Abstract
Recently it has been postulated that there is an atypical facioscapulohumeral muscular dystrophy (FSHD) phenotype with isolated axial myopathy. Involvement of paraspinal and limb muscles was evaluated in six patients with molecularly proven FSHD and a predominant bent spine phenotype. Consistent with the camptocormia phenotype, the most severely affected muscles in all six patients were the thoracic and lumbar spinal tract together with hamstrings. MRI disclosed severe axial muscle degeneration but mostly subclinical involvement of limb muscles. The involvement of hip extensor muscles in FSHD might considerably contribute to the clinical phenotype of camptocormia due to axial muscle involvement.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Camptocormia, also referred to as bent spine syndrome, is characterized by an abnormal posture of the trunk with marked flexion of the thoraco-lumbar spine, which increases during walking and abates in the recumbent position. Camptocormia may be associated with a variety of neurologic etiologies including movement disorders (Parkinson disease, dystonia [36]), motor neuron disorders (amyotrophic lateral sclerosis [21], spinal muscular atrophy), and myasthenia gravis [1]. There are also various myopathies with paraspinal muscle involvement leading to camptocormia. These occasionally include nemaline myopathy [45], inclusion body myositis [12], polymyositis [22], limb girdle dystrophy 2B [35], myotonic dystrophy type 2 [18], X-linked myopathy with postural muscle atrophy [46], central core diseases such as RYR1-related myopathy [15], valosin-containing protein myopathy [37], amyloid myopathy [8], and metabolic myopathies such as acid maltase deficiency (Pompe disease) [17], and carnitine deficiency.
However, myopathy with pure bent spine or camptocormia as the predominant clinical picture has been rarely reported [19, 35, 46]. This phenotype is impressive and may be misdiagnosed as the normal aging process [25].
Whereas the involvement of trunk muscles in FSHD has been postulated to be a common phenomenon [39], there has been only one recent case report on bent spine syndrome as the initial and leading symptom in FSHD [19]. Umapathy et al. [41] reported a 59-year-old woman presenting with camptocormia, scapular winging and mild facial and proximal weakness due to genetically verified FSHD with normal spine MRI.
Although MRI has been widely applied as an objective assessment of muscle status in a variety of muscular disorders, the imaging experience in FSHD is rather limited [7, 16, 18, 19, 29, 31, 34]. MRI provides additional information on subclinically involved muscle groups and sometimes leads to a typical diagnostic pattern of muscle disease [7, 18, 28, 34].
We present six cases of predominant axial myopathy as the initial manifestation of FSHD with the corresponding MRI and clinical data (clinical examination, genetics, electrophysiological investigations).
Methods
Subjects
Six consecutive patients with camptocormia in whom FSHD diagnosis was genetically verified were included into the present study. Clinical data are summarised in Table 1. Beside the son of patient 6, other family members were not investigated clinically and genetically. Age of onset was defined as the time when the patient first noticed functional impairment caused by the disease. There were no other possible causes of the bent spine syndrome like osteoporosis, degenerative disease of the spine, bed rest for a longer time period or steroid medication. In particular, the analysis of occupation of the patients gave no hint for increased risk of developing spinal pathology.
Molecular genetics
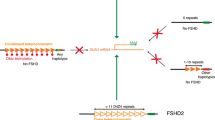
DNA was obtained from peripheral blood, digested with the restriction enzyme Eco RI, and double digested with Eco RI/Bln I [3]. The restriction fragments were detected by Southern blotting using the probe p13E-11. In unaffected individuals this method will show two 4q35 alleles of >38 Kb on the basis of an EcoRI DNA digestion. In individuals with FSHD, one of the two 4q35 alleles will be between 10 and 38 Kb. Genetic confirmation of FSHD1 was done by S. Jakubiczka, MD (Division of Laboratory Genetics at University hospital Magdeburg, Germany).
MRI
In each patient a standardised MRI was performed as follows using a 1.5-Tesla scanner (Siemens, Forchheim, Germany) with implemented quadrature body coil and a flexible table top extension. The patients were placed in a supine position with their arms beside the body. Separate examination areas ranged from (1) shoulder region including upper arm of both sides, (2) trunk muscles ranging from vertebra Th 3 to L 4, (3) hip/thigh and upper legs starting from vertebra L4, (4) lower legs. The lateral parts of the upper limbs, especially the forearms, were not available for analysis due to the maximum field of view being exceeded. The MRI sequence protocol included axial slices of the following sequences: T1-weighted Fast Spin Echo FSE (TR 474 ms, TE 11 ms, matrix 512, FoV 40 cm, slice thickness 5 mm, phase oversampling 100%), T2-weighted spectral fat suppressed (TIRM) (TR 5400 ms, TE 80 ms, TI 120 ms, matrix 512, FoV 40 cm, slice thickness 5 mm, phase oversampling 100%)—depending on the region the distance factor reached between 150 and 300%. The investigation time was about 45 min per patient. No contrast agent was administered.
Evaluation of MRI findings
The MRI scans were analysed by a neuroradiologist and a neurologist who were not aware of clinical findings. The scans were assessed for normal and abnormal muscle bulk (atrophy/hypertrophy) and for abnormal signal intensity on T1 weighted images. Changes were only considered to be significant if they were present in more than two slices. The muscles on either side of the body were evaluated separately to detect asymmetric affection.
The degree of fatty degeneration was scored according to a modified score as previously suggested [18, 30, 31] using a four-point semiquantitative visual scale (MRI score): 0, normal appearance; 1, discreet moth-eaten appearance with sporadic scattered T1 hyperintense areas; 2, moderate moth-eaten appearance with numerous scattered T1 hyperintense areas; 3, late moth-eaten appearance with numerous confluence areas of T1 hyperintensity; 4, complete fatty degeneration, replacement of muscle by fat.
The following muscle groups were evaluated: upper arm and shoulder girdle, thighs and hip girdle, lower leg and trunk (spinal and abdominal part).
Clinical evaluation
Another neurologist blinded for the MRI findings evaluated muscle strength in all patients by manual muscle testing using the MRC scale (Medical Research Council scale) [26].
Statistical analyses
The frequency of fatty infiltration, frequency of atrophy and degree of involvement measured by MRI score was evaluated for every muscle. For every affected muscle, appropriate individual MRI scores of affected patients was added. This total amount was divided through number of affected patients (mean MRI score, reported values are mean ± standard deviation). In addition, for every patient an individual MRI score (1, spine score; 2, upper leg/lower leg score; 3, arm score; 4, total MRI score) was calculated including the total amount of appropriate MRI muscle scores. Following other muscle MRI studies, the correlation between these individual MRI scores and clinical findings (age, age of onset, disease duration, allele fragment size) were calculated using the non-parametric Spearman rank analysis [31]. A p value of 0.05 or lower was considered significant (two-tailed testing). Asymmetrical involvement of corresponding muscles was evaluated.
Results
Clinical characteristics
All patients presented with camptocormia, which was the initial and predominant symptom of disease. The mean duration of symptoms experienced by the patients was 15.6 years (range 4–22, median 14) (Table 1). The median age of onset of muscle weakness was 55 years. The detected allele fragment size ranged from 21 to 34 kilo base pairs (normal range 41–350, FSHD <38) [43]. There was a striking predominance of female patients.
The symptoms progressed slowly in all cases and led to the need of intermediate or permanent walking aids in all patients. All patients reported difficulties keeping the trunk upright, especially while standing or carrying objects (e.g. tray). However, an upright sitting position was held without any difficulties.
Clinical examination revealed bent spine with predominance in the thoraco-lumbar region which disappeared in a supine position. There was mild to severe bilateral wasting of the paravertebral muscles. Facial weakness was absent in two patients, three patients showed a rather mild bilateral positive signe des cils, one patient suffered from mild bilateral non fluctuating ptosis. Prominent scapula (but not scapular winging) was detected in three patients, whereas an atrophy of shoulder girdle muscles appeared in two patients. Two patients showed mild to moderate weakness in the shoulder region, three patients presented hip girdle weakness and a foot drop with slight asymmetry. Three were generally not or only mildly affected but without functional relevance. Beevor’s sign was positive in three patients [4]. No patient suffered from respiratory distress.
Serum creatine-kinase levels at the time of investigation were normal in one, but slightly increased in five cases (approx. two times the upper normal limit). Electromyography of paravertebral muscles performed in four cases revealed myopathic findings consisting of a low amplitude pattern during voluntary effort; short duration, low voltage, polyphasic motor unit action potentials; and early recruitment. Diagnosis of FSHD was genetically established. There was no correlation between allele fragment size and age of initial manifestation of weakness (data not shown). Judging from the clinical picture as it was remembered by patients, family history for FSHD (with/without camptocormia) was negative in four patients. The daughter of patient 3 clinically suffered from undetermined muscle weakness without camptocormia. The son of patient 6 presented with a typical genetically proven FSHD phenotype.
Muscle MRI imaging
Trunk
The frequency of fatty muscle degeneration and mean MRI severity scores are shown in Table 2. Age of disease onset tended to be lower in patients with most severe erector spinae involvement (Table 3). Muscle MRI (T1 and T2 weighted) findings showed a selective and extensive fatty degeneration of the erector spinae muscles in all patients (Figs. 1, 2). The lumbar segments were more involved than the thoracic region showing complete (grade 4) degeneration in five patients. The lateral tract of the erector spinae system consisting of longissimus and iliocostalis muscles was mostly affected and resembled fatty pseudohypertrophy in four patients. Grade 3 degeneration was mainly assigned to muscles of the lumbar medial tract of erector spinae group (predominantly Mm. multifidii). There was severe and symmetric involvement in all cases. Quadratus lumborum and iliopsoas muscles were completely spared in five patients (Fig. 2). M. rectus abdominis seemed rather atrophic in two patients (Figs. 2, 3). Both presented with positive Beevor’s sign [4]. However, assessment of anterolateral abdominal muscles and M. rectus abdominis was limited due to ventilation artefacts.
No signs of muscle edema were detected on T2 weighted images. MRI of the spine showed mild lumbar spondylosis features in patient 4. Significant protrusions and degenerative changes of intervertebral discs were not found. There were neither gender-specific differences in spinal muscle involvement nor significant age-dependent effects on the number or severity of affected muscle groups.
Extremities
The degree of MRI changes was clearly milder in extremity muscles than in spinal muscles (Table 2). MRI showed evidence of mild to moderate fatty degeneration of shoulder girdle and forearm muscles in four of six patients with predominance in M. latissimus dorsi (mean MRI score 2.50 ± 0.83; Table 2). Muscle involvement of hip and lower body in MRI was shown in all patients (Table 2, Figs. 3, 4). The muscles most frequently and severely affected were the semimembranosus (frequency of fatty degeneration 92%, mean MRI score 3.27 ± 0.75) and gastrocnemius (frequency of fatty degeneration 75%, mean MRI score 3.00 ± 0.90). Adductor muscles and gluteus maximus were frequently affected as well; however, the degree was only moderate (mean MRI score 2.20 ± 1.23 and 1.83 ± 0.49, respectively). MRI revealed subclinical muscle affection in three patients. MRI score of lower extremity muscles significantly correlated with age at onset of symptoms (r = −0.9, p < 0.05), but not with disease duration (Table 3). No correlation with allele fragment size could be detected. However, these results have to be interpreted with great caution due to the small amount of patients.
Discussion
In classical FSHD phenotype muscle weakness is primarily restricted to facial and upper extremity muscles (especially scapula stabilizer and humeral muscles). Lower extremity involvement is common during disease progression and ranges from distal anterior leg weakness (typical footdrop) to hip girdle muscle weakness in advanced disease. Abdominal muscles rather frequently seem to be involved in FSHD patients [4].
Recently different atypical FSHD phenotypes presenting as isolated monomelic lower limb weakness [42, 44], limb girdle asymmetric weakness [5], and facial-sparing scapular myopathy [6, 14, 20] including isolated axial myopathy [19] have been described. Camptocormia has occasionally been reported in FSHD [19, 41, 47].
There are no systematic MRI studies on trunk muscle degeneration in FSHD. A study on muscle involvement in various myopathies showed fatty involvement of erector spinae muscles in a 47-year-old female FSHD patient (clinical data not reported) besides similar changes in gluteal muscles, vastus group, adductor muscles, semimembranosus and gastrocnemius (medial head) [34].
In our study the spine of all six patients was obviously bent in standing position. This was in accordance to muscle MRI findings ranging from severe to complete fatty, rather symmetric, degeneration of the longissimus, iliocostalis and medial spinal tract (Mm. multifidii etc.) muscles in all patients (Table 2, Figs. 1, 2). There were some residual muscle fibers of multifidii muscles in all patients. In our series the trunk flexor muscles, M. iliopsoas and M. quadratus lumborum seemed to be normal in five patients, but slightly reduced in one patient. Atrophy of abdominal muscles was seen in two patients who presented with positive Beevor’s sign [4].
There is no doubt that partial fatty degeneration and atrophy of erector spinae muscles in adults is an imaging feature described in the context of chronic low back pain [27], bed rest [10] and aging. In addition, focal myopathy of paravertebral muscles has been postulated in patients with Parkinson disease and multiple system atrophy as well [33, 36]. However, the severe, almost complete replacement of paravertebral muscles by fat in our study patients considerably exaggerates the MRI findings reported in the latter diseases [2, 9, 10]. In addition, they expand along the whole thoracic, lumbar and lumbosacral spine which is not common in degenerative spinal disorders [2, 9]. The hypothesis of a distinct specific myopathic change is confirmed by the finding of complete degeneration not only of multifidus muscles but of lateral erector spinae muscles (i.e. M. longissimus and M. iliocostalis) typically preserved with prolonged bed rest and low back pain [2, 10]. The morphologic muscle changes in the latter conditions commonly resemble neurogenic atrophy, whereas changes in our camptocormia patients fulfil the MRI criteria of myogenic amyotrophy (conserved muscle shape and size demonstrated by MRI) [23].
In patients with other myopathies (myotilinopathy, LGMD, desminopathy), probable subclinical fatty infiltration of the erector spinae muscles was found in 11 out of 18 investigated patients, in 7 out of 18 patients the grade of infiltration was severe. In contrast to our findings, oedema was frequently present in patients with hereditary myopathies, even in muscle groups with severe fatty infiltration [34].
The prevailing view is that axial weakness might simply explain camptocormia. However, there are other patients with complete erector spinae muscle fatty degeneration (e.g. in primary dysferlin deficiency) without the occurrence of camptocormia [35]. These cases are in line with our observation of some typical FSHD patients with fairly fatty degenerated erector spinae muscles but without any evidence of camptocormia (patients not included in the study because they did not meet inclusion criteria). Another study on patients with myotonic dystrophy type II showed fatty degeneration of thoracic and lumbosacral erector spinae muscles, whereas only 3 out of 14 showed clinical trunk muscle involvement [18]. Thus, axial weakness alone might not be sufficient to cause camptocormia in neuromuscular disease.
A quantitative analysis of upper body oscillations in FSHD patients revealed an increased range of body movement correlating with the clinical severity score [13]. This is in line with the previous finding that FSHD patients usually fall in a forward direction [11] which might be due to upper trunk and neck muscle weakness [13].
It has been shown that weakness of pelvic extensor muscles (gluteus, adductor magnus, semimembranosus) leads to increased pelvic tilt and hip flexion (hyperlordosis) and sagittal spinal imbalance in early onset FSHD [24]. Patients stand with the spine hyperextended to maintain an upright posture by a compensatory mechanism of strong back extensor muscles [24]. The additional fatty degeneration of pelvic extensor muscles, especially of M. semimembranosus, and to a milder extent, of adductor magnus and gluteus muscles, has been shown in all six patients with camptocormia phenotype (Table 2).
Thus, it can be suggested that only combined insufficiency of pelvic extensor and erector spinae muscles consecutively leads to camptocormia. This might explain why the tendency to lean forward becomes more pronounced with continued standing or walking whereas patients sitting and laying down are unremarkable. Patients prefer using a walking aid and, while standing, to extend their arms behind the back or to lay them on upper leg or lower abdomen to maintain gravitational balance. On the other hand, the aetiology of camptocormia does not necessarily include neuromuscular and movement disorders, but may occasionally arise due to spine deformities or idiopathic reason as well [1].
MRI provides additional information on subclinically involved muscle groups and sometimes leads to a typical diagnostic pattern of muscle disease [7, 18, 28, 34]. In our study, atrophy and fatty degeneration of lower extremity muscles were found in all investigated patients (Table 2). MRI revealed subclinical extremity involvement in 3 out of 6 patients. However, the degree of extremity involvement was in neither patient as severe as in erector spinae muscles. Although clinically asymmetric muscle involvement is commonly described in FSHD in our study only 11% of affected muscles showed asymmetry in MRI. This is in accordance to a previous study by Olsen et al. [31] who also found only an asymmetry rate of 15%. Often, the degree of involvement was larger at the distal end than in the remaining muscle as previously reported [16].
Both spinal and extremity muscle affection in MRI seemed to correlate with clinical severity as described previously [31], but the severity of muscle degeneration did not depend on disease duration. Posterior leg muscles have been described as clinically relatively spared in FSHD [38] or to be affected only in later stages of the disease [40]. However, it has previously been shown by MRI that hamstrings and semimembranosus and gastrocnemius muscles seem to be typically involved in FSHD [16, 31, 32]. This is consistent with our study (Table 2). There was mostly subclinical severe fatty degeneration of gastrocnemius muscle in five out of six patients. However, involvement of posterior leg muscles is not very specific as it has been shown in many other myopathies (desminopathy, myotilinopathy, LGMD2I, LGMD2A) as well [7, 34].
Conclusion
The study extends the spectrum of atypical clinical presentations of FSHD. Isolated or predominant weakness of spinal muscles leading to a camptocormia is a rare finding in FSHD and seems to represent a new phenotype of disease. Since the camptocormia phenotype is the initial clinical symptom it may cause diagnostic confusion. We postulate that the predominant spinal muscle involvement not necessarily leads to postural abnormality. In myopathic bent spine, insufficiency of spinal muscles might be accompanied by weakness of hip extensor muscles, especially of adductor and semimembranosus muscles, leading to an increased pelvic tilt. In the present study, MRI demonstrated not only severe fatty degeneration of thoracic and lumbar erector spinae muscle tract, but also involvement of hip extensors as well. Even though MRI indicated some subclinical muscle involvement (e.g. M. gastrocnemius), there seems to be a specific pattern of predominant abnormality of axial muscles in bent spine FSHD subgroup. As in other atypical forms of disease there was no correlation with allele fragment length. In addition the study shows that the axial phenotype is preserved for a long course of disease sparing other in FSHD typically involved muscle groups. The expanding clinical heterogeneity of FSHD is important to recognize so that DNA testing for FSHD has to be considered in undiagnosed patients with isolated bent spine syndrome.
References
Azher SN, Jankovic J (2005) Camptocormia: pathogenesis, classification, and response to therapy. Neurology 65:355–359
Bierry G, Kremer S, Kellner F, Abu Eid M, Bogorin A, Dietemann JL (2008) Disorders of paravertebral lumbar muscles: from pathology to cross-sectional imaging. Skeletal Radiol 37:967–977
Deidda G, Cacurri S, Piazzo N, Felicetti L (1996) Direct detection of 4q35 rearrangements implicated in facioscapulohumeral muscular dystrophy (FSHD). J Med Genet 33:361–365
Eger K, Jordan B, Habermann S, Zierz S (2010) Beevor’s sign in facioscapulohumeral muscular dystrophy: an old sign with new implications. J Neurol 257:436–438
Felice KJ, Moore SA (2001) Unusual clinical presentations in patients harboring the facioscapulohumeral dystrophy 4q35 deletion. Muscle Nerve 24:352–356
Felice KJ, North WA, Moore SA, Mathews KD (2000) FSH dystrophy 4q35 deletion in patients presenting with facial-sparing scapular myopathy. Neurology 54:1927–1931
Fischer D, Walter MC, Kesper K, Petersen JA, Aurino S, Nigro V, Kubisch C, Meindl T, Lochmuller H, Wilhelm K, Urbach H, Schroder R (2005) Diagnostic value of muscle MRI in differentiating LGMD2I from other LGMDs. J Neurol 252:538–547
Friedman Y, Paul JT, Turley J, Hazrati LN, Munoz D (2007) Axial myopathy due to primary amyloidosis. Muscle Nerve 36:542–546
Hadar H, Gadoth N, Heifetz M (1983) Fatty replacement of lower paraspinal muscles: normal and neuromuscular disorders. AJR Am J Roentgenol 141:895–898
Hides JA, Belavy DL, Stanton W, Wilson SJ, Rittweger J, Felsenberg D, Richardson CA (2007) Magnetic resonance imaging assessment of trunk muscles during prolonged bed rest. Spine 32:1687–1692
Horlings CG, Munneke M, Bickerstaffe A, Laverman L, Allum JH, Padberg GW, Bloem BR, van Engelen BG (2009) Epidemiology and pathophysiology of falls in facioscapulohumeral disease. J Neurol Neurosurg Psychiatry 80:1357–1363
Hund E, Heckl R, Goebel HH, Meinck HM (1995) Inclusion body myositis presenting with isolated erector spinae paresis. Neurology 45:993–994
Iosa M, Mazza C, Pecoraro F, Aprile I, Ricci E, Cappozzo A (2010) Control of the upper body movements during level walking in patients with facioscapulohumeral dystrophy. Gait Posture 31:68–72
Jardine PE, Koch MC, Lunt PW, Maynard J, Bathke KD, Harper PS, Upadhyaya M (1994) De novo facioscapulohumeral muscular dystrophy defined by DNA probe p13E–11 (D4F104S1). Arch Dis Child 71:221–227
Jungbluth H, Lillis S, Zhou H, Abbs S, Sewry C, Swash M, Muntoni F (2009) Late-onset axial myopathy with cores due to a novel heterozygous dominant mutation in the skeletal muscle ryanodine receptor (RYR1) gene. Neuromuscul Disord 19:344–347
Kan HE, Scheenen TW, Wohlgemuth M, Klomp DW, van Loosbroek-Wagenmans I, Padberg GW, Heerschap A (2009) Quantitative MR imaging of individual muscle involvement in facioscapulohumeral muscular dystrophy. Neuromuscul Disord 19:357–362
Katirji B, Kesner V, Hejal RB, Alshekhlee A (2008) Teaching NeuroImage: axial muscle atrophy in adult-onset Pompe disease. Neurology 70:e36
Kornblum C, Lutterbey G, Bogdanow M, Kesper K, Schild H, Schroder R, Wattjes MP (2006) Distinct neuromuscular phenotypes in myotonic dystrophy types 1 and 2: a whole body highfield MRI study. J Neurol 253:753–761
Kottlors M, Kress W, Meng G, Glocker FX (2010) Facioscapulohumeral muscular dystrophy presenting with isolated axial myopathy and bent spine syndrome. Muscle Nerve 42:273–275
Krasnianski M, Eger K, Neudecker S, Jakubiczka S, Zierz S (2003) Atypical phenotypes in patients with facioscapulohumeral muscular dystrophy 4q35 deletion. Arch Neurol 60:1421–1425
Kuncl RW, Cornblath DR, Griffin JW (1988) Assessment of thoracic paraspinal muscles in the diagnosis of ALS. Muscle Nerve 11:484–492
Kuo SH, Vullaganti M, Jimenez-Shahed J, Kwan JY (2009) Camptocormia as a presentation of generalized inflammatory myopathy. Muscle Nerve 40:1059–1063
Laroche M, Rousseau H, Mazieres B, Bonafe A, Joffre F, Arlet J (1989) Value of X-ray computed tomography in muscular pathology. Personal cases and review of the literature. Rev Rhum Mal Osteoartic 56:433–439
Lee CS, Kang SJ, Hwang CJ, Lee SW, Ahn YJ, Kim YT, Lee DH, Lee MY (2009) Early-onset facioscapulohumeral muscular dystrophy—significance of pelvic extensors in sagittal spinal imbalance. J Pediatr Orthop B 17
Mahjneh I, Marconi G, Paetau A, Saarinen A, Salmi T, Somer H (2002) Axial myopathy—an unrecognised entity. J Neurol 249:730–734
Medical Research Council (1976) Aids to the investigation of peripheral nerve injuries. Her Majesty’s Stationary Office, London
Mengiardi B, Schmid MR, Boos N, Pfirrmann CW, Brunner F, Elfering A, Hodler J (2006) Fat content of lumbar paraspinal muscles in patients with chronic low back pain and in asymptomatic volunteers: quantification with MR spectroscopy. Radiology 240:786–792
Mercuri E, Bushby K, Ricci E, Birchall D, Pane M, Kinali M, Allsop J, Nigro V, Saenz A, Nascimbeni A, Fulizio L, Angelini C, Muntoni F (2005) Muscle MRI findings in patients with limb girdle muscular dystrophy with calpain 3 deficiency (LGMD2A) and early contractures. Neuromuscul Disord 15:164–171
Mercuri E, Pichiecchio A, Allsop J, Messina S, Pane M, Muntoni F (2007) Muscle MRI in inherited neuromuscular disorders: past, present, and future. J Magn Reson Imaging 25:433–440
Mercuri E, Talim B, Moghadaszadeh B, Petit N, Brockington M, Counsell S, Guicheney P, Muntoni F, Merlini L (2002) Clinical and imaging findings in six cases of congenital muscular dystrophy with rigid spine syndrome linked to chromosome 1p (RSMD1). Neuromuscul Disord 12:631–638
Olsen DB, Gideon P, Jeppesen TD, Vissing J (2006) Leg muscle involvement in facioscapulohumeral muscular dystrophy assessed by MRI. J Neurol 253:1437–1441
Reilich P, Schramm N, Schoser B, Schneiderat P, Strigl-Pill N, Muller-Hocker J, Kress W, Ferbert A, Rudnik-Schoneborn S, Noth J, Lochmuller H, Weis J, Walter MC (2010) Facioscapulohumeral muscular dystrophy presenting with unusual phenotypes and atypical morphological features of vacuolar myopathy. J Neurol 257:1108–1118
Schabitz WR, Glatz K, Schuhan C, Sommer C, Berger C, Schwaninger M, Hartmann M, Hilmar Goebel H, Meinck HM (2003) Severe forward flexion of the trunk in Parkinson’s disease: focal myopathy of the paraspinal muscles mimicking camptocormia. Mov Disord 18:408–414
Schramm N, Born C, Weckbach S, Reilich P, Walter MC, Reiser MF (2008) Involvement patterns in myotilinopathy and desminopathy detected by a novel neuromuscular whole-body MRI protocol. Eur Radiol 18:2922–2936
Seror P, Krahn M, Laforet P, Leturcq F, Maisonobe T (2008) Complete fatty degeneration of lumbar erector spinae muscles caused by a primary dysferlinopathy. Muscle Nerve 37:410–414
Spuler S, Krug H, Klein C, Medialdea IC, Jakob W, Ebersbach G, Gruber D, Hoffmann KT, Trottenberg T, Kupsch A (2009) Myopathy causing camptocormia in idiopathic Parkinson’s disease: a multidisciplinary approach. Mov Disord 25:552–559
Stojkovic T, Hammouda el H, Richard P, de Munain AL, Ruiz-Martinez J, Gonzalez PC, Laforet P, Penisson-Besnier I, Ferrer X, Lacour A, Lacomblez L, Claeys KG, Maurage CA, Fardeau M, Eymard B (2009) Clinical outcome in 19 French and Spanish patients with valosin-containing protein myopathy associated with Paget’s disease of bone and frontotemporal dementia. Neuromuscul Disord 19:316–323
Tawil R, Figlewicz DA, Griggs RC, Weiffenbach B (1998) Facioscapulohumeral dystrophy: a distinct regional myopathy with a novel molecular pathogenesis. FSH Consortium. Ann Neurol 43:279–282
Tawil R, Van Der Maarel SM (2006) Facioscapulohumeral muscular dystrophy. Muscle Nerve 34:1–15
Tyler FH, Stephens FE (1950) Studies in disorders of muscle. II Clinical manifestations and inheritance of facioscapulohumeral dystrophy in a large family. Ann Intern Med 32:640–660
Umapathi T, Chaudhry V, Cornblath D, Drachman D, Griffin J, Kuncl R (2002) Head drop and camptocormia. J Neurol Neurosurg Psychiatr 73:1–7
Uncini A, Galluzzi G, Di Muzio A, De Angelis MV, Ricci E, Scoppetta C, Servidei S (2002) Facioscapulohumeral muscular dystrophy presenting isolated monomelic lower limb atrophy. Report of two patients with and without 4q35 rearrangement. Neuromuscul Disord 12:874–877
Upadhyaya M, Cooper DN (2002) Molecular diagnosis of facioscapulohumeral muscular dystrophy. Expert Rev Mol Diagn 2:160–171
van der Kooi AJ, Visser MC, Rosenberg N, van den Berg-Vos R, Wokke JH, Bakker E, de Visser M (2000) Extension of the clinical range of facioscapulohumeral dystrophy: report of six cases. J Neurol Neurosurg Psychiatr 69:114–116
Wallgren-Pettersson C, Laing N (1996) 40th ENMC Sponsored International Workshop: Nemaline Myopathy. 2–4 February 1996, Naarden, The Netherlands. Neuromuscul Disord 6:389–391
Windpassinger C, Schoser B, Straub V, Hochmeister S, Noor A, Lohberger B, Farra N, Petek E, Schwarzbraun T, Ofner L, Loscher WN, Wagner K, Lochmuller H, Vincent JB, Quasthoff S (2008) An X-linked myopathy with postural muscle atrophy and generalized hypertrophy, termed XMPMA, is caused by mutations in FHL1. Am J Hum Genet 82:88–99
Wood-Allum C, Brennan P, Hewitt M, Lowe J, Tyfield L, Wills A (2004) Clinical and histopathological heterogeneity in patients with 4q35 facioscapulohumeral muscular dystrophy (FSHD). Neuropathol Appl Neurobiol 30:188–191
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jordan, B., Eger, K., Koesling, S. et al. Camptocormia phenotype of FSHD: a clinical and MRI study on six patients. J Neurol 258, 866–873 (2011). https://doi.org/10.1007/s00415-010-5858-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-010-5858-z