:399–402
Hangai M, Yoshimura N, Hiroi K, Mandai M, Honda Y (1996) Inducible nitric oxide synthase in retinal ischemia-reperfusion injury. Exp Eye Res 63:501–509
Lee JS, Kang Decker N, Chatterjee S, Yao J, Friedman S, Shah V (2005) Mechanisms of nitric oxide interplay with Rho GTPase family members in modulation of actin membrane dynamics in pericytes and fibroblasts. Am J Pathol 166:1861–1870
Lopez-Talavera JC, Cadelina G, Olchowski J, Merrill W, Groszmann RJ (1996) Thalidomide inhibits tumor necrosis factor alpha, decreases nitric oxide synthesis, and ameliorates the hyperdynamic circulatory syndrome in portal-hypertensive rats. Hepatology 23:1616–1621
Moreira AL, Sampaio EP, Zmuidzinas A, Frindt P, Smith KA, Kaplan G (1993) Thalidomide exerts its inhibitory action on tumor necrosis factor alpha by enhancing mRNA degradation. J Exp Med 177:1675–1680
Warren JS (1990) Interleukins and tumor necrosis factor in inflammation. Crit Rev Clin Lab Sci 28:37–59
Dunzendorfer S, Schratzberger P, Reinisch N, Kahler CM, Wiedermann CJ (1997) Effects of thalidomide on neutrophil respiratory burst, chemotaxis, and transmigration of cytokine-and endotoxin-activated endothelium. Naunyn Schmiedebergs Arch Pharmacol 356:529–535
Majumdar S, Lamothe B, Aggarwal BB (2002) Thalidomide suppresses NF-kappa B activation induced by TNF and H2O2, but not that activated by ceramide, lipopolysaccharides, or phorbol ester. J Immunol 168:2644–2651
Yasui K, Kobayashi N, Yamazaki T, Agematsu K (2005) Thalidomide as an immunotherapeutic agent: the effects on neutrophil-mediated inflammation. Curr Pharm Des 11:395–401
Schroder JM, Sellhaus B, Wohrmann T, Kogel B, Zwingenberger K (1995) Inhibitory effects of thalidomide on cellular proliferation, endoneurial edema and myelin phagocytosis during early wallerian degeneration. Acta Neuropathol (Berl) 89:415–419
Acknowledgement
The author has full control of all primary data and agrees to allow Graefe’s Archive to review the data upon request.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study supported by Firot University Research Fund.
Rights and permissions
About this article
Cite this article
Aydoğan, S., Çeliker, Ü., Türkçüoğlu, P. et al. The effect of thalidomide on vascular endothelial growth factor and tumor necrosis factor-α levels in retinal ischemia/reperfusion injury. Graefes Arch Clin Exp Ophthalmol 246, 363–368 (2008). https://doi.org/10.1007/s00417-007-0663-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-007-0663-9