Abstract
Aim
Female genital mutilation (FGM) is a common practice especially performed in women with no anaesthesia or antibiotics and in absence of aseptic conditions. The aim of this systematic review is to explore and analyze for first time in the current literature, the clinical evidence related to the presence of infections in the practice of FGM.
Method
A systematic search of PubMed and Scopus was performed. A combination of the terms “female circumcision”, “genital mutilation”, “genital cutting” and “infection” were used. Studies reporting data on the infections related to patients with FGM were included.
Results
A total of 22,052 patients included, in the study, from African countries. The age ranged from 10 days to 20 years. The procedure was done by physicians, paramedical staff, and other specialties. Type I FGM was performed in 3,115 women while 5,894, 4,049 and 93 women underwent Type II, Type III and unknown type of FGM, respectively. Different types of infections were identified including UTIs, genitourinary tract infections, abscess formation and septicemia or even HIV infection. Moreover, most infections were identified in Type III FGM. The isolated pathogens in the different type of infections, were HIV, Clostridium tetani, Chlamydia trachomatis, Neisseria gonorrhoeae, Treponema pallidum, Candida albicans, Trichomonas vaginalis, HSV-2, Pseudomonas pyocyanea, Staphylococcus aureus. The univariate risk of infection ranged from 0.47 to 5.2.
Conclusion
A variety of infections can occur after FGM. The management of these complications in a low-income economy can be a great burden for the families.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The World Health Organization (WHO) defined as female genital mutilation (FGM) “all procedures involving partial or total removal of the external female genitalia or other injury to the female genital organs whether for cultural or other non-therapeutic reasons” [1]. FGM, otherwise known as female genital cutting or female genital circumcision, is a practice which was present from the antiquity. FGM was found in Egyptian mummies aged from 200 BC [2, 3]. FGM is observed through history in many cultures and countries, mostly in Africa, Indonesia, Australia and Asia and is connected with socio-cultural beliefs, attitudes, values and behaviours such as virginity, fidelity, hygiene, transition of girls into womanhood, tradition and cultural heritage [4, 5]. In Europe and North America, FGM was practiced in the nineteenth century as a remedy for various disorders like epilepsy, hysteria and masturbation [2].
The practice of FGM is mainly prevalent in countries of the Sub-Saharan Africa as well as in the Middle East [6]. There are no restrictions as far as the ethnic, religious or socio-economic class for the perpetuation of this practice [7]. According to the WHO, it is estimated that today the number of girls and women worldwide who are currently living with FGM is between 100 and 140 millions [8].
Female genital mutilation, according to WHO, is classified into four groups based on severity: Type I or clitoridectomy or sunna (excision of the prepuce and/or the clitoris), Type II (excision of the prepuce and clitoris with partial or total excision of the labia minora), Type III or infibulation or pharaonic (excision of the external genitalia with narrowing or stitching of the vaginal orifice) and Type IV (unclassified genital mutilation, including practices such as piercing, pricking, cauterizing or cutting of the vulva) [9].
Female genital mutilation is performed by trained circumcisers or midwives who travel from village to village to perform the cutting with no anaesthesia or antibiotics and in absence of aseptic conditions [7]. The used instruments are razors, knives or scissors which usually are reused [10]. Postoperatively, the extent of damage and the type of FGM indicates the respective wound care [7]. More specifically, Type I usually heals within few days, while girls with type III mutilation require bed rest for over 1 week with their thighs bounded together to guarantee proper healing of the scar [10].
Infections related to FGM are in correlation with the used operating equipment (such as unsterilized instruments), as well as with the traditional material used for haemostasis (cow dung, tree sap or ashes) [7, 11]. The aim of this systematic review is to explore and analyze for first time in the current literature, the clinical evidence related to the presence of infections in the practice of FGM.
Methods
Data sources
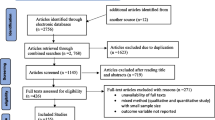
A systematic search of PubMed and Scopus was performed. A combination of the terms “female circumcision”, “genital mutilation”, “genital cutting” and “infection” were used (Fig. 1). The reference lists of the retrieved articles as well as the review papers of the relevant studies were also searched. A time limit was set to only include articles written after 1950. Studies before 1950 did not provide sufficient data and in addition the acquisition of the papers was difficult due to the oldness of the studies.
Study selection and data extraction
Studies reporting data on the infections related to patients with FGM were included in this review. Data reported in the form of letters, experimental studies and abstracts in scientific conferences were not included. Studies published in languages other than English, German, French, Italian, Greek and Spanish and abstracts in scientific conferences were excluded from this review (Fig. 2).
Data extracted from each of the included studies referred to the study design, the origin of the patients, the number of patients, the patients’ age when FGM was performed, the qualification of the person who performed the FGM, the type of FGM, the type of infection, the pathogen isolated and the frequency of the infection in the studied population. A two-tailed p value <0.05 was considered as statistically significant. Data regarding univariate model of testing for the association between the presence of FGM and infection were extracted. The p values, regarding cure, which were not provided by the included studies, were calculated with the use of the OpenEpi Software [12].
Results
After the detailed search process of the relevant literature, 21 individual articles were selected for inclusion in our review (Table 1). From those articles, two were prospective, two retrospective studies, while others were surveys, case control studies or case reports. This could be explained by the absence of relevant archived data, taboos, fear of each local society or even the denial to cooperate in such an effort.
Among the total 22,052 patients included in our review, the patients’ origin was mainly from Sub-Saharan Africa including 2,850, 3,706, 4,890, 1,741, 1,157, 770, 1,787 or 4,826 from Nigeria, Kenya, Sudan, Tanzania, Gambia, Somalia, Burkina Faso or Mali, respectively. The age of the girls who had undergone FGM ranged from 10 days to 20 years. No relevant data were found in 8/21 studies. Regarding the person who performed FGM, were physicians 128/797 (16 %), paramedical staff 46/797 (5.8 %), midwives 420/797 (52.7 %), traditional healers 49/797 (6.1 %), barbers 17/797 (2.1 %), older women 111/797 (14 %) or even family members 26/797 (3.3 %). In 11/21 studies relevant data were not provided. There were available data only for 13,847, in a total of 22,052 patients. Type I FGM was performed in 3,115/13,847 (22.5 %) women while 5,894/13,847 (42.6 %), 4,049/13,847 (29.2 %) and 93/13,847 (0.7 %) women underwent Type II, Type III and unknown type of FGM, respectively. No visible FGM was present in 696/13,847 (5 %).
Different types of infections were identified including acute local trauma infections (7/21) [2, 13–18], UTIs (3/21) [19–21], genitourinary tract infections (6/21) [21–26], abscess formation (1/21) [16] and septicemia (1/21) [16] or even HIV infection (5/21) [24, 25, 27–29]. Moreover, most infections were identified in Type III FGM as it is shown in five studies [15, 16, 18, 23, 30].
Regarding the isolated pathogens in the different type of infections, were HIV (5/21) [24, 25, 27–29], Clostridium tetani (2/21) [17, 23], Chlamydia trachomatis (4/21) [24, 25, 30, 31], Neisseria gonorrhoeae (2/21) [25, 30], Treponema pallidum (4/21) [24, 25, 30, 31], Candida albicans (3/21) [24, 25, 31], Trichomonas vaginalis (3/21) [24, 25, 31], HSV-2 (2/21) [25, 31], Pseudomonas pyocyanea (1/21) [13], and Staphylococcus aureus (1/21) [13].
The univariate risk of infection ranged from 0.47 to 5.2.
Discussion
It is estimated that more than 130 million women underwent some form of FGM, while almost more than two million procedures are currently performed every year [32]. FGM is generally performed at the age of 4–8 years [33]. In Malaysia, in the Democratic Republic of Congo and in Eritrea, it is also performed in newborn girls [33]. FGM, in some regions, is performed in groups of girls during a feast; while in others, individually at home [33]. Although the conditions in which FGM is usually performed are in relation to the financial status of the family, FGM is often performed under non-sterile conditions and without anaesthesia [7, 33].
The practise of FGM can lead both to direct medical and psychological complications [34]. Possible short-term consequences of FGM are acute haemorrhage, local and generalized infections (including HIV), acute pain, injury of neighbouring organs, as well as death [9]. Moreover, gynaecological infections, infertility, dyspareunia, chronic abdominal pain and complications during labour are the usual long-term complications [9, 35]. Psychological difficulties can also be presented as consequences of FGM [33]. Moreover, intervention on the genitals can cause painful memories [36]. Last but not least, cysts, recurrent bladder and urinary tract infections, renal calculi, incontinence, uterine and oviduct infections, infertility, clitoral malformations, painful menstruation (due to obstructed blood flow) and more generalized infections of the abdominal cavity were also described as long-term consequences [7, 37–39].
The most common infections were local including vaginitis caused by Trichomonas, Candida or Bacterial vaginosis and UTIs. According to Klouman, more than 73 % of the women questioned referred that have recovered in the past by vaginosis related to their FGM status, while Msuya showed the absence of statistical significance between FGM and the presence of any genitourinary tract infections. Knight et al. studied the rates of UTIs and recurrent UTIs and found to be 14/51 and 1/51, respectively.
Regarding local wound infections, El Dareer showed that were more often at Type II (138/386) than at Type III (13/2636). In the same study, the delayed complications such as vulvar abscesses, and chronic pelvic infections were the majority in patients with Type III FGM. Regarding the main pathogens identified, were found either in cases of local infections including Clostridium tetani, Pseudomonas pyocyanea and Staphylococcus aureus or in cases of vaginitis including Chlamydia trachomatis, Trichomonas vaginalis, Candida albicans, Neisseria gonorrhoeae, and Treponema pallidum. Moreover, it should be noticed that HIV, HBV and HSV were common findings.
It is difficult to clarify in the included studies how the infections reported appear to be directly related to FGM (e.g., local wound infection, tetanus) as it is not reported how these infections were identified (culture, serologies, survey, or medical records) and what were the observation periods for infection. In the case of the papers by Osifo and Almroth [17, 19], the surveillance time frame may have been short because the infections reported were acute. In the case of some of the other papers reporting the occurrence of sexually transmitted infections, the observation time frame was likely much longer and the relationship of the infection to the FGM is more tenuous. In the paper by Msuya et al. [25], a cross-sectional survey of women who presented to three large government primary health clinics was performed. Specimens were collected from the women for identification of several organisms, and blood was collected for serologic evidence of infection with HIV, syphilis and HSV-2. There was no association between FGM and prevalence of any of the infections analyzed. In this example, the prevalence of the reported infections was a reflection of the prevalence of these infections in young sexually active women in the community, rather than any association with FGM.
According to Elmusaraf, the rates of infection are higher in Type III of FGM. More specifically, the number of women with sexually transmitted diseases was more than five times up, on the group with Type III than that of Type I group. The rates of infection may be even higher as it is unknown what happened with the unreported cases. It should also be mentioned that HIV was more often in studies where the procedure has been performed by local healers a fact which could be explained by the use of unsterilized equipment.
The post-FGM scars are responsible for various problems in the sexual life of the infibulated women. The infibulated scars may cause pain during sexual intercourse [7]. Narrowing of the vagina ranging from complete to partial occlusion can also be a result of the post-FGM scars [40, 41]. Consequently, women with FGM may be at greater risk of HIV transmission caused by bleeding during coitus. Therefore, FGM may also represent a more efficient way of HIV transmission as the disruption of the genital epithelium and at the same time exposure to blood during sexual intercourse seem to increase the risk of HIV infection [42]. One study from Kenya examined the association between HIV infection and previous FGM [28], the problem with that type of analysis was that the link between FGM and HIV is more indirect, and not a direct relationship as implied in that review. In addition, since the analysis did not take into account confounding factors, the observed association could have been due to association of FGM with other factors known to increase the risk of HIV such as non-use of condoms or increased number of sexual partners.
Female genital mutilation can create complications during the labour and the delivery [7]. Prolonged labour, difficulties with vaginal assessment for progress of labour, increased perinatal mortality, perineal lesions, fistulas, episiotomies, post-partum haemorrhage and extended maternal hospital stay are described [19, 37, 43]. Perinatal complications are in direct correlation with the severity and the type of FGM [44].
Even though FGM is considered to have an ethnic background, health and psychological consequences are many [45]. FGM is embedded with the idea of social construction of sex and the standards of regulatory behaviour due to the reason that cause the procedure. Usually FGM is processing in communities, tribes or countries that women have a specific domestic role their rights are limited, and the joy of having sex is forbidden [46]. In addition, in some communities, FGM is considered a condition for a young girl to be regarded as a true woman and on this issue the social peer pressure can be very strong. Although, life in western countries can be an opportunity to eliminate such harmful traditions, there is still the tendency to maintain and to reinforce certain traditions which are even contrary to human rights in various immigrant communities [47]. This is happening because the refugees live with the fear that a forced return to their country of origin might stigmatize their daughter in the absence of FGM [33].
FGM seems now to be a worldwide threat for sexual abuse targeting children of immigrants from Middle East, Africa, Indonesia, Australia and other Muslim countries in Asia. In regard to this phenomenon, many countries have made laws to eliminate this harmful procedure. It is considered as disability and women are protected by the country reports on human rights [48–52].
With our study, we globally present, for first time ever, the infective consequences of FGM practice, in correlation to the patients’ characteristics and the type of FGM.
We tried to evaluate the infections caused by FGM, however, one could raise disagreements mainly for the underestimation of such complications. This could be explained by the low power of the included studies, as well as by the fact that no relevant data could be identified in all the studies caused by the fear, taboos or even denial of the local societies to report the complications of the procedure. For example in the paper by Momoh et al. [16], information was reported on 108 women who attended an African Well Woman Clinic in England many years after the FGM. The information on infectious complications (localized infection/abscess, septicemia) was obtained by a survey, in which the women were asked to recall complications many years later. Obviously, this type of study has the potential for serious recall bias, since the women reported history of infection years after the fact. The same is true of the study by Chalmers et al., in which Somali women residing in Canada were interviewed within 5 years of giving birth (i.e., many years after the FGM) and asked whether they had experienced “infection” immediately after mutilation or long-term “pelvic infections” [14]. In addition to concerns about recall bias, the questions that were asked of the women to elicit responses concerning infection were not reported, so it is unclear what is meant by “immediate infection” or “pelvic infection”. Although there is this probability of the recall bias, every piece of information on this issue should be evaluated. Moreover, we have done excerption of the detected microbial agents in women population from 21 studies who have had FGM previously in their life. After further evaluation were excluded due to the lack of substantial information on the type of infection, the pathogen isolated or whether there was connection of the infection and the practice of FGM. In addition, although many of the detected microbial agents are sexually transmitted and related to sexual behaviour, we believe that we should not be limited to the local infective consequences of FGM but also the infections which are the indirect result of this practice (due to low self esteem, submission to male partner or change of sexual behaviour).
In conclusion, the findings of our review indicate that FGM confirm the variety of infections that can occur after the practice of this form of mutilation. Furthermore, the presence of complications both medical and psychological indicates that more efforts are required to eliminate this ancient practice. Evidentiating the type and the frequencies of infections correlated to FGM, we present another strong reason for total elimination of this inhuman practice. The improvement in women’s socio-economic status as well as informative programs targeting both the local communities and the international public opinion are important in the attempt to eradicate the practice of FGM and its consequences in women’s health.
References
WHO/UNICEF/UNFPA (2008) Female genital mutilation: a joint WHO/UNICEF/UNFPA statement. Geneva WHO
Dirie MA, Lindmark G (1992) The risk of medical complications after female circumcision. East Afr Med J 69:479–482
The American College of Obstetricians and Gynecologists (2008) Female Genital Cutting: Clinical Management of Circumcised Women. Washington DTACoOaG
Gruenbaum E (2001) The female circumcision controversy: an anthropological perspective. University of Pennsylvania Press, Philadelphia
Kolucki B (2004) http://www.disabilityworld.org/01-03_04/women/fgm.shtml. KBFGMDWaDS
Mitike G, Deressa W (2009) Prevalence and associated factors of female genital mutilation among Somali refugees in eastern Ethiopia: a cross-sectional study. BMC Public Health 9:264
Nour NM (2008) Female genital cutting: a persisting practice. Rev Obstet Gynecol 1:135–139
Yoder PS, Abderrahim N, Zhuzhuni A (2004) Female genital cutting in the demographic and health surveys: a critical and comparative analysis. ORC Macro, Macro International, Inc., Calverton
World Health Organisation (WHO) (1995) Female genital mutilation. Report of The WHO Technical Working Group, Geneva
Davis G, Ellis J, Hibbert M, Perez RP, Zimbelman E (1999) Female circumcision: the prevalence and nature of the ritual in Eritrea. Mil Med 164:11–16
Kun KE (1997) Female genital mutilation: the potential for increased risk of HIV infection. Int J Gynaecol Obstet 59:153–155
Dean A, Sullivan Κ, Soe Μ (2008) OpenEpi: Open Source Epidemiologic Statistics for Public Health, Version 2.2.1. Updated 2008/04/06 [cited 2008/09/18]. http://www.OpenEpi.com
Adetoro OO, Ebomoyi E (1986) Health implications of traditional female circumcision in pregnancy. Asia Oceania J Obstet Gynaecol 12:489–492
Chalmers B, Hashi KO (2000) 432 Somali women’s birth experiences in Canada after earlier female genital mutilation. Birth 27:227–234
El Dareer A (1983) Complications of female circumcision in the Sudan. Trop Doct 13:131–133
Momoh C, Ladhani S, Lochrie DP, Rymer J (2001) Female genital mutilation: analysis of the first twelve months of a Southeast London specialist clinic. BJOG 108:186–191
Osifo DO, Evbuomwan I (2009) Female genital mutilation among Edo people: the complications and pattern of presentation at a pediatric surgery unit, Benin City. Afr J Reprod Health 13:17–25
Shandall AA (1967) Circumcision and infibulation of females: a general consideration of the problem and a clinical study of the complications in Sudanese women. Sudan Med J 5:178–212
Almroth L, Bedri H, El Musharaf S et al (2005) Urogenital complications among girls with genital mutilation: a hospital-based study in Khartoum. Afr J Reprod Health 9:118–124
el-Defrawi MH, Lotfy G, Dandash KF, Refaat AH, Eyada M (2001) Female genital mutilation and its psychosexual impact. J Sex Marital Ther 27:465–473
Knight R, Hotchin A, Bayly C, Grover S (1999) Female genital mutilation—experience of The Royal Women’s Hospital, Melbourne. Aust N Z J Obstet Gynaecol 39:50–54
Arbesman M, Kahler L, Buck GM (1993) Assessment of the impact of female circumcision on the gynecological, genitourinary and obstetrical health problems of women from Somalia: literature review and case series. Women Health 20:27–42
Hassan A (1995) Sudanese women’s struggle to eliminate harmful practices. Plan Parent Chall 2:17–8, 21–2
Klouman E, Manongi R, Klepp KI (2005) Self-reported and observed female genital cutting in rural Tanzania: associated demographic factors, HIV and sexually transmitted infections. Trop Med Int Health 10:105–115
Msuya SE, Mbizvo E, Hussain A, Sundby J, Sam NE, Stray-Pedersen B (2002) Female genital cutting in Kilimanjaro, Tanzania: changing attitudes? Trop Med Int Health 7:159–165
Okonofu FE, Larsen U, Oronsaye F, Snow RC, Slanger TE (2002) The association between female genital cutting and correlates of sexual and gynaecological morbidity in Edo State, Nigeria. BJOG 109:1089–1096
Brewer DD, Potterat JJ, Roberts JM Jr, Brody S (2007) Male and female circumcision associated with prevalent HIV infection in virgins and adolescents in Kenya, Lesotho, and Tanzania. Ann Epidemiol 17:217–226
Maslovskaya O, Brown JJ, Padmadas SS (2009) Disentangling the complex association between female genital cutting and HIV among Kenyan women. J Biosoc Sci 41:815–830
Yount KM, Abraham BK (2007) Female genital cutting and HIV/AIDS among Kenyan women. Stud Fam Plann 38:73–88
Elmusharaf S, Elkhidir I, Hoffmann S, Almroth L (2006) A case-control study on the association between female genital mutilation and sexually transmitted infections in Sudan. BJOG 113:469–474
Morison L, Scherf C, Ekpo G et al (2001) The long-term reproductive health consequences of female genital cutting in rural Gambia: a community-based survey. Trop Med Int Health 6:643–653
Hamoudi A, Shier M (2010) Late complications of childhood female genital mutilation. J Obstet Gynaecol Can 32:587–589
Jaeger F, Caflisch M, Hohlfeld P (2009) Female genital mutilation and its prevention: a challenge for paediatricians. Eur J Pediatr 168:27–33
Utz-Billing I, Kentenich H (2008) Female genital mutilation: an injury, physical and mental harm. J Psychosom Obstet Gynaecol 29:225–229
Leye EPR, Nienhuis G, Claeys P, Temmerman M (2006) Health care in Europe for women with genital mutilation. Health Care Women Int 27:362–378
Johansen RE (2006) Care for infibulated women giving birth in Norway: an anthropological analysis of health workers’ management of a medically and culturally unfamiliar issue. Med Anthropol Q 20:516–544
Momoh C (2004) Female genital mutilation. Curr Opin Obstet Gynecol 16:477–480
Nour NM (2004) Female genital cutting: clinical and cultural guidelines. Obstet Gynecol Surv 59:272–279
Obermeyer CM (2005) The consequences of female circumcision for health and sexuality: an update on the evidence. Cult Health Sex 7:443–461
Egwuatu VE, Agugua NE (1981) Complications of female circumcision in Nigerian Igbos. Br J Obstet Gynaecol 88:1090–1093
Iregbulem LM (1980) Post-circumcision vulval adhesions in Nigerians. Br J Plast Surg 33:83–86
European Study Group on Heterosexual Transmission of HIV (1992) Comparison of female to male and male to female transmission of HIV in 563 stable couples. BMJ 304:809–813
Merli C (2010) Male and female genital cutting among Southern Thailand’s Muslims: rituals, biomedical practice and local discourses. Cult Health Sex 12:725–738
Banks E, Meirik O, Farley T, Akande O, Bathija H, Ali M (2006) Female genital mutilation and obstetric outcome: WHO collaborative prospective study in six African countries. Lancet 367:1835–1841
Behrendt A, Moritz S (2005) Posttraumatic stress disorder and memory problems after female genital mutilation. Am J Psychiatry 162:1000–1002
Boyle EH (2002) Female genital cutting: cultural conflict in the global community. Johns Hopkins University Press, Baltimore
Snow RC, Slanger TE, Okonofua FE, Oronsaye F, Wacker J (2002) Female genital cutting in southern urban and peri-urban Nigeria: self-reported validity, social determinants and secular decline. Trop Med Int Health 7:91–100
FGM Joint Statement W, UNICEF and UNFPA, (1997, Page 4
Goodwin J-A, David J (2007) HtupofcidywsliscB, “Daily Mail, 2007-JAN-03. http://www.dailymail.co.uk/
Center for Reproductive Rights (2004) Legislation on Female Genital Mutilation in the United States CfRR. http://www.reproductiverights.org/
Megan C (2004) TiUSAoGM, Womensenews, 2004-FEB-19. http://www.womensenews.org/
US Department of State (2009) CRoHRPf-I, 25 February, sect. 5—attachment 1
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iavazzo, C., Sardi, T.A. & Gkegkes, I.D. Female genital mutilation and infections: a systematic review of the clinical evidence. Arch Gynecol Obstet 287, 1137–1149 (2013). https://doi.org/10.1007/s00404-012-2708-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-012-2708-5