Abstract
Background
The senses of smell and taste can be adversely affected by both tumour- and treatment-related factors amongst head and neck cancer patients. The consequences may negatively impact nutritional status as well as quality of life in this patient population.
Patients and methods
This prospective longitudinal follow-up study is consisted of 44 patients treated for oral cavity, oropharyngeal or hypopharyngeal cancer with tumour resection and microvascular free tissue transfer reconstruction at the Helsinki University Hospital, Helsinki, Finland. Thirty-nine (89%) of them also received radiotherapy. The senses of smell (odour detection, identification and threshold test) and taste (electrogustometry) and quality of life (UW-QOL) were evaluated preoperatively, and at 6 weeks, 3 months, 6 months and 12 months, postoperatively.
Results
There were higher scores in the odour detection values in the 6-week and 3-month tests compared with preoperative values for the tumour side. Other detection scores did not differ statistically from the preoperative values neither in the tumour nor the contralateral side. However, in the odour identification test, all posttreatment values were statistically significantly higher than pretreatment ones. In the olfactory threshold test, no statistically significant differences were found between pre- and posttreatment values. Electrogustometry values for the taste on the tumour side were statistically significantly impaired at 6 weeks (p < 0.05) and at 3 months (p < 0.01) compared with the pretreatment results. They were also impaired at 6 months and at 12 months, although the differences were not statistically significant. The quality of life was impaired after treatment in this patient series. However, the correlation between quality of life and sense of taste was found only at one time point (3 months) and only with contralateral side measurements.
Conclusions
We conclude that in oral and pharyngeal cancer patients the postoperative taste problems are related to the impairment on the taste sensation in the tongue but not with the sense of smell. Moreover, the impairment in the quality of life is not clearly related to the impaired sense of taste.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The aim of surgical treatment of oral and pharyngeal cancer consists of radical tumour resection with clear surgical margins and typically includes a neck dissection. Free tissue transfer reconstruction is an established and reliable technique for the reconstruction of the resultant tissue defect [1]. In addition, treatment is often supplemented by postoperative radiotherapy or chemoradiotherapy. Breathing, speaking, chewing, and swallowing can be impaired by both cancer- and treatment-related factors [2]. The sense of taste can also be affected by the various interventions to the upper aerodigestive area, as well as the tumour itself potentially destroying the oral mucosal lining, which encloses the taste buds [3]. The tumour may also disturb the food bolus preparation and prevent the aroma passing from the pharynx up to the nasal olfactory epithelium. Due to the close functional relationship between taste and smell sensations, it is obvious that flavor is a combination of both [4]. Self-reported disorders of smell and taste are thus quite common, especially in head and neck cancer patients, but often underestimated by health-care workers [5, 6]. Furthermore, these problems may have a negative impact on the patient’s nutritional status, which in turn is known to impair tissue healing and may predispose to complications [7, 8]. However, despite the clinical relevance of the problem, there are only a few studies in the medical literature focusing on objectively measured alterations in taste and smell among oral and pharyngeal cancer patients [5, 6].
The aim of this study was to evaluate both the sense of smell and taste amongst oral and pharyngeal cancer patients managed by surgical resection, free flap reconstruction and postoperative (chemo)radiotherapy. The effect of the disease itself and its treatment was studied by comparing preoperative and postoperative measurements during the 1-year follow-up period. Additionally, any possible correlation between the sensory changes and quality of life was evaluated.
Patients and methods
The study population consisted of 44 consecutive patients treated for carcinoma in the oral cavity, oropharynx or hypopharynx with curative intent by tumour resection and microvascular free tissue transfer reconstruction at the Helsinki University Hospital (HUH). A multidisciplinary head and neck tumour board evaluated the treatment modality for each case.
The sociodemographic and medical characteristics of the 44 patients are listed in Table 1. Mean age was 56.2 years (range 38–80). This study was part of a larger prospective study [2]. Smell and taste were measured prospectively together with a number of other functional parameters. The results of the other functional studies have been published previously [2]. Thirty-nine (89%) out of the 44 patients received radiotherapy, but the olfactory area was not included in the radiation field in any of the patients. The senses of smell and taste were evaluated preoperatively, and at 6 weeks, 3 months, 6 months and 12 months after treatment. For the assessment of smell, both a detection test and a threshold test were used. Fourteen patients died during the 1-year follow-up. After 6 weeks there were 39 patients, after 3 months 38, after 6 months 35 and after 12 months 30 patients still alive and eligible to be evaluated. The Research Ethics Board at HUH approved the study design and an institutional study permission was granted.
The odour detection and identification tests
The patient was asked to sniff seven different odours (coffee, cinnamon, spirit, vanillin, camphor, turpentine and gasoline). Detection without identification of the odour as well as specific odour identification were both assessed. Results were given as the sum of the positive answers. Each nostril (tumour side and contralateral side) was tested separately.
The olfactory threshold test
The detailed method has been published elsewhere [9, 10]. A commercially available smell test kit (Olfactory-Labs, Berkeley, California) was used. At each step of the test, the patient had to choose between the phenylethyl methyl ethyl carbinol sniff bottle and the control bottle containing the plain diluent, and was repeated three times. The weakest concentration at which the patient selected the correct bottle all three times was accepted as the olfactory threshold. The manufacturer expressed the concentrations using arbitrary logarithmic units (ALU). The concentrations in use were − 25, − 15, − 5, 5, 15, 25, 35, 45, and 55 ALU. A test result > 55 was awarded when the patient failed to smell the highest concentration. In the threshold test, the patient used both nostrils at the same time.
The gustatory test
The examination of the sense of taste was performed using electrogustometry (Rion TR-06 electrogustometer 6, Sensonics, Inc., Haddon Heights, NJ, USA). Both sides of the tongue were tested separately. The tongue was stimulated by a short electrical impulse and the intensity was gradually increased (10, 20, 40, 100, 200 and 400 µA) until the patient had a positive sensation. The value > 400 µA was awarded when the highest impulse did not cause a sensation. The positive sensation for the patients resembles the one when a battery is pressed against tongue. The tumour side and contralateral side were tested separately. An approximate value of 100 µA or less can be considered as normal taste sensation.
Quality of life
The University of Washington Quality-of-Life Questionnaire (UW-QOL) was used for the evaluation. Nine questions (pain, disfigurement, activity, recreation/entertainment, employment, chewing, swallowing, speech, shoulder disability) were rated according to a scale from 0 (the greatest dysfunction) to 100 (best function). For a final composite score (0–100), the total score (maximum 900) was divided by nine. The questionnaire was completed before operation and four times postoperatively (6 weeks, 3 months, 6 months and 12 months after operation).
Statistical analysis
Data analysis and statistical tests were performed with the help of a professional statistician. Descriptive analyses (frequency, mean, standard deviation, etc.) were calculated using NCSS 8 (LLC, Kaysville, Utah, USA). Differences between the groups were tested using Wilcoxon rank-sum test and the Spearman test was used to evaluate correlations. p values of < 0.05 were considered statistically significant.
Results
Odour detection and identification tests
In the preoperative detection test, the mean value ± standard deviation was 6.50 ± 0.65 on the tumour side and 6.72 ± 0.51 on the contralateral side and in the preoperative identification test 2.44 ± 1.50 and 2.38 ± 1.27, respectively. In the odour detection and identification tests, there were no statistically significant differences between the tumour and the contralateral side neither in the pre- and posttreatment measurements. There were higher scores in the odour detection values in the 6 weeks and 3-month tests compared with preoperative values for the tumour side. Other detection scores did not differ statistically from the preoperative values neither in the tumour nor the contralateral side. However, in the odour identification tests, all postoperative values were statistically significantly higher than preoperative ones. Differences between preoperative situation and each postoperative test (6 weeks, 3 months, 6 months and 12 months) are shown in Tables 2 and 3.
Olfactory threshold test
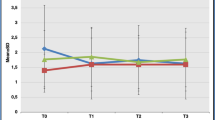
Almost two-thirds (63.9%) of patients had a preoperative threshold test result of less than 35 ALU (normal) and 36.1% between 35 and 55 ALU (hyposmia). In the postoperative measurements, only one patient (3.6%) at 6 weeks, one (3.2%) at 3 months, and two patients (10%) at 12 months had a value of more than 55 ALU (anosmia). There were no statistically significant differences between any of the preoperative and postoperative values (Fig. 1).
Sense of taste (electrogustometry)
The preoperative electrogustometry result of the contralateral side was 100 µA or less in 85.7% of tested patients compared with 64.3% in the tumour side (Fig. 2) (p < 0.05). In the tumour side, electrogustometry values were statistically significantly impaired after surgery at 6 weeks (p < 0.05) and at 3 months (p < 0.01) compared with the preoperative situation. Also, the 6 and 12 months measurements were impaired, although the differences were not statistically significant (Fig. 3). The changes in the contralateral values were not statistically significant (Fig. 4).
The correlations between electrogustometry values and body mass index, smoking years, alcohol consumption, the stage of disease, tumour size, tumour site, type of operation (mandibulotomy vs. non mandibulotomy, neck procedure) were calculated. Statistically significant correlations were found between preoperative taste on the tumour side and tumour size (p < 0.01) and between smoking years and taste on the contralateral side at 6 weeks (p < 0.05) and 6 months (p < 0.001) postoperatively. In other cases, either statistically significant correlation was not found or the number of cases in groups was too small for statistical testing.
Quality of life
The mean preoperative composite score was 83.5 and postoperatively at 6 weeks, and at 3, 6, and 12 months 76.2, 75.6, 76.6, and 79.8, respectively (p < 0.0001). A breakdown of the domain scores before and after operation and the significance of any change are presented in Table 4. These results have been previously reported in detail [11]. For this study, the correlation between electrogustometry result and quality of life was tested. The mean composite score and individual domain scores 3 (activity), 4 (recreation/entertainment), 6 (chewing) and 7 (swallowing) were tested separately. Only contralateral side taste at 3 months after treatment demonstrated a statistically significant correlation with the quality of life. This correlation was seen with the composite score (Spearman’s coefficient (r) = − 0.63, p < 0.05), and domain scores 3 (activity) (r = 0.55, p < 0.05), 6 (chewing) (r = − 0.58, p < 0.05) and 7 (swallowing) (r = − 0.63, p < 0.05). In all cases, the decreased sense of taste showed a correlation with a lower quality of life. The correlations between olfactory results and quality of life were not analyzed since the olfactory results improved after surgery, whereas the quality of life results were worse.
Discussion
This is a prospective follow-up study of a series of 44 patients with head and neck cancer affecting the upper aerodigestive tract who underwent multimodality therapy for their cancer. Olfactory and gustatory senses were tested to analyse the effect of their cancer treatment on these functions. We found that there is postoperatively impairment in the sense of taste but an improvement in the sense of smell.
We found that the sense of taste of the tumour side of the tongue tested by electrogustometry was preoperatively weaker than that of the contralateral side. This could be a sign of tumour-related disturbance of the sense of taste since the decreased taste was also positively correlated with the tumour size. As expected, the taste of the affected side decreased after surgery when it was tested by electrogustometry but the contralateral side remained unchanged. Three cranial nerves (CN) mediate the sense of taste. Facial nerve (CN VII) is responsible for the anterior two-thirds of the tongue and glossopharyngeal nerve (CN IX) for the posterior one-third. Vagal nerve (CN X) innervates the taste buds on the laryngeal surface of the epiglottis but its role in taste sensation is unclear. In addition, lingual nerve, a branch of the trigeminal nerve (CN V), supplies the sensory innervation to the tongue. It also carries the fibers from facial nerve via chorda tympani. Both the tumour and particularly its operative treatment can damage lingual and glossopharyngeal nerves. The study by Yamashita et al. suggested that radiotherapy results in damage to mainly the taste buds but not to the nerve fibers of the nerves involved in taste functions. In addition, they showed that the negative impact of radiotherapy on the sense of taste was at least partially reversible [12]. Mirza et al. [13] reported that radiotherapy for head and neck cancer impaired the sense of sour taste. In addition, they found that the amount of taste pores decreased during the treatment. In the study by Sandow et al. [14], 13 patients were treated by radiotherapy for primary tumours of the oropharynx. The study showed that the sense of taste was impaired during radiotherapy (the measurement was done at 1 month after the initiation of therapy), but it was normalized at 6 months after end of treatment. Mossman et al. [15] studied taste function in 13 patients treated by radiotherapy 1–7 years previously for tumours of the head and neck. They found subjective hypogeusia in 2 out of their 13 (15%) patients and objective taste impairment in 9 out of 13 (69%) patients. The taste impairment after radiotherapy can thus also be long lasting. In the present series, most patients received radiotherapy and therefore evaluation of its effect was impossible.
In the present series, there was no statistically significant decrease in the sense of smell according to either the detection test or the threshold test when preoperative and postoperative values were compared. In fact, the study subjects identified smells even better postoperatively than preoperatively. There are several potential explanations for this finding, e.g. stopping smoking, but further studies would be needed to clarify the mechanism of this phenomenon. Smoking has been shown to impair the sense of smell [16]. This effect is long lasting but reversible. The same study also showed that the improvement in smell function after the cessation of smoking is a slow phenomenon. Unfortunately, we did not record data on smoking cessation and its possible effects. We compared pre- and postoperative results and the study subjects served as their own controls. Therefore, although it is known that the sense of smell is age-related, we did not calculate age-related values for hyposmia and anosmia [17]. Both the odour detection and the identification tests and the olfactory threshold test were in clinical use at our department during the study period. Radiotherapy may negatively affect the sense of smell [18, 19]. However, while almost all (89%) patients in this series received radiotherapy, the olfactory area was not included in the radiation field. The low preoperative scores and the increase in the postoperative smell detection and threshold could be explained by the effect of the tumour smell itself intervening with olfactory functions.
The University of Washington Quality-of-Life Questionnaire (UW-QOL) is composed of nine questions, which deal with pain, disfigurement, activity, recreation/entertainment, employment, chewing, swallowing, speech, and shoulder disability. The correlation between the sense of taste and quality of life was found only at the 3-month time point and even then only with contralateral side measurements. Of the nine single domains, activity, recreation/entertainment, chewing and swallowing may be linked with taste. Thus, the sense of taste could not have a very strong impact on common scores. In addition, these four domains possibly linked with sense of taste were associated with several other factors, which may have a more significant predictive value. Therefore, it was understandable that the correlation between sense of taste and QOL scores was so weak.
Earlier studies have shown that smoking can impair the sense of taste and especially salty tasting [20, 21]. In the present study, 70% of the patients were smokers. Taste impairment was not correlated with the smoking status (nonsmoker, moderate smoker or heavy smoker) but instead it was correlated with smoking years. This correlation was seen on the contralateral side at 6 weeks and at 6 months. However, at 12 months the patient number was too small for statistical testing.
Electrogustometry has its own strengths and weaknesses. However, it is not expensive and not time consuming. In addition, its sensitivity and specificity have been reported to be too low for screening purposes [22]. In particular, the sensitivity seems low and for this reason, the percentage of patients suffering from taste problems may have been underestimated in the present series. However, electrogustometry is useful for follow-up studies, as in the present study [23]. Preoperative and postoperative values, the change between time points was quite reliably comparable when the patients serve as their own controls.
We conclude that the taste problems of oral and pharyngeal patients after free flap reconstruction and radiotherapy were purely taste-related without increased deficits in the sense of smell. Preoperatively, the tumour itself may have had some negative effect on the sense of taste but clearly stronger impairment was found after the treatment. Although these patients clearly have an impaired sense of taste on the tumour side and impaired quality of life, there was no statistically significant correlation between these two domains. Better understanding of the importance of senses of smell and taste on patient satisfaction, motivation to maintain sufficient eating pre-, peri- and posttreatment and during recovery, warrants further studies with larger series size.
References
de Bree R, Rinaldo A, Genden EM, Suarez C, Rodrigo JP, Fagan JJ et al (2008) Modern reconstruction techniques for oral and pharyngeal defects after tumor resection. Eur Arch Otorhinolaryngol 265(1):1–9
Markkanen-Leppanen M (2006) Functional outcome after free flap reconstructions in oral and pharyngeal cancer. Thesis. University of Helsinki, Helsinki, Finland. URN:ISBN:952-10-3047-X
Epstein JB, Barasch A (2010) Taste disorders in cancer patients: pathogenesis, and approach to assessment and management. Oral Oncol 46(2):77–81
Bhattacharyya N, Kepnes LJ (2015) Contemporary assessment of the prevalence of smell and taste problems in adults. Laryngoscope 125(5):1102–1106
Hong JH, Omur-Ozbek P, Stanek BT, Dietrich AM, Duncan SE, Lee YW et al (2009) Taste and odor abnormalities in cancer patients. J Support Oncol 7(2):58–65
Epstein JB, Smutzer G, Doty RL (2016) Understanding the impact of taste changes in oncology care. Support Care Cancer 24(4):1917–1931
Halyard MY (2009) Taste and smell alterations in cancer patients—real problems with few solutions. J Support Oncol 7(2):68–69
Farmer MN, Raddin RS, Roberts JD (2009) The relationship between taste, olfaction, and nutrition in the cancer population. J Support Oncol 7(2):70–72
Amoore JE, Ollman BG (1983) Practical test kits for quantitatively evaluating the sense of smell. Rhinology 21(1):49–54
Amoore JE, O’Neill RS (1986) Clinical olfactometry: improved convenience in squeeze-bottle kits; and a portable olfactometer. Chem Senses 11:576
Markkanen-Leppanen M, Makitie AA, Haapanen ML, Suominen E, Asko-Seljavaara S (2006) Quality of life after free-flap reconstruction in patients with oral and pharyngeal cancer. Head Neck 28(3):210–216
Yamashita H, Nakagawa K, Tago M, Nakamura N, Shiraishi K, Eda M et al (2006) Taste dysfunction in patients receiving radiotherapy. Head Neck 28(6):508–516
Mirza N, Machtay M, Devine PA, Troxel A, Abboud SK, Doty RL (2008) Gustatory impairment in patients undergoing head and neck irradiation. Laryngoscope 118(1):24–31
Sandow PL, Hejrat-Yazdi M, Heft MW (2006) Taste loss and recovery following radiation therapy. J Dent Res 85(7):608–611
Mossman K, Shatzman A, Chencharick J (1982) Long-term effects of radiotherapy on taste and salivary function in man. Int J Radiat Oncol Biol Phys 8(6):991–997
Frye RE, Schwartz BS, Doty RL (1990) Dose-related effects of cigarette smoking on olfactory function. JAMA 263(9):1233–1236
Simola M, Malmberg H (1998) Sense of smell in allergic and nonallergic rhinitis. Allergy 53(2):190–194
Ho WK, Kwong DL, Wei WI, Sham JS (2002) Change in olfaction after radiotherapy for nasopharyngeal cancer—a prospective study. Am J Otolaryngol 23(4):209–214
Holscher T, Seibt A, Appold S, Dorr W, Herrmann T, Huttenbrink KB et al (2005) Effects of radiotherapy on olfactory function. Radiother Oncol 77(2):157–163
Jackson JA (1967) Heavy smoking and sodium chloride hypogeusia. J Dent Res 46(4):742–744
Uota M, Ogawa T, Ikebe K, Arai Y, Kamide K, Gondo Y et al (2016) Factors related to taste sensitivity in elderly: cross-sectional findings from SONIC study. J Oral Rehabil 43(12):943–952
Ellegard EK, Hay KD, Morton RP (2007) Is electrogustometry useful for screening abnormalities of taste? J Laryngol Otol 121(12):1161–1164
Stillman JA, Morton RP, Hay KD, Ahmad Z, Goldsmith D (2003) Electrogustometry: strengths, weaknesses, and clinical evidence of stimulus boundaries. Clin Otolaryngol Allied Sci 28(5):406–410
Acknowledgements
The authors thank Timo Pessi, MSc for performing the statistical analyses.
Funding
This study was funded by the Helsinki University Hospital Research Fund (Grant number TYH2015204).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional research ethics committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The Research Ethics Board approved the study design and an institutional study permission was granted.
Informed consent
All patients filled in an informed consent form.
Rights and permissions
About this article
Cite this article
Lilja, M., Markkanen-Leppänen, M., Viitasalo, S. et al. Olfactory and gustatory functions after free flap reconstruction and radiotherapy for oral and pharyngeal cancer: a prospective follow-up study. Eur Arch Otorhinolaryngol 275, 959–966 (2018). https://doi.org/10.1007/s00405-018-4883-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-018-4883-x