Abstract.
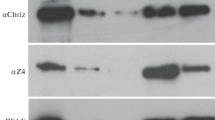
CHD1, an M r∼200,000 protein that contains a chromodomain (C), an ATPase/helicase-like domain (H) and a DNA-binding domain (D), was previously shown to be associated with decompacted interphase chromatin in mammalian cells and with transcriptionally active puffs and interbands in Drosophila polytene chromosomes. We now show by transient transfection experiments with genes expressing wild-type and mutant forms of CHD1 that both the C and H domains are essential for its proper association with chromatin. We also present evidence for an in vivo interaction between CHD1 and a novel HMG box-containing protein, SSRP1, which involves an amino-terminal segment of CHD1 that does not include the chromodomain. Immunocytochemical analyses indicated that CHD1 and SSRP1 colocalize in both mammalian nuclei and Drosophila polytene chromosomes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 6 September 1998; in revised form: 28 October 1998 / Accepted: 12 November 1998
Rights and permissions
About this article
Cite this article
Kelley, D., Stokes, D. & Perry, R. CHD1 interacts with SSRP1 and depends on both its chromodomain and its ATPase/helicase-like domain for proper association with chromatin. Chromosoma 108, 10–25 (1999). https://doi.org/10.1007/s004120050347
Issue Date:
DOI: https://doi.org/10.1007/s004120050347