Abstract
Purpose
We studied the efficacy of using pre-cesarean delivery (CD) temporary occlusion of internal iliac arteries with balloon catheters in case of placenta previa–accreta in terms of maternal and neonatal outcomes and to test accuracy of ultrasound (US) and magnetic resonance imaging (MRI) for prenatal diagnosis.
Methods
From March 2014 to January 2018, women with an US and/or MRI diagnosis of placenta previa–accreta and a planned delivery were enrolled and divided into two groups: balloon catheterization group (women treated with preoperative catheters and CD) and control group (women candidates to elective CD).
Results
37 patients were enrolled: 16 in balloon catheterization group and 21 in control group. Significant differences were detected in estimated blood loss. Prophylactic balloon catheterization could reduce intraoperative red blood cell transfusion. The incidence of hysterectomy was lower in balloon group. No statistical difference was found for neonatal outcomes. Both US and MRI have showed to be useful and complementary to diagnose placenta previa–accreta.
Conclusions
Temporal, perioperative, and prophylactic positioning of balloon vascular catheters is an effective method for managing severe hemorrhage caused by placenta previa–accreta as it reduced intraoperative blood loss, lessened perioperative hemostatic measures and intraoperative red cell transfusions, and reduced hysterectomies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Abnormally invasive placenta includes three pregnancy complications concerning the invasion of the placental villi into myometrium (placenta accrete and increta) or uterine serosa (placenta percreta) and, rarely, involves invasion of the surrounding organs. Abnormally invasive placenta may lead to major maternal and neonatal morbidity and mortality, representing a cause of obstetric hemorrhage [1, 2].
Incidence rate ranges between 1.7 and 90 per 10,000 [3,4,5], with an increase related to the rise in cesarean delivery (CD), which is a well-established risk factor [3, 4, 6]. However, abnormally invasive placenta affects up to 38% of primiparous women, suggesting that other important etiologic factors may play a role in the process [2, 7]. In fact, consistent with previous studies, advanced maternal age [3, 4, 6, 7], placenta previa [3, 4, 6, 8], hypertension [9], and female fetal sex [10] were significantly associated with abnormally invasive placenta.
Current prenatal diagnosis rests on subjective interpretation of “typical” sonographic findings or signs with two-dimensional (2D) gray-scale and color Doppler imaging [11].
The published literature is not univocal due to several problems in the definition, terminology, and diagnosis of this problem [12]. Therefore, to better define different imaging markers, consensus statements, elaborated by experts, have been published with the goal to provide standardized requirements for an ultrasound scan to diagnose placenta accreta disorders [13, 14].
Magnetic resonance imaging (MRI), although widely employed, has yet to demonstrate clearly a significant clinical application [15]. Therefore, MRI is up to now only recommended as an adjunct to ultrasound imaging by many professional bodies throughout the world including the Royal College of Obstetricians and Gynaecologists (RCOG) in the UK [16]. Irrespective of the imaging modality used, prenatal diagnosis of placenta disorders remains crucial [17] to plan the best case-management strategy to minimize the risks associated with clinical and, especially, surgical treatment, and to reduce maternal and perinatal morbidity.
Latest evidence of literature suggests that the management of women with placenta disorders by multidisciplinary teams in centers of excellence decreases maternal morbidity and mortality when compared with standard obstetric care [18,19,20,21]. Therefore, an adequate multidisciplinary team, including, in addition to the obstetrician gynecologist, an anesthesiologist, a radiologist, a hematologist, an urologist, a vascular surgeon, and a neonatologist, is the key to reduce complications.
Conventional management of this kind of abnormal placentation was cesarean hysterectomy with the placenta left in situ [22]. However, when there is a desire to preserve the uterus and fertility, alternative to hysterectomy is needed. Up to now, attempts to avoid hysterectomy include reducing intraoperative hemorrhage such as uterine compression sutures, intrauterine balloon tamponade, pelvic artery ligation, and spiral suturing of the lower uterine segment.
Since the first case of clinical use of aortic balloon catheter was reported in 1954 in Korea [23], the intravascular balloon catheter technique has been widely used to manage uncontrolled hemorrhage [24]. Recently, obstetrics has partnered with interventional radiology to use artery embolization and balloon catheter occlusion, which are appealing for their reversible nature and ability to reduce intraoperative blood loss. Data of literature report that the efficacy of prophylactic balloon catheterization (BC) of different target arteries is still debated [25,26,27], but BC of the internal iliac artery is less likely to induce complications such as limb and pelvic organ ischemia compared to aortic or common iliac artery BC [28].
In the present study, we evaluate the benefit of pre-CD balloon catheters in case of placenta accreta in terms of maternal and neonatal outcomes. Primary outcomes included estimated blood loss (EBL), duration of surgery, surgery- and catheterization-related complications, transfused blood product units, incidence of hysterectomy, number of intensive care unit (ICU) and neonatal intensive care unit (NICU) admissions, and Apgar scores at 1 and 5 min after birth. The accuracy of ultrasound (US) and MRI to diagnose placental accretism, with intraoperative and histopathological finding intraoperative, was analyzed as secondary outcomes.
Materials and methods
Between March 2014 and January 2018, all women with US and/or MRI diagnosis of placenta previa–accreta and a planned delivery were recruited. Those with a bleeding disorder or who underwent emergency CD or delivered before 32 week’s gestation were excluded. All the women in this study were fully informed of the benefits and complications of prophylactic internal iliac artery balloon catheterization by their doctors, and assigned to balloon catheterization group (cases) or control groups based on their willingness. Irrespective of treatment approach, signed consents were obtained. Because participants and doctors were aware of the assignment, the study was not double-blind so the data recorder and analyst were blinded to treatment groups to avoid bias.
The pregnancy-related characteristics considered for the study were: type of pregnancy (single or multiple), gestational age at CD, pregnancy complications (i.e., diabetes and preeclampsia), fertility treatments, loss of blood during pregnancy, placental localization, and modalities used to diagnose placenta accreta.
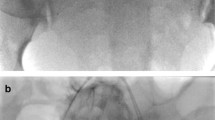
About 1 h before the scheduled CD, the balloon group underwent prophylactic internal iliac artery balloon catheterization with interventional radiology preoperatively, following the introduction, according to Seldinger’s standard technique, of two 6-Fr balloon catheters transfemoral bilaterally up to iliac bifurcation with position a “kissing balloon”.
These maneuvers were performed under local anesthesia and fluoroscopy’s guide to verify the correct balloon positioning, with time ranging between 5″ and 35″ with 65–72 kV and 0.5–0.7 mA/s. Fetal cardiac monitoring was performed immediately before and after the entire procedure.
After catheterization, CD was performed [29], during which balloons were inflated with normal saline as the uterine incision was made. After delivery, the placenta was delivered spontaneously or by cord traction. Once hemostasis was satisfied, the balloons were deflated and the uterus was sutured. Catheter removal was done after surgery completion by the same radiologist. Controls were treated the same way except for endovascular catheterization.
At least, two units of intraoperative of packed red blood cells for transfusion were required for all patients. In both groups, patients were administered uterotonic agents, such as oxytocin or carboprost trometamol, and hemostatic suturing and/or internal iliac artery (IIA) ligation were performed, if necessary. The indication for hysterectomy was uncontrolled bleeding despite the aforementioned surgical and medical interventions. In case of continuous not massive bleeding, the patient underwent to uterine arterial embolization (UAE). Statistical analyses were conducted using the Biostat statistical program. The estimated sample size for the study time duration was between 40 and 260 based on the published incidence rates of 1 in 2500 and 1 in 553 [30, 31]. To compare patients’ characteristics and maternal and fetal outcomes, we used T test and the χ2 test. P value of < 0.05 was considered statistically significant.
Results
From March 2014 to January 2018, 37 patients were enrolled. Sixteen cases underwent to prophylactic internal iliac artery balloon catheterization and CD (balloon group), and the remaining 21 cases underwent conventional CD (control group). Demographic and obstetrical characteristics of women from two groups presented no statistical differences (Table 1). Table 2 shows intraoperative data. Significant differences were observed in estimated blood loss (EBL) (P = 0.03), rate of hysterectomies, intraoperatively transfused RBC (P = 0.02), and the number of women transfused with any blood product (P = 0.02).
Among the two groups, no statistically significant differences were noted concerning postoperative recovery days (mean 4.5 ± 1.2 versus 4.1 ± 0.8 in balloon group and control group, respectively), recoveries in intensive care unit (ICU) (18% in balloon catheterization group and 19% in control group), and surgical complications. Neonatal outcomes were also not significantly different (Table 3).
Concerning accuracy of preoperative imaging in diagnosis of placenta accreta, ultrasound examination was performed on all 37 patients, while MRI was integrated only in 20 of these cases. As shown in Table 4, US was able to diagnose or suspect placenta accreta in 4/14 cases confirmed in operating room and/or histopathological examination with a sensitivity of 38% and a specificity of 75% (false positive in 4/23 cases). Concerning MRI, this technique was able to diagnose or suspect placental accretism in 6/20 cases with a sensitivity of 30% and a specificity of 79% (false positive in 4/12 cases). US and MRI were consistent with the diagnosis in 62% of cases.
Discussion
Prophylactic balloon (PB) catheterization is controversial and the literature reports different efficacies. A retrospective study of balloon catheter application for the abdominal aorta had postoperative complications 4.4% of the time, including arterial thrombosis and femoral nerve ischemic injury [27]. A Japanese study supported that BC for the common iliac artery was more effective than for the internal iliac artery due to the rich collateral uterine blood supply [28]. However, Mok et al. [32] stated that prophylactic internal iliac artery balloon catheterization could reduce intraoperative blood loss and improve the operating field due to decreased pulse pressure distal to the occlusion site.
Fan et al. [22] revealed that prophylactic internal iliac artery balloon catheterization can not only significantly reduce intraoperative blood loss for women with placenta accreta, especially with an anterior or anteroposterior placenta, but also lessen intraoperative RBC transfusion without serious adverse maternal or neonatal outcomes.
Recently, of 385 studies identified by Shahin et al., 69 (1811 patients, mean age 32.9 years, range 23–39 years) were included in a review and meta-analysis [33]. Mean gestational age at delivery was 35.1 weeks (range 27–38 weeks). Of 1395 patients who underwent endovascular intervention, 587 (42%) had placenta accreta, 254 (18%) placenta increta, and 313 (22%) placenta percreta. Prophylactic balloon occlusion of the internal iliac arteries (PBOIIA) was performed in 470 patients (33.6%), of the abdominal aorta (PBOAA) in 460 patients (33%), of the uterine artery (PBOUA) in 181 patients (13%), and of the common iliac arteries (PBOCIA) in 21 patients (1.5%). Primary embolization of the UA was performed in 246 patients (18%), of the pelvic collateral arteries in 12 patients (0.9%) and of the anterior division of the IIA in 5 patients (0.3%). Follow-up ranged from 0.5 to 42 months. Endovascular intervention was associated with less blood loss than no endovascular intervention (P < 0.001) with the lowest blood loss volume in patients who underwent PBOAA (P < 0.001). PBOAA was associated with a lower rate of hysterectomy (P = 0.030). Endovascular intervention did not result in increases in operative time or hospital stay. The study concludes that endovascular intervention is effective in controlling hemorrhage in abnormal placentation deliveries. PBOAA was associated with a lower rate of hysterectomy and less blood loss than other modalities.
In our study, we have reported several advantages related to the prophylactic positioning of endovascular catheters in the treatment of placenta accreta.
In particular, a statistically significant difference was found in EBL, percentage of hysterectomy, intraoperative transfusions in favor of balloon group. In addition, this technique resulted safe; in fact, no complications related to the use of endovascular catheters have been reported. Even with respect to the neonatal outcomes and the risk of fetal exposure to radiation, there are wide safety margins. In fact, the positioning of catheters at aortic bifurcation level, required fluoroscopy to a maximum time of 35 s, so the resulting fetal absorption dose would be about 0.7 mGy. In the literature, the risk of fetal malformation is significantly increased for doses higher than 0.15 Gy [34], while the risk of infantile cancer only differs by 0.05% among exposed and non-radiolabeled children [35].
The strengths of our study are several:
-
the reversibility of the adopted technique;
-
the chance of using the catheter, left in situ, in most cases for at least 12 h for checking post-partum bleeding;
-
the choice of performing both the radiological and the surgical procedures in the same operating room with the entire dedicated multidisciplinary team and a consequent reduction in operating times.
Another important key point is the prenatal diagnosis, essential for planning the optimal management strategy. Both ultrasonography and magnetic resonance imaging have proved useful and complementary in diagnosing placental placenta. An examination has not been better than the other, according to the literature’s data.
Based on our results, we propose the use of MRI not as a second level investigation in those cases suspected or inconclusive at US. In fact, it is important to perform both exams in patients with placenta previa and also in women with two previous CD, due to their high risk of placental abnormalities. The MRI also can properly evaluate the topography of the placenta and possible invasion of the surrounding organs. We should, however, highlight several weaknesses in the present study. First, a limited number of patients were recruited. In addition, the analysis is limited by including one center, but it is strong due to having the same multidisciplinary team to manage all women under the same management protocol. Ideally, this reduced potential operator-dependent bias.
Conclusion
In summary, based on our results and the reported latest evidence of literature, temporal, perioperative, and prophylactic positioning of balloon vascular catheters is a valid treatment for controlling and reducing blood loss and the need for transfusions in patients with placenta accreta. Even if, prospective randomized trials are needed to standardize the procedure and to draw accurate conclusions, concerning the feasibility of this procedure in clinical practice.
References
Oyelese Y, Smulian JC (2006) Placenta previa, placenta accreta, and vasa previa. Obstet Gynecol 107:927–941
Baldwin HJ, Patterson JA, Nippita TA, Torvaldsen S, Ibiebele I, Simpson J et al (2017) Maternal and neonatal outcomes following abnormally invasive placenta: a population-based record linkage study. Acta Obstet Gynecol Scand 96:1373–1381
Fitzpatrick KE, Sellers S, Spark P, Kurinczuk JJ, Brocklehurst P, Knight M (2012) Incidence and risk factors for placenta accreta/increta/percreta in the UK: a national case-control study. PLoS One 7:e52893
Miller DA, Chollet JA, Goodwin TM (1997) Clinical risk factors for placenta previa–placenta accreta. Am J Obstet Gynecol 177:210–214
Gielchinsky Y, Mankuta D, Rojansky N, Laufer N, Gielchinsky I, Ezra Y (2004) Perinatal outcome of pregnancies complicated by placenta accreta. Obstet Gynecol 104:527–530
Wu S, Kocherginsky M, Hibbard JU (2005) Abnormal placentation: twenty-year analysis. Am J Obstet Gynecol 192:1458–1461
Thurn L, Lindqvist P, Jakobsson M, Colmorn L, Klungsoyr K, Bjarnadóttir R et al (2015) Abnormally invasive placenta—prevalence, risk factors and antenatal suspicion: results from a large population-based pregnancy cohort study in the Nordic countries. BJOG 123:1348–1355
Hung TH, Shau WY, Hsieh CC, Chiu TH, Hsu JJ, Hsieh TT (1999) Risk factors for placenta accreta. Obstet Gynecol 93:545–550
Usta IM, Hobeika EM, Abu Musa AA, Gabriel GE, Nassar AH (2005) Placenta previa-accreta: risk factors and complications. Am J Obstet Gynecol 193:1045–1049
Khong T, Healy D, McCloud P (1991) Pregnancies complicated by abnormally adherent placenta and sex ratio at birth. BMJ 302:625–626
D’Antonio F, Iacovella C, Bhide A (2013) Prenatal identification of invasive placentation using ultrasound: systematic review and meta-analysis. Ultrasound Obstet Gynecol 42:509–517
Bhide A, Sebire N, Abuhamad A, Acharya G, Silver R (2017) Morbidly adherent placenta: the need for standardization. Ultrasound Obstet Gynecol 49:559–563
Alfirevic Z, Tang AW, Collins SL, Robson SC, Palacios-Jaraquemada J, Ad hoc International AIP Expert Group (2016) Pro forma for ultrasound reporting in suspected abnormally invasive placenta (AIP): an international consensus. Ultrasound Obstet Gynecol 47:276–278
Collins SL, Ashcroft A, Braun T et al (2016) Proposal for standardized ultrasound descriptors of abnormally invasive placenta (AIP). Ultrasound Obstet Gynecol 47:271–275
Committee Publications (2010) Society for maternal-fetal medicine, Belfort MA. Placenta accreta. Am J Obstet Gynecol 203:430–439
Royal College of Obstetricians and Gynecologists (2011) Placenta praevia, placenta praevia accreta and vasa praevia: diagnosis and management. In: Green Top Guideline No 27. RCOG, London
Jauniaux E, Bhide A, Kennedy A, Woodward P, Hubinont C, Collins S, Duncombe G, Klaritsch P, Chantraine F, Kingdom J, Grønbeck L, Rull K, Nigatu B, Tikkanen M, Sentilhes L, Asatiani T, Leung W, AIhaidari T, Brennan D, Kondoh E, Yang J, Seoud M, Jegasothy R, y Sosa SE, Jacod B, D’Antonio F, Shah N, Bomba-Opon D, Ayres-de-Campos D, Jeremic K, Kok TL, Soma-Pillay P, Mandić NT, Lindqvist P, Arnadottir TB, Hoesli I, Jaisamrarn U, Al Mulla A, Robson S, Cortez R (2018) FIGO consensus guidelines on placenta accreta spectrum disorders: Prenatal diagnosis and screening. Int J Gynecol Obstet 140:274–280. https://doi.org/10.1002/ijgo.12408
Eller AG, Bennett MA, Sharshiner M et al (2011) Maternal morbidity in cases of placenta accreta managed by a multidisciplinary care team compared with standard obstetric care. Obstet Gynecol 117:331–337
Chantraine F, Braun T, Gonser M, Henrich W, Tutschek B (2013) Prenatal diagnosis of abnormally invasive placenta reduces maternal peripartum hemorrhage and morbidity. Acta Obstet Gynecol Scand 92:439–444
Silver RM, Fox KA, Barton JR et al (2015) Center of excellence for placenta accreta. Am J Obstet Gynecol 212:561–568
Shamshirsaz AA, Fox KA, Salmanian B et al (2015) Maternal morbidity in patients with morbidly adherent placenta treated with and without a standardized multidisciplinary approach. Am J Obstet Gynecol 212(218):e1–e9
Fan Y, Gong X, Wang N, Mu K, Feng L, Qiao F, Chen S, Zeng W, Liu H, Wu Y, Zhou Q, Tian Y, Li Q, Yang M, Li F, He M, Beejadhursing R, Deng D (2017) A prospective observational study evaluating the efficacy of prophylactic internal iliac artery balloon catheterization in the management of placenta previa-accreta: a STROBE compliant article. Medicine (Baltimore) 96(45):e8276. https://doi.org/10.1097/MD.0000000000008276
Hughes CW (1954) Use of an intra-aortic balloon catheter tamponade for controlling intra-abdominal hemorrhage in man. Surgery 36:65–68
Belenkiy SM, Batchinsky AI, Rasmussen TE et al (2015) Resuscitative endovascular balloon occlusion of the aorta for hemorrhage control: past, present, and future. J Trauma Acute Care Surg 79(4 suppl 2):S236–S242
Chou MM, Kung HF, Hwang JI et al (2015) Temporary prophylactic intravascular balloon occlusion of the common iliac arteries before cesarean hysterectomy for controlling operative blood loss in abnormal placentation. Taiwan J Obstet Gynecol 54:493–498
Yamamoto N, Koga K, Akahane M et al (2015) Temporary balloon occlusion of the uterine arteries to control hemorrhage during hysterectomy in a case of uterine arteriovenous fistula. J Obstet Gynaecol Res 41:314–318
Wei X, Zhang J, Chu Q et al (2015) Prophylactic abdominal aorta balloon occlusion during caesarean section: a retrospective case series. Int J Obstet Anesth 27:3–8
Minas V, Gul N, Shaw E et al (2015) Prophylactic balloon occlusion of the common iliac arteries for the management of suspected placenta accreta/percreta: conclusions from a short case series. Arch Gynecol Obstet 291:461–465
Marino R, Capriglione S, Morosetti G, Di Angelo Antonio S, Miranda A, Pazzola M, Lopez S, Patrizi L, Angioli R, Stella P (2018) May intraperitoneal irrigation with Betadine improve cesarean delivery outcomes? Results of a 6 years’ single centre experience. J Matern Fetal Neonatal Med. 31(5):670–676. https://doi.org/10.1080/14767058.2017.1293036
ACOG Committee on Obstetric Practice (2002) ACOG Committee opinion. Number 266, January 2002: placenta accreta. Obstet Gynaecol 99(1):169–170
Committee Publications (2010) SMFM Belfort. Am J Obstet Gynecol 203(5):430–439
Mok M, Heidemann B, Dundas K et al (2008) Interventional radiology in women with suspected placenta accreta undergoing caesarean section. Int J Obstet Anesth 17:255–261
Shahin Y, Pang CL (2018) Endovascular interventional modalities for haemorrhage control in abnormal placental implantation deliveries: a systematic review and meta-analysis. Eur Radiol. https://doi.org/10.1007/s00330-017-5222-0
Patel SJ et al (2007) Imaging the pregnant patient for nonobstetric conditions: algorithms and radiation dose considerations. Radiograghics 27:1705–1722
Collough Mc et al (2007) Radiation exposure and pregnancy: when should we be concerned? Radiographics 27:909–917
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Gulino, F.A., Guardo, F.D., Zambrotta, E. et al. Placenta accreta and balloon catheterization: the experience of a single center and an update of latest evidence of literature. Arch Gynecol Obstet 298, 83–88 (2018). https://doi.org/10.1007/s00404-018-4780-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-018-4780-y