Abstract
Sex estimation of skeletal parts is of great value even in the DNA era. When computed tomography (CT) facilities were introduced to forensic institutes, new possibilities for sex estimation emerged. The aim of this study was to develop a CT-based method for sex estimation using the mandible. Twenty-five CT-based measurements of the mandible were developed and carried out on 3D reconstructions (volume rendering) and cross sections of the lower jaw of 438 adult individuals (214 males and 224 females). Intraobserver and interobserver variances of the measurements were examined using intraclass correlation coefficient (ICC) analysis. Five discriminant functions were developed using different states of completeness of the mandible. The success rates of these equations were cross validated twice. The measurements were found to be highly reliable (for intraobserver 0.838 < ICC < 0.995 and for interobserver 0.71 < ICC < 0.996). For a complete mandible, the correct classification rate was 90.8%. For incomplete mandibles, the correct classification rates varied from 72.9 to 85.6%. Cross-validation tests yielded similar success rates, for the complete mandible 89% and for the incomplete mandible 67.5 to 89%. We concluded that CT techniques are appropriate for estimating sex based on the mandible size and shape characteristics. Suggested discriminant functions for sex estimation are given with data on the correct classification rates.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sex estimation of skeletal material is one of the most fundamental tasks of forensic and physical anthropologists. Despite the revolutionary advancements in DNA methods in forensic science in recent years, the morphological methods used for estimating sex have retained their relevancy because of a number of reasons such as degradation of DNA under different forensic circumstances (e.g., fires) [1].
Various methods for sex estimation, based on different parts of the skeleton, have been reported [2–12]. Some of these methods rely on morphological features (descriptive), whereas others are based on measurements. The metric methods have a major advantage over the descriptive ones, since they are less dependent on the judgment of the observer [13]. A relatively new method, i.e., geometric morphometrics, has been applied for sex estimation to overcome the disadvantages of the morphologic method [14–17]. Although this method yields good results [16, 18], it requires both special equipment and a specialized researcher. Therefore, it is not applicable for most forensic and physical anthropologists.
Mandibles are both sexually dimorphic and durable (i.e., recovered intact or in an adequate condition) and are thus good candidates for sex estimation of unknown individuals [7, 15, 19–21]. Some studies have used metric characteristics of mandibles to create discriminant functions for sex identification [15, 19, 22]. These studies focused on standard measurements of the mandible such as mandibular angle, bicondylar and bigonial breadths, ramus height, and symphysis height. Other studies used descriptive methods, e.g., flexure of the ramus, shape of the chin, and gonial flaring [6, 23–26]. These methods, based either on continuous or discrete variables, suffer from various deficiencies; to wit, the variables for sex estimation were either arbitrarily selected or statistically picked from a small pool of measurements; the selected metric variables were limited by the available measuring tools (e.g., caliper); most methods were not cross-validated and did not respond to forensic needs (e.g., often only a fragment of the mandible is available); and they were constructed on samples derived from homogeneous populations. With regard to the latter, nowadays, most societies have become more heterogeneous owing to the increase in human mobility between countries and continents [27].
In recent years, recognizing the contribution of various imaging techniques, especially computed tomography (CT) scans, to postmortem investigation has increased [28–32], accompanied by studies that ensure the validity and reliability of these techniques [33–37]. Accordingly, CT is becoming a common diagnostic tool in many forensic institutes. Thus, the need for a CT-based method to estimate sex from skeletal remains has emerged. The aim of this study was to develop a CT-based method for sex estimation using the mandible, which overcomes much of past studies’ deficiencies.
Materials and methods
The study sample was derived from the current Israeli population. This population is particularly suitable for studying biological variation in heterogeneous populations due to its extensive mixture of people migrating to Israel from different parts of the world. The study design is retrospective. Head and neck CT scans of 438 individuals (214 males and 224 females), over the age of 20 years, were randomly selected from a pool of CT scans carried out between the years 2000 and 2012 at the Carmel Medical Center, Haifa, Israel (Brilliance 64, Philips Medical System, Cleveland, Ohio; slice thickness 0.9–3.0 mm, pixel spacing 0.3–0.5 mm, 120 kV, 250–500 mA, number of slices 150–950, and matrix 512 × 512). All CT scans were carried out for diagnostic purposes and for whom a CT exam was medically necessary. Inclusion criteria were as follows: age ≥20 years, intact lower incisors, and at least two teeth of the posterior unit (premolars and/or molars) on each side. Exclusion criteria included the absence of the lower incisors; dental implants and metal restorations that interfere with the measurement; prominent facial and mandibular asymmetry; cranio-facial, temporomandibular joint, or muscular disorders; trauma; previous surgery in the head and neck region (medical files or signs on the skull); and technically aberrant CT scans. This study was approved by the ethical board of the Carmel Medical Center (number 0066–11-CMC).
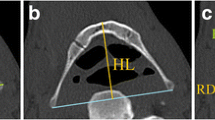
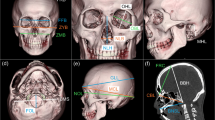
Two sets of measurements (Table 1) were taken using the Philips portal (thin client). The first set (n = 13) includes surface (external) linear measurements from a 3D reconstruction of the mandible, using the volume rendering application of the software (Fig. 1). The second set (n = 12) includes internal linear and area measurements from 2D images or cross sections of the mandible (Fig. 2). Measurements of the mandibular body and symphysis region were taken in relation to the mandibular plane.
Measurements of the mandible taken from a 3D model using the volume rendering technique. Anatomical definitions for each measurement appear in Table 1. Note that measurements of mandibular body height were taken when the mandible was positioned in the mandibular plane (the inferior margins of the mandibular body are positioned parallel to the horizontal plane) in a lateral view (i.e., ascending rami overlap)
Measurements of the mandible taken from lateral and cross-sectional images. Anatomical definitions for each measurement appear in Table 1. Note that cross sections of the mandibular body and symphysis were carried out perpendicular to the mandibular plane (the inferior margins of the mandibular body are positioned parallel to the horizontal plane). The antegonial notch is the space created between the inferior margin of the mandibular body and the mandibular plane
In many forensic/archeological cases, the mandible is incomplete; therefore, we calculated discriminant functions for sex estimation for five different states of completeness of the mandible (hereafter referred to as scenario I to V). Scenario I relates to a complete mandible; therefore, all CT-based measurements could be included in the regression analysis. Scenario II relates to half a mandible (from ramus to chin); thus, measurements of the ramus (length, width, and cross-sectional area (CSA)), body (length, heights, and CSAs), coronoid (height, width, and CSA), condyle (width), mandibular angle region (angle, width, and CSA), and antegonial notch area could be included in the regression analysis. Scenario III relates to a fracture of the mandible where only the mandibular arch (without rami) exists. Here, three external measurements, body height at the premolar and molar regions and chin width, and six internal measurements of the symphysis and chin (heights, thicknesses, and areas) could be included in the regression analysis. Scenario IV included the ramus alone (from coronoid and condyle to the mandibular angle). Here, five external measurements could be included in the regression analysis, which are ramus length and width, coronoid length and width, condyle width, and two internal measurements ramus width CSA and coronoid width CSA. In scenario V, a small fragment of the mandibular body was included. Thus, two external measurements and two internal measurements were included in the forward analysis, which are the mandibular body heights at the premolar and molar regions and their CSAs, respectively.
Statistical analysis
Data were analyzed using SPSS 21.0 software. Significance was set at p < 0.05. Intraobserver and interobserver variations were examined on 15 individuals using the intraclass correlation coefficient (ICC) analysis. For intraobserver variation, measurements were taken twice with a 2-week interval between each by the same researcher (TS). For interobserver variation, measurements were taken by an additional independent researcher (either HM or VS). ICC was interpreted according to the Cicchetti [38] categorization system, <0.40 poor agreement, 0.40–0.59 fair agreement, 0.60–0.74 good agreement, and 0.75–1 excellent agreement.
General summary information, i.e., mean and standard deviation (SD) for each measurement, were obtained via descriptive statistics. An independent sample t test was used to examine the significance of differences between males and females for each measurement. The rate (%) of sexual dimorphism for each measurement was calculated as follows: % dimorphism = [(mean males − mean females)/mean females] × 100. Discriminant functions for sex estimation and their success rates were calculated using logistic regression (forward analysis). Odds ratios (OR) and 95% confident intervals (CIs) were given for variables included in the discriminant equations.
Validation
To examine the validity of the success rates of the suggested discriminant functions for sex estimation, two cross-validation tests were conducted. Random sampling was carried out, using the function RAND (Excel 2013), to select 40 individuals for the test group, i.e., each of the 438 individuals received a random number; the 20 males and 20 females with the lowest numbers were included in the test group. The obtained discriminant functions, based on 398 individuals, were used to estimate the sex of the individuals in the test group. This procedure was carried out twice.
Results
The studied population (n = 438) consisted of 49% males and 51% females with no significant differences (p > 0.05) in age (53.3 ± 19.9 and 56.2 ± 20.6 years, respectively). ICC values for intraobserver and interobserver variations are presented in Table 2. The intraobserver variation of both external and internal measurements showed excellent results (0.905 < ICC < 0.991 and 0.838 < ICC < 0.986, respectively). The interobserver variation of external measurements showed excellent results for all measurements (0.85 < ICC < 0.996), except for two, coronoid width and chin width, which yielded good results (0.71 and 0.715, respectively). Most internal measurements (10 out of 12) showed excellent results (0.763 < ICC < 0.98), except for two (the chin area and the antegonial notch area), which yielded good results (0.741 < ICC < 0.785).
Significant differences between males and females were found for all mandibular external measurements and for most of the internal measurements (Table 3). For all measurements, except for mandibular angle, males have a greater means than females (Table 3). Sexual dimorphism rates varied from 1.6 to 103.1%. The most dimorphic traits were the antegonial notch area (103%), the chin width (22.3%), the body height CSAs (premolar 15.7% and molar regions 16.6%), the symphysis area (13.9%), the ramus length (13.5%), the body height at the molar region (11.1%), the coronoid height (10.8%), the body height at the premolar region (10.5%), the condyle width (10.4%), and the symphysis height (10%). A logistic regression analysis (forward method) was carried out separately for each scenario (I–V). In scenario I, a complete mandible, 6 out of the 25 measurements, were included in the discriminant function, which are ramus length, coronoid height, chin width, bigonial breadth, symphysis height, and antegonial notch area (Table 4), with a successful classification rate reaching 90.8% (similarly for males and females) (Table 5). In scenario II, half mandible, 4 out of 16 measurements, were included in the discriminant function, which are the ramus length, the coronoid height, the condyle width, and the antegonial notch area (Table 4). The classification rate using these measurements was 85.6%, with similar rates for males and females (Table 5). In scenario III, when only the mandibular arch (mandible without rami) was considered for analysis, only three internal measurements out of nine were included in the discriminant function, which are the chin height, the chin width, and the symphysis height (Table 4). The successful classification rate was 79.1%, with a slightly higher correct classification rate for males (80.3%) than for females (77.8%) (Table 5). Scenario IV describes a situation where only the ramus was considered for sex estimation (from coronoid and condyle to the mandibular angle). Two out of seven measurements were included in the discriminant function, which are the ramus length and the coronoid height (Table 4). A correct classification rate of 82.2% (with similar rates for males and females) was achieved (Table 5). Scenario V included a fragment of the mandibular body. Only two measurements out of four were included in the discriminant function, which are the body height at the premolar region and its CSA (Table 4). A correct classification rate of 72.9% (76.9% for males and 68.3% for females) was achieved (Table 5). Table 5 presents the discriminant functions for sex estimation with correct classification rates for the five scenarios of the mandibular state of completeness.
Cross-validation analysis revealed that the fit of our models to a sample of observations, which was not used to estimate the model, was high (Table 6), yielding a mean success rate of 89% for scenarios I (complete mandible) and II (half mandible).
Discussion
The current study provides a series of discriminant functions for sex estimation based on measurements taken from CT scans of the mandibles. Each function was constructed based on a different state of completeness of the mandible. Our study shows a high rate of success discrimination for complete (90.8%) and partially preserved (half) mandibles (85.6%). Successful classification rates of previous methods using different features of the mandibles vary from 59 to 94% [14, 18, 20, 22, 24, 30]. The only study where a successful classification rate greater than ours (94.2%) was reported by Loth and Henneberg [23], who relied on mandibular ramus flexure. However, researchers who tested their method found much lower accuracy rates (66–85.8%) [24, 25, 39]. Additionally, geometric morphometric analysis of the ramus flexure [16] showed that the accuracy of sex estimation using this feature is low and that it has better classification characteristics for males than for females. Considering the rate of sexual dimorphism in mandible features (Table 3), it is clear that discrimination between the sexes based on a single trait is problematic.
The predictive rates of previous studies are lower than ours for two main reasons: (1) variables for sex estimation were either arbitrarily selected or were statistically taken from a small number of measurements and (2) various size and shape characteristics of the mandible could not be utilized by the traditional measuring tools (e.g., CSA and bone thickness).
Our method exhibits several major advantages over previous methods. First, the suggested method enables forensic anthropologists to change from descriptive evaluations (e.g., the robusticity rate, the gonial eversion magnitude) to numeric ones. Second, measurements included in the discriminant equations for sex estimation were taken from a large pool of mandibular measurements, tested statistically for their discrimination power. Third, it enables access to morphological features (e.g., CSA of the mandibular body) not possible by traditional measuring tools (e.g., caliper). Fourth, it provides clear knowledge on the success rates for males and females from a heterogeneous population that has undergone cross-validation. Fifth, it is more adequate for forensic needs because it covers different states of completeness of the mandible.
Limitations of the study
The discriminant functions were developed based on a given population. Although the study population is heterogeneous, the equations should be tested on other populations as well. Although the presented functions can be applied to mandibles of all ages, their applications for elderly individuals should be carried out carefully to ensure that they meet the inclusion criteria (e.g., intact incisors, the presence of molars or premolars at the measured location).
Conclusions
A simple, reliable, and valid method is suggested for forensic scientists for estimating sex, using CT scans of mandibles retrieved from a modern western industrial society. Five discriminant functions, based on mandibular measurements, were constructed to cover various conditions of completeness of the mandible. The greater the completeness of the mandible is, the higher the rate of success discrimination (up to 90.8%) will be. This method is not age dependent and has specific inclusion and exclusion criteria.
References
Jobling MA, Gill P (2004) Encoded evidence: DNA in forensic analysis. Nat Rev Genet 5:739–751
Giles E, Elliot O (1963) Sex determination by discriminant function analysis of crania. Am J Phys Anthropol 21:53–68
Steele DG (1976) The estimation of sex on the basis of the talus and calcaneus. Am J Phys Anthropol 45:581–588
Buikstra JE, Ubelaker DH (1994) Standards for data collection from human skeletal remains. Arkansas Archaeological Survey, Research Series No, Fayetteville, p 44
Rogers T, Saunders S (1994) Accuracy of sex determination using morphological traits of the human pelvis. Journal of Forensic Science 39:1047–1056
Bass W (1995) Human osteology: a laboratory and field method. Thomas, Springfield
Steyn M, İşcan MY (1998) Sexual dimorphism in the crania and mandibles of south African whites. Forensic Sci Int 98:9–16
Bruzek J (2002) A method for visual determination of sex, using the human hip bone. Am J Phys Anthropol 117:157–168
Albanese J (2003) A metric method for sex determination using the hipbone and the femur. J Forensic Sci 48:263–273
Dayal M, Spocter M, Bidmos M (2008) An assessment of sex using the skull of black south Africans by discriminant function analysis. Homo 59:209–221
Dabbs GR, Moore-Jansen PH (2010) A method for estimating sex using metric analysis of the scapula. J Forensic Sci 55:149–152
Urbanová P, Hejna P, Zátopková L, Šafr M (2013) What is the appropriate approach in sex determination of hyoid bones? J Forensic Legal Med 20:996–1003
Sierp I, Henneberg M (2015) The difficulty of sexing skeletons from unknown populations. Journal of Anthropology 2015: Article ID 908535, 13 pages
Franklin D, Freedman L, Milne N, Oxnard C (2006) A geometric morphometric study of sexual dimorphism in the crania of indigenous southern Africans. S Afr J Sci 102:229–238
Franklin D, O'Higgins P, Oxnard C (2008) Sexual dimorphism in the mandible of indigenous south Africans: a geometric morphometric approach. S Afr J Sci 104:101–106
Oettlé A, Pretorius E, Steyn M (2005) Geometric morphometric analysis of mandibular ramus flexure. Am J Phys Anthropol 128:623–629
Kimmerle EH, Ross A, Slice D (2008) Sexual dimorphism in America: geometric morphometric analysis of the craniofacial region. J Forensic Sci 53:54–57
Vance V, Steyn M (2013) Geometric morphometric assessment of sexually dimorphic characteristics of the distal humerus. Homo 64:329–340
Giles E (1964) Sex determination by discriminant function analysis of the mandible. Am J Phys Anthropol 22:129–135
Waldron T (1987) The relative survival of the human skeleton: implications for palaeopathology. In: Boddinton A, Garland AN, Jana-way RC (eds) Death, decay and reconstruction: approaches to archaeology and forensic science. Manchester University Press, Manchester, pp 55–64
Arnay-de-la-Rosa M, González-Reimers E, Fregel R, Velasco-Vázquez J, Delgado-Darias T, González AM, Larruga J (2007) Canary Islands aborigine sex determination based on mandible parameters contrasted by amelogenin analysis. J Archaeol Sci 34:1515–1522
Martin E (1936) A study of an Egyptian series of mandibles, with special reference to mathematical methods of sexing. Biometrika 28:149–178
Loth SR, Henneberg M (1996) Mandibular ramus flexure: a new morphologic indicator of sexual dimorphism in the human skeleton. Am J Phys Anthropol 99:473–485
Kemkes-Grottenthaler A, Löbig F, Stock F (2002) Mandibular ramus flexure and gonial eversion as morphologic indicators of sex. Homo 53:97–111
Balci Y, Yavuz M, Cağdir S (2005) Predictive accuracy of sexing the mandible by ramus flexure. Homo 55:229–237
Hu K, Koh K, Han S, Shin K, Kim H (2006) Sex determination using nonmetric characteristics of the mandible in Koreans. J Forensic Sci 51:1376–1382
Bade KJ (2004) Legal and illegal immigration into Europe: experiences and challenges. European Review 12:339–375
Leth PM (2009) Computerized tomography used as a routine procedure at postmortem investigations. Am J Forensic Med Pathol 30:219–222
Thali MJ, Braun M, Wirth J, Vock P, Dirnhofer R (2003) 3D surface and body documentation in forensic medicine: 3-D/CAD photogrammetry merged with 3D radiological scanning. J Forensic Sci 48:1356–1365
Thomsen AH, Jurik AG, Uhrenholt L, Vesterby A (2009) An alternative approach to computerized tomography (CT) in forensic pathology. Forensic Sci Int 183:87–90
Thali MJ, Yen K, Schweitzer W, Vock P, Boesch C, Ozdoba C, Schroth G, Ith M, Sonnenschein M, Doernhoefer T, Scheurer E, Plattner T, Dirnhofer R (2003) Virtopsy, a new imaging horizon in forensic pathology: virtual autopsy by postmortem multislice computed tomography (MSCT) and magnetic resonance imaging (MRI)—a feasibility study. J Forensic Sci 48:386–403
Dirnhofer R, Jackowski C, Vock P, Potter K, Thali MJ (2006) VIRTOPSY: minimally invasive, imaging-guided virtual autopsy. Radiographics 26:1305–1333
Uysal SELMA, Gokharman D, Kacar M, Tuncbilek I, Kosa U (2005) Estimation of sex by 3D CT measurements of the foramen magnum. J Forensic Sci 50:1310–1314
Thali M, Yen K, Plattner T, Schweitzer W, Vock P, Ozdoba C, Dirnhofer R (2002) Charred body: virtual autopsy with multi-slice computed tomography and magnetic resonance imaging. J Forensic Sci 47:1326–1331
Verhoff MA, Ramsthaler F, Krähahn J, Deml U, Gille RJ, Grabherr S, Thali MJ, Kreutz K (2008) Digital forensic osteology—possibilities in cooperation with the Virtopsy® project. Forensic Sci Int 174:152–156
Ramsthaler F, Kettner M, Gehl A, Verhoff MA (2010) Digital forensic osteology: morphological sexing of skeletal remains using volume-rendered cranial CT scans. Forensic Sci Int 195:148–152
Roberts IS, Benamore RE, Benbow EW, Lee SH, Harris JN, Jackson A, Mallett S, Patankar T, Peebles C, Roobottom C, Traill ZC (2012) Post-mortem imaging as an alternative to autopsy in the diagnosis of adult deaths: a validation study. Lancet 379:136–142
Cicchetti DV (1994) Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol Assess 6:284–290
Hill CA (2000) Technical note: evaluating mandibular ramus flexure as a morphological indicator of sex. Am J Phys Anthropol 111:573–577
Acknowledgements
The authors wish to thank the Dan David Foundation for their financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tunis, T.S., Sarig, R., Cohen, H. et al. Sex estimation using computed tomography of the mandible. Int J Legal Med 131, 1691–1700 (2017). https://doi.org/10.1007/s00414-017-1554-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-017-1554-1