Abstract
Alterations in complex lipids may be involved in pathophysiology of schizophrenia spectrum disorders. Previously, we demonstrated importance of detecting lipid metabolism dysregulation by acylcarnitine (ACs) profile analysis in patients with first-episode psychosis (FEP). The aim of this study was to adopt lipidomics to identify serum glycerophospholipids (GPLs) and sphingomyelins (SMs) for describing FEP status before and after 7-month antipsychotic treatment. Using mass spectrometry and liquid chromatography technique, we profiled 105 individual lipids [14 lysophosphatidylcholines (LysoPCs), 76 phosphatidylcholines (PCs) and 15 SMs] in serum samples from 53 antipsychotic-naïve FEP patients, 44 of them were studied longitudinally and from 37 control subjects (CSs). Among the identified and quantified metabolites one LysoPC was elevated, and contrary the levels of 16 PCs as well as the level of one SM were significantly (p ≤ 0.0005) reduced in antipsychotic-naïve FEP patients compared to CSs. Comparison of serum lipids profiles of FEP patients before and after 7-month antipsychotic treatment revealed that 11 GPLs (2 LysoPCs, 9 PCs), and 2 SMs were found to be significantly changed (p ≤ 0.0005) in which GPLs were up-regulated, and SMs were down-regulated. However, no significant differences were noted when treated patient’s serum lipid profiles were compared with CSs. Our findings suggest that complex lipid profile abnormalities are specifically associated with FEP and these discrepancies reflect two different disease-related pathways. Our findings provide insight into lipidomic information that may be used for monitoring FEP status and impact of the treatment in the early stage of the schizophrenia spectrum disorder.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Schizophrenia (SCH) is a heterogeneous disorder with multi-etiopathogenetic nature. A number of hypotheses have been highlighted to explain the pathological processes regarding SCH spectrum psychoses. The molecular changes are associated with disturbed neurotransmission [1], imbalance in immune and oxidative systems as well as metabolic shifts both in the central nervous system and periphery [2,3,4]. In addition, abnormalities in the level, metabolism, and genetic regulation of bioactive lipids have been found in brain tissue as well as in the periphery [5], supporting the membrane hypothesis of SCH [6]. Therefore, treatment strategies targeting abovementioned systems, involving also metabolomics changes may have impact on patient stratification and personalized medicine approaches.
Metabolomics, targeted to assay of low-molecular compounds (metabolites) of the human body, is a novel challenge to find disease-related new biomarkers or to identify disease- or intervention-derived shifts in metabolism [7]. Rapidly growing part in metabolomics is lipidomics comprising of a wide spectrum of lipid metabolism-derived or lipid metabolism-related species in body fluids and providing new insights into the pathogenesis of diseases. Metabolism/catabolism of each class of lipids comprises multiple steps, and therefore, generates a large array of intermediate molecules or metabolites with diverse physicochemical properties [8]. Thus, it is crucial to accurately identify and quantify these molecules. Using the flow injection analysis tandem mass spectrometry [(FIA)-MS/MS] and liquid chromatography [(LC)-MS/MS] technique, we previously reported that patients in the early stage of the SCH spectrum disorder demonstrate acylcarnitine (ACs) profile alterations before and after 7-month antipsychotic treatment [9]. ACs are essential compounds for the metabolism of fatty acids and represent intermediates of mitochondrial fatty acid β-oxidation [8]. Our findings indicated that antipsychotic-naïve FEP patients are characterized by different changes in the profile of short-chain ACs (SCACs) and long-chain ACs (LCACs) [9]. To be more specific, the levels of LCACs (C16:0, C16:1, C16:1-OH, C18:1, C18:2) were elevated and the levels of SCACs (C3, C5) demonstrated reduced formation of these molecules in drug-naïve FEP patients compared with that of CSs. These changes in ACs profile were accompanied by elevated levels of pro-inflammatory cytokines. Furthermore, treatment with antipsychotic drugs reversed the levels of LCACs, SCACs, and biomarkers of low-grade inflammation to that measured in CSs [9].
In this study, using partially the same sample, we extended to quantification of further lipid classes and focused on glycerophospolipids (GPLs) and sphingolipids (SLs) (e.g. SMs) which are crucial cellular and intracellular compounds. They are required for active membrane transport and for several enzymatic processes, membrane formation, and cell signaling. Disruption of their metabolism leads to several diseases, with diverse metabolic, neurological, and psychiatric consequences. Among blood lipid-related species, several GPLs have been recently highlighted as potential biomarkers of cardiovascular disease [10]. Furthermore, recent reports, including our data [11], have shown just a few individual molecular species among phosphatidylcholines (PCs) and LysoPCs have impact as potential novel biomarkers for cardiovascular risk and total mortality [12].
GPLs consist of the following parts: a glycerol backbone, esterified by two long-chain fatty acids at carbons 1 and 2 (C1 and C2), phosphoric acid esterified to the C3 hydroxyl group of glycerol and usually an alcohol head group esterified to the phosphate group. Prominent parts of GPLs family are PCs containing choline as an alcohol head group. The PCs’ structural role in plasma membrane and other membranes of cellular compartments as well as in blood lipoproteins is indispensable. PCs are important in membrane-mediated cell signaling [13]. The partial hydrolysis of PCs removing a fatty acid residue (e.g., arachidonic acid, docosahexaenoic acid) from carbons 2 by phospholipase A2 results in genesis of LysoPCs [14]. This has great impact as first, LysoPCs represent a reservoir for arachidonic acid known as a central precursor for lipid-related potent signaling molecules; second, LysoPCs generated from PCs belong to bioactive lipids involved in monocyte recruitment, vascular smooth muscle cell proliferation, and endothelial dysfunction [15]. In addition, PCs and LysoPCs are tightly related prominent members of the GPLs family. SLs consist of aliphatic amino alcohol sphingosine, fatty acid residue, and a head group. SMs, the members of SLs family, have phosphocholine or phosphoethanolamine as a head group.
Several studies reported altered levels of PCs in SCH regarding the brain tissues, blood plasma, or blood serum [6, 16,17,18,19,20,21]. However, some authors did not find significant difference in phospholipids on the basis of global lipidomics profiling [22, 23]. Data concerning metabolic profiling of SMs regarding SCH spectrum disorders suggest that sphingolipid species are altered in the brain of patients with SCH [24, 25]. Furthermore, disrupted expression of genes encoding enzymes involved in SLs metabolism [26] and a network analysis of candidate genes highlighted the involvement of myelin-related pathways in SCH [27]. Lipidomic studies of FEP are, at present, relatively limited. To our best knowledge there is only one study about blood plasma GPLs, plasmalogens [28], and few research groups have studied serum targeted metabolomic profiling for drug-naïve FEP patients or subjects with sub-threshold psychosis comprising PCs, LysoPCs, and SMs [29,30,31]. In addition, findings suggest that during the early stage of the illness dysregulation in the bioactive lipid pathways may be compensated, or reversed by treatment [26, 29] and blood metabolite profiles predict future weight gain and other metabolic abnormalities in psychotic patients [30].
Therefore, it is apparent that further studies are needed by using comprehensive approach for assay of circulating PCs, their derivatives LysoPCs, and SMs. Together with existing data it allows to shed more light on impact of intriguing part of lipid metabolism-derived species like GPLs, LysoPCs, and SMs in pathogenesis of SCH as well as in metabolic profile of FEP patients. Accordingly, the identification of notable changes in PCs, LysoPCs, and SMs levels or profiles compared with that of healthy people during disease initiation or progression could provide diagnostic and prognostic information as well as aid for treatment decisions of SCH spectrum disorders. For example, individual PCs and LysoPCs molecules differ both in their functional properties and their susceptibility to oxidative stress due to diverse composition and distribution of unsaturated and saturated fatty acids on the glycerol backbone [32]. Focusing on lipid molecular species rather than lipid classes may thus be an efficient way to uncover their role in the pathogenesis of diseases including SCH spectrum disorders.
Taking into account the aforementioned context, the objective of this study was to characterize general profiles of GPLs and SMs as well as to find the specific shifts in these biomolecule-related profiles in patients with FEP before and after 7-month antipsychotic treatment compared to CSs and to evaluate the effect of treatment on these metabolite levels.
Materials and methods
Participants
People with first psychotic episode (53 patients) were recruited at the time of their first clinical contact for psychotic symptoms at the Psychiatric Clinic of Tartu University Hospital, Estonia. The patients were recruited if they met the following criteria: men or women aged between 18 and 45; had experienced the FEP; the duration of their untreated psychosis had been less than 3 years; no antipsychotic use prior to the study. Participants with psychosis were allowed to receive anti-anxiety medication the night before first blood sample was collected. Patients were excluded from the study if they had psychotic disorders owing to a general medical condition or had substance induced psychosis. FEP diagnoses were based on clinical interviews according to the International Classification of Diseases, Tenth Edition (ICD-10) criteria [33]. Among the patients, the frequencies of various diagnoses were as follows: acute polymorphic psychotic disorder without symptoms of schizophrenia (F23.0, n = 9); acute polymorphic psychotic disorder with symptoms of schizophrenia (F23.1, n = 10); acute schizophrenia-like psychotic disorder (F23.2, n = 14); other acute predominantly delusional psychotic disorder (F23.3, n = 1); paranoid schizophrenia (F20.09, n = 14); undifferentiated schizophrenia (F20.39, n = 4), and schizotypal disorder (F21, n = 1). F20.09, F20.39, and F21 category patients had experienced psychotic symptoms for longer than 1 month.
Forty-four patients participated at the point of follow-up in the study. Seven patients had lost contact with health-services, two refused to take antipsychotic medications and they were excluded from the follow-up analysis. History of antipsychotic medication use was collected according to reviews of patients’ medical charts. No restrictions were made in terms of usage of specific pharmacological agent due to naturalistic and longitudinal study design and patients were treated with various antipsychotic medications according to clinically relevant circumstances. During the second blood collection 14 patients received quetiapine (among them 7 cases as only antipsychotic treatment), 11 patients received aripiprazole (3 cases as only antipsychotic treatment), 14 were treated with olanzapine (10 cases as only antipsychotic treatment), 5 patients were assigned to risperidone (4 case as only treatment), 4 patients to sertindole (2 case as only antipsychotic treatment), 3 patients to ziprasidone, 2 patients to clozapine (in both cases clozapine was administered in the combination with other psychotropic drugs), and 3 patients to perphenazine (2 case as only treatment). At the time of the follow-up blood collection the mean theoretical chlorpromazine dose equivalent [34] was 363 ± 165 (range 80–780) mg. Thirty-three patients were treated only with antipsychotics, but eleven patients additionally needed mood stabilizers, antidepressants, or hypnotics.
The control group was recruited using advertisements and 37 mentally healthy subjects (with residence in the catchment area of the Psychiatric Clinic of Tartu University Hospital) participated in the study as CSs. They were interviewed by experienced psychiatric doctors to avoid the inclusion of CSs with mental disorders. Exclusion criteria for the control group also included psychotic disorders among close relatives. Additional exclusion criteria for all participants were as follows: history of diabetes, neurological, and immune-related diseases. As it was a naturalistic study, substance abuse was not exclusion criteria for either group. Partially FEP group and entirely CSs group were subjected to the versatile characterization by previous studies [9, 35,36,37].
Procedure
Fasting blood samples, clinical, and BMI data of the FEP patients were assessed at two time points: on admission and after the follow-up period (mean duration 7.2 ± 0.7 months). The time duration between two occasions consisted of initial stabilization of acute psychotic symptoms (took approximately a month) and further 6-month continuous treatment with antipsychotics. Range and severity of psychopathology was assessed using the Brief Psychiatric Rating Scale (BPRS) [38]. The BPRS consists of 18 symptoms and each item is measured on a seven-point Likert scale from “not present” to “extremely severe”. A total score was used as the outcome.
Fasting blood samples and BMI data from CSs were collected cross-sectionally.
Blood collection and clinical laboratory measurements
Fasting serum samples of FEP patients from two different time points (before and after 7-month antipsychotic treatment) and CSs (one time) were collected to metabolomic measurement. Blood samples were collected using standard venipuncture technique between 09:00–11:00 a.m. Blood (5 ml) was sampled in anticoagulant-free tubes and kept for 1 h at 4 °C (for platelet activation) before serum was isolated (centrifugation at 2000 rpm for 15 min at 4 °C). Samples were collected during 5 years’ period of time, and recruitment to control group and FEP patients’ group was carried out in parallel. Serum samples were frozen and stored in identical conditions at − 20 °C for 3- to 8 years before determination of biomarkers.
Measurement of GPLs and SMs
Metabolite concentrations were determined using the targeted metabolomics kit AbsoluteIDQ™ p150 (BIOCRATES Life Sciences AG, Innsbruck, Austria) applying the flow injection analysis tandem mass spectrometry [(FIA)-MS/MS] as well as liquid chromatography [(LC)-MS/MS] technique as described previously [9, 37]. The metabolomics data set contained 14 LysoPCs, 76 PCs, and 15 SMs (complete list of the analysed metabolites is demonstrated in Supplementary Tables S-1–S-3). In addition, metabolite sums and ratios that were assumed to indicate a certain metabolic state or process were calculated. The assays were performed according to the manufacturer’s manual UM-P180. Identification and quantification of GPLs and SMs were achieved using multiple reactions monitoring along with internal standards. Calculation of metabolite concentrations was automatically performed by MetIDQ™ software (BIOCRATES Life Sciences AG). Data quality was checked based on the level of detection (LOD) (see also special comments at the bottom of Supplementary Tables). Median and range of all measured GPLs and SMs are given in the Supplementary Table S-1–S-3. GPLs are divided on the basis of ester (a) and ether (e) bonds in the glycerol moiety, whereas “aa” (diacyl) denotes that at positions sn-1 and sn-2 of glycerol moiety is a fatty acid residue and “ae” (acyl-alkyl) denotes that one the residues is acyl residue but second is a alkyl residue. For example, PC-aa-C32:1 denotes a phosphatidylcholine with 32 carbons in the two fatty acid side chains and a single double bond in one of them or SM-C20:2 denotes a sphingomyelin with 20 carbons and two double bounds in fatty acid side chain.
Statistical analyses
In the first step, all data were examined for normality of distribution using the Shapiro–Wilk test. In the next step, normally distributed data (age, BMI, psychopathology score) were analyzed using the Student’s t test. Dichotomous data (sex and smoking status) were analyzed using the Chi square test. Non-normally distributed data (levels of the metabolic markers) were analyzed using the Mann–Whitney U test to establish preliminary metabolic profile differences between groups (FEP patients’ pre-treatment or post-treatment condition vs. CSs). Wilcoxon Matched Pairs Test was used to demonstrate within-group (FEP patients pre- vs. post-treatment condition) GPLs and SMs serum level differences. For within-subjects’ analyses patients were paired one by one. Because of large number of simultaneous comparisons, meaningful differences were determined by setting the significance level using the Bonferroni procedure. This resulted in a corrected critical p value for the GPLs and SMs between- and within-group level differences of ≤ 0.0005. The effect sizes (η2) for significant non-parametric test results were calculated (the values of squared standardized test statistics (Z) were divided by the total number of observations on which Z was based). Effect sizes ≥ 0.14 were interpreted as large [39]. A non-parametric Levene’s test was used to verify the equality of variances between and within the groups [40]. Thereafter, we used general linear models (GLM) to confirm biomarkers levels’ differences between the groups (i.e. drug-naïve FEP patient vs. CSs), and within the group (i.e. drug-naïve FEP patient vs. FEP patients after treatment). Prior to analyses, the metabolic data were log10-transformed to reduce the heterogeneity of variance commonly associated with biomarker data. Categorical (disease, sex, smoking status) and continuous (age) covariates were used in the GLM to compare biomarkers levels (dependent variables) between groups. To determine which type of model best fits the data, backward variable elimination was used. Each subsequent step removed the least significant variable in the model until all remaining variables had individual p values smaller than 0.05. F tests were used to further compare the fits of linear models and analyze significant (disease or treatment) main effects in the final models and partial eta2 values (the proportion of the effect in addition to error variance that is attributable to the effect) were established for the final models. Partial eta2 values more than 0.26 were defined as large effects [39]. The statistical analyses were performed using Statistica software [41] for Windows.
Results
General description of the study groups
Clinical and demographic features of all the participants included in the study are summarized in Table 1. There were no statistically significant differences between antipsychotic-naïve FEP patients and CSs in terms of age (t(88) = 1.10, p = 0.28), sex (\(\chi _{{(1)}}^{2}\) = 2.57, p = 0.11), tobacco use (\(\chi _{{(1)}}^{2}\) = 3.04, p = 0.08), or mean (± SD) values of BMI (22.9 ± 3.1 and 23.0 ± 3.1, respectively; t(88) = − 0.21, p = 0.84). With regard to substance abuse, 17 patients (14 males) and one CS (male) (\(\chi _{{(1)}}^{2}\) = 8.32, p = 0.004) reported having tried or habitually used cannabis at some point during their lifetime. None of the study participants met cannabis or alcohol dependence criteria. During the 7-month treatment psychopathology (BPRS) score decreased (t(43) = 11.23, p < 0.00001) and BMI increased significantly (t(43) = − 7.36, p < 0.00001) in FEP patients group.
Profiles of GPLs and SMs in FEP patients before and after antipsychotic treatment
FEP induced alterations in levels of GPLs and SMs
First, to clarify baseline differences in GPLs and SMs profile of FEP patients compared to CSs we used Mann–Whitney U test. From 90 circulating GPLs the level of one LysoPC (LysoPC-a-C20:4) was significantly elevated and on the contrary, the levels of 11 PC-aa-Cs (PC-aa-C30:0, PC-aa-C32:1, PC-aa-C32:2, PC-aa-C34:2, PC-aa-C34:3, PC-aa-C34:4, PC-aa-C36:1, PC-aa-C36:2, PC-aa-C36:3, PC-aa-C36:6, PC-aa-C38:3), the levels of 5 PC-ae-Cs (PC-ae-C34:2, PC-ae-C36:2, PC-ae-C36:3, PC-ae-C40:2, PC-ae-C40:4) as well as the level of one SM (SM-C20:2) were significantly reduced in antipsychotic-naïve FEP patients compared to CSs (Table 2, and detailed results are presented in the Supplementary Table S-1). According to non-parametric Levene’s test, all biomarker level’s differences that reached statistical significance had equal variances (Supplementary Table S-1).
In addition ratio of Total_PC-aa/Total_LysoPC-a and PC-aa-C36:3/PC-aa-C36:4 were diminished, and ratio of LysoPC-a-C20:4/LysoPC-a-C20:3 was elevated in FEP patients before treatment compared to CSs (Table 2). All these differences survived the Bonferroni correction (p value less or equal to 0.0005) and displayed large effects (effect size units (η2) range was 0.14–0.42).
To evaluate the simultaneous main effect of the FEP on abovementioned metabolite levels, alternative methodological approach (GLM) was used. All these metabolites (Table 3) had substantial contribution to the model fit. The final model demonstrated a large main effect of the disease (F(18,67) = 5.48, p < 0.00001, partial eta2 = 0.60) on the GPLs and SMs levels. The strongest associations emerged between the reduced levels of PCs (especially PC-aa-C32:2, PC-aa-C34:3, PC-aa-C34:4, PC-aa-C36:2, PC-aa-C36:3) and the presence of the FEP.
Antipsychotic treatment induced alterations in levels of GPLs and SMs in FEP patients
Comparison of PCs and SMs levels before and after treatment (Wilcoxon matched pairs test) with antipsychotic drugs revealed that the levels of two LysoPC-a-C (LysoPC-a-C14:0, LysoPC-a-C20:3), Total_PC-aa, and 9 PCs (PC-aa-C32:2, PC-aa-C34:3, PC-aa-C34:4, PC-aa-C36:1, PC-aa-C36:2, PC-aa-C36:3, PC-aa-C36:6, PC-aa-C38:3, PC-aa-C40:5) as well as the ratios between PC-aa-C36:3/PC-aa-C36:4, and Total_PC-aa/Total_SM were significantly increased after 7-month treatment (Table 4, and for more detailed results see Supplementary Table S-2). To the contrary, the levels of two SMs (SM-(OH)-C16:1, SM-C18:0), and the ratios of LysoPC-a-C16:0/LysoPC-a-C16:1 and LysoPC-a-C20:4/LysoPC-a-C20:3 were lower after treatment. All these differences within the group survived the correction for multiple comparison (p ≤ 0.0005) and they all displayed a large effect (effect size range η2 = 0.14–0.31). With respect to SM-(OH)-C16:1 level difference, non-parametric Levene’s test verified that the homogeneity of variance was violated (F(1,70) 4.41, p = 0.04) (Supplementary Table S-2).
To demonstrate the main effect of the 7-month antipsychotic treatment on concentrations of the serum GPLs and SMs, repeated measures GLM was used. Aforementioned set of metabolites that demonstrated statistically significant change after antipsychotic treatment were entered into analysis and backward elimination procedure was implemented to remove valueless variables and to establish a combination of significantly changed GPLs and SMs levels. The best model comprised of two LysoPC-a-C (LysoPC-a-C14:0, LysoPC-a-C20:3) and nine PCs (PC-aa-C32:2, PC-aa-C34:3, PC-aa-C34:4, PC-aa-C36:1, PC-aa-C36:2, PC-aa-C36:3, PC-aa-C36:6, PC-aa-C38:3, PC-aa-C40:5) and confirmed that antipsychotic treatment caused elevation in these metabolite levels in FEP patient group (F(11,76) = 4.05, p = 0.0001, partial eta2 = 0.37). The most prominent effects emerged between treatment and elevation of PC-aa-C36:2 and PC-aa-C36:3.
Comparison of levels of GPLs and SMs in FEP patients treated for 7 months with antipsychotic drugs and in CSs
Among GPLs 14 (2 LysoPCs, 1 PC-aa, 11 PC-ae) and among SMs two metabolites demonstrated differences when FEP patients’ serum levels were compared to CSs (Supplementary Table S-3). However, these differences as well as calculated metabolite ratio comparisons did not survive Bonferroni correction. This suggested that after the 7-month antipsychotic treatment most of the GPLs and SMs of FEP patients returned to the level comparable to CSs.
Discussion
Lipids serve various physiological functions in the living organisms. They are essential for the assembling of cellular membranes, the storage of metabolic energy, and as signaling molecules in cell metabolism. In addition, lipids also are implicated as playing direct and causal roles in the pathophysiology of human diseases, including cardiovascular diseases [10, 12], metabolic diseases [42], chronic inflammatory processes [43], and various neuropsychiatric diseases [44]. Alterations of lipid profiles can precede the onset of diseases or be a consequence of the side-effect of antipsychotic treatment in SCH spectrum disorder [16, 19, 29, 45]. Still, to get deeper look of lipid metabolism in SCH-related changes, including effect of treatment, the shifts/levels of the individual compounds of several lipid metabolic groups like GPLs and SMs should be evaluated. Among GPLs a subgroup of greatest interest is PCs, a class of phospholipids with choline headgroup, being a major component of biomembranes. In postmortem study on thalamus homogenate a decrease of PCs and crucial myelin membrane components SMs and galactocerebrosides in SCH patients was demonstrated [16]. Study on postmortem prefrontal white matter in bipolar disorder (BD) and SCH patients showed significantly lower PC and PC20:4n6, phosphatidylethanolamine (PE) 22:5n6 and PC22:5n6 levels in the BD group and lower PC20:3n6, PE22:5n6 and PC22:5n6 levels in the SCH group, compared to CSs. Higher levels of PE22:0, PE24:1, and PE20:2n6 in the BD group and PE22:1 in the SCH group, compared to CSs were found [20]. In lipidomics study Schwarz et al. [17], using white and gray matter as well as red blood cell samples (including seven samples from drug-naïve first onset patients), found statistically significant shifts in levels of free fatty acids and PCs in gray and white matter and significantly elevated ceramides in white matter of both SCH and BD samples compared to CSs.
Naturally it raises a question about the profile of the circulating serum levels of PCs in FEP patients. Recent thorough lipidomic study on serum of FEP patients established on the basis of cluster analysis several strongest peaks in some lipidomic clusters containing also PCs, e.g., PC(34:1), PC(34:2), PC(36:1), PC(36:2), PC(36:3), PC(36:4), PC(38:3) [30]. They also noticed that the case group had lower levels of PCs with the most marked decline at the 2-month follow-up time point in lipidomic cluster 12 containing also PCs (38:4). Present study, involving drug-naïve FEP patients, established that FEP is accompanied by a significant decline of the levels of PC-aa-C30:0, PC-aa-C32:1, PC-aa-C32:2, PC-aa-C34:2, PC-aa-C34:3, PC-aa-C34:4, PC-aa-C36:1, PC-aa-C36:2, PC-aa-C36:3, PC-aa-C36:6, PC-aa-C38:3, and calculated Total PC-aa. Furthermore, the levels of PC-ae-C34:2, PC-ae-C36:2, PC-ae-C36:3, PC-ae-C40:2, PC-ae-C40:4, and SM-C20:2 were also reduced if FEP patients compared to CSs. Contrarily, the level of LysoPC-a-C20:4 was elevated in patients when compared to CSs (Table 2, Table S-1). All these metabolites demonstrated large effect sizes while two groups were compared. Subsequently, using the GLM model, we confirmed the strong main effect of the disease (p = 0.0000001, partial eta2 = 0.60) for all of these biomolecule level alterations. The most salient biomolecules in the model were PC-aa-C32:2, PC-aa-C34:3, PC-aa-C34:4, PC-aa-C36:2 and PC-aa-C36:3.
Next, the 7-month antipsychotic treatment of FEP patients induced significant increase in the levels of several PCs and two LysoPCs, but decreased the levels of SM-(OH)-C16:1 and SM-C18:0 (Table 4, Table S-2). According to GLM, the best model comprised of PC-aa-C32:2, PC-aa-C34:3, PC-aa-C34:4, PC-aa-C36:1, PC-aa-C36:2, PC-aa-C36:3, PC-aa-C36:6, PC-aa-C38:3, PC-aa-C40:5, LysoPC-a-C14:0, and LysoPC-a-C20:3 verified that antipsychotic treatment caused elevation in these lipid metabolite levels in FEP patient group (p = 0.0001, partial eta2 = 0.37) with the most prominent effects for elevation of PC-aa-C36:2, and PC-aa-C36:3. Interestingly, systemic statistical approach to compare FEP patients treated for 7 months and CSs shows that the data for most GPLs and SMs did not differ. Our findings concerning PCs are in agreement with the study that plasma levels of plasmalogens (PCs and PE plasmalogens) were significantly lower in FEP patients compared to CSs [28].
Decline in the levels of mentioned PCs has evidently several reasons. PCs are principal phospholipids circulating in plasma, as components of the blood lipoproteins, especially the high-density lipoprotein (HDL). Recent review and meta-analysis highlighted that first-episode non-affective psychosis patients have subclinical dyslipidemic profile comprising also the lower levels of HDL [46]. Next, in animals PCs belong mainly to diacyls (PC-aa), containing typically C16 and C18 saturated fatty acids, C18:1, C18:2 monounsaturated fatty acids, and C18, C20, C22, C24 polyunsaturated fatty acids. Thus, PC-aa-C34:3 may contain C18:1, PC-aa-C32:2 and PC-aa-C36:2 may contain C18:2, and PC-aa-C36:3 may contain C18:1 and C18:2. Recently, we showed that the levels of circulating LCACs (C16:0, C16:1, C16:1-OH, C18:1, and C18:2) were elevated and these biomarkers’ elevation was related to high serum concentration of pro-inflammatory cytokine interleukin (IL-)6, and epidermal growth factor in FEP patients [9]. According to these results, FEP is accompanied by lipidomic shifts characterized by elevated conversion of PC-aa-C32:2, PC-aa-C34:3, and PC-aa-C36:2 that leads to lower levels of them and taken up by carnitine C16, C16:1, C18:1, and C18:2 contribute to elevation of the levels of acylcarnitines C16, C16:1, C18:1, and C18:2.
Moreover, we also established elevated level of LysoPC-a-C20:4 in FEP patients. LysoPCs are formed by action of both phospholipases and acyltransferases. For example, a significant amount of blood plasma LysoPCs is formed by lecithin-cholesterol acyltransferase (LCAT), which is secreted from the liver. This enzyme transfers the fatty acid of position sn-2 of PC to the free cholesterol in plasma, with formation of cholesteride and LysoPC. As PC-aa-C34:4 may contain an arachidonic acid residue (C20:4) it might be assumed that the decreased level, seen in antipsychotic-naïve FEP patients, should lead to elevation of the levels of LysoPC-a-20:4, which contains a chain (C20:4) at the sn-1 position. It should be noted that lysophospholipids (LysoPC-a-C20:4) have a role in lipid signaling by action on lysophospholipid receptors (LPL-R), members of the G protein-coupled receptor family of integral membrane proteins. In addition, recently it was suggested that LCAC C18:1 suppresses glycine (Gly) transport by Gly transporter GlyT2, related to the nociceptive pathways [47]. The tight relation of nociceptive and inflammatory processes is well known. Thus, lipidomic-profile shifts, particularly intensive use of certain GPLs (seen as reduced levels of PC-aa-32:2, PC-aa-34:3, PC-aa-34:4, PC-aa-36:1, PC-aa-36:2, PC-aa-36:3, PC-ae-36:3), and elevation of LCACs (C16:0, C16:1, C16:1-OH C18:1, C18:2) [9] together with excess supply of LysoPC-a-C20:4 refer to some impairments in cell membrane structure and function in antipsychotic-naïve patients with FEP.
Additionally, a certain mitochondrial dysfunctionality is also involved due to circumstance that mitochondria are a functional “cross-road” in whole body metabolism (lipids, glucose, amino acids). Recently we showed elevated hexose levels in FEP patients [9], where we established decline of PCs. Both facts refer to certain metabolic shifts in FEP patients comprising lipids and glucose. It is known that mitochondrial 3-β-hydroxybutyrate dehydrogenase converting acetoacetate into keto body beta-hydroxybutyrate needs PCs as an allosteric activator [48].
We found that FEP induced a significant decline of SM-C20:2 (it denotes a SM with 20 carbons and two double bounds in fatty acid side chain) compared to CSs. Furthermore, the GLM confirmed an effect of the disease on decreased level of SM-C20:2 (Table 3), what makes sense to highlight some aspects. Data about metabolic profiling of SMs are still quite limited. Some papers referred to SLs abnormalities in psychiatric disorders [26] and revealed altered PCs and SMs profile in the prefrontal cortex of a patient with SCH [25]. Ceramide and SMs are evidently associated with the development of metabolic syndrome. Hanamatsu and colleagues [49] showed in obese persons that the high levels of serum SMs with saturated acyl chains (C18:0, C20:0, C22:0, and C24:0), but not SMs containing unsaturated acyl chains, were closely associated with the development of metabolic syndrome (MetS) and might serve as novel biomarkers of MetS and associated diseases. As highlighted in the recent thorough review, already small shifts in phospholipid levels appear to have large implications on MetS-related parameters like lipid profiles, obesity, and insulin resistance [50].
In the previous article, using partially same sample, we found that 7 months of antipsychotic drug treatment caused growth in BMI and imbalance in metabolic biomarkers (C-peptide and leptin levels increased, and adiponectin level decreased) which point to problems in insulin processing already in the early stage of disorder [36]. Now we demonstrated that antipsychotic treatment leads to some shifts in certain types of SMs, although effects remain limited and affected types of SMs comprised from saturated and unsaturated fatty acids (Tables 4, 5).
It is worth to empasize that after 7-month follow-up period GPLs and SMs profiles were comparable between FEP patients and CSs. Previously we indicated that antipsychotics had therapeutic effects on FEP patients clinical symptoms and at the same time treatment revealed desirable impact on the measured biomolecule levels. None of the biomolecule level difference between treated patients group and CSs was statistically significant after Bonferroni correction was applied to adjust for multiple comparisons. However, the absence of statistically significant differences between treated FEP patients and CSs could be caused by two effects: reduction in mean differences between groups and increased or decreased within-subjects variation in after-treatment samples leading to misleading results. FEP patients and CSs are samples from two different populations and sampling variation may account for differences in the sample mean or median between groups. Since we are using a within-subjects design the change in variance associated with individual differences may occur. To investigate these possibilities, we performed biomarker level homogeneity of variance tests across all studied conditions. Our results demonstrated that 96.2% (101 out of 105) and 94.3% (99 out of 105) biomarkers had equal variance when FEP patients’ data obtained before and after treatment were compared to CSs data sets (Supplemantary Table S-1, and Table S-3). In addition we identified that homogeneity of within-subject variances was equal for 100 biomarkers out of 105 (95.2%). Our findings verified that estimations of intra-individual and inter-individual variations were fairly similar across biomolecule serum concentrations. Homogeneity of variance was not robustly violated which in turn confirmed our statistically significant results derived from the non-parametric tests to compare two related or different samples’ central tendencies and GLM models.
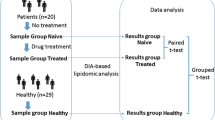
Research of changes of both GPLs and SMs in psychotic disorders deserves strong attention in the future. First, in tissues some of PC membrane functions are shared with structurally related SMs. Second, the synthesis of SMs involves special stage of enzymatic transfer of a phosphocholine from PCs to a ceramide. Third, a recent review proposed evidence for a bidirectional homeostatic crosstalk between SLs and GPLs, whereas the dysregulation of the balance between them results in lipotoxicity-induced metabolic stress associated with pathophysiology of common metabolic diseases [51]. Schematic overview of the main findings of this study can be found in Fig. 1.
This study does have some limitations. In particular, the limited cohort size may create generalization problems. Small sample size in our study arose from the rarity of first episode, antipsychotic-naïve patients. Moreover, fewer patients underwent the follow-up assessments compared with the number of those included in the baseline evaluations. In addition, we collected data from CSs at one point in time and did not control their health condition or metabolite levels after the same follow-up period as was done for the FEP patients group. In this study, patients’ treatment did not have any restrictions in terms of usage of specific pharmacological agents and the pharmacological therapy was altered during the study according to clinically relevant circumstances. However, because of our relative small sample size we could not establish direct effects of any specific active substance on the measured biomolecules. Furthermore, this is a single-centre study of an observational nature and the most important limitation of this kind of research is the possibility of confounding variables, for instance participants’ lifestyle, sleep problems, cannabis and alcohol use. Moreover, there could be bias due to unmeasured confounders that only can be evaluated by a randomized controlled trial. Our results reflect ‘real-world’ situation rather than highly selected trial sample, which might allow for an even more valid conclusion with greater relevance to daily clinical routine. Furthermore, although it has been shown that with consistent handling, frozen serum samples can be stored for later quantitative analysis with minor effects on quantitative lipid composition for most of the biologically relevant lipid species in humans [52], there is no consensus in relation to the maximum storage time and temperature for ensuring the integrity of the samples which contains low-molecular weight compounds. Finally, further studies are needed to help gain insights into the underlying biological processes driving the observed changes in GPLs and SMs in drug-naïve FEP patients and those patients who had used the long-term antipsychotic treatment.
Conclusions
Our results revealed that FEP manifestation is significantly associated with the reduced levels of specific SM (SM-C20:2) and 16 particular PCs as well as elevation of the level of LysoPC-a-C20:4. As these signatures of biomolecules may be dynamic in terms of the course of disease and antipsychotic treatment, we also investigated the change in molecular signature in FEP patient sample after 7-month treatment with antipsychotics. The results showed that FEP onset characteristic molecular pattern in relation to GPLs and SMs was reversed by antipsychotic drug treatment. Our results extend our previous findings that FEP is characterized by peripheral biomarker signature. Further studies in relation to these findings using larger number of participants in clinical trials are required for the development of molecular tools, which could be useful for detecting early stage of the FEP or to monitor antipsychotic treatment in patients with SCH spectrum disorders.
References
Howes O, McCutcheon R, Stone J (2015) Glutamate and dopamine in schizophrenia: an update for the 21st century. J Psychopharmacol (Oxf) 29:97–115. https://doi.org/10.1177/0269881114563634
Upthegrove R, Manzanares-Teson N, Barnes NM (2014) Cytokine function in medication-naive first episode psychosis: a systematic review and meta-analysis. Schizophr Res 155:101–108. https://doi.org/10.1016/j.schres.2014.03.005
Fraguas D, Díaz-Caneja CM, Rodríguez-Quiroga A, Arango C (2017) Oxidative stress and inflammation in early onset first episode psychosis: a systematic review and meta-analysis. Int J Neuropsychopharmacol 20:435–444. https://doi.org/10.1093/ijnp/pyx015
Pillinger T, Beck K, Gobjila C, Donocik JG, Jauhar S, Howes OD (2017) Impaired glucose homeostasis in first-episode schizophrenia: a systematic review and meta-analysis. JAMA Psychiatry 74:261–269. https://doi.org/10.1001/jamapsychiatry.2016.3803
Schneider M, Levant B, Reichel M, Gulbins E, Kornhuber J, Müller CP (2017) Lipids in psychiatric disorders and preventive medicine. Neurosci Biobehav Rev 76:336–362. https://doi.org/10.1016/j.neubiorev.2016.06.002
Horrobin DF, Glen AIM, Vaddadi K (1994) The membrane hypothesis of schizophrenia. Schizophr Res 13:195–207. https://doi.org/10.1016/0920-9964(94)90043-4
Zhang A, Sun H, Wang X (2012) Serum metabolomics as a novel diagnostic approach for disease: a systematic review. Anal Bioanal Chem 404:1239–1245. https://doi.org/10.1007/s00216-012-6117-1
Han X (2016) Lipidomics: comprehensive mass spectrometry of lipids. Wiley, Hoboken
Kriisa K, Leppik L, Balõtšev R, Ottas A, Soomets U, Koido K, Volke V, Innos J, Haring L, Vasar E, Zilmer M (2017) Profiling of acylcarnitines in first episode psychosis before and after antipsychotic treatment. J Proteome Res 16:3558–3566. https://doi.org/10.1021/acs.jproteome.7b00279
Stegemann C, Pechlaner R, Willeit P, Langley SR, Mangino M, Mayr U, Menni C, Moayyeri A, Santer P, Rungger G, Spector TD, Willeit J, Kiechl S, Mayr M (2014) Lipidomics profiling and risk of cardiovascular disease in the prospective population-based bruneck study. Circulation 129:1821–1831. https://doi.org/10.1161/CIRCULATIONAHA.113.002500
Paapstel K, Kals J, Eha J, Tootsi K, Ottas A, Piir A, Jakobson M, Lieberg J, Zilmer M (2018) Inverse relations of serum phosphatidylcholines and lysophosphatidylcholines with vascular damage and heart rate in patients with atherosclerosis. Nutr Metab Cardiovasc Dis 28:44–52. https://doi.org/10.1016/j.numecd.2017.07.011
Sigruener A, Kleber ME, Heimerl S, Liebisch G, Schmitz G, Maerz W (2014) Glycerophospholipid and sphingolipid species and mortality: the ludwigshafen risk and cardiovascular health (LURIC) study. PLoS One. https://doi.org/10.1371/journal.pone.0085724
Kanno K, Wu MK, Agate DS, Fanelli BJ, Wagle N, Scapa EF, Ukomadu C, Cohen DE (2007) Interacting proteins dictate function of the minimal start domain phosphatidylcholine transfer protein/starD2. J Biol Chem 282:30728–30736. https://doi.org/10.1074/jbc.M703745200
Lipid Library website (2009) https://web.archive.org/web/20090531115217/http://www.lipidlibrary.co.uk/Lipids/pc/index.htm. Accessed 27 Apr 2018
Park JY, Lee S-H, Shin M-J, Hwang G-S (2015) Alteration in metabolic signature and lipid metabolism in patients with angina pectoris and myocardial infarction. PLoS One 10:e0135228. https://doi.org/10.1371/journal.pone.0135228
Schmitt A, Wilczek K, Blennow K, Maras A, Jatzko A, Petroianu G, Braus DF, Gattaz WF (2004) Altered thalamic membrane phospholipids in schizophrenia: a postmortem study. Biol Psychiatry 56:41–45. https://doi.org/10.1016/j.biopsych.2004.03.019
Schwarz E, Prabakaran S, Whitfield P, Major H, Leweke FM, Koethe D, McKenna P, Bahn S (2008) High throughput lipidomic profiling of schizophrenia and bipolar disorder brain tissue reveals alterations of free fatty acids, phosphatidylcholines, and ceramides. J Proteome Res 7:4266–4277. https://doi.org/10.1021/pr800188y
He Y, Yu Z, Giegling I, Xie L, Hartmann AM, Prehn C, Adamski J, Kahn R, Li Y, Illig T, Wang-Sattler R, Rujescu D (2012) Schizophrenia shows a unique metabolomics signature in plasma. Transl Psychiatry 2:e149. https://doi.org/10.1038/tp.2012.76
Weber-Fahr W, Englisch S, Esser A, Tunc-Skarka N, Meyer-Lindenberg A, Ende G, Zink M (2013) Altered phospholipid metabolism in schizophrenia: a phosphorus 31 nuclear magnetic resonance spectroscopy study. Psychiatry Res Neuroimaging 214:365–373. https://doi.org/10.1016/j.pscychresns.2013.06.011
Ghosh S, Dyer RA, Beasley CL (2017) Evidence for altered cell membrane lipid composition in postmortem prefrontal white matter in bipolar disorder and schizophrenia. J Psychiatr Res 95:135–142. https://doi.org/10.1016/j.jpsychires.2017.08.009
Aquino A, Alexandrino GL, Guest PC, Augusto F, Gomes AF, Murgu M, Steiner J, Martins-de-Souza D (2018) Blood-based lipidomics approach to evaluate biomarkers associated with response to olanzapine, risperidone, and quetiapine treatment in schizophrenia patients. Front Psychiatry. https://doi.org/10.3389/fpsyt.2018.00209
Orešič M, Tang J, Seppänen-Laakso T, Mattila I, Saarni SE, Saarni SI, Lönnqvist J, Sysi-Aho M, Hyötyläinen T, Perälä J, Suvisaari J (2011) Metabolome in schizophrenia and other psychotic disorders: a general population-based study. Genome Med 3:19. https://doi.org/10.1186/gm233
Yang J, Chen T, Sun L, Zhao Z, Qi X, Zhou K, Cao Y, Wang X, Qiu Y, Su M, Zhao A, Wang P, Yang P, Wu J, Feng G, He L, Jia W, Wan C (2013) Potential metabolite markers of schizophrenia. Mol Psychiatry 18:67–78. https://doi.org/10.1038/mp.2011.131
Fukuzako H, Kodama S (1999) Changes in levels of phosphorus metabolites in temporal lobes of drug-naive schizophrenic patients. Am J Psychiatry 4:1205–1208
Matsumoto J, Sugiura Y, Yuki D, Hayasaka T, Goto-Inoue N, Zaima N, Kunii Y, Wada A, Yang Q, Nishiura K, Akatsu H, Hori A, Hashizume Y, Yamamoto T, Ikemoto K, Setou M, Niwa S (2011) Abnormal phospholipids distribution in the prefrontal cortex from a patient with schizophrenia revealed by matrix-assisted laser desorption/ionization imaging mass spectrometry. Anal Bioanal Chem 400:1933. https://doi.org/10.1007/s00216-011-4909-3
Narayan S, Head SR, Gilmartin TJ, Dean B, Thomas EA (2009) Evidence for disruption of sphingolipid metabolism in schizophrenia. J Neurosci Res 87:278–288. https://doi.org/10.1002/jnr.21822
Rietkerk T, Boks MPM, Sommer IEC, Jong S de, Kahn RS, Ophoff RA (2009) Network analysis of positional candidate genes of schizophrenia highlights myelin-related pathways. Mol Psychiatry 14:353–355. https://doi.org/10.1038/mp.2008.86
Kaddurah-Daouk R, McEvoy J, Baillie R, Zhu H, Yao KJ, Nimgaonkar VL, Buckley PF, Keshavan MS, Georgiades A, Nasrallah HA (2012) Impaired plasmalogens in patients with schizophrenia. Psychiatry Res 198:347–352. https://doi.org/10.1016/j.psychres.2012.02.019
McEvoy J, Baillie RA, Zhu H, Buckley P, Keshavan MS, Nasrallah HA, Dougherty GG, Yao JK, Kaddurah-Daouk R (2013) Lipidomics reveals early metabolic changes in subjects with schizophrenia: effects of atypical antipsychotics. PLoS One 8:e68717. https://doi.org/10.1371/journal.pone.0068717
Suvitaival T, Mantere O, Kieseppä T, Mattila I, Pöhö P, Hyötyläinen T, Suvisaari J, Orešič M (2016) Serum metabolite profile associates with the development of metabolic co-morbidities in first-episode psychosis. Transl Psychiatry 6:e951. https://doi.org/10.1038/tp.2016.222
O’Gorman A, Suvitaival T, Ahonen L, Cannon M, Zammit S, Lewis G, Roche HM, Mattila I, Hyotylainen T, Oresic M, Brennan L, Cotter DR (2017) Identification of a plasma signature of psychotic disorder in children and adolescents from the Avon Longitudinal Study of Parents and Children (ALSPAC) cohort. Transl Psychiatry 7:e1240. https://doi.org/10.1038/tp.2017.211
Philippova M, Resink T, Erne P, Bochkov V (2014) Oxidised phospholipids as biomarkers in human disease. Swiss Med Wkly. https://doi.org/10.4414/smw.2014.14037
World Health Organization (1992) The ICD-10 classification of mental and behavioural disorders: clinical descriptions and diagnostic guidelines. World Health Organization, Geneva
Gardner DM, Murphy AL, O’Donnell H, Centorrino F, Baldessarini RJ (2010) International consensus study of antipsychotic dosing. Am J Psychiatry 167:686–693. https://doi.org/10.1176/appi.ajp.2009.09060802
Haring L, Koido K, Vasar V, Leping V, Zilmer K, Zilmer M, Vasar E (2015) Antipsychotic treatment reduces psychotic symptoms and markers of low-grade inflammation in first episode psychosis patients, but increases their body mass index. Schizophr Res 169:22–29. https://doi.org/10.1016/j.schres.2015.08.027
Balõtšev R, Haring L, Koido K, Leping V, Kriisa K, Zilmer M, Vasar V, Piir A, Lang A, Vasar E (2017) Antipsychotic treatment is associated with inflammatory and metabolic biomarkers alterations among first-episode psychosis patients: a 7-month follow-up study. Early Interv Psychiatry. https://doi.org/10.1111/eip.12457
Leppik L, Kriisa K, Koido K, Koch K, Kajalaid K, Haring L, Vasar E, Zilmer M (2018) Profiling of amino acids and their derivatives biogenic amines before and after antipsychotic treatment in first-episode psychosis. Front Psychiatry. https://doi.org/10.3389/fpsyt.2018.00155
Overall JE, Gorham DR (1962) The brief psychiatric rating scale. Psychol Rep 10:799–812. https://doi.org/10.2466/pr0.1962.10.3.799
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. L. Erlbaum Associates, Hillsdale
Nordstokke DW, Zumbo BD (2010) A new nonparametric levene test for equal variances. Psicol Int J Methodol Exp Psychol 31:401–430
TIBCO Software Inc (2017) Statistica (data analysis software system), version 13. TIBCO Software Inc., Palo Alto
Tonks KT, Coster AC, Christopher MJ, Chaudhuri R, Xu A, Gagnon-Bartsch J, Chisholm DJ, James DE, Meikle PJ, Greenfield JR, Samocha-Bonet D (2016) Skeletal muscle and plasma lipidomic signatures of insulin resistance and overweight/obesity in humans. Obesity 24:908–916. https://doi.org/10.1002/oby.21448
Chiurchiù V, Leuti A, Maccarrone M (2018) Bioactive lipids and chronic inflammation: managing the fire within. Front Immunol. https://doi.org/10.3389/fimmu.2018.00038
Sethi S, Hayashi MA, Sussulini A, Tasic L, Brietzke E (2017) Analytical approaches for lipidomics and its potential applications in neuropsychiatric disorders. World J Biol Psychiatry 18:506–520. https://doi.org/10.3109/15622975.2015.1117656
Kaddurah-Daouk R, Krishnan KRR (2009) Metabolomics: a global biochemical approach to the study of central nervous system diseases. Neuropsychopharmacology 34:173–186. https://doi.org/10.1038/npp.2008.174
Misiak B, Stańczykiewicz B, Łaczmański Ł, Frydecka D (2017) Lipid profile disturbances in antipsychotic-naive patients with first-episode non-affective psychosis: a systematic review and meta-analysis. Schizophr Res 190:18–27. https://doi.org/10.1016/j.schres.2017.03.031
Carland JE, Mansfield RE, Ryan RM, Vandenberg RJ (2013) Oleoyl-l-carnitine inhibits glycine transport by GlyT2. Br J Pharmacol 168:891–902. https://doi.org/10.1111/j.1476-5381.2012.02213.x
Koren D, Palladino A (2016) Chap. 3—hypoglycemia. In: Weiss RE, Refetoff S (eds) Genetic diagnosis of endocrine disorders, 2nd edn. Academic Press, San Diego, pp 31–75
Hanamatsu H, Ohnishi S, Sakai S, Yuyama K, Mitsutake S, Takeda H, Hashino S, Igarashi Y (2014) Altered levels of serum sphingomyelin and ceramide containing distinct acyl chains in young obese adults. Nutr Diabetes 4:e141. https://doi.org/10.1038/nutd.2014.38
van der Veen JN, Kennelly JP, Wan S, Vance JE, Vance DE, Jacobs RL (2017) The critical role of phosphatidylcholine and phosphatidylethanolamine metabolism in health and disease. Biochim Biophys Acta BBA Biomembr 1859:1558–1572. https://doi.org/10.1016/j.bbamem.2017.04.006
Rodriguez-Cuenca S, Pellegrinelli V, Campbell M, Oresic M, Vidal-Puig A (2017) Sphingolipids and glycerophospholipids—the “ying and yang” of lipotoxicity in metabolic diseases. Prog Lipid Res 66:14–29. https://doi.org/10.1016/j.plipres.2017.01.002
Zivkovic AM, Wiest MM, Nguyen UT, Davis R, Watkins SM, German JB (2009) Effects of sample handling and storage on quantitative lipid analysis in human serum. Metabolomics 5:507. https://doi.org/10.1007/s11306-009-0174-2
Acknowledgements
This research was supported by the European Union through the European Regional Development Fund (Project no. 2014-2020.4.01.15-0012), and grants from the Estonian Research Foundation (IUT 20-41, IUT 20-42). The funding sources do not have any role in the writing of the manuscript or the decision to submit it for the publication.
Author information
Authors and Affiliations
Contributions
LH, EV and MZ designed the study. LH, LL, MP and SJ contributed to data collection and carried out the literature search. MZ and KK were responsible for metabolomics measurements. LH, EV and MZ co-wrote the first draft of the report. LL, MP, SJ, KK, LH, EV, MZ critically revised the manuscript. EV and MZ obtained the research funding. All authors reviewed the manuscript for important intellectual content and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
The protocol of this study was approved by the Ethics Review Committee on Human Research of the University of Tartu (Estonia). All subjects gave written informed consent in accordance with the Declaration of Helsinki.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Leppik, L., Parksepp, M., Janno, S. et al. Profiling of lipidomics before and after antipsychotic treatment in first-episode psychosis. Eur Arch Psychiatry Clin Neurosci 270, 59–70 (2020). https://doi.org/10.1007/s00406-018-0971-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-018-0971-6