Abstract
Nasopharyngeal carcinoma (NPC) patients may have persistent or recurrent neck masses following radiotherapy. The clinicopathological features for these two subgroups are not fully characterized, however. In this study, we reviewed our experiences in patients with neck masses underwent either local excision or neck dissection. Between 1990 and 2004, 37 NPC patients who had persistent (n = 18) or recurrent (n = 19) neck masses following radiotherapy were enrolled. The clinical and pathological parameters were measured. Squamous cell carcinoma was found in the resected specimens of 72.2% of the patients (13/18) with persistent neck mass and 89.5% of the analogs with the recurrent form (17/19). Extra-nodal tumor extension was noted in 53.8% (7/13) of persistent neck malignancies and 64.7% (11/17) of the recurrent variants. At the time of the neck surgery, individuals with persistent neck malignancies had fewer concomitant distant failures (23.1%, 3/13) as compared to their counterparts with the recurrent form (58.8%, 10/17) (P = 0.07). At the end of the follow-up, however, the rates of local and/or distant failures were both high for the persistent (92.3%, 12/13) and recurrent groups (76.5%, 13/17). In conclusion, following radiotherapy for NPC, both persistent and recurrent neck masses were associated with a high rate of squamous cell carcinoma. Although patients with persistent neck malignancies have significantly fewer additional sites of failures at the time of their neck surgeries, they tend to develop local and/or distant recurrences later during follow-up.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nasopharyngeal carcinoma (NPC) is highly radiosensitive, with radiotherapy alone or in combination with chemotherapy being recognized as the mainstay treatment [4]. Despite therapeutic or prophylactic irradiation of the neck, however, persistent or recurrent neck masses may appear in some of the NPC patients during follow-up. Reviewing the literature, the incidence of neck failures post radiotherapy is reportedly around 6–18% [3, 5, 10]. Neck dissection is recommended as the treatment of choice for cervical metastasis in NPC after radiotherapy [19, 22]. In clinical practice, however, it may be difficult to define the nature of these persistent/recurrent neck masses before surgery by image study or aspiration cytology because of post-irradiation changes, such as induration and fibrosis, in cervical soft tissues [19, 22]. Furthermore, the disease nature and clinical outcome for NPC patients with persistent or recurrent neck masses after radiotherapy are not well characterized. To provide better insight into this clinical problem, therefore, in this study we retrospectively analyzed our experience treating NPC patients presenting with persistent or recurrent neck masses after radiotherapy.
Materials and methods
From January 1990 to June 2004, 37 NPC patients suffering either persistent or recurrent neck mass after radiotherapy underwent neck surgery in the National Cheng Kung University Hospital and were enrolled in this study. The primary nasopharyngeal site was evaluated with either indirect nasopharyngoscopy or flexible endoscopic examination and supposed to be clinically free of disease by experienced otolaryngologists before surgery. Patients with concomitant local disease, proved by nasopharyngeal biopsy, were excluded from this study. All the patients received irradiation of the nasopharynx and bilateral neck, with a mean dose of 72.0 ± 8.6 Gy delivered to the primary tumor and 67.7 ± 5.6 Gy to the neck. After review of all the medical records, the tumor staging was retrospectively reclassified according to the criteria proposed by the American Joint Committee on Cancer in 2002 [8]. Basic data for these patients are presented in Table 1.
The 37 patients were classified into persistent or recurrent groups according to the clinical manifestations of their neck masses. The former, who still had clinically detectable neck node(s) 6 months after completing the radiotherapy, was defined as persistent group. The latter group, or the recurrent group, was defined by neck masses that appeared after radiotherapy or that had resolved completely within 6 months but reappeared during subsequent follow-ups. The potential sites of distant metastasis were evaluated in all cases at the time of neck surgery. Distant metastasis was confirmed either by biopsy or serial image studies. The choice of surgical treatment for the neck disease between neck dissection and local excision depended on clinical condition and/or patient request. For patients who already had distant metastasis, neck surgery was performed with curative intent (a) when medical oncologists requested a tissue proof of the neck mass; or (b) when the patient was a candidate for combined neck surgery and metastasectomy. Neck dissections were generally performed to include neck level I to V, with sparing of spinal accessory nerve if feasible. All resected specimens were routinely sent for pathological examination. Presence of extra-nodal tumor extension into neck soft tissue was especially specified in the pathological reports.
The persistent and recurrent groups were compared for clinical features of the neck disease, operative and pathological findings, and evidence of local and/or systemic failures. Statistical Product and Service Solutions software (Version 10.0; SPSS Inc, Chicago, IL, USA) was used for analysis of the results. The Wilcoxon rank sum test, Fisher’s exact test and Chi-square test were used for comparison of the variables between subgroups. A probability level of less than 0.05 (P < 0.05) was considered to indicate a significant difference.
Results
Demographic data and clinical parameters for the 37 patients are presented in Table 1. According to our grouping criteria, 18 and 19 of the patients had persistent and recurrent neck masses, respectively. The age range for the 32 male and 5 female patients at surgery was 26–71 years (mean 47.2 ± 11.5). No statistically significant between-group difference was noted comparing age, sex and initial tumor stage. Detectable neck masses were unilateral in all subjects. On local examination, most were solitary neck masses in both the persistent (55.6%, 10/18) and recurrent (63.1%, 12/19) groups; however, location differed between the two. In the persistent group, the commonest location for the neck mass was level II (n = 12; 52.2%). By contrast, recurrent-group neck distribution was wider (levels I–V).
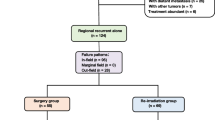
Surgical procedures and pathological results for the 37 NPC patients are summarized in Fig. 1. Twelve subjects (66.7%; n = 18) from the persistent group received neck dissection, including five cases with modified type I and seven cases with radical neck dissection. Twelve patients (63.2%; n = 19) from the recurrent analog received neck dissection, including four cases with modified type I and eight cases with radical neck dissection. Malignant cells were detected in 13 (72.2%; n = 18) and 17 (89.5%; n = 19) of resected specimens from the persistent and recurrent groups, respectively. The histological classification of the tumor included nonkeratinizing undifferentiated squamous cell carcinoma (World Health Organization [WHO]-III [pathological classification of NPC]) in 29 patients, nonkeratinizing differentiated squamous cell carcinoma (WHO-II) in 6 patients and keratinizing squamous cell carcinoma (WHO-I) in 2 patients. Thus, a high overall malignancy rate of 81.1% (30/37) was noted for this sample of patients presenting with persistent/recurrent neck mass after radiotherapy. Neck malignancy was confirmed from neck dissection for six of the persistent group with solitary neck mass by local examination. Pathological examination revealed that three (50%; n = 6) had multiple nodal involvements. In the recurrent group, neck dissection confirmed malignancy in seven of the solitary neck-mass cases, while six (87.5%, n = 7) also had multiple nodal involvements from pathology. For the malignant specimens in the persistent group, extra-nodal tumor extension was noted in seven cases (53.8%; n = 13); while 11 (64.7%; n = 17) of the malignant specimens in the recurrence group also had extra-nodal tumor extension, with an overall extra-nodal extension rate of 60% (18/30).
At the time of neck surgery, only 3 (23.1%) of the 13 patients with persistent neck disease suffered from concomitant distant metastasis. No local recurrence was noted. By contrast, distant metastasis were demonstrated in 10 of the 17 patients (58.8%) with recurrent neck disease (P = 0.07; Table 2). However, nine more patients with persistent neck disease developed local and/or distant failures during follow-up. By contrast, in the recurrent group only three more patients developed local and/or distant failures (Table 2). After a follow-up from neck surgery of 26.3 months (standard deviation 19.1 months), 12 (92.3%; n = 13) and 13 (76.5%; n = 17) of the persistent and recurrent group, respectively, suffered local recurrence and/or distant metastasis (Table 2). In addition, four subjects from the persistent group and two from the recurrent analog suffered from loco-regional recurrence after the neck surgery.
Eight (26.7%) of the 30 patients confirmed to have neck malignancies remained alive at the end of this study, with their follow-up periods ranging from 2.1 to 8.5 years (median = 4.5 years). The 5-year disease-free and overall survival for all patients with persistent or recurrent neck malignancies was 39.2 and 41.6%, respectively. The 5-year disease-free/overall survivals were 31.1/33.4 and 44.2/50.5% for the persistent and recurrent groups, respectively (Fig. 2a,b). There was no significant difference between the two groups in term of the 5-year disease-free or the overall survival rates. Among the patients who received different types of neck procedures, we did not observe an overall survival benefit in those patients who received the more extensive surgeries, such as modified type I or radical neck dissection (Fig. 3).
Discussion
Persistent or recurrent neck masses may appear in some NPC patients during follow-up. It may be difficult to define the nature of these neck masses, however, due to post-irradiation changes in neck soft tissues [19, 22]. Although it is reported that ultrasound-guided fine needle aspiration cytology may be helpful in differentiating malignant lymphadenopathy from benign inflammatory nodes [2, 13], in clinical practice, the confirmation of cervical malignancy may still require some form of surgical interventions. In a previous report, cervical malignancy was confirmed by radical neck dissection for 88.2% (45/51) of NPC patients who presented with persistent/recurrent neck mass after radiotherapy without evidence of local recurrence before neck surgeries [19]. Our results also show that 81.1% of NPC patients with the presenting symptom of persistent/recurrent neck mass do have cervical malignancies.
Cervical malignancies in NPC patients following radiation are reported to be mainly located at levels II, III and V [3, 12, 21]. In this study, during clinical examination, it is not surprising to note that persistent neck masses in NPC patients post radiotherapy appear to be located mainly at level II (52.2%; n = 12), which is the commonest site of cervical metastasis characteristic of NPC. However, in the recurrent group, neck masses were distributed more widely at all neck areas (levels I–V), suggesting that the regular lymphatic drainage route may be altered following irradiation. It is noted that two (33.3%) of the six recurrent level I (submental) neck masses were pathologically confirmed to be tumor-bearing nodes. This demonstrates that the risk of nodal recurrence in the submental triangle following radiation is not negligible. It has also been documented that 28% (7/25) of submental nodal enlargements in a sample of NPC patients post radiotherapy harbored malignancies, as confirmed by ultrasound-guided fine-needle aspiration cytology [2].
In most of our patients, a solitary neck mass was discovered during preoperative examination for both the persistent and recurrent groups (55.5% and 63.1%, respectively). However, multiple nodal involvement was noted in three (50%; n = 6) and six (87.5%) of the persistent and recurrent group who underwent neck dissection, respectively, implying that many of the malignant lymph nodes that recur after radiotherapy may be difficult to detect clinically [12, 22]. In addition, extra-nodal tumor extension was noted in 60% (18/30) of all malignant specimens. In a previous study which included 43 NPC patients who received radical neck dissection following radiotherapy for control of persistent/recurrent neck disease, multiple nodal involvement was confirmed in 75.9% (22/29) of single nodal cases. Extracapsular spread was found in 70% (28/40) of all malignant specimens [20]. Thus, we also advocate neck dissection as the surgical procedure of choice for control of neck disease in NPC patients following radiotherapy.
It is interesting to note that, at the time of surgery, the patients with persistent neck disease had significantly fewer distant failures (23.1%, 3/13) compared to analogs with the recurrent variant (58.8%, 10/17; P < 0.05). Only three cases with concomitant distant metastasis were noted in the persistent group. However, during follow-up, local and/or distant failures developed in nine more persistent cases (Table 2). By contrast, only three of the recurrent group developed local and/or distant failures during follow-up. Thus, if the 13 cases with concomitant distant failures at the time of neck surgery were excluded, 12 (71%) of the remaining 17 NPC patients who were confirmed to have cervical metastasis (n = 30) developed local and/or distant failures following neck surgery. This failure rate (71%) is somewhat higher than the range reported in the NPC literature (29–55%) after salvage neck dissection [13, 21, 22]. A plausible explanation for this discrepancy might be that, in this study, a greater proportion of the patients without concomitant local and/or distant failures at the time of neck surgery (59%, 10/17) belong to the nodal persistence group as compared to the sample population in the other reports (12.9–31.4%) [19, 20, 21, 22]. And, as we observed, patients with persistent neck diseases tend to manifest local and/or distant failures later than analogs with the recurrent form. Thus, meticulous investigation to detect local and/or distant failures is both mandatory at the time before and following salvage neck surgery. Nasopharyngeal swab [18] and quantitative real-time PCR [16] may be helpful for detection of early local recurrence and assist in the identification of those at high risk for distant metastasis. Adjuvant therapies should also be considered postoperatively in these patients with high risk of local and/or distant failures, such as low dose metronomic chemotherapy [23].
Previous literature had reported that NPC patients with distant metastasis had poor survival after the diagnosis of metastasis, with the median survival ranged from 5 to 11 months [1, 15]. Thus, when NPC patients with neck failures were identified concomitantly with distant metastasis at our institution, chemotherapy was generally recommended for most patients. However, it is shown recently that a significant proportion of individuals with synchronous early stage locoregional failures can achieve long-term disease control and survival after aggressive surgical salvage with or without combined radiotherapy [6]. For NPC patients with distant metastasis confined to the lung, long-term survival is also possible [11]. In line with this, three of our patients with single lung metastasis also underwent lobectomy for control of distant metastasis. Although these three patients died from uncontrolled loco-regional recurrence during follow-up, none of them manifested evidence of lung disease after lobectomy. The survival time after the salvage surgery ranged from 11.5 to 25.4 months. Our finding also supports that single lung metastasis in NPC patients can be controlled with appropriate surgery. Thus, in selective cases with distant metastasis those are considered appropriate candidates for metastasectomy [7, 17], combined surgical approaches for loco-regional relapse, such as nasopharyngectomy [9, 14, 24] and/or neck dissection, should be considered.
Conclusion
Appearance of both persistent and recurrent neck masses in NPC patients after radiotherapy is associated with a high rate of malignancy and concomitant local and/or distant failures. Multiple nodal involvement and extra-nodal tumor extension are frequently observed in patients where neck malignancy has been confirmed. Although patients with persistent neck disease have significantly fewer concomitant local and/or distant failures at the time of neck surgery, additional sites of recurrence tend to manifest later in the follow-up periods.
References
Ahmad A, Stefani S (1986) Distant metastases of nasopharyngeal carcinoma: a study of 256 male patients. J Surg Oncol 33:194–197
Ahuja AT, Leung SF, Teo P, Ying M, King W, Metreweli C (1999) Submental metastasis from nasopharyngeal carcinoma. Clin Radiol 54:25–28
Bedwinek JM, Perez CA, Keys DJ (1980) Analysis of failure after definitive irradiation for epidermoid carcinoma of the nasopharynx. Cancer 45:2725–2729
Chan AT, Teo PM, Huang DP (2004) Pathogenesis and treatment of nasopharyngeal carcinoma. Semin Oncol 31:794–801
Chen WZ, Zhou DL, Luo KS (1989) Long-term observation after radiotherapy for nasopharyngeal carcinoma (NPC). Int J Radiat Oncol Biol Phys 16:311–314
Chua DT, Wei WI, Sham JS, Cheng AC, Au G (2003) Treatment outcome for synchronous locoregional failures of nasopharyngeal carcinoma. Head Neck 25:585–594
Davidson RS, Nwogu CE, Brentjens MJ, Anderson TM (2001) The surgical management of pulmonary metastasis: current concepts. Surg Oncol 10:35–42
Greene FL, Page DL, Fleming ID, Fritz A, Balch CM, Haller DG, Morrow M (2002) AJCC cancer staging manual, 6th edn. Springer, New York
Hao SP, Tsang NM, Chang CN (2002) Salvage surgery for recurrent nasopharyngeal carcinoma. Arch Otolaryngol Head Neck Surg 128:63–67
Hsu MM, Tu SM (1983) Nasopharyngeal carcinoma in Taiwan: clinical manifestations and results of therapy. Cancer 52:362–368
Hui EP, Leung SF, Au JS, Zee B, Tung S, Chua D, Sze WM, Law CK, Leung TW, Chan AT (2004) Lung metastasis alone in nasopharyngeal carcinoma: a relatively favorable prognostic group. Cancer 101:300–306
Khoo MLC, Soo KC, Lim DTH, Fong KW, Goh CHK, Sethi DS (1999) The pattern of nodal recurrence following definitive radiotherapy for nasopharyngeal carcinoma. Aust N Z J Surg 69:354–356
King WWK, Teo PML, Li AKC (1992) Patterns of failure after radical neck dissection for recurrent nasopharyngeal carcinoma. Am J Surg 164:599–602
King WW, Ku PK, Mok CO, Teo PM (2000) Nasopharyngectomy in the treatment of recurrent nasopharyngeal carcinoma: a twelve-year experience. Head Neck 22:215–222
Leung SF, Teo PM, Shiu WW, Tsao SY, Leung TW (1991) Clinical features and management of distant metastases of nasopharyngeal carcinoma. J Otolaryngol 20:27–29
Lin JC, Wang WY, Chen KY, Wei YH, Liang WM, Jan JS, Jiang RS (2004) Quantification of plasma Epstein-Barr virus DNA in patients with advanced nasopharyngeal carcinoma. N Engl J Med 350:2461–2470
Mineo TC, Ambrogi V, Tonini G, Bollero P, Roselli M, Mineo D, Nofroni I (2003) Long-term results after resection of simultaneous and sequential lung and liver metastases from colorectal carcinoma. J Am Coll Surg 197:386–391
Tsang NM, Chuang CC, Tseng CK, Hao SP, Kuo TT, Lin CY, Pai PC (2003) Presence of the latent membrane protein 1 gene in nasopharyngeal swabs from patients with mucosal recurrent nasopharyngeal carcinoma. Cancer 98:2385–2392
Wei WI, Lam KH, Ho CM, Sham JST, Lau SK (1990) Efficacy of radical neck dissection for the control of cervical metastasis after radiotherapy for nasopharyngeal carcinoma. Am J Surg 160:439–442
Wei WI, Ho CM, Wong MP, Ng WF, Lau SK, Lam KH (1992) Pathological basis of surgery in the management of postradiotherapy cervical metastasis in nasopharyngeal carcinoma. Arch Otolaryngol Head Neck Surg 118:923–929
Wei WI, Ho WK, Cheng AC, Wu X, Li GK, Nicholls J, Yuen PW, Sham JS (2001) Management of extensive cervical nodal metastasis in nasopharyngeal carcinoma after radiotherapy. Arch Otolaryngol Head Neck Surg 127:1457–1462
Yen KL, Hsu LP, Sheen TS, Chang YL, Hsu MM (1997) Salvage neck dissection for cervical recurrence of nasopharyngeal carcinoma. Arch Otolaryngol Head Neck Surg 123:725–729
Young SD, Whissell M, Noble JC, Cano PO, Lopez PG, Germond CJ (2006) Phase II clinical trial results involving treatment with low-dose daily oral cyclophosphamide, weekly vinblastine, and rofecoxib in patients with advanced solid tumors. Clin Cancer Res 12:3092–3098
Yu KH, Leung SF, Tung SY, Zee B, Chua DT, Sze WM, Law SC, Kam MK, Leung TW, Sham JS, Lee AW, Au JS, Hui EP, Sze WK, Cheng AC, Yau TK, Ngan RK, Wong FC, Au GK, Chan AT; Hong Kong Nasopharyngeal Carcinoma Study Group (2005) Survival outcome of patients with nasopharyngeal carcinoma with first local failure: a study by the Hong Kong Nasopharyngeal Carcinoma Study Group. Head Neck 27:397–405
Acknowledgment
This work was supported by grants from the National Science Council, Taiwan (NSC 93-2314-B-006-040 to JRH and NSC 93-2320-B006-077 to YTJ).
Conflict of interest statement
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, CY., Tsai, ST., Jin, YT. et al. Outcome of surgical management of persistent or recurrent neck mass in patients with nasopharyngeal carcinoma after radiotherapy. Eur Arch Otorhinolaryngol 265 (Suppl 1), 69–74 (2008). https://doi.org/10.1007/s00405-007-0552-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-007-0552-1