Abstract
Here, we review the cerebrospinal fluid (CSF) candidate markers with regard to their clinical relevance as potential surrogates for disease activity, prognosis assessment, and predictors of treatment response. We searched different online databases such as MEDLINE and EMBASE for studies on schizophrenia and CSF. Initial studies on cerebrospinal fluid in patients with schizophrenia revealed increased brain–blood barrier permeability with elevated total protein content, increased CSF-to-serum ratio for albumin, and intrathecal production of immunoglobulins in subgroups of patients. Analyses of metabolites in CSF suggest alterations within glutamatergic neurotransmission as well as monoamine and cannabinoid metabolism. Decreased levels of brain-derived neurotrophic factor and nerve growth factor in CSF of first-episode patients with schizophrenia reported in recent studies point to a dysregulation of neuroprotective and neurodevelopmental processes. Still, these findings must be considered as non-specific. A more profound characterization of the particular psychopathological profiles, the investigation of patients in the prodromal phase or within the first episode of schizophrenia promoting longitudinal investigations, implementation of different approaches of proteomics, and rigorous adherence to standard procedures based on international CSF guidelines are necessary to improve the quality of CSF studies in schizophrenia, paving the way for identification of syndrome-specific biomarker candidates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Schizophrenia is the most severe and impairing mental disorder. The life-time-risk amounts to 0.5–1% [51] and worldwide incidence rates of 15 per 100,000 have been reported [85]. Schizophrenia is characterized by disorganization of thought and behavior, along with hallucinations of different modalities, paranoid and delusional beliefs, and mood disturbance, resulting in impairment of core functions of the self. Due to the frequency of severe impairment and the chronic course of the disease, schizophrenia has an enormous impact not only on the health and lifestyle of patients and their relatives but also on the global economic burden of disease [151]. With regard to clinical phenotype, the subtypes of schizophrenia appear markedly heterogeneous, and this might also be true for their etiologies and pathomechanisms. The diagnosis of schizophrenia, however, still fully relies on clinical criteria; there are no sub-type specific and not even disease-specific neuroimaging or biochemical markers. Cerebrospinal fluid (CSF) obviously reflects biochemical changes within the central nervous system (CNS) more directly than does serum [147] and, therefore, CSF might be a useful source of potential biochemical markers of schizophrenia. Several recent studies investigated such biomarker candidates of schizophrenia in CSF [47, 129–131], unhappily without identifying promising one. The present article reviews the current knowledge on CSF parameters that might be related to aspects of the pathophysiology of schizophrenia.
Basic principles of CSF analysis
CSF is a clear, colorless fluid, which occupies the space between arachnoid mater and pia mater, the subarachnoid space, the ventricles, and the central canal of brain stem and spinal cord. The CSF total volume remains essentially constant at 100–150 ml. However, CSF has a high turnover and is continuously being produced at a rate of about 500 ml per day, predominantly within the choroid plexus, and also continuously being drained, predominantly via arachnoid granulations into the bloodstream. CSF is usually obtained by lumbar puncture, which is generally well tolerated if performed by an experienced clinician using a small-gauge non-traumatic needle [115].The CSF proteins mainly derive from the blood. The Blood-CSF barrier limits the passage of molecules into CSF [108]. Accordingly, the protein concentration in CSF is lower by two to three orders of magnitude compared to serum [133]. The protein content approximates 0.3% of plasma protein content (15–40 mg/dl) while the glucose concentration amounts to 50–70% of plasma glucose concentrations (50–80 mg/dl) [15–17]. Since albumin is produced only in the liver, the ratio of CSF albumin to serum albumin (Qalb) can be used as a measure of Blood-CSF barrier permeability. Standardized age-corrected reference ranges exist for this purpose [112]. Normal CSF contains no cells, although up to 4 leukocytes per microliter are considered non-specific.
CSF basic findings in schizophrenia
With regard to psychiatric disorders, the role of CSF analyses so far is confined to the exclusion of neurological causes [70, 108]. Several studies in patients with schizophrenia reported alterations of basic CSF parameters like total protein content, Qalb, total glucose content, lactate concentration, immunoglobulins, and cellular elements (e.g., [11]).
Total cell count
While the total CSF cell count showed no significant differences between patients with schizophrenia and controls, in several studies by Nikkila and co-workers, acutely psychotic patients were observed to show an increased proportion of activated lymphoid cells as well as elevated proportions of monocytes and macrophages, indicating an activation of microglial cells [98, 99]. With antipsychotic treatment, the number of macrophages was reported to decline.
Glucose and lactate
In drug-naïve patients with first-onset schizophrenia, increased CSF glucose levels and decreased lactate and acetate concentrations were reported, which returned to normal in over half of the patients after short-term treatment with antipsychotic medication [44].
Blood-CSF barrier
Several studies reported increased Blood-CSF barrier permeability in fractions of about 30% of patients with schizophrenia [9, 63]. Axelsson et al. [7] reported increased Qalb in 7 out of 25 patients with schizophrenia as a possible indicator of Blood-CSF barrier impairment; in the subgroup with elevated Qalb, the onset of psychosis occurred 20 years earlier than in patients with normal Qalb, suggesting that Blood-CSF barrier impairment might contribute to the development of psychosis at least in some individuals. Generally, elevation of Qalb is a non-specific finding [20], which could also be attributed to a reduced rate of exchange of CSF [102, 103] and is encountered in a wide range of disorders [20]. In psychiatric diseases, the Blood-CSF barrier dysfunction appears to be more pronounced in schizophrenia than in affective disorders [108]. Still, the mechanisms underlying Blood-CSF barrier dysfunction in a subgroup of patients with schizophrenia remain unclear. Schwarz et al. [132] reported Blood-CSF barrier dysfunction in schizophrenia to be associated with high levels of the soluble intercellular adhesion molecule-1 (sICAM-1), suggesting inflammatory mechanisms. A possible influence of antipsychotic therapy was investigated by Ben-Shachar and colleagues: Administration of haloperidol or chlorpromazine in rats lead to an elevated iron uptake into the brain, indicating a possible affection of Blood-CSF barrier function by neuroleptic treatment [12]. Treatment with antipsychotic or antidepressant drugs might also indirectly contribute to elevated Qalb via an increase in total body weight since increased epidural fat storage and increased central venous pressure [140] both tend to reduce the pressure gradient between CSF and venous compartment, contributing to a reduced CSF exchange rate and an increased CSF protein concentration [20].
Immunoglobulins
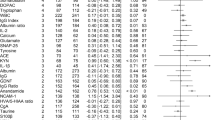
Immunoglobulin G synthesis within CSF (oligoclonal bands) indicates an intrathecal immune reaction [128]. Increased immunoglobulin G concentrations within the CSF can also be caused by an impaired Blood-CSF barrier that may be a consequence of traumatic injury, inflammation, or degenerative spine disease. The IgG-index accounts for CSF–blood barrier permeability by relating the intrathecal IgG production to the CSF/serum albumin ratio (IgG-Index = [IgG(Liquor)/IgG(Serum)]/[Albumin(Liquor)/Albumin(Serum)]; [17]). Increased immunoglobulin G levels in schizophrenia have been described by Toorey and colleagues [145], but this finding could not be reproduced ([1, 19]; Table 1).
Considering the relatively long period in which the majority of studies have been conducted, the different and partly out-dated methodological approaches, as well as the diversity of reported results, basic CSF findings reported in some patients with schizophrenia most likely represent non-specific changes.
Metabolic marker candidates
Glutamatergic metabolites
Decreased levels of gamma-glutamylglutamine and taurine as well as increased levels of isoleucine were found in CSF of 26 drug-free patients with schizophrenia using high-pressure liquid chromatography and gas chromatography/mass spectrometry [31]. A possible hypothesis of dysregulation of glutamatergic neurotransmission would also be supported by studies analyzing d-serine. d-serine is an endogenous agonist of the glycine site on the N-methyl-D-aspartate (NMDA) glutamate receptor (see Fig. 1). Decreased levels of d-serine and reduced d-serine-to-total-serine ratio but elevated levels of total and L-serine were found in serum of 42 patients with schizophrenia [40]. At the same time, d-serine level and d-/l-serine ratio in plasma have been shown to increase with clinical improvement [100]. Reduced d-serine-to-total-serine ratio was also found in CSF of male, first-episode, drug-naïve patients with schizophrenia [39], possibly due to increased levels of D-amino acid oxidase (DAAO; it degrades d-serine) and decreased serine racemase levels (converts l-serine to d-serine), which have been demonstrated in a post-mortem study of brain tissue [13]. However, the expression of DAAO and serine racemase as well as corresponding mRNA levels seem to vary considerably depending on the brain region investigated [153] and administration of haloperidol did not affect serine racemase or DAAO levels in rats [153]. One study reported unaltered levels and relative ratios of d-serine in CSF before and after 6 weeks of treatment with olanzapine 10 mg/die [37]; Table 2.
Molecular mechanisms being involved in the pathogenesis of schizophrenia, as indicated by the CSF findings. Endocannabinoids leave the post-synaptic cell and activate presynaptic CB1 receptors, which than inhibit Ca2+ influx and thereby decrease the probability of release of neurotransmitter vesicles [155]. Thus, the reported increased CSF anandamide in schizophrenia may contribute to the reduction of—in some regions possibly abnormal increased—neurotransmitter secretion, and therefore, might represent a protective factor, which was found to be negatively correlated with severity of psychosis [38]. d-serine binds to the glycine/d-serine-binding site on the N-methyl-D-aspartate (NMDA) glutamate receptor. In conjunction with the binding of glutamate, this leads to opening of the ligand-gated Ca2+-receptor-channel [93]. Decreased levels of d-serine in schizophrenia might support the notion of the altered—presumably reduced—functioning of the NMDA receptor, which is modulated by both glutamate from the synaptic cleft and the d-serine stemming from astrocytes. As a part of the Trans-SNARE complexes (together with synaptobrevin and syntaxin), SNAP-25 is involved in the Ca2+-triggered exocytosis of neurotransmitters [25]. The reported increased CSF levels of SNAP-25 in schizophrenia would support the notion of the dysfunctional synaptic secretion of neurotransmitters. The binding of the brain-derived neurotrophic factor (BDNF) to the post-synaptic TrkB receptors causes depolarization of the post-synaptic neuron. This process is mediated through opening of Na+ channels and concomitant activation of Ca2+ channels [80]. The activation of TrkB receptors enhances also NMDA receptor opening, resulting in an overall increase of the intracellular Ca2+ concentration. In astroglia, the BDNF binding to the TrkB-T1 receptors activates the release of Ca2+ from the intracellular stores, thereby increasing intracellular Ca2+ concentration [113, 116]. By its binding to the TrkA receptors, the nerve growth factor (NGF) initiates an intracellular signal cascade [160], thereby enhancing and coordinating the neurogenesis and neural survival. Decreased CSF levels of both BDNF and NGF indicate alterations in the process of neurogenesis in schizophrenia. Increased levels of S100b can be attributed to the increased activation of astrocytes, possibly as a sign of a genuine glia cells dysfunction or their response within an inflammatory reaction. Glial fibrillary acidic protein (GFAP) was not found to be altered in schizophrenia, suggesting that the increase in S100B might indeed be caused by an active secretion of S100b from astrocytes rather than by a destruction of astrocytes, oligodendrocytes or neurons [119]. Furthermore, no alterations in the levels of tau protein and neuron-specific enolase (NSE) could be found, indicating that no major neuro-axonal damage occurs
Fatty acids
Polyunsaturated fatty acids (PUFAs), particularly docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA), are linked with several aspects of neural function, including neurotransmission, membrane fluidity, ion-channel and enzyme regulation, and gene expression [159]. Decreased blood levels of omega-3 fatty acids have been associated with a number of neuropsychiatric disorders including schizophrenia, and some supplementation studies suggest benefits in terms of decreasing disease risk and reducing symptoms, particularly in patients with a short illness history [4]. Still, results remain inconclusive [58] and no investigations of PUFAs or their metabolites in CSF have been conducted.
Monoamine metabolites
The concentrations of dopamine, dihydroxyphenylacetic acid (DOPAC), norepinephrine, 5-hydroxytryptamine (5-HT), and 5-hydroxyindolacetic acid (5-HIAA) did not differ in first-onset drug-naive schizophrenia patients when compared to controls [86]. Treatment with haloperidol, 20 mg/die [59], or olanzapine, 10 mg/die [124], was reported to elevate the homovanillic acid (HVA) concentration in the CSF and significantly increase the ratio between HVA and 5-HIAA, a serotonin metabolite, in adult patients with schizophrenia although a correlation with clinical effectiveness could not be definitely established. In contrast, a similar regime (haloperidol and clozapine) did not significantly change CSF monoamine concentrations or the ratio between HVA and 5-HIAA in a group of drug-free patients with childhood-onset schizophrenia [52]. Furthermore, treatment with clozapine had similar effects both in childhood-onset and later-onset schizophrenia.
Cannabinoid metabolites
The endogenous cannabinoid anandamide is a bioactive lipid that binds to cannabinoid receptors. Anandamide concentrations were shown to be increased both in prodromal states [64] and during acute episodes [38, 75] of schizophrenia; Table 2. CSF anandamide appeared to be negatively correlated with severity of psychosis [38], suggesting that anandamide elevation might reflect a compensatory adaptation to the disease state. Patients with schizophrenia who consumed cannabis had lower CSF concentrations of anandamide when compared to both more frequently consuming healthy subjects, or less frequently consuming schizophrenic patients, indicating that intensive cannabis abuse might down-regulate anandamide signaling in patients with schizophrenia and therefore increase the risk of disease manifestation [74]. The neuroimaging data suggest that first-episode schizophrenia patients who use cannabis show a more pronounced cortical thinning than non-using patients, particularly in brain regions known for their high density of CB1 receptors (see Fig. 1), such as the anterior cingulate cortex and the dorsolateral prefrontal cortex [109]. Thus, it seems advisable to discriminate between the endogenous cannabinoid system, which indeed seems to be altered in schizophrenia (as indicated by increased CSF anandamide or increased density of CB1 receptor binding in some brain areas; though possibly having also protective role to some extent, see above), and the exogenous cannabis consumption, which is a risk factor enhancing the manifestation and progression of disorders from the schizophrenia spectrum, possibly by negatively affecting the endogenous cannabinoid system [36].
Glycogen synthase kinase 3 (GSK-3)
Whereas GSK-3 proteins were originally identified as key regulatory enzymes in glucose metabolism, nowadays it is well-known that GSK-3s (two isoforms, GSK-3alpha and GSK-3beta) mediate various signaling pathways, in particular the growth factor and Wnt signaling pathways. GSK-3s has a number of transcription factors as substrates, thereby being involved in regulation of multiple neurodevelopmental processes including neurogenesis, neuronal migration, neuronal polarization, and axon growth and guidance [49]. The investigation of GSK-3alpha and GSK-3beta mRNA levels, GSK-3beta protein levels, or total GSK-3 (alpha + beta) enzyme activity in lymphocytes of patients with schizophrenia revealed no differences compared to controls [95]. Therefore, determination of GSK-3 in CSF could provide additional information regarding the pathogenetic role and possible therapeutic consequences in schizophrenia [65]. Both GSK-3beta levels [67] and GSK-3 activity [66] were reported to be lowered in post-mortem frontal cortex of patients with schizophrenia, possibly suggesting that low GSK-3 in post-mortem brain of schizophrenic patients might be a late consequence of perinatal neurodevelopmental insult in schizophrenia [68]. Still, post-mortem studies do not consistently show GSK-3 alterations in patients with schizophrenia [94]. Only one study investigated GSK-3beta in CSF in alive patients with schizophrenia, using Western-blot analysis and showing decreased levels ([69]; Table 2). However, only 6 patients were studied, the patients had a long duration of illness, in average over 30 years, and the difference in GSK-3beta protein levels was only marginally significant (P = 0.048), so that further studies are necessary.
Inflammatory marker candidates
Interleukins
Several studies observed neuroimmunological abnormalities in patients with schizophrenia, including alterations of various cytokines with a wide range of immunomodulatory, neurodevelopmental, and neuroregulatory functions [71]. Particularly, proinflammatory cytokines seem to be altered in some cases of schizophrenia ([71]; Table 3). Increased CSF levels of both interleukin-6 (IL-6) [150] and interleukin-2 (IL-2) [84] have been reported. In one study [84], patients with a later relapse, examined both while treated (haloperidol) and after drug withdrawal (up to 6 weeks), had higher levels of CSF IL-2 than patients who did not relapse. Therefore, increased CSF IL-2 levels were proposed to predict disease progress. No similar association could be found for blood levels, suggesting independent regulation of blood and CSF cytokine levels. Elevated levels of CSF IL-2, but not interleukin-1 alpha (IL-1α), were also found in antipsychotic-free patients with schizophrenia [77]. Other studies reported no alteration of CSF IL-1, IL-2, or IL-6 levels [8, 34, 61, 110].
Antiphospholipid antibodies
One study demonstrated different patterns of antiphospholipid antibodies in CSF of patients with schizophrenia compared to serum indicating possible intrathecal synthesis [136].
Infectious agents
Cytomegalovirus, toxoplasma gondii, HSV 1, HSV 2, Epstein Barr virus
Various infectious agents have been discussed as candidate pathogenic agents in schizophrenia, including toxoplasma gondii, cytomegalovirus [3, 96, 158], as well as herpes simplex virus (HSV) type 1, 2, and 6 [97, 157]. Elevated levels of serum immunoglobulin G antibodies against these microbial agents were observed both in individuals at high risk for psychosis and in patients with schizophrenia and were suggested to be linked to the severity of psychotic symptoms. Leweke and colleagues reported increased levels of CSF IgG antibodies to both cytomegalovirus and toxoplasma gondii in untreated individuals with recent-onset schizophrenia whereas levels of immunoglobulin M antibodies were not altered [73]; Table 3). There was no difference in the levels of antibodies against HSV 1, HSV 2, and Epstein Barr virus. Interestingly, patients who received neuroleptic treatment had lower levels of antibodies to cytomegalovirus and toxoplasma gondii, suggesting that treatment with neuroleptics could influence B-cell immunology associated with immunoglobulin synthesis in the CNS. So far, conclusive data are missing, and the link between immunology and schizophrenia remains to be elucidated.
Neurotrophic factors
The neurotrophins (nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), neurotrophin 3, neurotrophin 4, and neurotrophin 5) are proteins with high affinity to receptors of the tyrosine kinase family (TrkA, B, C). By regulating synaptic activity and neurotransmitter synthesis (see Fig. 1), they stimulate and control cellular proliferation, migration, differentiation, and the survival of the neurons not only during the embryo- and organogenesis, but also during the nerve regeneration, enabling, and supporting neural plasticity [32]. Alterations in the expression of the neurotrophins were suggested to contribute to the pathology of schizophrenia, indicating the deficiency of endogenous neuroprotective mechanisms.
Brain-derived neurotrophic factor (BDNF)
Decreased serum BDNF has repeatedly been observed in drug-naïve first-episode schizophrenic patients [23, 26, 114]. Jindal and colleagues showed that patients with schizophrenia had lower serum BDNF levels than patients with non-schizophrenia psychosis that had lower BDNF levels than healthy controls [57]. In a post-mortem brain tissue of patients with schizophrenia, increased BDNF levels and decreased levels of neurotrophin 3 in cortical areas as well as decreased levels of BDNF in hippocampus were found [32]. In another post-mortem study, decreased BDNF levels in prefrontal cortex and in the CSF were found. Also, a significant negative correlation between BDNF and cortisol was observed both in prefrontal cortex tissue and in CSF. Within the same study, these findings could be replicated in an animal model of schizophrenia [50]; Table 4. In one study, serum and CSF BDNF levels were correlated with each other, both showing a negative correlation with the scores of baseline PANSS positive symptom subscalesclinical scores of positive symptoms [106].
Nerve growth factor (NGF)
Kale et al. [60] reported decreased levels of NGF in both CSF and serum in drug-naïve first-episode patients with schizophrenia; Table 4). CSF studies on further neurotrophic factors such as neurotrophin-3, neurotrophin-4, and novel neurotrophin-1 are lacking.
Neuronal marker candidates
Patients with schizophrenia exhibit minor abnormalities on cerebral imaging including enlarged third or lateral ventricles or reduced volume of temporal lobe structures [107], which could be related to causes or consequences of the disease. The current longitudinal investigations suggest that particularly frontal and temporal cortical areas show progressive thinning across the course of the illness [149], although these changes might rather be attributed to the reduction of the neuropil, and not mainly to the degeneration of the neurons (see below the chapter about the glial marker candidates). Also different expression levels of myelin basic protein and myelin oligodendrocyte protein in patients with schizophrenia were found in both CSF and post-mortem mediodorsal thalamus, suggesting alterations within oligodendrocyte and cytoskeleton assembly [82].To elucidate the role of neurodegeneration, several studies investigated particularly various CSF markers of neuronal damage in schizophrenia.
Tau protein
Tau protein is a microtubule-associated phospho-protein that is primarily localized in axons promoting microtubule assembly and stability. After neuronal damage, tau is released into extracellular space from where it diffuses into the CSF [152]. Elevation of CSF tau has been reported in a wide range of neurodegenerative diseases, with highest levels observed in diseases with a rapid neuro-axonal degeneration like Creutzfeldt-Jakob disease (CJD) [101]. In patients with schizophrenia, CSF tau levels are usually normal [126], providing evidence against major neuro-axonal degeneration in schizophrenia; Table 4. On a clinical level, normal CSF tau in elderly patients with cognitive deficits and possible schizophrenia may help to delineate Alzheimer’s disease (AD), which is often accompanied by an early elevation of CSF tau protein [18].
Neuron-specific enolase (NSE)
Enolase is a glycolytic enzyme, consisting of three subunits (α, β and γ), which converts 2-phospho-glycerate to phosphoenolpyruvate [21]. In the CNS, the αγ and γγ isoforms are mainly localized within neurons and are therefore called neuron-specific enolase (NSE). Like tau protein, NSE was observed to be elevated in conditions of neuronal injury [156] with highest CSF levels observed in conditions of rapid neuro-axonal degeneration [10]. In line with observations on tau protein, several studies reported normal CSF NSE in schizophrenia [33, 127, 139]; Table 4. Elevated NSE levels were reported by a Chinese study, which however, included a disproportionate low number of controls (n = 9) [76]. The present CSF findings regarding tau protein and NSE do not support the notion of major neuro-axonal damage in schizophrenia, although a slow-going, persistent degenerative process, concerning both neuropil and neural tissue, as suggested by neuroimaging findings, cannot be ruled out.
Glial marker candidates
S100b
S100B, a calcium-binding astrocyte-specific cytokine, presents a marker of astrocytic activation [119], Fig. 1. It is an acidic calcium-binding protein with a molecular weight of 21 kD [161]. S100b exerts various autocrine and paracrine effects on glia, neurons, and microglia. It was suggested to play a role in the regulation of cerebral energy metabolism as it stimulates the enzymatic activity of fructose-1,6-bisphosphate aldolase [162]. S100b influences the integrity of the cytoskeleton by inhibiting the assembly of microtubules and type III intermediary filaments [14]. Whereas in nanomolar levels, it acts as a neurotrophic factor and was suggested to promote neuronal survival in brain development and following neuronal injury, overproduction to micromolar levels was shown to exacerbate neuroinflammation and neuronal dysfunction [148]. Increased serum or CSF concentrations of S100b have been reported in a wide range of neurological diseases associated with neuronal damage and glial activation, including cerebral ischemia, subarachnoid hemorrhage, multiple sclerosis, AD, or traumatic brain injury [24, 35, 53, 89]. In patients with schizophrenia, several studies consistently observed S100b to be elevated in CSF ([122, 139]; Table 4) or blood [72, 78, 121, 123, 125, 154]. So far, it is unclear whether increased levels of S100B in CSF and serum play a specific pathophysiological role in schizophrenia. An association of schizophrenia with certain S100B haplotypes has been observed, which may lead to a tendency for increased S100B expression [79]. Findings from neuropathological and imaging studies reveal a reduction of neuropil in schizophrenia that may have a progressive component [135]. Neuronal cell sizes as well as dendrite and synapse numbers are decreased while the total number of neurons appears to remain unchanged [134]. Astrocytes influence dendrites and synapses via glutamate-induced modulation [5]. A dysfunction of astrocytes could be linked to the above-mentioned alterations of neuropil and might therefore represent an independent pathogenetic factor in the development of schizophrenia [120]. On the other hand, increased astrocyte activation, as indicated by increased CSF S100b concentrations, might be an unsuccessful attempt of the brain to fight an unknown pathogenic mechanism, such as inflammation of unidentified origin (as suggested by Rothermundt and colleagues, [119]). So far, no correlation of CSF S100b with clinical parameters like duration of disease or clinical scores was observed [120, 139].
Glial fibrillary acidic protein (GFAP)
GFAP is the major structural component of the intermediate filament of fibrillary astrocytes [54]. High CSF levels of GFAP were observed in conditions of acute CNS injury with disintegration of astroglial cells [6, 41]. Chronic brain disorders with (reactive) gliosis such as AD, vascular dementia, or multiple sclerosis also entailed an increase of GFAP levels [28, 88]. Consequently, GFAP has been proposed to be both a marker of CNS tissue disintegration and astrogliosis [28, 105, 117]. In a study of patients with schizophrenia, Steiner et al. [139] observed CSF S100b to be elevated, whereas no significant alteration of GFAP was found, indicating that GFAP may be less sensitive than S100b to detect an alteration of astrocyte function in schizophrenia.
Proteomic analyses in the identification of possible CSF biomarkers
VGF23-62, transthyretin, apolipoprotein A-IV
Jiang and colleagues studied the CSF proteome in patients with schizophrenia by two-dimensional gel electrophoresis and matrix-assisted laser desorption/ionization mass spectrometry (MALDI-MS). They report altered levels of CSF apolipoprotein A-IV in patients with schizophrenia, as well as altered levels of haptoglobin, fibrinogen, and complement component 3 [56]. Using similar methodology, the upregulation of apolipoprotein E, apolipoprotein A1, and prostaglandin-H2 D-isomerase has been reported, supporting the hypothesis of disturbed cholesterol and phospholipid metabolism in schizophrenia [83]. Using surface-enhanced laser desorption/ionization (SELDI), Huang and colleagues observed an increase of the 40-amino acid VGF23–62 peptide and a decrease of transthyretin in CSF of first-onset, drug-naïve patients with schizophrenia. Corresponding results were found post-mortem in brain samples [45]; Table 5). No differences regarding the schizophrenia biomarker panel were found when compared with patients with obsessive-compulsive disorder or Alzheimer’s disease. Although the function of the VGF23–62 sequence is still unknown, there is data to suggest that the full-length neuroprotein VGF is involved in the control of food intake, body weight and reproduction [55], and also in mediating antidepressant responses. The transcription of VGF is regulated by both brain-derived neurotrophic factor (BDNF) and serotonin. VGF can be upregulated also by antidepressant drugs and voluntary exercise and is reduced in animal models of depression [142]. Furthermore, VGF enhances hippocampal synaptic plasticity as well as neurogenesis in the dentate gyrus [2]. Regarding CSF transthyretin, over 90% is secreted from the choroid plexus, and the rest derives from blood [111]. Since transthyretin is responsible for thyroxine transport [137], decreased transthyretin concentrations may reduce the transport of thyroxine in the brain, possibly abetting the development of psychotic symptoms. A proteomic and metabolic profile with increased glucose and VGF23-62 peptide and decreased lactate and transthyretin, similar to the one described above for first-onset schizophrenia, was also reported for patients in the initial prodromal state of psychosis, suggesting more or less continuous alterations in the CSF metabolome and proteome along a gradient “healthy state—prodromal symptoms of the psychosis—first manifestation of the schizophrenia” [46]; Table 5. In this study, the presence of biochemical alterations in the individuals with prodromal symptoms did not correlate with the risk of developing schizophrenia. Still, this method can be applied to isolate molecular biomarkers in CSF that might support diagnosis at an early stage and characterize mechanisms of disease progression by tracking alterations of parameters along the course of the disease.
Synaptosome-associated protein of 25 kDa (SNAP-25)
SNAP-25 is expressed in particular subsets of neurons being located on the cytoplasmic face of the plasma membrane in synaptic terminals and throughout the axon. It forms a stable ternary “Trans-SNARE” complex with two other exocytotic proteins, syntaxin and the synaptic vesicle protein synaptobrevin, which are essential for Ca2+-regulated exocytosis, therefore regulating presynaptic vesicle trafficking and synaptic secretion and being directly involved in the release of neurotransmitters [42]. Based on the studies of different SNAP-25 polymorphisms in patients with schizophrenia, the integrity of SNAP-25 was shown to be associated with clinical response [91], weight gain during antipsychotic treatment [91, 92], and executive functioning [138]. SNAP-25 was reported to be elevated in CSF of patients with schizophrenia ([143, 144]; Table 5) suggesting possible dysfunction of the synaptic secretion due to altered release of dopamine, serotonin, and glutamate [27]. These reported changes in the plasma membrane may also be in line with the above-mentioned assumption of the phospholipid dysfunction in schizophrenia, as reflected by alterations in the expression level of apolipoprotein A-IV, apolipoprotein E, and apolipoprotein A1.
Summary and conclusion
Initial studies on CSF in patients with schizophrenia revealed impaired brain–blood permeability with elevated total protein content and increased CSF/serum ratio for albumin. In some patients, intrathecal production of immunoglobulins was observed. In search of more specific biomarkers, metabolic, inflammatory, glial, neuronal, and neurotrophic mechanisms associated with schizophrenia were investigated [47, 104, 130]. Analyses of metabolites in CSF suggest alterations within glutamatergic neurotransmission as well as monoamine and cannabinoid metabolism. Increased cytokine levels in subgroups of patients with schizophrenia indicate a possible pathogenetic role of inflammatory, neuroimmunologic, and neurodevelopmental processes. Decreased levels of BDNF and NGF reported in recent CSF studies suggest a dysregulation within neuroprotective and neurodevelopmental pathways in first-episode patients with schizophrenia. So far, no changes of neuronal markers such as tau protein and neuron-specific enolase are reported, making major neuro-axonal damage in schizophrenia unlikely. CSF “metabolome” and “proteome” analysis in recent years revealed increased levels of glucose and VGF23-62 peptide and decreased levels of lactate and transthyretin.
Still, the discovery of “potential biomarker candidates” (PBC, [81]) or even disease-specific biomarkers in schizophrenia is at the very beginning. This could at least partially be a consequence of the heterogeneousness with regard to age, gender, medication, subtype, and duration of schizophrenia in groups of patients included by the majority of existing studies. The comparability of studies is further impeded by methodological heterogeneity. Only few CSF studies compared schizophrenia with other mental disorders such as bipolar or depressive disorders, which limits assessment of specificity of the reported CSF changes in schizophrenia. Given the additional methodological problems of proteomic procedures including a high vulnerability to outside influences like conditions of CSF collection and storage [118], the comparability of the various studies seems to be low and their relevance, therefore, questionable. A more integrated and more profound understanding of the relationship between the single parameters and their pathophysiological impact is mostly missing, and present findings must be considered as non-specific.
A number of steps appear to be necessary in order to bring forward the research of CSF in schizophrenia. In view of schizophrenia being a highly heterogeneous disease, a careful selection of patients to achieve homogenous cohorts is necessary. The older findings concerning the total cell count, the Blood-CSF barrier, and the immunoglobulins derive from investigations that used inconsistent and to some extent obsolete methodology with regard to the patients selection and the CSF analyses, are difficult to replicate nowadays, and must be considered, particularly with regard to the recent methodology improvements, with caution. One method to come forward could be to capture the psychopathology on a syndrome level by registering subscores for paranoia, disorganization, cognitive deficits, or negative symptoms such as anhedonia or listlessness (e.g., within “Positive and Negative Syndrome Scale”, PANSS). A more profound characterization of the particular psychopathological profiles would allow analyses between single parameters and distinct psychopathological entities, alleviating comparison between different patient cohorts. The investigation of patients in the prodromal phase or within the first episode of schizophrenia would promote longitudinal investigations, which are still lacking, although they represent the only way to identify the prognostic impact and test the suitability of potential biomarkers.
Also regarding proteomics, rigorous adherence to standard procedures based on international CSF guidelines [87, 141] is necessary to improve the quality of proteomic studies and to allow frequently and more precise detection of possible marker candidates, preferably within multicenter projects in order to increase the number of patients included. The search for single proteins at different molecular sizes should be advanced, paving the way for identification of syndrome-specific biomarker candidates, and possibly making a step toward improving diagnosis, treatment, and monitoring of schizophrenia in the future by CSF analyses.
References
Albrecht P, Torrey EF, Boone E, Hicks JT, Daniel N (1980) Raised cytomegalovirus-antibody level in cerebrospinal fluid of schizophrenic patients. Lancet 2:769–772
Alder J, Thakker-Varia S, Bangasser DA, Kuroiwa M, Plummer MR, Shors TJ, Black IB (2003) Brain-derived neurotrophic factor-induced gene expression reveals novel actions of VGF in hippocampal synaptic plasticity. J Neurosci 23:10800–10808
Amminger GP, McGorry PD, Berger GE, Wade D, Yung AR, Phillips LJ, Harrigan SM, Francey SM, Yolken RH (2007) Antibodies to infectious agents in individuals at ultra-high risk for psychosis. Biol Psychiatry 61:1215–1217
Amminger GP, Schafer MR, Papageorgiou K, Klier CM, Cotton SM, Harrigan SM, Mackinnon A, McGorry PD, Berger GE (2010) Long-chain omega-3 fatty acids for indicated prevention of psychotic disorders: a randomized, placebo-controlled trial. Arch Gen Psychiatry 67:146–154
Araque A, Parpura V, Sanzgiri RP, Haydon PG (1999) Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci 22:208–215
Aurell A, Rosengren LE, Karlsson B, Olsson JE, Zbornikova V, Haglid KG (1991) Determination of S-100 and glial fibrillary acidic protein concentrations in cerebrospinal fluid after brain infarction. Stroke 22:1254–1258
Axelsson R, Martensson E, Alling C (1982) Impairment of the blood-brain barrier as an aetiological factor in paranoid psychosis. Br J Psychiatry 141:273–281
Barak V, Barak Y, Levine J, Nisman B, Roisman I (1995) Changes in interleukin-1 beta and soluble interleukin-2 receptor levels in CSF and serum of schizophrenic patients. J Basic Clin Physiol Pharmacol 6:61–69
Bauer K, Kornhuber J (1987) Blood-cerebrospinal fluid barrier in schizophrenic patients. Eur Arch Psychiatry Neurol Sci 236:257–259
Beaudry P, Cohen P, Brandel JP, Delasnerie-Laupretre N, Richard S, Launay JM, Laplanche JL (1999) 14–3-3 protein, neuron-specific enolase, and S-100 protein in cerebrospinal fluid of patients with Creutzfeldt-Jakob disease. Dement Geriatr Cogn Disord 10:40–46
Bechter K, Reiber H, Herzog S, Fuchs D, Tumani H, Maxeiner HG (2010) Cerebrospinal fluid analysis in affective and schizophrenic spectrum disorders: identification of subgroups with immune responses and blood-CSF barrier dysfunction. J Psychiatr Res 44:321–330
Ben-Shachar D, Livne E, Spanier I, Leenders KL, Youdim MB (1994) Typical and atypical neuroleptics induce alteration in blood-brain barrier and brain 59FeCl3 uptake. J Neurochem 62:1112–1118
Bendikov I, Nadri C, Amar S, Panizzutti R, De Miranda J, Wolosker H, Agam G (2007) A CSF and postmortem brain study of d-serine metabolic parameters in schizophrenia. Schizophr Res 90:41–51
Bianchi R, Giambanco I, Donato R (1993) S-100 protein, but not calmodulin, binds to the glial fibrillary acidic protein and inhibits its polymerization in a Ca(2 +)-dependent manner. J Biol Chem 268:12669–12674
Blennow K, Fredman P, Wallin A, Gottfries CG, Karlsson I, Langstrom G, Skoog I, Svennerholm L, Wikkelso C (1993) Protein analysis in cerebrospinal fluid. II. Reference values derived from healthy individuals 18–88 years of age. Eur Neurol 33:129–133
Blennow K, Fredman P, Wallin A, Gottfries CG, Langstrom G, Svennerholm L (1993) Protein analyses in cerebrospinal fluid. I. Influence of concentration gradients for proteins on cerebrospinal fluid/serum albumin ratio. Eur Neurol 33:126–128
Blennow K, Fredman P, Wallin A, Gottfries CG, Skoog I, Wikkelso C, Svennerholm L (1993) Protein analysis in cerebrospinal fluid. III. Relation to blood-cerebrospinal fluid barrier function for formulas for quantitative determination of intrathecal IgG production. Eur Neurol 33:134–142
Blennow K, Vanmechelen E, Hampel H (2001) CSF total tau, Abeta42 and phosphorylated tau protein as biomarkers for Alzheimer’s disease. Mol Neurobiol 24:87–97
Bock E (1978) Immunoglobulins, prealbumin, transferrin, albumin, and alpha2-macroglobulin in cerebrospinal fluid and serum in schizophrenic patients. Birth Defects Orig Artic Ser 14:283–295
Brettschneider J, Claus A, Kassubek J, Tumani H (2005) Isolated blood-cerebrospinal fluid barrier dysfunction: prevalence and associated diseases. J Neurol 252:1067–1073
Brown KW, Kynoch PA, Thompson RJ (1980) Immunoreactive nervous system of specific enolase (14–3-2 protein) in human serum and cerebrospinal fluid. Clin Chim Acta 101:257–264
Bruetsch WL, Bahr MA, Skobba JS, Dieter WJ (1942) The group of dementia praecox patients with an increase of the protein content of the cerebrospinal fluid. J Nerv Ment Dis 95:669–679
Buckley PF, Pillai A, Evans D, Stirewalt E, Mahadik S (2007) Brain derived neurotropic factor in first-episode psychosis. Schizophr Res 91:1–5
Buttner T, Weyers S, Postert T, Sprengelmeyer R, Kuhn W (1997) S-100 protein: serum marker of focal brain damage after ischemic territorial MCA infarction. Stroke 28:1961–1965
Chapman ER (2002) Synaptotagmin: a Ca(2 +) sensor that triggers exocytosis? Natl Rev Mol Cell Biol 3:498–508
Chen da C, Wang J, Wang B, Yang SC, Zhang CX, Zheng YL, Li YL, Wang N, Yang KB, Xiu MH, Kosten TR, Zhang XY (2009) Decreased levels of serum brain-derived neurotrophic factor in drug-naive first-episode schizophrenia: relationship to clinical phenotypes. Psychopharmacology (Berl) 207:375–380
Corradini I, Verderio C, Sala M, Wilson MC, Matteoli M (2009) SNAP-25 in neuropsychiatric disorders. Ann N Y Acad Sci 1152:93–99
Crols R, Saerens J, Noppe M, Lowenthal A (1986) Increased GFAp levels in CSF as a marker of organicity in patients with Alzheimer’s disease and other types of irreversible chronic organic brain syndrome. J Neurol 233:157–160
Delisi LE, Weinberger DR, Potkin S, Neckers LM, Shiling DJ, Wyatt RJ (1981) Quantitative determination of immunoglobulins in CSF and plasma of chronic schizophrenic patients. Br J Psychiatry 139:513–518
Dencker SJ, Zethraeus S (1961) Sex differences in total protein content of cerebrospinal fluid. Acta Psychiatr Scand 36:76–82
Do KQ, Lauer CJ, Schreiber W, Zollinger M, Gutteck-Amsler U, Cuenod M, Holsboer F (1995) gamma-Glutamylglutamine and taurine concentrations are decreased in the cerebrospinal fluid of drug-naive patients with schizophrenic disorders. J Neurochem 65:2652–2662
Durany N, Michel T, Zochling R, Boissl KW, Cruz-Sanchez FF, Riederer P, Thome J (2001) Brain-derived neurotrophic factor and neurotrophin 3 in schizophrenic psychoses. Schizophr Res 52:79–86
Egan MF, el-Mallakh RS, Suddath RL, Lohr JB, Bracha HS, Wyatt RJ (1992) Cerebrospinal fluid and serum levels of neuron-specific enolase in patients with schizophrenia. Psychiatry Res 43:187–195
el-Mallakh RS, Suddath RL, Wyatt RJ (1993) Interleukin-1 alpha and interleukin-2 in cerebrospinal fluid of schizophrenic subjects. Prog Neuropsychopharmacol Biol Psychiatry 17:383–391
Fassbender K, Schmidt R, Schreiner A, Fatar M, Muhlhauser F, Daffertshofer M, Hennerici M (1997) Leakage of brain-originated proteins in peripheral blood: temporal profile and diagnostic value in early ischemic stroke. J Neurol Sci 148:101–105
Fernandez-Espejo E, Viveros MP, Nunez L, Ellenbroek BA, Rodriguez de Fonseca F (2009) Role of cannabis and endocannabinoids in the genesis of schizophrenia. Psychopharmacology (Berl) 206:531–549
Fuchs SA, De Barse MM, Scheepers FE, Cahn W, Dorland L, der Velden MG, Klomp LW, Berger R, Kahn RS, de Koning TJ (2008) Cerebrospinal fluid d-serine and glycine concentrations are unaltered and unaffected by olanzapine therapy in male schizophrenic patients. Eur Neuropsychopharmacol 18:333–338
Giuffrida A, Leweke FM, Gerth CW, Schreiber D, Koethe D, Faulhaber J, Klosterkotter J, Piomelli D (2004) Cerebrospinal anandamide levels are elevated in acute schizophrenia and are inversely correlated with psychotic symptoms. Neuropsychopharmacology 29:2108–2114
Hashimoto K, Engberg G, Shimizu E, Nordin C, Lindstrom LH, Iyo M (2005) Reduced d-serine to total serine ratio in the cerebrospinal fluid of drug naive schizophrenic patients. Prog Neuropsychopharmacol Biol Psychiatry 29:767–769
Hashimoto K, Fukushima T, Shimizu E, Komatsu N, Watanabe H, Shinoda N, Nakazato M, Kumakiri C, Okada S, Hasegawa H, Imai K, Iyo M (2003) Decreased serum levels of d-serine in patients with schizophrenia: evidence in support of the N-methyl-D-aspartate receptor hypofunction hypothesis of schizophrenia. Arch Gen Psychiatry 60:572–576
Hayakawa T, Ushio Y, Mori T, Arita N, Yoshimine T, Maeda Y, Shimizu K, Myoga A (1979) Levels in stroke patients of CSF astroprotein, an astrocyte-specific cerebroprotein. Stroke 10:685–689
Hodel A (1998) Snap-25. Int J Biochem Cell Biol 30:1069–1073
Hoerster SA Jr, Hillman FA, Bohls SW, Lara FY, Thurman N (1963) Cerebrospinal fluid in mental diseases (a study using paper electrophoresis). Dis Nerv Syst 24:357–360
Holmes E, Tsang TM, Huang JT, Leweke FM, Koethe D, Gerth CW, Nolden BM, Gross S, Schreiber D, Nicholson JK, Bahn S (2006) Metabolic profiling of CSF: evidence that early intervention may impact on disease progression and outcome in schizophrenia. PLoS Med 3:e327
Huang JT, Leweke FM, Oxley D, Wang L, Harris N, Koethe D, Gerth CW, Nolden BM, Gross S, Schreiber D, Reed B, Bahn S (2006) Disease biomarkers in cerebrospinal fluid of patients with first-onset psychosis. PLoS Med 3:e428
Huang JT, Leweke FM, Tsang TM, Koethe D, Kranaster L, Gerth CW, Gross S, Schreiber D, Ruhrmann S, Schultze-Lutter F, Klosterkotter J, Holmes E, Bahn S (2007) CSF metabolic and proteomic profiles in patients prodromal for psychosis. PLoS One 2:e756
Hunnerkopf R, Grassl J, Thome J (2007) Proteomics: biomarker research in psychiatry. Fortschr Neurol Psychiatry 75:579–586
Hunter R, Jones M, Malleson A (1969) Abnormal cerebrospinal fluid protein and gammaglobulin levels in 256 patients admitted to a psychiatric unit. J Neurol Sci 9:11–38
Hur EM, Zhou FQ (2010) GSK3 signalling in neural development. Nat Rev Neurosci 11:539–551
Issa G, Wilson C, Terry AV Jr, Pillai A (2010) An inverse relationship between cortisol and BDNF levels in schizophrenia: data from human postmortem and animal studies. Neurobiol Dis 39:327–333
Jablensky A (1997) The 100-year epidemiology of schizophrenia. Schizophr Res 28:111–125
Jacobsen LK, Frazier JA, Malhotra AK, Karoum F, McKenna K, Gordon CT, Hamburger SD, Lenane MC, Pickar D, Potter WZ, Rapoport JL (1997) Cerebrospinal fluid monoamine metabolites in childhood-onset schizophrenia. Am J Psychiatry 154:69–74
Jesse S, Steinacker P, Cepek L, von Arnim CA, Tumani H, Lehnert S, Kretzschmar HA, Baier M, Otto M (2009) Glial fibrillary acidic protein and protein S-100B: different concentration pattern of glial proteins in cerebrospinal fluid of patients with Alzheimer’s disease and Creutzfeldt-Jakob disease. J Alzheimers Dis 17:541–551
Jessen KR, Mirsky R (1980) Glial cells in the enteric nervous system contain glial fibrillary acidic protein. Nature 286:736–737
Jethwa PH, Ebling FJ (2008) Role of VGF-derived peptides in the control of food intake, body weight and reproduction. Neuroendocrinology 88:80–87
Jiang L, Lindpaintner K, Li HF, Gu NF, Langen H, He L, Fountoulakis M (2003) Proteomic analysis of the cerebrospinal fluid of patients with schizophrenia. Amino Acids 25:49–57
Jindal RD, Pillai AK, Mahadik SP, Eklund K, Montrose DM, Keshavan MS (2010) Decreased BDNF in patients with antipsychotic naive first episode schizophrenia. Schizophr Res 119:47–51
Joy CB, Mumby-Croft R, Joy LA (2006) Polyunsaturated fatty acid supplementation for schizophrenia. Cochrane Database Syst Rev 3:CD001257
Kahn RS, Davidson M, Knott P, Stern RG, Apter S, Davis KL (1993) Effect of neuroleptic medication on cerebrospinal fluid monoamine metabolite concentrations in schizophrenia. Serotonin-dopamine interactions as a target for treatment. Arch Gen Psychiatry 50:599–605
Kale A, Joshi S, Pillai A, Naphade N, Raju M, Nasrallah H, Mahadik SP (2009) Reduced cerebrospinal fluid and plasma nerve growth factor in drug-naive psychotic patients. Schizophr Res 115:209–214
Katila H, Hurme M, Wahlbeck K, Appelberg B, Rimon R (1994) Plasma and cerebrospinal fluid interleukin-1 beta and interleukin-6 in hospitalized schizophrenic patients. Neuropsychobiology 30:20–23
Kirch DG, Kaufmann CA, Papadopoulos NM, Martin B, Weinberger DR (1985) Abnormal cerebrospinal fluid protein indices in schizophrenia. Biol Psychiatry 20:1039–1046
Kirch DG, Alexander RC, Suddath RL, Papadopoulos NM, Kaufmann CA, Daniel DG, Wyatt RJ (1992) Blood-CSF barrier permeability and central nervous system immunoglobulin G in schizophrenia. J Neural Transm Gen Sect 89:219–232
Koethe D, Giuffrida A, Schreiber D, Hellmich M, Schultze-Lutter F, Ruhrmann S, Klosterkotter J, Piomelli D, Leweke FM (2009) Anandamide elevation in cerebrospinal fluid in initial prodromal states of psychosis. Br J Psychiatry 194:371–372
Koros E, Dorner-Ciossek C (2007) The role of glycogen synthase kinase-3beta in schizophrenia. Drug News Perspect 20:437–445
Kozlovsky N, Belmaker RH, Agam G (2001) Low GSK-3 activity in frontal cortex of schizophrenic patients. Schizophr Res 52:101–105
Kozlovsky N, Belmaker RH, Agam G (2000) Low GSK-3beta immunoreactivity in postmortem frontal cortex of schizophrenic patients. Am J Psychiatry 157:831–833
Kozlovsky N, Nadri C, Agam G (2005) Low GSK-3beta in schizophrenia as a consequence of neurodevelopmental insult. Eur Neuropsychopharmacol 15:1–11
Kozlovsky N, Regenold WT, Levine J, Rapoport A, Belmaker RH, Agam G (2004) GSK-3beta in cerebrospinal fluid of schizophrenia patients. J Neural Transm 111:1093–1098
Kranaster L, Koethe D, Hoyer C, Meyer-Lindenberg A, Leweke FM (2011) Cerebrospinal fluid diagnostics in first-episode schizophrenia. Eur Arch Psychiatry Clin Neurosci 261:529–530
Kronfol Z, Remick DG (2000) Cytokines and the brain: implications for clinical psychiatry. Am J Psychiatr 157:683–694
Lara DR, Gama CS, Belmonte-de-Abreu P, Portela LV, Goncalves CA, Fonseca M, Hauck S, Souza DO (2001) Increased serum S100B protein in schizophrenia: a study in medication-free patients. J Psychiatry Res 35:11–14
Leweke FM, Gerth CW, Koethe D, Klosterkotter J, Ruslanova I, Krivogorsky B, Torrey EF, Yolken RH (2004) Antibodies to infectious agents in individuals with recent onset schizophrenia. Eur Arch Psychiatry Clin Neurosci 254:4–8
Leweke FM, Giuffrida A, Koethe D, Schreiber D, Nolden BM, Kranaster L, Neatby MA, Schneider M, Gerth CW, Hellmich M, Klosterkotter J, Piomelli D (2007) Anandamide levels in cerebrospinal fluid of first-episode schizophrenic patients: impact of cannabis use. Schizophr Res 94:29–36
Leweke FM, Giuffrida A, Wurster U, Emrich HM, Piomelli D (1999) Elevated endogenous cannabinoids in schizophrenia. Neuroreport 10:1665–1669
Li S, Wu H, Guo H, Zhao Z (2006) Neuron-specific Enolase and myelin basic protein in cerebrospinal fluid of patients with first episode schizophrenia. J Huazhong Univ Sci Technolog Med Sci 26:228–230
Licinio J, Seibyl JP, Altemus M, Charney DS, Krystal JH (1993) Elevated CSF levels of interleukin-2 in neuroleptic-free schizophrenic patients. Am J Psychiatry 150:1408–1410
Ling SH, Tang YL, Jiang F, Wiste A, Guo SS, Weng YZ, Yang TS (2007) Plasma S-100B protein in Chinese patients with schizophrenia: comparison with healthy controls and effect of antipsychotics treatment. J Psychiatry Res 41:36–42
Liu J, Shi Y, Tang J, Guo T, Li X, Yang Y, Chen Q, Zhao X, He G, Feng G, Gu N, Zhu S, Liu H, He L (2005) SNPs and haplotypes in the S100B gene reveal association with schizophrenia. Biochem Biophys Res Commun 328:335–341
Manabe T (2002) Does BDNF have pre- or postsynaptic targets? Science 295:1651–1653
Martins-de-Souza D (2010) Is the word ‘biomarker’ being properly used by proteomics research in neuroscience? Eur Arch Psychiatry Clin Neurosci 260:561–562
Martins-de-Souza D, Maccarrone G, Wobrock T, Zerr I, Gormanns P, Reckow S, Falkai P, Schmitt A, Turck CW (2010) Proteome analysis of the thalamus and cerebrospinal fluid reveals glycolysis dysfunction and potential biomarkers candidates for schizophrenia. J Psychiatry Res 44:1176–1189
Martins-De-Souza D, Wobrock T, Zerr I, Schmitt A, Gawinecka J, Schneider-Axmann T, Falkai P, Turck CW (2010) Different apolipoprotein E, apolipoprotein A1 and prostaglandin-H2 D-isomerase levels in cerebrospinal fluid of schizophrenia patients and healthy controls. World J Biol Psychiatry 11:719–728
McAllister CG, van Kammen DP, Rehn TJ, Miller AL, Gurklis J, Kelley ME, Yao J, Peters JL (1995) Increases in CSF levels of interleukin-2 in schizophrenia: effects of recurrence of psychosis and medication status. Am J Psychiatry 152:1291–1297
McGrath J, Saha S, Welham J, El Saadi O, MacCauley C, Chant D (2004) A systematic review of the incidence of schizophrenia: the distribution of rates and the influence of sex, urbanicity, migrant status and methodology. BMC Med 2:13
Miller C, Kirchmair R, Troger J, Saria A, Fleischhacker WW, Fischer-Colbrie R, Benzer A, Winkler H (1996) CSF of neuroleptic-naive first-episode schizophrenic patients: levels of biogenic amines, substance P, and peptides derived from chromogranin A (GE-25) and secretogranin II (secretoneurin). Biol Psychiatry 39:911–918
Mischak H, Apweiler R, Banks RE, Conaway M, Coon J, Dominiczak A, Ehrich JH, Fliser D, Girolami M, Hermjakob H, Hochstrasser D, Jankowski J, Julian BA, Kolch W, Massy ZA, Neusuess C, Novak J, Peter K, Rossing K, Schanstra J, Semmes OJ, Theodorescu D, Thongboonkerd V, Weissinger EM, Van Eyk JE, Yamamoto T (2007) Clinical proteomics: a need to define the field and to begin to set adequate standards. Proteomics Clin Appl 1:148–156
Misu T, Takano R, Fujihara K, Takahashi T, Sato S, Itoyama Y (2009) Marked increase in cerebrospinal fluid glial fibrillar acidic protein in neuromyelitis optica: an astrocytic damage marker. J Neurol Neurosurg Psychiatry 80:575–577
Mollenhauer B, Cepek L, Bibl M, Wiltfang J, Schulz-Schaeffer WJ, Ciesielczyk B, Neumann M, Steinacker P, Kretzschmar HA, Poser S, Trenkwalder C, Otto M (2005) Tau protein, Abeta42 and S-100B protein in cerebrospinal fluid of patients with dementia with Lewy bodies. Dement Geriatr Cogn Disord 19:164–170
Müller N, Ackenheil M (1995) Immunoglobulin and albumin content of cerebrospinal fluid in schizophrenic patients: relationship to negative symptomatology. Schizophr Res 14:223–228
Muller DJ, Klempan TA, De Luca V, Sicard T, Volavka J, Czobor P, Sheitman BB, Lindenmayer JP, Citrome L, McEvoy JP, Lieberman JA, Honer WG, Kennedy JL (2005) The SNAP-25 gene may be associated with clinical response and weight gain in antipsychotic treatment of schizophrenia. Neurosci Lett 379:81–89
Musil R, Spellmann I, Riedel M, Dehning S, Douhet A, Maino K, Zill P, Muller N, Moller HJ, Bondy B (2008) SNAP-25 gene polymorphisms and weight gain in schizophrenic patients. J Psychiatry Res 42:963–970
Mustafa AK, Kumar M, Selvakumar B, Ho GP, Ehmsen JT, Barrow RK, Amzel LM, Snyder SH (2007) Nitric oxide S-nitrosylates serine racemase, mediating feedback inhibition of d-serine formation. Proc Natl Acad Sci USA 104:2950–2955
Nadri C, Dean B, Scarr E, Agam G (2004) GSK-3 parameters in postmortem frontal cortex and hippocampus of schizophrenic patients. Schizophr Res 71:377–382
Nadri C, Kozlovsky N, Agam G, Bersudsky Y (2002) GSK-3 parameters in lymphocytes of schizophrenic patients. Psychiatry Res 112:51–57
Niebuhr DW, Millikan AM, Cowan DN, Yolken R, Li Y, Weber NS (2008) Selected infectious agents and risk of schizophrenia among U.S. military personnel. Am J Psychiatry 165:99–106
Niebuhr DW, Millikan AM, Yolken R, Li Y, Weber NS (2008) Results from a hypothesis generating case-control study: herpes family viruses and schizophrenia among military personnel. Schizophr Bull 34:1182–1188
Nikkila HV, Muller K, Ahokas A, Miettinen K, Rimon R, Andersson LC (1999) Accumulation of macrophages in the CSF of schizophrenic patients during acute psychotic episodes. Am J Psychiatry 156:1725–1729
Nikkila HV, Muller K, Ahokas A, Rimon R, Andersson LC (2001) Increased frequency of activated lymphocytes in the cerebrospinal fluid of patients with acute schizophrenia. Schizophr Res 49:99–105
Ohnuma T, Sakai Y, Maeshima H, Hatano T, Hanzawa R, Abe S, Kida S, Shibata N, Suzuki T, Arai H (2008) Changes in plasma glycine, l-serine, and d-serine levels in patients with schizophrenia as their clinical symptoms improve: results from the Juntendo University Schizophrenia Projects (JUSP). Prog Neuropsychopharmacol Biol Psychiatry 32:1905–1912
Otto M, Wiltfang J, Tumani H, Zerr I, Lantsch M, Kornhuber J, Weber T, Kretzschmar HA, Poser S (1997) Elevated levels of tau-protein in cerebrospinal fluid of patients with Creutzfeldt-Jakob disease. Neurosci Lett 225:210–212
Oxenstierna G, Bergstrand G, Bjerkenstedt L, Sedvall G, Wik G (1984) Evidence of disturbed CSF circulation and brain atrophy in cases of schizophrenic psychosis. Br J Psychiatry 144:654–661
Oxenstierna G, Bergstrand G, Edman G, Flyckt L, Nyback H, Sedvall G (1996) Increased frequency of aberrant CSF circulation in schizophrenic patients compared to healthy volunteers. Eur Psychiatry 11:16–20
Pennington K, Cotter D, Dunn MJ (2005) The role of proteomics in investigating psychiatric disorders. Br J Psychiatry 187:4–6
Petzold A, Keir G, Green AJ, Giovannoni G, Thompson EJ (2004) An ELISA for glial fibrillary acidic protein. J Immunol Methods 287:169–177
Pillai A, Kale A, Joshi S, Naphade N, Raju MS, Nasrallah H, Mahadik SP (2010) Decreased BDNF levels in CSF of drug-naive first-episode psychotic subjects: correlation with plasma BDNF and psychopathology. Int J Neuropsychopharmacol 13:535–539
Puri BK (2010) Progressive structural brain changes in schizophrenia. Expert Rev Neurother 10:33–42
Raedler TJ, Wiedemann K (2006) CSF-studies in neuropsychiatric disorders. Neuro Endocrinol Lett 27:297–305
Rais M, van Haren NE, Cahn W, Schnack HG, Lepage C, Collins L, Evans AC, Hulshoff Pol HE, Kahn RS (2010) Cannabis use and progressive cortical thickness loss in areas rich in CB1 receptors during the first five years of schizophrenia. Eur Neuropsychopharmacol 20:855–865
Rapaport MH, McAllister CG, Pickar D, Tamarkin L, Kirch DG, Paul SM (1997) CSF IL-1 and IL-2 in medicated schizophrenic patients and normal volunteers. Schizophr Res 25:123–129
Reiber H (2001) Dynamics of brain-derived proteins in cerebrospinal fluid. Clin Chim Acta 310:173–186
Reiber H, Felgenhauer K (1987) Protein transfer at the blood cerebrospinal fluid barrier and the quantitation of the humoral immune response within the central nervous system. Clin Chim Acta 163:319–328
Reichardt LF (2003) Neurobiology: signals that make waves. Nature 426:25–26
Rizos EN, Rontos I, Laskos E, Arsenis G, Michalopoulou PG, Vasilopoulos D, Gournellis R, Lykouras L (2008) Investigation of serum BDNF levels in drug-naive patients with schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry 32:1308–1311
Roos KL (2003) Lumbar puncture. Semin Neurol 23:105–114
Rose CR, Blum R, Pichler B, Lepier A, Kafitz KW, Konnerth A (2003) Truncated TrkB-T1 mediates neurotrophin-evoked calcium signalling in glia cells. Nature 426:74–78
Rosengren LE, Ahlsen G, Belfrage M, Gillberg C, Haglid KG, Hamberger A (1992) A sensitive ELISA for glial fibrillary acidic protein: application in CSF of children. J Neurosci Methods 44:113–119
Rosenling T, Slim CL, Christin C, Coulier L, Shi S, Stoop MP, Bosman J, Suits F, Horvatovich PL, Stockhofe-Zurwieden N, Vreeken R, Hankemeier T, van Gool AJ, Luider TM, Bischoff R (2009) The effect of preanalytical factors on stability of the proteome and selected metabolites in cerebrospinal fluid (CSF). J Proteome Res 8:5511–5522
Rothermundt M, Ahn JN, Jorgens S (2009) S100B in schizophrenia: an update. Gen Physiol Biophys 28 Spec No Focus:F76–F81
Rothermundt M, Falkai P, Ponath G, Abel S, Burkle H, Diedrich M, Hetzel G, Peters M, Siegmund A, Pedersen A, Maier W, Schramm J, Suslow T, Ohrmann P, Arolt V (2004) Glial cell dysfunction in schizophrenia indicated by increased S100B in the CSF. Mol Psychiatry 9:897–899
Rothermundt M, Missler U, Arolt V, Peters M, Leadbeater J, Wiesmann M, Rudolf S, Wandinger KP, Kirchner H (2001) Increased S100B blood levels in unmedicated and treated schizophrenic patients are correlated with negative symptomatology. Mol Psychiatry 6:445–449
Rothermundt M, Ponath G, Glaser T, Hetzel G, Arolt V (2004) S100B serum levels and long-term improvement of negative symptoms in patients with schizophrenia. Neuropsychopharmacology 29:1004–1011
Sarandol A, Kirli S, Akkaya C, Altin A, Demirci M, Sarandol E (2007) Oxidative-antioxidative systems and their relation with serum S100 B levels in patients with schizophrenia: effects of short term antipsychotic treatment. Prog Neuropsychopharmacol Biol Psychiatrty 31:1164–1169
Scheepers FE, Gispen-de Wied CC, Westenberg HG, Kahn RS (2001) The effect of olanzapine treatment on monoamine metabolite concentrations in the cerebrospinal fluid of schizophrenic patients. Neuropsychopharmacology 25:468–475
Schmitt A, Bertsch T, Henning U, Tost H, Klimke A, Henn FA, Falkai P (2005) Increased serum S100B in elderly, chronic schizophrenic patients: negative correlation with deficit symptoms. Schizophr Res 80:305–313
Schönknecht P, Hempel A, Hunt A, Seidl U, Volkmann M, Pantel J, Schröder J (2003) Cerebrospinal fluid tau protein levels in schizophrenia. Eur Arch Psychiatry Clin Neurosci 253:100–102
Schroeter ML, Abdul-Khaliq H, Krebs M, Diefenbacher A, Blasig IE (2009) Neuron-specific enolase is unaltered whereas S100B is elevated in serum of patients with schizophrenia–original research and meta-analysis. Psychiatry Res 167:66–72
Schuller EA, Benabdallah S, Sagar HJ, Reboul JA, Tompe LC (1987) IgG synthesis within the central nervous system. Comparison of three formulas. Arch Neurol 44:600–604
Schwarz E, Bahn S (2008) Biomarker discovery in psychiatric disorders. Electrophoresis 29:2884–2890
Schwarz E, Bahn S (2008) Cerebrospinal fluid: identification of diagnostic markers for schizophrenia. Expert Rev Mol Diagn 8:209–216
Schwarz E, Bahn S (2008) The utility of biomarker discovery approaches for the detection of disease mechanisms in psychiatric disorders. Br J Pharmacol 153(Suppl 1):S133–S136
Schwarz MJ, Ackenheil M, Riedel M, Muller N (1998) Blood-cerebrospinal fluid barrier impairment as indicator for an immune process in schizophrenia. Neurosci Lett 253:201–203
Seehusen DA, Reeves MM, Fomin DA (2003) Cerebrospinal fluid analysis. Am Fam Physician 68:1103–1108
Selemon LD, Goldman-Rakic PS (1999) The reduced neuropil hypothesis: a circuit based model of schizophrenia. Biol Psychiatry 45:17–25
Selemon LD, Mrzljak J, Kleinman JE, Herman MM, Goldman-Rakic PS (2003) Regional specificity in the neuropathologic substrates of schizophrenia: a morphometric analysis of Broca’s area 44 and area 9. Arch Gen Psychiatry 60:69–77
Sokol DK, O’Brien RS, Wagenknecht DR, Rao T, McIntyre JA (2007) Antiphospholipid antibodies in blood and cerebrospinal fluids of patients with psychosis. J Neuroimmunol 190:151–156
Southwell BR, Duan W, Alcorn D, Brack C, Richardson SJ, Kohrle J, Schreiber G (1993) Thyroxine transport to the brain: role of protein synthesis by the choroid plexus. Endocrinology 133:2116–2126
Spellmann I, Muller N, Musil R, Zill P, Douhet A, Dehning S, Cerovecki A, Bondy B, Moller HJ, Riedel M (2008) Associations of SNAP-25 polymorphisms with cognitive dysfunctions in Caucasian patients with schizophrenia during a brief trail of treatment with atypical antipsychotics. Eur Arch Psychiatry Clin Neurosci 258:335–344
Steiner J, Bielau H, Bernstein HG, Bogerts B, Wunderlich MT (2006) Increased cerebrospinal fluid and serum levels of S100B in first-onset schizophrenia are not related to a degenerative release of glial fibrillar acidic protein, myelin basic protein and neurone-specific enolase from glia or neurones. J Neurol Neurosurg Psychiatry 77:1284–1287
Sugerman HJ, DeMaria EJ, Felton WL 3rd, Nakatsuka M, Sismanis A (1997) Increased intra-abdominal pressure and cardiac filling pressures in obesity-associated pseudotumor cerebri. Neurology 49:507–511
Teunissen CE, Petzold A, Bennett JL, Berven FS, Brundin L, Comabella M, Franciotta D, Frederiksen JL, Fleming JO, Furlan R, Hintzen RQ, Hughes SG, Johnson MH, Krasulova E, Kuhle J, Magnone MC, Rajda C, Rejdak K, Schmidt HK, van Pesch V, Waubant E, Wolf C, Giovannoni G, Hemmer B, Tumani H, Deisenhammer F (2009) A consensus protocol for the standardization of cerebrospinal fluid collection and biobanking. Neurology 73:1914–1922
Thakker-Varia S, Alder J (2009) Neuropeptides in depression: role of VGF. Behav Brain Res 197:262–278
Thompson PM, Kelley M, Yao J, Tsai G, van Kammen DP (2003) Elevated cerebrospinal fluid SNAP-25 in schizophrenia. Biol Psychiatry 53:1132–1137
Thompson PM, Rosenberger C, Qualls C (1999) CSF SNAP-25 in schizophrenia and bipolar illness. A pilot study. Neuropsychopharmacology 21:717–722
Toorey EF, Peterson MR, Brannon WL, Carpenter WT, Post RM, Van Kammen DP (1978) Immunoglobulins and viral antibodies in psychiatric patients. Br J Psychiatry 132:342–348
Torrey EF, Albrecht P, Behr DE (1985) Permeability of the blood-brain barrier in psychiatric patients. Am J Psychiatry 142:657–658
Tumani H, Teunissen C, Sussmuth S, Otto M, Ludolph AC, Brettschneider J (2008) Cerebrospinal fluid biomarkers of neurodegeneration in chronic neurological diseases. Expert Rev Mol Diagn 8:479–494
Van Eldik LJ, Wainwright MS (2003) The Janus face of glial-derived S100B: beneficial and detrimental functions in the brain. Restor Neurol Neurosci 21:97–108
van Haren NE, Schnack HG, Cahn W, van den Heuvel MP, Lepage C, Collins L, Evans AC, Hulshoff Pol HE, Kahn RS (2011) Changes in cortical thickness during the course of illness in schizophrenia. Arch Gen Psychiatry 68:871–880
van Kammen DP, McAllister-Sistilli CG, Kelley ME, Gurklis JA, Yao JK (1999) Elevated interleukin-6 in schizophrenia. Psychiatry Res 87:129–136
Vasic N, Wolf RC (2006) How early is it possible to detect and to treat schizophrenia? Nervenheilkunde 25:351–358
Verheecke P (1975) On the tau-protein in cerebrospinal fluid. J Neurol Sci 26:277–281
Verrall L, Walker M, Rawlings N, Benzel I, Kew JN, Harrison PJ, Burnet PW (2007) d-Amino acid oxidase and serine racemase in human brain: normal distribution and altered expression in schizophrenia. Eur J Neurosci 26:1657–1669
Wiesmann M, Wandinger KP, Missler U, Eckhoff D, Rothermundt M, Arolt V, Kirchner H (1999) Elevated plasma levels of S-100b protein in schizophrenic patients. Biol Psychiatry 45:1508–1511
Wilson RI, Nicoll RA (2002) Endocannabinoid signaling in the brain. Science 296:678–682
Wunderlich MT, Lins H, Skalej M, Wallesch CW, Goertler M (2006) Neuron-specific enolase and tau protein as neurobiochemical markers of neuronal damage are related to early clinical course and long-term outcome in acute ischemic stroke. Clin Neurol Neurosurg 108:558–563
Yolken R (2004) Viruses and schizophrenia: a focus on herpes simplex virus. Herpes 11(Suppl 2):83A–88A
Yolken RH, Torrey EF (2008) Are some cases of psychosis caused by microbial agents? A review of the evidence. Mol Psychiatry 13:470–479
Young G, Conquer J (2005) Omega-3 fatty acids and neuropsychiatric disorders. Reprod Nutr Dev 45:1–28
Yuan J, Yankner BA (2000) Apoptosis in the nervous system. Nature 407:802–809
Zimmer DB, Cornwall EH, Landar A, Song W (1995) The S100 protein family: history, function, and expression. Brain Res Bull 37:417–429
Zimmer DB, Van Eldik LJ (1986) Identification of a molecular target for the calcium-modulated protein S100. Fructose-1, 6-bisphosphate aldolase. J Biol Chem 261:11424–11428
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vasic, N., Connemann, B.J., Wolf, R.C. et al. Cerebrospinal fluid biomarker candidates of schizophrenia: where do we stand?. Eur Arch Psychiatry Clin Neurosci 262, 375–391 (2012). https://doi.org/10.1007/s00406-011-0280-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-011-0280-9