Abstract
Inflammatory serum parameters are intensely investigated in the search of biomarkers for disease activity and treatment response in multiple sclerosis (MS). A reason for contradictory results might be the timing of blood collection for analyzing serum concentrations of inflammatory parameters which are subject to diurnal changes. We included 34 untreated patients with relapsing-remitting MS and 34 age- and sex-matched healthy controls. 12 MS patients showed acute disease activity in corresponding MRI scans. Blood samples were obtained at 7.00, 11.00 am, 2.30, 6.00 and 9.30 pm within 1 day. We determined serum levels of cortisol and inflammatory markers including soluble tumor necrosis factor-beta (sTNF-β), soluble TNF-Receptor-1 (sTNF-R1) and -2 (sTNF-2), soluble vascular adhesion molecule-1 (sVCAM-1) and soluble intercellular adhesion molecule-1 (sICAM-1) by ELISA. We observed significantly higher serum levels of sTNF-R1 (p < 0.001) and sTNF-R2 (p < 0.001) in the morning and a significant decline of sICAM-1 (p < 0.005) and sVCAM-1 (p < 0.001) in the afternoon in both, MS patients and healthy controls. Comparison of diurnal serum levels between MS patients with active versus with non-active disease revealed significantly higher serum levels of sVCAM-1 (p < 0.05) around noon and in the early afternoon in MS patients with active disease. A significant decline of sICAM-1 (p < 0.05) in the afternoon was seen in MS patients with active and non-active disease. Our data indicate that increased awareness of potential diurnal serum concentration changes of biomarkers can eliminate one major cause of biased data as they occur in most of the investigated immunological parameters.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory disease of the human central nervous system (CNS). Although MS pathogenesis is heterogeneous, autoreactive T helper-1 cells are considered to play an important role. Blood-brain barrier (BBB) breakdown and recruitment of activated immune cells from the peripheral blood into the CNS are early events in the development of inflammatory MS lesions [1]. Adhesion molecules, such as intercellular cell adhesion molecule (ICAM) -1, -2, -3, very late activation antigen-4 (VLA-4), leukocyte-function antigen-1 (LFA-1), and vascular cell adhesion molecule-1 (VCAM-1) regulate adherence and diapedesis of immune cells across the microvascular endothelium of the BBB into the CNS [2].
Current anti-inflammatory, immunomodulatory and immunosuppressive treatments aim to delay disease progression during early stages of MS and immunological parameters including cytokines, cell adhesion molecules (AM) or cytokine receptors have intensively been investigated to identify biomarkers for disease activity and treatment responsiveness [3–8]. Adhesion molecules have attracted further interest in MS research since the approval of natalizumab [2]. Natalizumab is a monoclonal antibody directed against the alpha-4 subunit of VLA-4. Changes in surface expression patterns on immune cells from MS patients have been suggested potential biomarkers for monitoring efficacy and side effects during Natalizumab treatment [3]. Serum concentrations of soluble AM, cytokines and shedded cytokine receptors from MS patients have been investigated in numerous studies and found to be altered [3–8]. However, some of the results are contradictory. Diurnal changes in expression levels and serum concentrations of the immunologic parameters are a possible explanation for the observed differences as they might lead to systemic errors due to sampling on different time points for different cohorts [9].
In this regard the known circadian pattern of cortisol should be considered [10]. During the course of a day cortisol causes variations in many immune functions, thus, playing an important role in diurnal changes of immunological markers both in healthy people as well as in MS patients [11]. Moreover, elevated cortisol levels were observed in MS subjects compared to healthy controls and a clinical benefit of night-time administration of glucocorticoid therapy for management of MS relapses has recently been demonstrated [12, 13].
Similar to cortisol, various leukocyte-mediated immune functions show nocturnal increases. In addition, circadian variations in cell adhesion molecule expression have been observed in normal human leukocytes [9]. Our study was initiated to investigate a potential circadian periodicity of expression levels of various disease relevant cytokines, AM, and cytokine receptors including soluble tumor necrosis factor beta (sTNF-β), soluble forms of its receptors (sTNF-R) 1 and 2, soluble ICAM-1 (sICAM-1), and soluble VCAM-1 (sVCAM-1) in MS.
A rhythmic alteration of these immune parameters in relation to cortisol rhythmicity may be clinically relevant, since they have the potential to modulate leukocyte-induced pathogenesis associated with disease progression [4].
ICAM-1 and VCAM-1 are involved in leukocyte trafficking across endothelia, TNF-β has been shown to lead to indirect damage of the myelin sheaths [14], and elevated serum levels of sTNF-R1 have been found in MS patients with stable but not with active disease [6, 7]. However, it is unknown whether these molecules underlie diurnal alternations with potential pathogenic and/or therapeutic relevance. Moreover, changing serum levels in the course of the day and variable blood collection times might account for contradictory results between different studies on immunological markers in MS.
Methods
We included 34 untreated patients with clinically definite RRMS according to the revised McDonald criteria [15] and 34 age- and sex-matched healthy controls in this study. All 34 patients were without any clinically defined disease activity. All patients underwent magnetic resonance imaging (MRI) scans within 2 weeks before blood sampling. Twelve MS patients showed gadolinium enhancing lesions in corresponding MRI scans. These 12 MS patients were defined as “patients with active disease”. The other 22 MS patients were defined as “patients with non-active disease”. Baseline criteria of MS patients are shown in Table 1. Exclusion criteria were immunomodulatory therapy within 3 months or treatment with immunosuppressive drugs within 6 months prior to inclusion into the study. Further exclusion criteria were severe diseases courses, or pregnancy. All patients gave written informed consent and the study was approved by the local Ethics Committee.
Venous blood was collected five times at 7.00, 11.00 am, 2.30, 6.00 and 9.30 pm within one day and serum parameters were determined by ELISA according to the manufacturers’ instructions (Cortisol: Molecular Devices, Sunnyvale, CA, USA; sTNF-β: Bender MedSystems, Vienna, Austria; sTNF-R1 and sTNF-R2: Medgenix Biosource, Fleurus, Belgium; sICAM-1: Diaclone, Besancon, France; sVCAM-1: Bender MedSystems, Vienna, Austria). The blood sampling time points were selected on the basis of the diurnal cortisol rhythm.
Statistics
Mann–Whitney U test was performed to compare active with non-active MS and healthy individuals with MS-patients, as not all variables fulfilled parametric distribution. Different time periods were compared with each other by Wilcoxon test. Due to multiple pair-wise testing Bonferroni correction for multiple comparisons was applied. For doing so, the significance level was divided by the number of tests used. In all other cases a significance level of 0.05 was used. Data analysis was performed using SPSS™ for Windows™ version 18.0.
Results
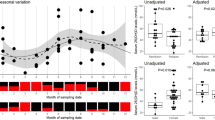
We determined serum concentrations of selected inflammatory parameters at five defined time points of the day to investigate potential diurnal concentration changes. The known circadian rhythmicity of serum cortisol served as reference parameter. Differences in serum concentrations were evaluated between MS patients and healthy controls (Fig. 1; Table 2) and between MS patients with active versus non-active disease at the time of blood collection (Fig. 2; Table 3).
Comparison of the median of serum levels of cortisol, soluble tumor necrosis factor-beta (sTNF-β), soluble forms of the TNF receptors-1 and -2 (sTNF-R1, sTNF-R2), soluble intercellular cell adhesion molecule-1 (sICAM-1), and soluble vascular cell adhesion molecule-1 (sVCAM-1) between multiple sclerosis patients (n = 34) and healthy donors (n = 34) at 7, 11 am, 2.30, 6, 9.30 pm
Comparison of the median of serum levels of cortisol, soluble tumor necrosis factor-beta (sTNF-β), the soluble forms of TNF receptors-1 and -2 (sTNF-R1, sTNF-R2), soluble intercellular cell adhesion molecule-1 (sICAM-1), and soluble vascular cell adhesion molecule-1 (sVCAM-1) between patients with active (n = 12) and patients with non-active multiple sclerosis (n = 22) at 7,11 am, 2.30, 6, 9.30 pm
MS patients versus healthy donors
Serum levels of cortisol showed a clear circadian rhythmicity with a significant and continuing decrease (p < 0.001) from about 25 to 5 μg/dl between 7.30 am and 9.30 pm, in MS patients and healthy controls (Fig. 1a; Table 2). There was no significant difference in serum cortisol concentrations of MS patients (22.00 μg/dl at 7.30 am and 3.60 μg/dl at 9.30 pm) and controls (19.25 μg/dl at 7.30 am and 4.80 μg/dl at 9.30 pm) at the five time points.
No circadian changes could be evidenced regarding serum levels of sTNF-β (Fig. 1d; Table 2), but all five measurements revealed almost fourfold higher sTNF-β levels (p < 0.01) in MS patients (median ~ 80 pg/ml) compared to healthy subjects (median ~ 20 pg/ml). Contrary to their ligand, circadian concentration changes became evident for the two TNF receptors. Patients and controls showed a significant decrease from about 3,700 to 3,200 pg/dl in sTNF-R2 serum levels (p < 0.001, Fig. 1c; Table 2) and a similar trend with a decrease from about 900 to 800 pg/ml in sTNR-R1 serum levels (Fig. 1b; Table 2) over the course of the day. However the main decrease of sTNR-R1 and sTNF-R2 serum levels was shown between 7:30 am and 2.30 pm in contrast to a relatively stable course between 2:30 and 9:30 pm in MS patients as well as in healthy controls. The serum concentrations of both parameters did not differ significantly between MS patient and controls at all time-points.
Both groups showed similar serum levels of sICAM-1 (Fig. 1e; Table 2) and sVCAM-1 (Fig. 1f; Table 2) between 7.30 am to 9.30 pm and a discrete and significant decline in serum levels of both soluble AM at 6.00 pm.
Patients with active MS versus patients with non-active MS
A significant decline in serum levels of cortisol (p < 0.001, Fig. 2a; Table 3) between 7.30 am and 9.30 pm was seen in both MS groups. Decreased serum concentrations for sTNF-R1 (Fig. 2b; Table 3) between 7.30 am and 2.30 pm and relatively stable course between 2:30 and 9:30 pm was shown for the two patient groups. Analysis of differences in circadian rhythmicity for sTNF-R2 (Fig. 2c; Table 3) revealed a significant decline in serum levels for sTNF-R2 between 7.30 am and 2.30 pm but only in patients with non-active MS. Increased serum concentrations for sICAM-1 (Fig. 2e; Table 3) between 7.30 and 11.00 am were observed in patients with active MS only, whereas a significant decline between 2.30 and 6.00 pm was evident in both patient groups. (Fig. 2e; Table 3).
No significant circadian changes in serum levels of sTNF-β and sVCAM-1 were observed.
Comparison of serum concentrations between the two patient groups revealed that sTNF-R1, sTNF-R2, sTNF-β and sICAM-1 levels showed no significant differences, but serum concentrations of cortisol and sVCAM-1 were higher in patients with active MS (Fig. 2; Table 3) compared to MS patients without gadolinium enhancing-lesions. The observed differences in serum concentrations were significant for sVCAM-1 (p < 0.05) at 11.00 am and 2.30 pm (Fig. 2f; Table 3).
Discussion
Inflammatory processes are considered major pathogenic mechanisms during onset and early stages of MS. Delay in disease progression via immunomodulatory and immunosuppressive drugs is still the primary treatment strategy for this life-long disease [16]. Given the immune-mediated background of MS, promising candidates for reliable and informative biomarkers on treatment response or unresponsiveness, include inflammatory parameters like cytokines, their receptors, chemokines, or adhesion molecules. These molecules are either membrane bound or released, respectively shedded, by immune competent cells. Soluble forms thereof can conveniently be investigated in patient sera. To date, numerous studies have been published and many potential biomarkers have been nominated, however, with conflicting results [3–8].
In our opinion some of the conflicting data could be the result from diurnal changes in serum concentrations of the investigated biomarkers and uncoordinated times of blood collections between study groups. Regarding the known circadian rhythmicity of cortisol and its effect on immune functions, we postulate that any immunological parameter can potentially be involved in diurnal concentration changes.
We serially investigated serum levels of five potential biomarkers in MS at defined times from early morning to late evening. In addition to the expected circadian rhythmicity of cortisol, we found diurnal changes in serum concentrations in four out of the five parameters. Both in MS patients and healthy controls, sTNF-R1 levels were elevated in the morning, sTNF-R2 levels showed a continuous and significant decrease in the course of the day, and sICAM-1 and sVCAM-1 levels were significantly reduced in the late afternoon. Only sTNF-β serum levels remained unaffected by the time of day.
These findings confirm our hypothesis that the time-point of blood drawings is relevant for clinical studies and comparisons of serum concentrations of immunological markers in different cohorts. Former studies, for example, reported higher serum levels of sTNF-R1 in MS patients with stable disease compared to patients with active disease [6, 7]. In contrast, in our study there were no significant differences of TNF-R1 levels between both patient groups but a general decrease in serum concentrations during the whole day. We could also have found nearly significant differences, if we had compared sTNF-R1 levels of blood collected at 7 am from the one patient group with serum levels of blood collected at 11 am from the other patient group.
Members of the TNF receptor superfamily and their soluble forms play a major role in the pathogenesis of MS [14]. Our results reveal a significant decline in serum levels of sTNF-R2 and a similar trend of sTNF-R1 levels in MS patients and controls which might be relevant for further studies. The importance of our findings supported by the fact that pharmacological modulation of TNF receptor signaling has recently been suggested a novel target for modulating the course of MS [17].
Lacking circadian rhythmicity of sTNF-β serum levels indicates little relevance for the time of blood collections for this parameter. However, the about twofold higher sTNF-β levels of MS patients compared to healthy subjects confirms a prominent role of sTNF-β in MS pathogenesis. Tumor necrosis factor-β has been detected in brain lesions and cerebrospinal fluid of patients with MS, has been shown to indirectly damage the myelin sheath, and TNF-β expression has been reported to be increased in peripheral immune cells from MS patients [14, 18].
Although increased in MS patients compared to controls, we did not observe differences in sTNF-β levels between patients with active or non-active disease. This is in line with a previous study showing that sTNF-β serum levels are no markers for subclinical disease activity [19].
Both, sICAM-1 and sVCAM-1 serum levels show circadian rhythmicity which is by far not as pronounced as that of cortisol or sTNF-R2. However, it should be considered for future study designs, as serum concentrations exhibited a significant decrease in the late afternoon in MS patients and controls. Intercellular adhesion molecule-1 is expressed on endothelial cells and leukocytes whereas VCAM-1 is mainly expressed on endothelium. Both AM bind to two different integrins on the surface of immune cells, leukocyte function associated antigen-1 (LFA-1) and very late activation antigen-4 (VLA-4) [20, 21]. Together, they play important roles in immune cell transmigration across the BBB into the CNS and are considered to be critically involved in the pathogenesis of MS [22]. Natalizumab (Tysabri™), for instance, is a therapeutic antibody specific for VLA-4 on peripheral blood mononuclear cells and has been proven to be efficient in the treatment of relapsing-remitting MS [2].
In previous studies with MS patients a positive correlation of sICAM-1 serum and sVCAM-1 CSF and serum levels but an inverse correlation of sICAM-1 CSF levels with clinical and MRI disease activity was reported [5, 6, 21]. Here, we can show a trend towards higher sVCAM-1 serum concentrations in MS patients with active disease compared to patients without active lesions. These differences were significant in the morning and early afternoon and indicated different diurnal serum levels between the two patient groups. Again, this demonstrates that comparing data of different time points might be biased by diurnal concentration changes. Furthermore, the increased levels of sVCAM-1 at a specific time of the day in patients with active MS could be a hint that the recruitment of activated peripheral blood mononuclear cells across the BBB might also underlie a certain circadian rhythmicity.
This brings us directly to another important topic. According to our results, different time points of treatment application may differently influence the circadian rhythmicity of immunological markers and, therefore, may have a different impact on disease modulation.
The role of cortisol with its immunomodulatory function and pronounced circadian rhythmicity due to endocrine regulation via the hypothalamic–pituitary–adrenal system has been investigated in several studies in MS patients [23, 24]. It is still unclear whether there is an endocrine insufficiency in MS patients. Apart from that, high-dose corticoid therapy is beyond controversy first-line treatment of MS relapses but, recently, the optimal time point of application has been under debate [12, 13].
The trend to higher cortisol levels in the serum from patients with active MS indicates a certain interrelation between disease activity and circadian rhythmicity and could be a sign for autoregulation of immunomodulation in the active phase of MS. The significant decrease over the course of the day with lowest levels in the night as we found also in MS patients with active disease could be an explanation for the recently demonstrated clinical benefit of night-time administration of corticoid steroid therapy in the management of MS relapses [12, 13].
Similarly, a recent study suggested modulation of signal transduction pathways of molecules such as sTNF-R1 and -R2 to be part of the therapeutic effect of interferon (IFN)-β [25]. This and our findings of circadian rhythmicity of sTNF-R1 and -R2 serum levels implicate that the time-point of IFN-β application and potentially other disease modifying drugs, too, might contribute to its therapeutic efficacy. Further studies are necessary to obtain additional facts on the suspected interaction of administration time points of the respective drugs on disease modulation and treatment side effects.
In conclusion, our data show that the circadian rhythmicity of immunological markers such as sICAM-1, sVCAM-1, sTNF-R1 and sTNF-R2 should be considered as a possible explanation for contradictory results from studies on inflammatory markers in MS. However, the interpretation of our results is restricted by relatively small sample size of a special subgroup of MS patients with long disease duration, low EDSS and without treatment. Thus a larger cohort is needed to prove our data.
Certainly we are aware, that further factors such as different ELISA systems might contribute to the above mentioned discrepancies, but increased awareness of the possibility of diurnal serum concentration changes of biomarkers could potentially eliminate a major cause of biased data.
Finally, our data are supportive for the hypothesis that optimization of exact time of drug application in MS might be important to enhance treatment efficacy or to reduce treatment side effects.
References
Noseworthy JH et al (2000) Multiple sclerosis. N Engl J Med 343:938–952
Engelhardt B, Kappos L (2008) Natalizumab: targeting alpha4-integrins in multiple sclerosis. Neurodegener Dis 5:16–22
Wipfler P et al (2011) Adhesion molecules are promising candidates to establish surrogate markers for natalizumab treatment. Mult Scler 17:16–23
Kraus J et al (1998) Soluble and cell surface ICAM-1 as markers for disease activity in multiple sclerosis. Acta Neurol Scand 98:102–109
Kraus J et al (2002) Cell surface bound and soluble adhesion molecules in CSF and blood in multiple sclerosis: correlation with MRI-measures of subclinical disease severity and activity. J Neuroimmunol 122:175–185
Rieckmann P, Altenhofen B, Riegel A, Kallmann B, Felgenhauer K (1998) Correlation of soluble adhesion molecules in blood and cerebrospinal fluid with magnetic resonance imaging activity in patients with multiple sclerosis. Mult Scler 4:178–182
Hartung HP, Reiners K, Archelos JJ et al (1996) Circulating adhesion molecules and tumor necrosis factor receptor in multiple sclerosis: correlation with magnetic resonance imaging. Ann Neurol 38:186–193
McDonnell GV, McMillan SA, Douglas JP, Droogan AG, Hawkins SA (1998) Raised CSF levels of soluble adhesion molecules across the clinical spectrum of multiple sclerosis. J Neuroimmunol 85:186–192
Niehaus GD et al (2002) Circadian variation in cell-adhesion molecule expression by normal human leukocytes. Can J Physiol Pharmacol 80:935–940
Young MW (2000) The tick–tock of the biological clock. Sci Am 282:64–71
Petrovsky N, McNair P, Harrison LC (1998) Diurnal rhythms of pro-inflammatory cytokines: regulation by plasma cortisol and therapeutic implications. Cytokine 10:307–312
Glass-Marmor L et al (2007) Chronotherapy using corticosteroids for multiple sclerosis relapses. J Neurol Neurosurg Psychiatry 78:886–888
Glass-Marmor L et al (2009) Immunomodulation by chronobiologically based glucocorticoids treatment for multiple sclerosis relapses. J Neuroimmunol 210:124–127
Laske C et al (2001) Induction of sTNF-R1 and sTNF-R2 by interferon beta-1b in correlation with clinical and MRI activity. Acta Neurol Scand 103:105–113
Polman CH, Reingold SC, Edan G et al (2005) Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol 58:840–846
Pilz G, Wipfler P, Ladurner G et al (2008) Modern multiple sclerosis treatment—what is approved, what is on the horizon. Drug Discov Today 13:1013–1025
Brambilla R, Ashbaugh JJ, Magliozzi R et al (2011) Inhibition of soluble tumour necrosis factor is therapeutic in experimental autoimmune encephalomyelitis and promotes axon preservation and remyelination. Brain 134:2736–2754
Redford EJ, Hall SM, Smith KJ (1995) Vascular changes and demyelination induced by the intraneural injection of tumour necrosis factor. Brain 118:869–878
Kraus J, Kuehne BS, Tofghi J et al (2002) Serum cytokine levels do not correlate with disease activity and severity assessed by brain MRI in multiple sclerosis. Acta Neurol Scand 105:300–308
Archelos JJ, Hartung HP (1997) The role of adhesion molecules in multiple sclerosis: biology, pathogenesis and therapeutic implications. Mol Med Today 3:310–321
Giovannoni G, Lai M, Thorpe J, Kidd D, Chamoun V, Thompson AJ, Miller DH, Feldmann M, Thompson EJ (1997) Longitudinal study of soluble adhesion molecules in multiple sclerosis: correlation with gadolinium enhanced magnetic resonance imaging. Neurology 48:1557–1565
Engelhardt B (2006) Molecular mechanisms involved in T cell migration across the blood-brain barrier. J Neural Transm 113:477–485
Michelson D, Stone L, Galliven E et al (1994) Multiple sclerosis is associated with alterations in hypothalamic-pituitary-adrenal axis function. J Clin Endocrinol Metab 79:848–853
Grasser A, Moller A, Backmund H et al (1996) Heterogeneity of hypothalamic–pituitary–adrenal system response to a combined dexamethasone-CRH test in multiple sclerosis. Exp Clin Endocrinol Diabet 104:31–37
Reuss R et al (2009) Interferon beta-1a induces tumor necrosis factor receptor 1 but decreases tumor necrosis factor receptor 2 leukocyte mRNA levels in relapsing-remitting multiple sclerosis. NeuroImmunoModulation 16:171–176
Conflicts of interest
We certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
Ethical standard
This study has been approved by the appropriate ethics committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wipfler, P., Heikkinen, A., Harrer, A. et al. Circadian rhythmicity of inflammatory serum parameters: a neglected issue in the search of biomarkers in multiple sclerosis. J Neurol 260, 221–227 (2013). https://doi.org/10.1007/s00415-012-6622-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-012-6622-3