Abstract
Objectives
To determine the incidence of Ureaplasma urealyticum and Ureaplasma parvum (UP) in symptomatic and asymptomatic women of reproductive age and to estimate antibiotic susceptibility of ureaplasma isolates.
Materials and methods
This study included 424 ureaplasma positive women of 1,370 tested women who visited gynecological practices during 2010. Cervicovaginal or urethral swab specimens from each patient were obtained for cultivation and molecular typing by RT-PCR.
Results
Ureaplasma spp. was identified by cultivation in 424 (34.4 %) cases, of which 79.0 % were from women with symptoms and 21.0 % from women without symptoms. Among ureaplasma positive women, 121 (28.5 %) were pregnant. Genotyping was successful in 244 strains, and the majority of samples were identified as UP (92.6 %). Among genotyped isolates, there were 79.5 % from symptomatic and 20.5 % from asymptomatic women; 29.9 % from pregnant and 70.1 % from non-pregnant women. There was no difference in the incidence of ureaplasma type regarding symptoms. Antibiotic susceptibility of 424 ureaplasma isolates identified by cultivation showed that all strains were susceptible to doxycycline, josamycin, erythromycin, tetracycline, clarithromycin and pristinamycin, but there was lower susceptibility to quinolone antibiotics, i.e., 42.9 and 24.5 % isolates were susceptible to ofloxacin and ciprofloxacin, respectively.
Conclusion
This study shows that UP was the most frequent isolated ureaplasma species (92.6 %). Regarding antibiotic susceptibility, quinolones are not the best choice for the treatment of ureaplasma infections, while macrolides and tetracyclines are still effective.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urogenital ureaplasma in women of childbearing age is associated with urethritis, pelvic inflammatory disease, pregnancy complications, premature birth and infertility but is also very frequent in healthy women. Mycoplasmas are the smallest known free living microorganisms of 200–300 nm in size. Taxonomically, mycoplasmas belong to the class of Mollicutes together with ureaplasma, also called T (tiny) mycoplasmas because they produce a very small colony size of only 50–60 μm in diameter [1]. In humans, urogenital ureaplasma infection is caused by two species, Ureaplasma urealyticum (UU) and Ureaplasma parvum (UP), with a total of 14 serotypes. UU has characteristics of biovar T960T (or biovar 2 or A), and includes 10 large genomic serovars (0.88–1.2 Mbps): 2, 4, 5, 7, 8, 9, 10, 11, 12 and 13, while UP has characteristics of parvo biovar (or biovar 1 or B) and includes four serovars: 1, 3, 6 and 14 [2, 3].
In the urogenital system UU and UP are considered pathogenic isolates. In women, the urogenital tract infection can be asymptomatic or with mild to severe symptoms, while in men ureaplasma commonly causes urethritis. If the infection spreads to the uterus or fallopian tubes in women or in the small bowel it may lead to pelvic inflammatory disease. The role of ureaplasma in pregnant women is of particular importance as the infection may lead to complications in pregnancy resulting in premature labor, miscarriage or stillborn child [2, 4–9]. Undiagnosed and untreated infection can also lead to infertility. When determining the clinical significance of ureaplasma infection, the differentiation of colonization and infection is necessary because of the high prevalence of ureaplasmas in the healthy population (ureaplasma 70–80 %, mycoplasmas 30–40 %) [10]. The presence of more than 104 CFU in a sample is an additional criterion to distinguish colonization from infection.
Although considered “gold standard”, culture methods could not differentiate species, UU from UP. Therefore, it is important to introduce molecular methods such as real-time polymerase chain reaction (RT-PCR), which enables distinguishing ureaplasma species [11, 12]. The objectives of this study were to determine the incidence of UU and UP in symptomatic and asymptomatic women of reproductive age and to estimate antibiotic susceptibility of ureaplasma isolates.
Materials and methods
Study population
The study was carried out at Gynecological Practices of Primary Health Centers in Zagreb, Croatia which are collaborating institutions of the Croatian National Institute of Public Health. Of 1,370 women who visited gynecological practices during 2010, 471 women were positive for ureaplasma isolates. Only 424 isolates were included in the analysis as for 47 isolates complete medical records were not available. Of the total number of ureaplasma positive women 121 (28.5 %) were pregnant. Women were divided in two groups, 89 (21.0 %) asymptomatic women who came to the gynecological examination because of a routine check-up, or disease conditions that were not related to urogenital system, and 335 (79.0 %) symptomatic women who had at least one of the following symptoms: non-specific pain and tension in the lower abdomen, increased vaginal discharge, dispareunia, burning and frequent urination. The age of symptomatic women ranged from 16 to 64 years (mean 31.6; median 30), while the age of asymptomatic women ranged from 20 to 68 years (mean 30.7; median 30).
Specimen collection
Cervicovaginal or urethral swab specimens were collected for microbiological and molecular analyses. All specimens were obtained before antibiotic treatment. Ureaplasma cultivation was performed at the Department of Bacteriology, while the RT-PCR analysis was performed at the Department of Molecular Diagnostics of the Croatian National Institute of Public Health.
Bacterial detection and antimicrobial susceptibility
Ureaplasma identification was done by cultivation on agar and liquid medium, as well as with a commercial Mycoplasma IST 2 (BioMerieux, Marcy l’Etoile, France). The test is based on the principle of metabolic inhibition of sensitive strains. This test allows the cultivation, identification, determination of the indicative number of bacteria and determination of the susceptibility of the isolates to antibiotics [13, 14].
Co-infection with other microorganisms including Gardnerella vaginalis (GV), beta-haemolytic Group B Streptococcus (GBS), Candida albicans (CA) was identified according to routine laboratory methods, including API-tests (BioMérieux SA Marcy l’Etoile, France). Chlamydia trachomatis (CT) identification was done with COBAS AMPLICOR CT/NG Test (Roche, Basel, Switzerland) and according to Airell et al. [15].
Real-time PCR
Two hundred eighty six samples for which the Mycoplasma IST 2 test result was ≥104 CFU were used in subsequent identification with RT-PCR. Ureaplasma isolates were stored at −20 °C until DNA isolation procedure with the commercial assays (QIAamp DNA Mini Kit; QIAGEN GmbH, Hilden) according to the manufacturer’s instructions. Differentiating strains of UU of UP was performed as described by Mallard et al. [12].
Statistical analysis
Data and statistical analyses were performed in the Division of Molecular Medicine, Rudjer Boskovic Institute using GraphPad Prism (version 4.00) (GraphPad Software, San Diego, CA, USA). Methods of descriptive statistics, Chi-square (χ2) test and Fishers exact test were used. The P values of <0.05 were considered statistically significant.
Results
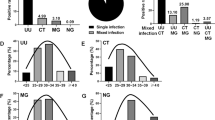
Of the 424 samples, 286 (67.4 %) were analyzed by RT-PCR. In 42/286 (14.6 %) samples the PCR failed to identify ureaplasma species, due to technical difficulties, while 244 samples were successfully genotyped (Table 1). There were 18 (7.4 %) samples identified as UU and 226 (92.6 %) as UP. Of 18 UU cases, 15 (83.3 %) were isolated in symptomatic and 3 (16.7 %) in asymptomatic women, while of 226 UP cases 179 (79.2 %) and 47 (20.8 %) were isolated in symptomatic and asymptomatic women, respectively. There were no statistically significant difference between the incidence of UU and UP among symptomatic and asymptomatic women (P = 0.676).
In the group of pregnant women, UU and UP were isolated in 4/18 (22.2 %) and 69/226 (30.5 %) women, respectively. In the group of women who were not pregnant, UU was isolated in 14/18 (77.8 %), while UP was isolated in 157/226 (69.5 %) women (Table 1). There were no statistically significant difference between the incidence of UU and UP among pregnant and non-pregnant women (P = 0.459).
Table 2 presents subgroups of samples including both pregnancy and symptoms data. Statistical significance (Chi-square test P = 0.022) was observed in UU and UP isolation in the non-pregnant subset where UU appeared more often in asymptomatic infections than UP. However, this difference is probably due to very limited number of asymptomatic infections in this subset (n = 6). Using Fishers exact test, which is more suitable for small sample sizes, statistical difference was not found (P = 0.077). Subset of samples including only pregnant women did not indicate statistically significant difference of ureaplasma species isolation in women with or without symptoms (P = 0.138).
In a cohort of symptomatic women who were not pregnant, UP was found in 153 of 157 (97.5 %) cases, while UU in 12 of 14 (85.7 %) cases. In a cohort of asymptomatic women who were not pregnant, UP was found in 4 of 157 (2.5 %) cases, while UU in 2 of 14 (14.3 %) cases.
In a cohort of symptomatic pregnant women UP was found in 26 of 69 (37.7 %) cases, while UU in 3 of 4 (75.0 %) cases. In asymptomatic pregnant women UP was found in 43 of 69 (62.3 %) cases, while UU in 1 of 4 (25.0 %) cases.
In genotyped ureaplasma isolates, simultaneously associated microorganisms (Table 3) were GV (20.1 %), CA (11.1 %), GBS (4.1 %), and CT (3.7 %). However, there were no significant differences between UU and UP species in co-isolated microorganisms.
Antibiotic susceptibility of 424 ureaplasma isolates identified by cultivation (Table 4) showed that all strains were sensitive to doxycycline, josamycin, erythromycin, tetracycline, clarithromycin, and pristinamycin. Only one ureaplasma isolate showed moderate susceptibility to azithromycin. In addition, there was some resistance to quinolone antibiotics.
The antibiotic susceptibility of 244 ureaplasma genotyped by RT-PCR showed that all strains of UU and UP were sensitive to doxycycline, josamycin, erythromycin, tetracycline, clarithromycin and pristinamycin even though some isolates were resistant to quinolone antibiotics (Table 4). Lower sensitivity was observed to ofloxacin (42.9 %) and ciprofloxacin (24.5 %). One UU isolate (0.3 %) showed moderate susceptibility to azithromycin. Regarding genotyped ureaplasma isolates, there were 72.2 % UU isolates resistant to ciprofloxacin,
Discussion
In recent years urogenital ureaplasmas are frequently isolated in women of childbearing age. Prior to this study the prevalence of different ureaplasma species was not well studied for the Croatian population. This study showed 34.3 % positive samples of urogenital tract in the female population. In comparison to our findings, Zdrodowska-Stefanow et al. [16] found slightly lower ureaplasma prevalence (29.8 % of 541 women tested), while Kechagia et al. [17] found slightly higher prevalence (37.0 % of 369 women tested). Molecular based studies have shown that most of ureaplasma isolates thus far considered UU were actually UP [18–20]. According to Kacerovský et al. [21], the prevalence of UP in a cohort of healthy women who were not pregnant was 57 %, which is a much higher prevalence than those of other genital mycoplasmas, viruses, Chlamydia or GBS infections.
In this study, most ureaplasma isolates were identified as UP (92.6 %) as expected [18, 21, 22]. We found no significant differences between UP and UU positive individuals regarding symptoms or pregnancy. There are evidence of adverse impact of ureaplasma infection on the course and outcome of pregnancy [2, 4], and that ureaplasma infection in infants is associated with low birth weight and perinatal mortality [23]. Govender et al. [24] found high prevalence of ureaplasma among women aged ≥26 years. However, the association between colonization of Mycoplasma hominis, UU or UP and premature births was not confirmed. The results of this study showed that the UU and UP were almost equally often isolated in women who were pregnant or not (22.2 vs. 30.5 %).
Women of reproductive age often suffer from urogenital infections. In this study the incidence of other concomitant bacteria and fungi with ureaplasma was also investigated. Common isolated microorganisms with ureaplasma were GV (20.1 %), CA (11.1 %), GBS (4.1 %), and CT (3.7 %). The findings are consistent with a study of Vogel et al. [25] who found GV the most commonly isolated with ureaplasma. In our study, CA has been isolated in women with UP (11.9 %), but not in patients with UU. Since CA causes unpleasant clinical symptoms, we believe that co-infection with CA and UP should be further investigated on a large number of samples. Despite the significant role of CT infection in genitourinary tract, in our study it was found in only 3.7 % of women.
In this study, all strains of ureaplasma isolates identified by cultivation showed sensitivity to doxycycline, josamycin, erythromycin, tetracycline, clarithromycin and pristinamycin, although some resistance to quinolones was also observed. Thus, limited sensitivity was observed with ofloxacin (42.9 %) and ciprofloxacin (24.5 %). One UU isolate (0.3 %) showed moderate resistance to azithromycin. In addition, regarding the genotyped ureaplasma isolates, where 72.2 % of UU isolates were resistant to ciprofloxacin, it seems that quinolones are not the best choice for treatment of ureaplasma isolates. These findings are consistent with the study of Bayraktar et al. [23] who estimated higher resistance to quinolones of UU, i.e., 86.2 % to ofloxacin and 92.6 % to ciprofloxacin. The study of Mares et al. [26] showing resistance to ofloxacin (16.1 %) and ciprofloxacin (53.8 %) also support the findings in our study. However, macrolides and tetracyclines are still highly effective in treatment of ureaplasma infection.
Conclusion
In this study, both species of ureaplasma, UP and UU, appeared in women regardless of symptoms or pregnancy with UP being the predominant species in the Croatian population. However, due to small number of UU samples it is so far impossible to determine differences in pathogenicity between UP and UU, and further large scale studies should be done to elucidate potential differences. Regarding the relatively high resistance to quinolones in the Croatian population, alternative antibiotics should be considered for treatment of women with ureaplasma infection.
References
Waites KB, Taylor-Robinson D (2011) Mycoplasma and ureaplasma. In: Versalovic J, Carroll KC, Funke G, Jorgensen JH, Landry ML (eds) Manual of clinical microbiology, 10th edn. ASM Press, Washington DC, pp 974–985
Cassell GH, Waites KB, Watson HL, Crouse DT, Harasawa R (1993) Ureaplasma urealyticum intrauterine infection: role in prematurity and disease in newborns. Clin Microbiol Rev 6:69–87
Waites KB, Katz B, Schelonka RL (2005) Mycoplasmas and ureaplasmas as neonatal pathogens. Clin Microbiol Rev 18:757–789. doi:10.1128/CMR.18.4.757-789.2005
Taylor-Robinson D, Bébéar C (1997) Antibiotic susceptibilities of mycoplasmas and treatment of mycoplasmal infections. J Antimicrob Chemother 40:622–630. doi:10.1093/jac/40.5.622
Glass JI, Lefkowitz EJ, Glass JS, Heiner CR, Chen EY et al (2000) The complete sequence of the mucosal pathogen Ureaplasma urealyticum. Nature 407:757–762. doi:10.1038/35037619
Zhang W, Wu Y, Yin W, Yu M (2002) Study of isolation of fluoroquinolone-resistant Ureaplasma urealyticum and identification of mutant sites. Chin Med J 115:1573–1575
Daxboeck F, Zitta S, Stadler M, Iro E, Krause R (2005) Mycoplasma hominis and Ureaplasma urealyticum in patients with sterile pyuria. J Infect 51:54–58. doi:10.1016/j.jinf.2004.06.010
Witt A, Berger A, Gruber CJ, Petricevic L, Apfalter P et al (2005) Increased intrauterine frequency of Ureaplasma urealyticum in women with preterm labor and preterm premature rupture of the membranes and subsequent cesarean delivery. Am J Obstet Gynecol 193:1663–1669. doi:10.1016/j.ajog.2005.03.067
Pararas MV, Skevaki CL, Kafetzis DA (2006) Preterm birth due to maternal infection: causative pathogens and modes of prevention. Eur J Clin Microbiol Infect Dis 25:562–569. doi:10.1007/s10096-006-0190-3
Clegg A, Passey M, Yoannes M, Michael A (1997) High rates of genital mycoplasma infection in the highlands of Papua New Guinea determined both by culture and by a commercial detection kit. J Clin Microbiol 35:197–200
Robertson JA, Stemke GW, Davis JW, Harasawa R, Thirkell D et al (2002) Proposal of Ureaplasma parvum sp. nov. and emended description of Ureaplasma urealyticum (Shepard et al. 1974) Robertson et al. 2001. Int J Syst Evol Microbiol 52:587–597
Mallard K, Schopfer K, Bodmer T (2005) Development of real-time PCR for the differential detection and quantification of Ureaplasma urealyticum and Ureaplasma parvum. J Microbiol Methods 60:13–19. doi:10.1016/j.mimet.2004.08.005
Kilic D, Basar MM, Kaygusuz S, Yilmaz E, Basar H et al (2004) Prevalence and treatment of Chlamydia trachomatis, Ureaplasma urealyticum, and Mycoplasma hominis in patients with non-gonococcal urethritis. Jpn J Infect Dis 57:17–20
Xie X, Zhang J (2006) Trends in the rates of resistance of Ureaplasma urealyticum to antibiotics and identification of the mutation site in the quinolone resistance-determining region in Chinese patients. FEMS Microbiol Lett 259:181–186. doi:10.1111/j.1574-6968.2006.00239.x
Airell A, Ottosson L, Bygdeman SM, Carlberg H, Lidbrink P et al (2000) Chlamydia trachomatis PCR (Cobas Amplicor) in women: endocervical specimen transported in a specimen of urine versus endocervical and urethral specimens in 2-SP medium versus urine specimen only. Int J STD AIDS 11:651–658
Zdrodowska-Stefanow B, Kłosowska WM, Ostaszewska-Puchalska I, Bułhak-Kozioł V, Kotowicz B (2006) Ureaplasma urealyticum and Mycoplasma hominis infection in women with urogenital diseases. Adv Med Sci 51:250–253
Kechagia N, Bersimis S, Chatzipanagiotou S (2008) Incidence and antimicrobial susceptibilities of genital mycoplasmas in outpatient women with clinical vaginitis in Athens, Greece. J Antimicrob Chemother 62:122–125. doi:10.1093/jac/dkn158
Ekiel AM, Friedek DA, Romanik MK, Jóźwiak J, Martirosian G (2009) Occurrence of Ureaplasma parvum and Ureaplasma urealyticum in women with cervical dysplasia in Katowice, Poland. J Korean Med Sci 24:1177–1181. doi:10.3346/jkms.2009.24.6.1177
Xiao L, Glass JI, Paralanov V, Yooseph S, Cassell GH et al (2010) Detection and characterization of human ureaplasma species and serovars by real-time PCR. J Clin Microbiol 48:2715–2723. doi:10.1128/JCM.01877-09
Tang J, Zhou L, Liu X, Zhang C, Zhao Y et al (2011) Novel multiplex real-time PCR system using the SNP technology for the simultaneous diagnosis of Chlamydia trachomatis, Ureaplasma parvum and Ureaplasma urealyticum and genetic typing of serovars of C. trachomatis and U. parvum in NGU. Mol Cell Probes 25:55–59. doi:10.1016/j.mcp.2010.12.001
Kacerovský M, Boudys L (2008) Preterm premature rupture of membranes and Ureaplasma urealyticum. Ceska Gynekol 73:154–159
Beeton ML, Chalker VJ, Kotecha S, Spiller OB (2009) Comparison of full gyrA, gyrB, parC and parE gene sequences between all Ureaplasma parvum and Ureaplasma urealyticum serovars to separate true fluoroquinolone antibiotic resistance mutations from non-resistance polymorphism. J Antimicrob Chemother 64:529–538. doi:10.1093/jac/dkp218
Bayraktar MR, Ozerol IH, Gucluer N, Celik O (2010) Prevalence and antibiotic susceptibility of Mycoplasma hominis and Ureaplasma urealyticum in pregnant women. Int J Infect Dis 14:e90–e95. doi:10.1016/j.ijid.2009.03.020
Govender S, Theron GB, Odendaal HJ, Chalkley LJ (2009) Prevalence of genital mycoplasmas, ureaplasmas and chlamydia in pregnancy. J Obstet Gynaecol 29:698–701. doi:10.3109/01443610903184033
Vogel I, Thorsen P, Hogan VK, Schieve LA, Jacobsson B et al (2006) The joint effect of vaginal Ureaplasma urealyticum and bacterial vaginosis on adverse pregnancy outcomes. Acta Obstet Gynecol Scand 85:778–785. doi:10.1080/00016340500442423
Mareş MNV (2011) High prevalence of fluoroquinolones resistance in Ureaplasma and Mycoplasma strains isolated from infertile women under initial evaluation in North-east Romania. Romanian Biotechnol Lett 16:5858–5862
Acknowledgments
Authors thank Dr Irena Tabain for technical assistance, Dr Štefica Findri-Guštek and Dr Jean Chedid for sample collection. This research has been partially supported by the Ministry of Science, Education and Sports (Grant Number: 098-0982464-2510).
Conflict of interest
The authors have declared that no competing interests exist.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hunjak, B., Sabol, I., Vojnović, G. et al. Ureaplasma urealyticum and Ureaplasma parvum in women of reproductive age. Arch Gynecol Obstet 289, 407–412 (2014). https://doi.org/10.1007/s00404-013-2980-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-013-2980-z