Abstract
Evidence suggests that patients with head and neck cancer (HNC) are susceptible to post-traumatic stress disorder (PTSD). However, research is yet to examine predictors of PTSD symptoms in this patient group. The objective of this study was to investigate whether coping strategies at HNC diagnosis were related to outcomes of post-traumatic stress and health-related quality of life (HRQL) 6 months later. Sixty-five patients with HNC completed an assessment of coping, distress, and health-related quality of life at diagnosis and again 6 months later, and an assessment of post-traumatic stress at 6 months. Correlations and regression analyses were performed to examine relationships between coping and outcomes over time. Regression analyses showed that denial, behavioural disengagement and self-blame at diagnosis predicted post-traumatic stress symptoms. Self-blame at diagnosis also predicted poor HRQL. Results have implications for the development of psychological interventions that provide alternative coping strategies to potentially reduce PTSD symptoms and improve HRQL.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Head and neck cancer (HNC) and its treatment are associated with disruption to patient health-related quality of life (HRQL), often resulting in problems with speech, swallowing, eating and breathing, and disfigurement [1]. These factors have implications for patient psychological well-being [2]. The rates of depression and anxiety are higher in patients with HNC than any other cancer patient population [3], and preliminary evidence suggests that patients are susceptible to symptoms of post-traumatic stress disorder (PTSD) [4].
PTSD is a psychiatric disorder that can develop after experiencing a traumatic event, involving physical harm or the threat of physical harm. Symptoms of PTSD are grouped into four broad categories, including heightened arousal, intrusive thoughts (re-experiencing the traumatic event), avoidance, and negative changes in cognitions and mood [5]. These symptoms cause clinically significant distress and impairment in important domains of function [5]. A diagnosis of cancer is recognised as an event capable of eliciting PTSD by the American Psychiatric Association [6]. However, HNC may have greater potential to produce PTSD compared to other cancers [7] given that the disease is not only life threatening, but also associated with highly aversive treatments applied to the head and neck region, which can have an enduring impact on basic functions and appearance.
To date, only two studies have investigated HNC-related PTSD. The first included both HNC and lung cancer patients and found that 22 % met criteria for PTSD 6 months post-diagnosis [8]. The most recent study found that 12 % of patients with HNC met the criteria for PTSD between 4 and 16 weeks post-diagnosis [7]. The lower rate of PTSD in this study may be attributable to the shorter time frame.
These findings suggest that more studies are needed to determine the prevalence of PTSD in patients with HNC and the factors that may contribute to its development. A recent meta-analysis that investigated the prevalence of cancer-related PTSD also concluded that more research to identify factors contributing to PTSD vulnerability is needed [9]. One factor that has been associated with a range of outcomes in HNC patients is coping [10]. Coping has been defined as “constantly changing cognitive and behavioral efforts to manage specific external and/or internal demands that are appraised as taxing or exceeding the resources of the person” [11].
An important distinction in coping is between behaviours characterised by engagement and those characterised by disengagement [12]. Engagement-focused coping strategies include those aimed at actively dealing with a stressor (e.g. problem-focused coping, support seeking and emotion regulation), while disengagement-focused strategies are characterised by avoidance or escape from the stressor and/or its associated distressing emotions [13]. Examples of disengagement/avoidant coping strategies include denial, self-distraction, and alcohol and drug use.
Available research suggests that when coping with an HNC diagnosis, engagement-focused coping strategies are related to positive outcomes, including benefit finding [14, 15] and lower distress [16]. Conversely, the use of disengagement/avoidant coping strategies has been related to low HRQL, anxiety, and depression. There is more evidence to support the adverse impact of disengagement coping than there is to support the beneficial effects of engagement coping in patients with HNC [17], as has been found in other cancer groups [18]. Another coping strategy that has been related to poor psychological adjustment is self-blame. Behavioural self-blame can occur when individuals perceive that they could have done something differently to prevent or change their situation. This strategy may be particularly relevant to HNC patients, for whom cigarette smoking and alcohol use are established risk factors [19]. Indeed, self-blame has been correlated with lower HRQL [20] and continued smoking in this patient group [21]. Self-blame has also been found to predict depression 6–8 months post-treatment for HNC [17]. However, no prospective study has examined whether self-blame is associated with HNC patient HRQL or PTSD.
The objective of the present study was to investigate the influence of coping strategies at HNC diagnosis on PTSD symptoms and HRQL 6 months post-diagnosis. The focus was on coping with diagnosis in particular, given that it is a time of high distress [22] and few prospective studies have examined whether coping at this time point is related to future outcomes.
Methods
Participants
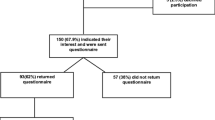
Participants were a consecutive sample of patients with HNC recruited from a head and neck outpatient clinic at a hospital in New Zealand, between February and October 2013. Patients were invited to take part in the study if they had a diagnosis of primary epithelial head and neck cancer (carcinoma in the pharynx, larynx, oral cavity, sinonasal cavity), or an advanced (metastatic) skin cancer in the head and neck region, within 3 weeks prior to attendance. Patients were excluded if they were unable to speak or read English, if they were to be treated with palliative intent, or if they had conditions that would interfere with participation (including cognitive impairment, physical disability, or psychiatric conditions).
Participant characteristics are presented in Table 1.
Ninety-one patients completed the questionnaires at diagnosis. Between diagnosis and 6 months follow-up, 15 patients died. Sixty-five of the 76 remaining participants returned follow-up questionnaires (response rate = 88 %). No systematic differences were observed between those who returned the questionnaires and those who did not, with respect to gender, ethnicity, marital status, cancer site, cancer stage, or treatment modality. Reasons for failure to return the follow-up questionnaire included: feeling distressed (n = 3), patients being non-contactable (n = 5), and issues with the mailing system (sent but not received; n = 3).
Power analysis
In a previous research, avoidance coping at diagnosis predicted depressive symptoms with a medium effect size (r = 0.34) [23]. G-power [24] indicated that 49 patients would be needed to detect a similar effect (setting power at 0.80 and α at 0.05).
Procedure
Informed consent was obtained from all individual participants included in the study. Participants were asked to complete a booklet of questionnaires (assessing demographics, coping, HRQL, and distress) after diagnosis at their multi-disciplinary clinic visit. They were mailed another questionnaire booklet 6 months later which contained the same assessments, as well as an assessment of PTSD symptoms. Information on medical and treatment characteristics was collected from patient medical records. Approval was obtained from the University Human Participants Ethics Committee and the relevant District Health Board Research Review Committee.
Measures
Functional Assessment Of Cancer Therapy-Head and Neck (FACT-H&N)
The FACT-H&N is a validated self-report instrument that assesses HRQL in patients with HNC, including physical, social, emotional, and functional well-being, as well as head and neck-related symptoms [25]. Each item is rated on a 0–4 scale. Scores on each subscale are summed to create a total HRQL score, with higher scores indicative of better quality of life. The reliability was satisfactory in this study (α = 0.78).
General Health Questionnaire (GHQ-12)
The GHQ-12 is a 12-item questionnaire to identify distress in both general and clinical populations [26]. Higher scores indicate higher distress, with scores greater than 15 indicating clinically significant distress symptoms. It is a valid measure for detecting depressive symptoms in patients with cancer [27] and has good internal consistency and test–retest reliability [26]. Cronbach’s alpha of the scale in this study was 0.91. Distress was assessed to control for scores at diagnosis when investigating the relationship between coping and subsequent PTSD.
Brief COPE
The Brief COPE is a valid and reliable self-report scale to assess different ways of coping [28]. Subscales include self-distraction (in this study, α = 0.50), active coping (α = 0.78), denial (α = 0.80), substance use (α = 0.96), emotional support (α = 0.84), instrumental support (α = 0.68), behavioural disengagement (α = 0.45), venting (α = 0.61), positive reframing (α = 0.70), planning (α = 0.73), humour (α = 0.81), acceptance (α = 0.62), religion (α = 0.93), and self-blame (α = 0.74). These reliabilities are consistent with other research in patients with HNC [14].
Post-Traumatic Stress Disorder Symptom Scale (PSS-SR)
The PTSD Symptom Scale Self-Report version (PSS-SR) assesses the presence and severity of PTSD symptoms according to DSM-III-R criteria [29]. Respondents are asked to answer each item in relation to a single identified traumatic event (in this case HNC). Scores can range from 0 to 51, and scores higher than 13 indicate PTSD. The PSS-SR has satisfactory internal consistency, high test–retest reliability, and good concurrent validity [29]. The scale had good reliability (α = 0.95). PTSD symptoms were not assessed at diagnosis, as symptoms need to be present for at least 4 weeks to detect PTSD caseness.
Analysis
Data were analysed using SPSS version 21. Independent samples t tests were used to investigate differences in PTSD and HRQL between patients with an early cancer stage (TI–TII) versus a later cancer stage (TIII–TIV), patients who had surgery versus radiotherapy, and patients who had single modality treatment versus combined modality treatment. Pearson product–moment correlations were used to examine relationships between baseline coping strategies and follow-up PTSD and HRQL. Following this, hierarchical multiple regression analyses were used to examine which coping strategies had the capacity to predict each outcome at 6 months follow-up. Cancer stage and baseline scores were entered in the first step, and coping strategies correlated with the outcome of interest at p < 0.01 or less were entered in step two (to limit the number of variables in each regression model). A two-sided p value less than 0.05 was considered to be statistically significant. Missing data were addressed using pairwise deletion.
Results
Descriptives
Twelve of 64 patients had scores indicative of PTSD at 6 months follow-up (19 %). Age was not significantly correlated with post-traumatic stress or HRQL at diagnosis or follow-up, and there was no significant difference between men and women. There were no significant differences in post-traumatic stress or HRQL (at diagnosis or follow-up) between patients who had surgery and those who had radiotherapy, and there were also no differences between those who had single modality treatment and those who had combined modality treatment. At diagnosis, patients with an early stage cancer (TI–TII) reported significantly higher HRQL (M = 123.67, SD = 18.23) than patients with a later stage cancer (TIII–TIV) (M = 114.27, SD = 21.01) [t(89) = 2.21, p = 0.029]. This difference in HRQL remained at 6 months (stage TI–TII M = 122.83, SD = 17.02; stage TIII–TIV M = 108.13, SD = 24.15; t(63) = 2.81, p = 0.007). There was no significant difference between patients with different stages of cancer with respect to post-traumatic stress. No significant difference was observed between patients with HNC and those with an advanced skin cancer in the head and neck region with respect to post-traumatic stress or HRQL.
Associations between coping, PTSD, and HRQL
Denial, substance use, behavioural disengagement, venting, and self-blame at diagnosis were significantly correlated with lower HRQL and higher post-traumatic stress at follow-up (see Table 2). Interestingly, the use of humour was also positively correlated with post-traumatic stress scores.
The inter-correlations between these coping strategies revealed some moderate associations. Denial was positively correlated with substance use (r = 0.350, p < 0.001) and self-blame (r = 0.236, p = 0.020). Substance use (r = 0.290, p = 0.004) and venting (r = 0.276, p = 0.006) were also positively correlated with self-blame. No other significant correlations were identified.
Regression analyses
The first regression model presented in Table 3 explained 53 % of the variance in patient’s post-traumatic stress [adjusted R 2 = 48 %; F(6, 55) = 10.371, p < 0.001]. Baseline distress scores explained 24 % of this variance, and coping strategies explained an additional 26 %. Denial, behavioural disengagement, and self-blame each made a unique statistically significant contribution to the model.
The second regression model presented in Table 3 explained 73 % of the variance in HRQL [adjusted R 2 = 71 %; F(7, 49) = 17.74, p < 0.001]. Baseline HRQL and cancer stage contributed 59 % of the variance, and the coping variables contributed an additional 14 %. Self-blame was the only coping strategy to make a unique statistically significant contribution to the model.
Discussion
This is the first prospective study to demonstrate that coping strategies at diagnosis predict the development of post-traumatic stress symptoms in patients with HNC and one of only few studies to show that coping strategies predict HRQL. Rates of post-traumatic stress were 19 % 6 months following diagnosis, which represents a sizeable proportion of patients and is consistent with the rates of 12 and 22 % as shown in previous research [7, 8]. PTSD symptoms at 6 months were predicted by self-blame, denial, and behavioural disengagement, even when controlling for general distress, suggesting that the results are not solely attributable to a general negative affect.
This research adds to previous literature regarding the effects of denial and avoidant coping in cancer. While some research has found denial to be related to lower levels of distress, other studies have demonstrated a link with lower HRQL [30]. Few longitudinal studies have been conducted into the relationship between coping and PTSD in cancer. However, research in other patient groups suggests that avoidant coping is linked to more severe PTSD [31]. This may be because avoidance often results in a paradoxical increase in intrusive thoughts about a stressor [32]. The correlation between the use of humour and more PTSD symptoms found in this study is curious, although consistent with other findings [33]. More research is required to see if this is a robust finding.
The use of self-blame as a coping strategy at diagnosis predicted PTSD symptoms and low HRQL, both of which are new findings in patients with HNC. Previously, a prospective relationship has only been shown between self-blame and depression [17]. However, self-blame has been associated with distress in patients with other cancer types [34], including lung, breast, and prostate cancer [35, 36]. The deleterious effects of self-blame on patient psychological well-being are likely attributable to negative self-evaluation and increased self-focused attention [37], each of which have been implicated as precursors for mood disorders [38], particularly depression.
These results have implications for the development of psychological interventions that aim to address the use of avoidant/disengagement coping and self-blame at diagnosis in patients with HNC. As part of this, it would be important to introduce routine screening following diagnosis using a brief coping questionnaire. This would enable identification of patients engaging in ineffective coping behaviours, who would most benefit from psychological support. Targeted psycho-educational interventions could then be delivered, involving one or more sessions with a psychologist or nurse specialist, in which patients could be encouraged to replace their coping strategies with more adaptive techniques, such as problem solving, relaxation, goal setting, communication, and the development of support networks.
If psychological interventions are successful at modifying patient coping behaviours, they may result in improved HRQL and fewer symptoms of PTSD following treatment. While interventions to improve coping have proven beneficial in patients with other types of cancer [39], there is an absence of methodologically sound studies testing psychological interventions for patients with HNC [40]. Efforts to reduce PTSD symptoms and increase HRQL could have a number of clinical benefits for this group, given well-documented associations linking PTSD and low HRQL to rehospitalisation, disease relapses, symptom intensity, and morbidity and mortality [41, 42].
There are several limitations to this study. First, to ensure that we captured patients’ subjective understanding of their HRQL, it may have been appropriate to also include a global quality of life scale. Second, a limitation of using the Brief COPE is that subscales can have low reliability [43]. Finally, PTSD symptoms were assessed at 6 months follow-up only, which prevented investigation of changes in PTSD over time.
In conclusion, this prospective study is the first to demonstrate a link between the use of avoidant coping strategies and self-blame at HNC diagnosis, and post-traumatic stress and low HRQL 6 months later. Future research could investigate whether identifying patients engaging in these strategies and providing coping-based psychological interventions can reduce the likelihood of post-traumatic stress and improve HRQL.
References
List MA, Bilir SP (2004) Functional outcomes in head and neck cancer. Semin Radiat Oncol 14:178–189
Haisfield-Wolfe M, McGuire DB, Soeken K, Geiger-Brown J, De Forge BR (2009) Prevalence and correlates of depression among patients with head and neck cancer: a systematic review of implications for research. Oncol Nurs Forum 36:E107–E125. doi:10.1188/09.ONF.E107-E125
Singer S, Krauß O, Keszte J et al (2012) Predictors of emotional distress in patients with head and neck cancer. Head Neck 34:180–187. doi:10.1002/hed.21702
Kangas M, Milross C, Taylor A, Bryant RA (2013) A pilot randomized controlled trial of a brief early intervention for reducing posttraumatic stress disorder, anxiety and depressive symptoms in newly diagnosed head and neck cancer patients. Psychooncology 22:1665–1673. doi:10.1002/pon.3208
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders (DSM-IV), text revised, 4th edn. American Psychiatric Association, Washington
American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders (DSM-IV), 4th edn. American Psychiatric Association, Washington
Posluszny DM, Dougall AL, Johnson JT et al (2015) Posttraumatic stress disorder symptoms in newly diagnosed patients with head and neck cancer and their partners. Head Neck 37:1282–1289. doi:10.1002/hed.23760
Kangas M, Henry JL, Bryant RA (2005) Predictors of posttraumatic stress disorder following cancer. Health Psychol 24:579–585
Abbey G, Thompson SB, Hickish T, Heathcote D (2015) A meta-analysis of prevalence rates and moderating factors for cancer-related post-traumatic stress disorder. Psychooncology 24:371–381. doi:10.1002/pon.3654
Howren MB, Christensen AJ, Karnell LH, Funk GF (2013) Psychological factors associated with head and neck cancer treatment and survivorship: evidence and opportunities for behavioral medicine. J Consult Clin Psychol 81:299–317. doi:10.1037/a0029940
Lazarus RS, Folkman S (1984) Stress, appraisal, and coping. Springer, New York
Carver CS, Connor-Smith J (2010) Personality and coping. Annu Rev Psychol 61:679–704. doi:10.1146/annurev.psych.093008.100352
Skinner EA, Edge J, Altman J, Sherwood H (2003) Searching for the structure of coping: a review and critique of category systems for classifying ways of coping. Psychol Bull 129:216–269
Llewellyn CD, Horney DJ, McGurk M, Weinman J, Herold J, Altman K, Smith HE (2013) Assessing the psychological predictors of benefit findings in patients with head and neck cancer. Psychooncology 22:97–105. doi:10.1002/pon.2065
Cavell S, Broadbent E, Donkin L, Gear K, Morton RP (2016) Observations of benefit finding in head and neck cancer patients. Eur Arch Otorhinolaryngol 273(2):479–485. doi:10.1007/s00405-015-3527-7
Karnell LH, Christensen AJ, Rosenthal EL, Magnuson JS, Funk GF (2007) Influence of social support on health-related quality of life outcomes in head and neck cancer. Head Neck 29:143–146
Llewellyn CD, McGurk M, Weinman J (2007) Illness and treatment beliefs in head and neck cancer: is Leventhal’s common sense model a useful framework for determining changes in outcomes over time? J Psychosom Res 63:17–26
Yang H, Brothers BM, Andersen BL (2008) Stress and quality of life in breast cancer recurrence: moderation or mediation of coping? Ann Behav Med 35:188–197. doi:10.1007/s12160-008-9016-0
Hashibe M, Brennan P, Benhamou S et al (2007) Alcohol drinking in never users of tobacco, cigarette smoking in never drinkers, and the risk of head and neck cancer: pooled analysis in the International Head and Neck Cancer Epidemiology Consortium. J Natl Cancer Inst 99:777–789
Scharloo M, Baatenburg de Jong RJ, Langeveld TP, van Velzen-Verkaik E, Doorn-op den Akker MM, Kaptein AA (2005) Quality of life and illness perceptions in patients with recently diagnosed head and neck cancer. Head Neck 27:857–863
Christensen AJ, Moran PJ, Ehlers SL, Raichle K, Karnell L, Funk G (1999) Smoking and drinking behavior in patients with head and neck cancer: effects of behavioral self-blame and perceived control. J Behav Med 22:407–418
Neilson K, Pollard A, Boonzaier A et al (2013) A longitudinal study of distress (depression and anxiety) up to 18 months after radiotherapy for head and neck cancer. Psychooncology 22:1843–1848. doi:10.1002/pon.3228
de Leeuw J, de Graeff A, Ros WJ, Blijham GH, Hordijk GJ, Winnubst JA (2000) Prediction of depressive symptomatology after treatment of head and neck cancer: the influence of pre-treatment physical and depressive symptoms, coping, and social support. Head Neck 22:799–807
Faul F, Erdfelder E, Lang AG, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Cella DF, Tulsky DS, Gray G et al (1993) The Functional Assessment of Cancer Therapy scale: development and validation of the general measure. J Clin Oncol 11:570–579
Goldberg D (1992) General Health Questionnaire (GHQ-12). NFER-Nelson, Windsor
Reuter K, Harter M (2001) Screening for mental disorders in cancer patients—discriminant validity of HADS and GHQ-12 assessed by standardized clinical interview. Int J Methods Psychiatr Res 10:86–96. doi:10.1002/mpr.103
Carver CS (1997) You want to measure coping but your protocol’s too long: consider the Brief COPE. Int J Behav Med 4:92–100
Foa EB, Riggs DS, Dancu CV, Rothbaum BO (2006) Reliability and validity of a brief instrument for assessing post-traumatic stress disorder. J Trauma Stress 6:12–25. doi:10.1002/jts.2490060405
Vos MS, de Haes JC (2007) Denial in cancer patients, an explorative review. Psychooncology 16:12–25
Otis JD, Keane TM, Kerns RD (2003) An examination of the relationship between chronic pain and post-traumatic stress disorder. J Rehabil Res Dev 40:397–405
Najmi S, Wegner DM (2008) Thought suppression and psychopathology. In: Elliot A (ed) Handbook of approach and avoidance motivation. Erlbaum, Mahwah, pp 447–459
Aarstad AK, Aarstad HJ, Olofsson J (2008) Personality and choice of coping predict quality of life in head and neck cancer patients during follow-up. Acta Oncol 47:879–890. doi:10.1080/02841860701798858
Malcarne VL, Compas BE, Epping-Jordan JE, Howell DC (1995) Cognitive factors in adjustment to cancer: attributions of self-blame and perceptions of control. J Behav Med 18:401–417
Houldin AD, Jacobsen B, Lowery BJ (1996) Self-blame and adjustment to breast cancer. Oncol Nurs Forum 23:75–79
Else-Quest NM, LoConte NK, Schiller JH, Hyde JS (2009) Perceived stigma, self-blame, and adjustment among lung, breast and prostate cancer patients. Psychol Health 24:949–964. doi:10.1080/08870440802074664
Frisch MB (1998) Quality of life therapy and assessment in health care. Clin Psychol Sci Pract 5:19–40. doi:10.1111/j.1468-2850.1998.tb00132.x
Smith TW, Ingram RE, Roth DL (1985) Self-focused attention and depression: self-evaluation, affect, and life stress. Motiv Emotion 9:381–389
Gaston-Johansson F, Fall-Dickson J, Nanda J, Ohly K, Stillman S, Krumm S, Kennedy JM (2000) The effectiveness of the comprehensive coping strategy program on clinical outcomes in breast cancer autologous bone marrow transplantation. Cancer Nurs 23:277–285
Semple C, Parahoo K, Norman A, McCaughan E, Humphris G, Mills M (2013) Psychosocial interventions for patients with head and neck cancer. Cochrane Database Syst Rev. doi:10.1002/14651858.CD009441.pub2
Cavalcanti-Ribeiro P, Andrade-Nascimento M, Morais-de-Jesus M et al (2012) Post-traumatic stress disorder as a comorbidity: impact on disease outcomes. Expert Rev Neurother 12:1023–1037
Mehanna HM, Morton R (2006) Deterioration in quality-of-life (10-year) survivors of head and neck cancer. Clin Otolaryngol 31:204–211
Hankins M, Foster C, Hulbert-Williams N, Breckons M (2013) The Brief COPE in cancer research. Psychooncology 22:23
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicting interests to disclose.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Richardson, A.E., Morton, R.P. & Broadbent, E. Coping strategies predict post-traumatic stress in patients with head and neck cancer. Eur Arch Otorhinolaryngol 273, 3385–3391 (2016). https://doi.org/10.1007/s00405-016-3960-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-016-3960-2