Abstract
Purpose
The purpose of the study is to compare the newborns weight in singleton term birth following transfer of thawed blastocysts–frozen on either day 5 or day 6 after in vitro fertilization.
Method
The retrospective study included 1444 frozen–thawed blastocyst transfer (FBT) cycles resulting in live singleton births between Jan 2013 and Dec 2016. The main outcomes measured were absolute birth weight, z-score adjusted for gestational age and gender, and incidence of large-for-gestational-age (LGA) newborns. Generalized linear model (GLM) and logistic regression were used in multivariate analyses.
Result(s)
Both the absolute birth weight (3416.49 ± 404.74 vs 3349.22 ± 416.17) and the z-score (0.6 ± 0.93 vs 0.41 ± 0.93) were significantly higher on day 6 FBT in comparison with day 5 FBT. The incidence of LGA newborns was also increased on day 6 FBT (22.8 vs 14.7%, P = 0.006). Adjusted for maternal age, BMI, PCOS diagnosis, present of vanishing twin, and embryo quality, the odds ratio (95% confidence interval) for LGA on day 6 FBT comparing with day 5 FBT was 1.76 (1.18–2.64).
Conclusion(s)
Day 6 FBT is associated with increased birth weight and contributes to the incidence of LGA newborns in FBT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Registry data showed that the use of embryo transfer at the blastocyst stage is increasing along with selective embryo transfer (SET) in recent years [1]. In comparison with the initial use of cleavage-stage embryo transfer, blastocyst transfer not only favors the implantation rate and live birth rate per transfer [2], but also allows for the control of multiple pregnancies in combination with SET. However, only approximately half of the embryos subjected to extended culture reach blastocyst stage [3], and some of these embryos are slower to progress and showed a delayed blastulation. Practically, both day 5 and day 6 blastocysts are considered for transfer, but the latter is often associated with poor clinical outcomes in fresh cycles. However, evidence in frozen–thawed cycles suggested that the pregnancy rates of cryopreserved blastocysts are comparable regardless of developmental speed before cryopreservation [4, 5]. Lower pregnancy rates after day 6 transfers may be due to the asynchrony between embryo development and the stimulated endometrium [6]. Some authors suggested an extended culture and vitrification strategy for delayed blastocyst on day 5, to achieve a better synchronization between embryo development and endometrial receptivity [7]. These studies may encourage the transfer of day 6 blastocyst in frozen–thawed cycles.
Recent studies listed a series of factors that may affect the neonatal outcomes following IVF, including ovarian stimulation [8], frozen–thawed transfer [9], culture medium [10], and an extended period of culture [11]. These data highlighted the role of epigenetic reprogramming occurred during pre-implantation development and implied that the effects of unfavorable factors may extend beyond implantation. In patients undergoing day 6 blastocyst transfer, the delayed blastulation may reflect the intrinsic flaw of embryo quality or a suboptimal pre-implantation environment. Therefore, birth resulted from delayed blastocyst may have a different neonatal outcome measuring by birth weight and likelihoods of small for gestational age (LGA) or large for gestational age (SGA), even though the pregnancy rates on day 6 and day 5 transfer were comparable in frozen–thawed cycles. However, effects of day 6 transfer on neonatal outcomes are less clear.
The aim of the present study was to explore the association between day 6 embryo transfers and the birth weight in full-term singletons resulting from frozen–thawed transfers.
Materials and methods
Study subjects
Institutional Review Board approval for this retrospective study was obtained from the Ethical Committee of the Medical College Xiamen University. Informed consent was not necessary, because the research was based on non-identifiable records as approved by the ethics committee.
All patients who underwent FBT in the affiliated Chenggong Hospital of Xiamen University in the period between January 2013 and December 2016 were accessed for potential inclusion. Due to the transfer policies in our clinic, day 6 blastocysts were only transferred in frozen–thawed cycles and no mixed transferred of day 6 and day 5 embryos occurred. Only cycles resulting in singleton live birth were included. Exclusion criteria were preterm birth (gestational week < 37), patients reporting maternal gestational hypertension or diabetes, neonatal abnormalities, and unknown perinatal outcomes due to incomplete record.
In stimulation cycles, all patients were treated with agonist or antagonist protocol with the use of FSH or hMG as previously described [12]. The hCG was administrated when ultrasonography revealed at least one follicle measuring ≥ 18 mm in mean diameter. Oocyte retrieval was scheduled for 34–36 h after hCG administration under transvaginal ultrasound guidance.
Laboratory protocol
Oocytes were inseminated using either conventional IVF or ICSI. In IVF cycles, cumulus–oocyte complexes were transferred to fertilization culture medium (K-SIFM, Cook) and inseminated with 1–1.5 × 105 spermatozoa for 4 h. Oocytes for ICSI were denuded 2 h after ovum pickup, and sperm injection was performed 4 h after retrieval. All oocytes were transferred to embryonic development medium (K-SICM, Cook) after insemination and the pronuclei were identified 17–18 h later. On day 3, the embryos were assigned quality grades and embryos for blastocyst culture were transferred to blastocyst medium (K-SIBM, Cook). The Gardner scale [13] was used to evaluate the quality of blastocysts. Top quality embryos for transfer were defined as expanded blastocysts with good inner cell mass and good trophectoderm (≥ 3AA), and embryos with poor morphological score (≤ 4CC) or low expansion grade (grades 1–2) were not considered for cryopreservation or transfer. On day 5, expanded blastocysts (grades 3–4) with acceptable morphological score were cryopreserved for subsequent transfer and slow development blastocysts were allowed for extended culture until day 6. Day 6 embryos were cryopreserved if they met the aforementioned morphological criteria.
For vitrification, blastocoelic volume was reduced. Blastocysts were equilibrated for 4 min in 10 ml equilibration solution containing 7.5% dimethyl sulfoxide and 7.5% ethylene glycol, and then washed in vitrification solution (15% dimethyl sulfoxide, 15% ethylene glycol, 10 mg/mL Ficoll-70, and 0.6 M sucrose) for 30–40 s before being immersed in liquid nitrogen. While the blastocysts thawed, blastocysts were directly immersed in no. 1 thawing solution (TS) containing 1 M sucrose at 37 °C for 1 min and then incubated in no. 2 TS at room temperature for 3 min. After being washed in no. 3 and no. 4 TS at room temperature for 5 min each, the blastocysts were cultured for 1 h before FBT. Survival of thawed embryos was accessed under an inverted microscope. The blastocysts that appeared to be severely damaged or showed a completely degenerated cellular content were considered not to have survived. The re-expansion of the blastocoele cavity was evaluated immediately before transfer.
Endometrium preparation
Either natural cycles (NC) or hormone replacement treatment (HRT) cycles were used for endometrial preparation. In NC cycles, growth of follicles was monitored under transvaginal ultrasonography from cycle day 9 to 11. Since the diameter of leading follicle reached 1.4 mm, LH and estradiol measurements were scheduled for every 3 days. Intramuscular progesterone injections (40 mg/day) were started at the day of ovulation and FBT was performed on the 5th day after ovulation. In HRT cycles, endometrial priming with daily oral estradiol valerate (5–6 mg) began on cycle day 1 and lasted for 14–15 days. Progesterone injection (60–100 mg) was administrated as soon as the endometrial thickness reached 7–8 mm. FBT was scheduled after 6 day of progesterone therapy. Embryo transfer was performed with a Guardia Access Embryo Transfer catheter (K-JETS-7019-SIVF, Cook, IN, USA) under transabdominal ultrasound guidance. Luteal support continued until 10 weeks of pregnancy.
Statistical analysis
To calculate the proportion of small for gestational age (SGA) and large for gestational age (LGA) and z-score adjusted for gestational age and gender, we used the latest published international birth weight Ref. [14]. z-score was defined as the weight of the individual child minus the media weight of a reference population of children born at the same gestational age and of the same gender divided by the standard deviation from the same reference population. LGA was defined as newborns with a birth weight > 90th percentile for that gestational age and gender. SGA was defined as newborns with a birth weight < 10th percentile for that gestational age and gender.
For continuous variables, normality plots and a Shapiro–Wilk test were used for normality testing, and a T test or Mann–Whitney U test was used for comparison. Dichotomous variables were analyzed by a Chi-square test or Fisher’s exact test, as appropriate. The non-parametric Spearman test was used for bivariate correlations. Generalized linear model (GLM) was used to analyze the effect on z-scores, and logistic regression analyses were used to access the effect on the incidence of SGA/LGA. The same set of covariates was used for all multivariate analyses. The maternal age, BMI, parity (0 vs ≥ 1), PCOS diagnosis [15], and present of vanishing twin [16] were selected as covariates due to previous knowledge. The multivariate analyses also included parameters that differ significantly between groups. Paternal age, order of transfer, duration of infertility, and insemination protocols were included. Although the number of embryos cryopreserved and the blastulation rates were also significantly different between groups, these parameters were logically associated with day 6 transfer. Adding them into the model only slightly reduced the odds ratio, but resulted in a poor fit (Hosmer and Lemeshow test, P = 0.057). Therefore, we did not consider them as independent factors in our final model. Finally, the quality of embryos transferred (at least on top quality embryo transferred) was considered, as patients receiving day 6 transfer might bias toward receiving transfer with lower scored embryos.
All calculations were performed with SPSS (version 19; IBM).
Result
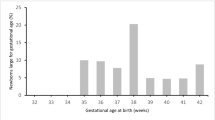
The present study included 1444 FBT cycles resulting term singleton live birth. The mean maternal age of the population was 30.58 ± 3.82 years. The average birth weight of the newborns was 3357.2 ± 411.7 g. Comparing to the reference population, 3.7% (n = 54) of the cases were identified as SGA and 15.7% (n = 226) were identified as LGA.
Among the FBT cycles, 1273 cycles were transferred with day 5 embryos (D5 group), whereas 171 cycles were transferred with day 6 embryos (D6 group). The comparisons in patient characteristics and cycle outcomes between two groups are shown in Table 1. Patient characteristics were comparable between groups, except that the cycles transferred with day 6 embryos had a higher paternal age and a longer duration of infertility. In stimulation cycles, more cycles were inseminated with ICSI in D6 group than that in D5 group (P < 0.001). A lower blastulation rate was also observed in D6 group. Correspondingly, the number of blastocysts cryopreserved for future transfer was lower in D6 group. In transfer cycles, order of transfer was the only parameter differed significantly between groups. Data indicated that patients in D6 group had more previous attempts of embryo transfer.
Significant differences were detected in both absolute birth weight and z-score adjusted for gestational age and gender (P < 0.05). The difference in z-score [95% confidence interval (CI)] was 0.19 (0.043, 0.339). The incidence of LGA was also significantly increased in D6 group (P < 0.001).
In a GLM model adjusted for aforementioned covariates, the associated between day 6 embryo transfer and increased z-score remained significant (B = 0.189, P = 0.012). The adjusted population marginal means (95% CI) for z-score in D5 group and D6 group were 0.35 (0.24, 0.46) and 0.54 (0.37, 0.70), respectively.
Controlling for the same set of covariates included in GLM model, the logistic regression analyses showed that the adjusted odds ratio (95% CI) for LGA comparing day 6 transfer with day 5 transfer was 1.76 (1.18, 2.64). The P value for the Hosmer and Lemeshow test was 0.762 (Table 2).
Discussion
Many studies have compared the development competence of delayed day 6 blastocysts with that of day 5 blastocysts in terms of pregnancy rates and live birth rates following IVF [5, 17]. However, whether the babies derived from delayed blastocysts differ to those derived from day 5 blastocysts in terms of neonatal outcomes is less clear. In the present study, we found an increase in both absolute birth weight and z-score adjusted for gender and gestational age among singleton newborns derived from day 6 FBT in comparison with those derived from day 5 FBT. After adjustment for covariates, day 6 FBT was associated with approximately 1.7 fold increases in the incidence of LGA. The results suggested that the effect of delayed development in the blastocyst stage may extend beyond implantation.
Our conclusion conflicted with two previous retrospective studies performed in smaller cohorts. In 71 deliveries resulted from FBT with embryos vitrificated at day 5, 6 and 7, Hiraoka et al. reported no significant difference in terms of gestational age, preterm delivery rate, and birth weight [18]. However, the results were confounded by the presence of twins and only 26 cases of day 6 transfer were included. Wang et al. compared the effects of day 5 and day 6 FBT on neonatal outcomes in single birth and twin birth, respectively, in a population including 515 cases and only found that the birth weight at day 6 group was prone to be heavier regardless the birth group [19]. Both studies are potentially under power and neither of them reported the outcomes with adjustment for gestational age and gender. Taking our study into account, data regarding the birth weight of full-term singletons born after transfer of day 6 blastocysts remained limited.
Although the absolute difference in birth weight was relatively small when comparing births after day 6 FBT with those after day 5 FBT, a significant increase in the incidence of LGA may be of concerns regarding long-term health effects. Cumulative evidences suggest that infants being born LGA are prone to develop metabolic syndrome and cardiovascular disease, such as diabetes mellitus and hypertensive status [20, 21]. A recent genome-wide DNA methylation study comparing children born LGA and born appropriate for gestational age suggested that LGA is associated with altered DNA methylation pattern in genes related to cardiovascular disease [22], suggesting links among epigenetic modification during early development, LGA at delivery, and origin of chronic disease in adulthood.
The conventional morphological assessment widely accepted as the gold standard in embryo selection stands on the hypothesis that morphological events of embryos and their timing reflect the intrinsic quality of embryos. Morphokinetic studies in recent years further support this idea by linking morphological phenotypes with molecular programs in the early development [23] and embryo ploidy [24]. A recent paper demonstrated that the global methylation levels of high-quality blastocysts are similar, while the methylation levels of low-quality blastocysts are divergent [25], suggesting a link between DNA methylation and embryo viability. Nevertheless, whether morphological observations had any implication on birth outcomes is unclear. The timing of blastulation has been associated with embryo competence along with many other morphological/morphokinetic parameters [26]. Our results may lead to the questions: whether the poor scored embryos leading to live births with different neonatal outcomes? And whether the embryo selection affects adulthood long-term health outcome?
According to systemic reviews and meta-analyses [11, 27], extending in vitro culture to blastocyst stage resulted in higher risks of LGA in comparison with cleavage-stage culture. In addition, birth following blastocyst transfer also had a lower incidence of SGA in comparison with those from spontaneous conception [28]. Taken together, evidence supported the idea that extended culture may trigger genetic and epigenetic changes in trophodermal cells and contribute to increased birth weight. Therefore, the effects of day 6 FBT may be confounded by the extra duration of in vitro culture. In animals, both delayed blastocyst formation and an extra day culture could increase the incidence of apoptosis, decrease the ICM:TE cell ratio, and influence the gene expression and diameter of blastocysts [29]. However, pig blastocysts formed at day 7 appeared to have a higher frequency of apoptosis than blastocysts formed at day 6 with one extra day of culture and one extra day of culture on blastocysts formed at day 7 further increased the prevalence to low-quality blastocyst development [29]. This animal study implied that the effects of delayed blastulation and extended in vitro culture on blastocysts could be cumulative.
For patients whose embryos only reach blastocyst stage on day 6, Wirleitner et al. compared possible strategies for the transfer of slow blastulation and/or delayed cavitation embryos [7]. The authors recommended that the best strategy for non-top quality or delayed embryos was prolonged culture before vitrification on day 6, as the improvement in live birth rates was even more pronounced. However, day 5 FBT yielded a comparable live birth rates than day 6 FBT (21.4 vs 28.3%) and avoided an extra day of in vitro culture [7]. As delayed blastocyst with extended culture duration may be associated with neonatal LGA, further study regarding the effects of prolonged culture on the neonatal outcome of delayed blastocyst may allow one balance the advantage of day 6 FBT against the potential risk of long-term health effect.
The present study was limited by the retrospective design. Although the patients reporting gestational hypertension or diabetes were excluded, the study design did not allow for controlling important variables which affect neonatal outcomes during pregnancy. As all patients with delayed blastocyst received day 6 FBT in the present study, whether alternative transfer strategies affect the birth weight outcomes in these patients is still no known.
In conclusion, our results indicated that patients with delayed blastocysts transferred on day 6 delivered babies of higher risks of LGA. The finding is potentially of concerns regarding the long-term health following IVF. The results also suggested heterogeneity among patients undergoing blastocyst transfer in terms of neonatal outcomes. In previous studies comparing the neonatal outcomes of blastocyst transfer with cleavage-stage transfer [2, 22], the patients undergoing day 5 or day 6 blastocyst transfer were often subjected to the same study group. Conversely, some author found that “more LGA deliveries were observed after blastocyst compared with cleavage-stage transfer in frozen cycles and no differences between the two groups in fresh cycles” [30]. The differences in birth weight between patients undergoing day 5 and day 6 blastocysts implied that the including patients with delayed blastocysts might contribute to the wide heterogeneity of the studies.
References
Kushnir VA, Barad DH, Albertini DF, Darmon SK, Gleicher N (2017) Systematic review of worldwide trends in assisted reproductive technology 2004–2013. Reprod Biol Endocrinol. https://doi.org/10.1186/s12958-016-0225-2
Glujovsky D, Farquhar C, Retamar AMQ, Sedo CRA, Blake D (2016) Cleavage stage versus blastocyst stage embryo transfer in assisted reproductive technology. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd002118.pub5
Pfeifer S, Fritz M, Goldberg J, McClure RD, Lobo R, Thomas M, Pisarska M, Widra E, Schattman G, Licht M, Sandlow J, Collins J, Cedars M, Rosen M, Racowsky C, Vernon M, Davis O, Odem R, Barnhart K, Gracia C, Thornton K, Catherino W, Dumesic D, Rebar R, La Barbera A, Med ASR, Med ASR, Technology SAR (2013) Blastocyst culture and transfer in clinical-assisted reproduction: a committee opinion. Fertil Steril 99(3):667–672. https://doi.org/10.1016/j.fertnstert.2013.01.087
Sunkara SK, Siozos A, Bolton VN, Khalaf Y, Braude PR, El-Toukhy T (2010) The influence of delayed blastocyst formation on the outcome of frozen–thawed blastocyst transfer: a systematic review and meta-analysis. Hum Reprod 25(8):1906–1915. https://doi.org/10.1093/humrep/deq143
El-Toukhy T, Wharf E, Walavalkar R, Singh A, Bolton V, Khalaf Y, Braude P (2011) Delayed blastocyst development does not influence the outcome of frozen–thawed transfer cycles. BJOG Int J Obstet Gynaecol 118(13):1551–1556. https://doi.org/10.1111/j.1471-0528.2011.03101.x
Poulsen V, Ingerslev HJ, Kirkegaard K (2017) Elective embryo transfers on day 6 reduce implantation compared with transfers on day 5. Hum Reprod 32(6):1238–1243. https://doi.org/10.1093/humrep/dex059
Wirleitner B, Schuff M, Stecher A, Murtinger M, Vanderzwalmen P (2016) Pregnancy and birth outcomes following fresh or vitrified embryo transfer according to blastocyst morphology and expansion stage, and culturing strategy for delayed development. Hum Reprod 31(8):1685–1695. https://doi.org/10.1093/humrep/dew127
Pereira N, Elias RT, Christos PJ, Petrini AC, Hancock K, Lekovich JP, Rosenwaks Z (2017) Supraphysiologic estradiol is an independent predictor of low birth weight in full-term singletons born after fresh embryo transfer. Hum Reprod 32(7):1410–1417. https://doi.org/10.1093/humrep/dex095
Kaartinen N, Kananen K, Huhtala H, Keranen S, Tinkanen H (2016) The freezing method of cleavage stage embryos has no impact on the weight of the newborns. J Assist Reprod Genet 33(3):393–399. https://doi.org/10.1007/s10815-015-0642-8
Kleijkers SHM, Mantikou E, Slappendel E, Consten D, van Echten-Arends J, Wetzels AM, van Wely M, Smits LJM, van Montfoort APA, Repping S, Dumoulin JCM, Mastenbroek S (2016) Influence of embryo culture medium (G5 and HTF) on pregnancy and perinatal outcome after IVF: a multicenter RCT. Hum Reprod 31(10):2219–2230. https://doi.org/10.1093/humrep/dew156
Wang XL, Du MZ, Guan YC, Wang BJ, Zhang JW, Liu ZH (2017) Comparative neonatal outcomes in singleton births from blastocyst transfers or cleavage-stage embryo transfers: a systematic review and meta-analysis. Reprod Biol Endocrinol. https://doi.org/10.1186/s12958-017-0255-4
Cai JL, Liu LL, Zhang JW, Qiu HL, Jiang XM, Li P, Sha AG, Ren JZ (2017) Low body mass index compromises live birth rate in fresh transfer in vitro fertilization cycles: a retrospective study in a Chinese population. Fertil Steril 107(2):422. https://doi.org/10.1016/j.fertnstert.2016.10.029
Gardner DK, Schoolcraft WB (1999) Culture and transfer of human blastocysts. Curr Opin Obstet Gynecol 11(3):307–311
Villar J, Ismail LC, Victora CG, Ohuma EO, Bertino E, Altman DG, Lambert A, Papageorghiou AT, Carvalho M, Jaffer YA, Gravett MG, Purwar M, Frederick IO, Noble AJ, Pang RY, Barros FC, Chumlea C, Bhutta ZA, Kennedy SH, Growth IFN (2014) International standards for newborn weight, length, and head circumference by gestational age and sex: the Newborn Cross-Sectional Study of the INTERGROWTH-21st Project. Lancet 384(9946):857–868
Sterling L, Liu J, Okun N, Sakhuja A, Sierra S, Greenblatt E (2016) Pregnancy outcomes in women with polycystic ovary syndrome undergoing in vitro fertilization. Fertil Steril 105(3):791. https://doi.org/10.1016/j.fertnstert.2015.11.019
Magnus MC, Ghaderi S, Morken NH, Magnus P, Romundstad LB, Skjrven R, Wilcox AJ, Haberg SE (2017) Vanishing twin syndrome among ART singletons and pregnancy outcomes. Hum Reprod 32(11):2298–2304. https://doi.org/10.1093/humrep/dex277
Sunkara SK, Siozos A, Bolton V, Khalaf Y, Braude P, El-Toukhy T (2010) The influence of delayed blastocyst formation on the outcome of frozen–thawed blastocyst transfer: a systematic review and meta-analysis. Hum Reprod 25:I205–I205
Hiraoka K, Hiraoka K, Miyazaki M, Fukunaga E, Horiuchi T, Kusuda T, Okano S, Kinutani M, Kinutani K (2010) Perinatal outcomes following transfer of human blastocysts vitrified at day 5, 6 and 7. Hum Reprod 25:I60–I60
Wang X, Zhen JR, Sun ZY, Yu Q, Deng CY, Zhou YZ, Wang HB, He FF (2016) Effects of fifth day (D5) or sixth day (D6) frozen–thawed blastocysts on neonatal outcomes. Zygote 24(5):684–691. https://doi.org/10.1017/S0967199415000696
Pocobelli G, Dublin S, Enquobahrie DA, Mueller BA (2016) Birth weight and birth weight for gestational age in relation to risk of hospitalization with primary hypertension in children and young adults. Matern Child Health J 20(7):1415–1423. https://doi.org/10.1007/s10995-016-1939-7
Belbasis L, Savvidou MD, Kanu C, Evangelou E, Tzoulaki I (2016) Birth weight in relation to health and disease in later life: an umbrella review of systematic reviews and meta-analyses. BMC Med. https://doi.org/10.1186/s12916-016-0692-5
Lin XH, Wu DD, Gao L, Zhang JY, Pan HT, Wang H, Li C, Zhang P, Guo MX, Wu YT, Tan YJ, Jin L, Xiang YQ, Li JX, Sheng JZ, Huang HF (2016) Altered DNA methylation in neonates born large-for-gestational-age is associated with cardiometabolic risk in children. Oncotarget 7(52):86511–86521. https://doi.org/10.18632/oncotarget.13442
Wong CC, Loewke KE, Bossert NL, Behr B, De Jonge CJ, Baer TM, Pera RAR (2010) Non-invasive imaging of human embryos before embryonic genome activation predicts development to the blastocyst stage. Nat Biotechnol 28(10):1115–U1199. https://doi.org/10.1038/nbt.1686
Zaninovic N, Irani M, Meseguer M (2017) Assessment of embryo morphology and developmental dynamics by time-lapse microscopy: is there a relation to implantation and ploidy? Fertil Steril 108(5):722–729. https://doi.org/10.1016/j.fertnstert.2017.10.002
Li GQ, Yu Y, Fan Y, Li CR, Xu XC, Duan JL, Li R, Kang XJ, Ma X, Chen XP, Ke YW, Yan J, Lian Y, Liu P, Zhao Y, Zhao HC, Chen YY, Sun XF, Liu JQ, Qiao J, Liu J (2017) Genome wide abnormal DNA methylome of human blastocyst in assisted reproductive technology. J Genet Genom 44(10):475–481. https://doi.org/10.1016/j.jgg.2017.09.001
Desai N, Ploskonka S, Goodman L, Attaran M, Goldberg JM, Austin C, Falcone T (2016) Delayed blastulation, multinucleation, and expansion grade are independently associated with live-birth rates in frozen blastocyst transfer cycles. Fertil Steril 106(6):1370–1378. https://doi.org/10.1016/j.fertnstert.2016.07.1095
Martins WP, Nastri CO, Rienzi L, van der Poel SZ, Gracia CR, Racowsky C (2016) Obstetrical and perinatal outcomes following blastocyst transfer compared to cleavage transfer: a systematic review and meta-analysis. Hum Reprod 31(11):2561–2569. https://doi.org/10.1093/humrep/dew244
Ernstad EG, Bergh C, Khatibi A, Kallen KBM, Westlander G, Nilsson S, Wennerholm UB (2016) Neonatal and maternal outcome after blastocyst transfer: a population-based registry study. Am J Obstet Gynecol. https://doi.org/10.1016/j.ajog.2015.12.040
Lin T, Lee JE, Ogani RK, Kim SY, Cho ES, Jeong YD, Bael JJ, Jin DI (2017) Delayed blastocyst formation or an extra day culture increases apoptosis in pig blastocysts. Anim Reprod Sci 185:128–139. https://doi.org/10.1016/j.anireprosci.2017.08.012
Alviggi C, Conforti A, Carbone IF, Borrelli R, De Placido G, Guerriero S (2017) Does cryopreservation influence perinatal outcome after blastocyst-stage vs cleavage-stage transfer—a systematic review and meta-analysis. Ultrasound Obstet Gynecol. https://doi.org/10.1002/uog.18942
Acknowledgements
The authors would like to thank all the staff, nurses, and physicians at the Reproductive Medicine Center for their support in generating this manuscript. This work was supported by the National Natural Science Foundation of China (Grant number 81302454); the Science and Technology Project funding in Xiamen City (Grant number 3502Z20144039); and the Natural Science Foundation of Fujian Province (Grant number 2016D025).
Author information
Authors and Affiliations
Contributions
JC: protocol/project development, data collection or management, manuscript writing/editing. LL: protocol/project development, data collection or management, manuscript writing/editing. YX: protocol/project development, data collection or management, manuscript writing/editing. ZL: protocol/project development, data collection or management, manuscript writing/editing. XJ: data collection or management, manuscript writing/editing. PL: data collection or management, manuscript writing/editing. AS: data collection or management, manuscript writing/editing. JR: data analysis, manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Institutional Review Board approval for this retrospective study was obtained from the Ethical Committee of the Medical College Xiamen University. This paper does not contain any studies with human participants performed by any of the authors. As a retrospective research, the study was in accordance with the 1964 Helsinki Declaration and its later amendments ethical standards.
Informed consent
For this type of study formal consent is not required. Informed consents were not required by the Institutional Ethical Review Board.
Rights and permissions
About this article
Cite this article
Cai, J., Liu, L., Xu, Y. et al. Day 6 blastocyst is associated with increased birth weight in full-term singleton newborns after frozen–thawed transfer. Arch Gynecol Obstet 298, 397–403 (2018). https://doi.org/10.1007/s00404-018-4808-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-018-4808-3