Abstract
Purpose
To assess the effects of the combination of pelvic floor rehabilitation and intravaginal estriol administration on stress urinary incontinence (SUI), urogenital atrophy and recurrent urinary tract infections in postmenopausal women.
Methods
Two-hundred-six postmenopausal women with urogenital aging symptoms were enrolled in this prospective randomized controlled study. Patients were randomly divided into two groups and each group consisted of 103 women. Subjects in the treatment group received intravaginal estriol ovules, such as 1 ovule (1 mg) once daily for 2 weeks and then 2 ovules once weekly for a total of 6 months as maintenance therapy plus pelvic floor rehabilitation. Subjects in the control group received only intravaginal estriol in a similar regimen. We evaluated urogenital symptomatology, urine cultures, colposcopic findings, urethral cytologic findings, urethral pressure profiles and urethrocystometry before, as well as after 6 months of treatment.
Results
After therapy, the symptoms and signs of urogenital atrophy significantly improved in both groups. 61/83 (73.49%) of the treated patients, and only 10/103 (9.71%) of the control patients referred a subjective improvement of their incontinence. In the patients treated by combination therapy with estriol plus pelvic floor rehabilitation, we observed significant improvements of colposcopic findings, and there were statistically significant increases in mean maximum urethral pressure (MUP), in mean urethral closure pressure (MUCP), as well as in the abdominal pressure transmission ratio to the proximal urethra (PTR).
Conclusions
Our results showed that combination therapy with estriol plus pelvic floor rehabilitation was effective and should be considered as a first-line treatment for symptoms of urogenital aging in postmenopausal women.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
10–40% [1] of all postmenopausal women were found to have symptoms related to urogenital atrophy but only about 25% of these patients presented the problem to their physicians and a very few indeed underwent hormone replacement therapy (HRT) [2].
Symptoms and signs of urogenital integrity disorders involving the lower urinary tract, genital tract and pelvic floor become evident after menopause increasing with advancing age [1].
In the West, 8% of the total population has urogenital problems [3] and in the United States 20 million women, who do not undergo HRT, have these socially disabling symptoms [4].
The symptoms relating to urogenital aging may be categorized into two groups: those localized to the lower urinary tract (urethra and bladder) and those confined to the vagina and vulva. The first group includes frequency and urgency, nocturia, dysuria, recurrent urinary tract infections and urinary incontinence; the second group comprises vaginal dryness, itching, burning and dyspareunia.
It is now well demonstrated that the urogenital organs are highly sensitive to the influence of estrogen. In fact, estrogen receptors have been found in the urethra and bladder trigone [5], as well as in the round ligaments and levator ani muscles [4, 6].
In the vagina, the progressive decline of estrogens during climacterium induces an atrophy of the mucosa, which becomes thinner.
Similarly in the urethra and bladder, the reduction of estrogens produces the atrophy of the mucosae, causing nocturia, dysuria and urinary urgency.
The efficacy of estrogen replacement therapy (ERT) on urogenital complaints has already been clearly demonstrated by numerous clinical studies [7–11].
Estriol is considered to be free of the potential risks associated with the systemic use of estrogenic therapy [12], moreover, it has been definitively demonstrated that intravaginal estriol does not stimulate the proliferation of endometrium [13]. Its vaginal absorption is swift and effective, and bypasses the first hepatic inactivation and more stable circulating levels are achieved with respect to the oral route.
Several studies have shown the efficacy of intravaginal estriol in the treatment of urogenital atrophy [13, 15–20], of recurrent urinary tract infections [21, 22] and of climacteric symptoms [15, 18].
We have already demonstrated the efficacy and safety of intravaginal estriol administration on urinary incontinence, urogenital atrophy and recurrent urinary tract infections in postmenopausal women [11].
Several conservative treatment options are available for the management of stress urinary incontinence (SUI), e.g., physical therapies, behavioral modification, and pharmacological intervention [23]. Pelvic floor rehabilitation is prescribed as a first-line treatment for women with SUI, particularly in cases of urinary incontinence with poor-quality perineal testing results or inverted perineal command [26].
The aim of our study was to investigate the effects of the combination of pelvic floor rehabilitation (pelvic floor muscle training and electrical stimulation) and intravaginal estriol on urinary incontinence, urogenital atrophy and recurrent urinary tract infections in postmenopausal women.
Materials and methods
Study area
The study was carried out from May 2005 to April 2010 in Sassari, the largest district of northern Sardinia with 122,803 inhabitants and a density of 219 inhabitants/km2. The populations included 11,269 women of age range 55–70 years old.
Sampling
Sample size was calculated on the basis of prevalence of urinary incontinence, urogenital atrophy and recurrent urinary tract infections in postmenopausal women, increased by 10%. Consequently, our estimates were safeguarded at an optimal level of precision (5%) against the possible effect of (a) disease reduction since the previous study and (b) the non-response numbers.
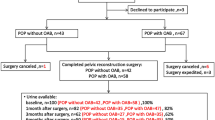
The theoretical sample size was 576. All the women were issued with an information leaflet explaining the aim of the study and requesting their participation. One-hundred ten (19.10%) women chose not to participate, hence 466 (80.90%) women were evaluated.
An initial assessment (screening visit or visit 1) including the recent history of somatic symptoms, complete medical and surgical history, and a complete physical and gynecological examination was performed to determine the subject’s suitability for the study in accordance with inclusion and exclusion criteria.
If the subject was suitable for the study an appointment was made for her to return for the baseline visit (visit 2). During the baseline visit the determination of vaginal pH, colposcopic examination, vaginal and urethral smears, urodynamic examination, and a further check of compliance of subject with inclusion and exclusion criteria were performed.
All patients presented symptoms and signs of urinary stress incontinence, vaginal atrophy, and histories of recurrent urinary tract infections. None of the patients had received estrogen treatment prior to the study. The patients with previous hysterectomy were eligible for the study too.
Exclusion criteria for the study were pathologies or anatomical lesions of the urogenital tract such as uterovaginal prolapse, cystocele, and rectocele of grade II or III according to Baden and Walker classification (HWS), the presence of severe systemic disorders, thromboembolic diseases, biliary lithiasis, previous breast or uterine cancer, abnormal uterine bleeding and body mass index (BMI) ≥25 kg/m2.
The diagnosis of genuine stress urinary incontinence was confirmed by the direct visualization of loss of urine from the urethra during the standard stress test and by urodynamic investigation. Patients with detrusor overactivity and abnormal maximal cystometric capacity were excluded from the study.
Hence, the study reports data on 206 postmenopausal women with urogenital aging symptoms (Fig. 1).
At this point, the eligible women signed informed consent and were randomly assigned into a treatment and a control group.
Each group consisted of 103 women. Randomization was obtained using sets of sequenced, sealed, opaque envelopes, each containing the bottle number to be given to each patient.
Subjects in the group I were given intravaginal estriol ovules: 1 ovule (1 mg) once daily for 2 weeks (Colpogyn, Angelini, Rome, Italy) and then 2 ovules once weekly as maintenance therapy for a total of 6 months plus pelvic floor muscle training group and electrical stimulation.
Subjects in the group II received only estriol in a similar regimen.
Pelvic floor muscle training and electrical stimulation were performed as explained by Castro et al. [25].
We did not use a placebo group because all the women enrolled had urogenital atrophy requiring local estrogen therapy. Recently, Archer [27] showed the efficacy and tolerability of local estrogen therapy for urogenital atrophy. We have already demonstrated the advantage of estriol versus placebo [11].
We evaluated the urogenital symptomatology, urine cultures, colposcopic findings, urethral cytologic findings, urethral pressure profile, and urethrocystometry before, as well as after 6 months of treatment.
The women enrolled in the study complained of the main symptoms of stress urinary incontinence, such as urine loss with physical exertion, coughing, sneezing and intercourse, and symptoms of genital atrophic conditions, including vaginal dryness and dyspareunia.
The subjective evaluation of incontinence disorders was based on the patient’s description regarding the effect of the incontinence with respect to activities at home and at work (Incontinence Impact Questionnaire) [28]. The urinary incontinence complaints were assessed as: none, mild, moderate, and severe.
The therapy efficacy on urinary incontinence complaints was assessed as follows: women who changed from ‘mild/moderate/severe’ to ‘none’ were classified as ‘cured’. Women who changed from ‘moderate’ to ‘mild’ or from ‘severe’ to ‘moderate/mild’ were classified as ‘improved’. Women who did not change from pretreatment were classified as ‘no change’. Women who changed from ‘none’ to ‘mild/moderate/severe’ or from ‘mild’ to ‘moderate/severe’ or from ‘moderate’ to ‘severe’ were classified as ‘worse’.
The symptoms relating to urogenital atrophy such as vaginal dryness and dyspareunia were classified as follows: none, moderate or severe at each visit. The therapy efficacy on atrophic condition complaints was assessed as described above.
The gynaecological evaluation included vaginal atrophy and pH assessment.
The authors visually assessed the degree of urogenital atrophic conditions as none, moderate or severe at each visit, taking into account pallor, petechiae, friability and vaginal dryness (no, yes) as objective evidence of estrogen deficiency.
Vaginal pH was measured using an indicator strip, and midstream urine specimens were obtained at the beginning of the study and after 6 months of treatment.
Significant bacteriuria was considered to be present if the midstream urine culture yielded ≥105 colony-forming units (CFU) per milliliter.
The decrease in thickness of pavement epithelium of portio was subjectively assessed as present or absent.
We performed Schiller’s test in all patients and visually evaluated the possible presence of petechiae.
Vaginal and urethral smears were taken during colposcopic examination by the cytobrush sampling technique from the upper lateral vaginal walls and from the distal urethral walls. After preparation with a cytology fixative, the smears were sent to the pathologist for preparation and staining according to Papanicolau.
The effect of estriol on the vaginal and urethral epithelium was estimated by means of the karyopyknotic index (KPI), defined as the percentage of superficial cells found in the total population of the squamous cells examined [29], which is considered as a reliable cellular index for the determination of estrogen activity [30].
We calculated the urethral pressure profile and urethrocystometry at the beginning of the study and after 6 months of treatment according to the criteria of the International Continence Society (ICS), using the Phoenix Plus videourodynamic machine (Albyn Medical LTD, Dingwall, Scotland).
Urethral and intravesical pressures were measured in the supine position by a catheter equipped with a microtransducer two-way standard (Albyn Medical LTD, Dingwall, Scotland). Vesical filling was performed with saline solution at a constant speed of 50 ml/min. By three reproducible urethral pressure profiles, the mean maximum urethral pressure (MUP), the mean maximum urethral closure pressure (MUCP) were calculated. The abdominal pressure transmission ratio (PTR) to the urethra was calculated as a/b × 100 where a is the urethral pressure increase and b is the intravesical pressure increase during coughing.
At the beginning of the study, the women received a diary in which they were asked to record the occurrence of localized or systemic side effects. Six months later we reviewed these diaries to assess the treatment compliance.
Our Ethic Board Committee approved the study.
Statistical analysis
It is a common feature to have dropout patients in studies designed such as our. Nevertheless, we decided to include the eventual dropout patients in statistical analysis.
In the statistical elaboration of the data concerning the dropouts, the unavailable outcome data were assumed to be the worst case, i.e., the parameters taken into account at the baseline were considered to have remained unchanged.
All the data were computed in a database. The analysis was carried out using an one-way Anova for means observation and χ2 with collinearity for dichotomous variables. All statistical significant figures apply to the after-treatment measurements.
Results
The characteristics of the patients are summarized in Table 1. The groups I and II were homogenous for age, vaginal parity, menopause duration, and duration of urinary incontinence.
The dropout patients consisted of 9 women in the group I and 11 in the group II which interrupted the treatment before the 6 months established in the protocol. Among them 11 patients (5 in the group I and 6 in the group II) experienced discomfort during vaginal treatment, 9 (4 in the group I and 5 in the group II) experienced localized adverse reactions, such as vaginal irritation and burning.
Before starting the therapy, 83/103 patients in group I versus 103/103 patients in group II presented symptoms of stress incontinence ranging from mild to moderate.
After 6 months, 61/83 (73.49%) of the treated patients, referred subjective improvement of their incontinence (43 totally continent and 18 significantly improved), while only 10/103 (9.71%) of the control patients reported this improvement (P < 0.01) (Table 2).
Before starting the therapy, all the 206 patients had presented urogenital atrophy ranging from moderate to severe and had suffered from vaginal dryness; 75 in the treatment group and 65 in the control group referred symptoms of dyspareunia.
Subsequently, on clinical examination symptoms and signs of urogenital atrophy, vaginal dryness, and dyspareunia improved significantly in the treatment group in comparison to the control group (Table 2).
At baseline, 52 women (50.48%) had presented significant bacteriuria (Escherichia coli in all cases) in the group I, and 48 (46.60%) in the group II (Escherichia coli in all cases). After treatment, there was a significant quantity of bacteria in the urine of 10/52 patients (19.23%) and in 21/48 (43.75%), respectively (P < 0.001).
The mean vaginal pH at baseline was 5.53 ± 0.86 for the group I and 5.36 ± 0.74 for the group II. After therapy, vaginal pH was 4.34 ± 0.76 and 4.62 ± 0.81, respectively (P < 0.05) (Table 2).
In both groups, a statistically significant improvement of colposcopic parameters, and a significant rise in KPI were found after treatment in the vaginal and urethral epithelium (Table 3).
In the subjects treated with estriol and rehabilitation, there were statistically significant increases in mean MUP and MUCP and a significant increase in mean PTR in comparison to the estriol group (Table 4).
Urethrocystometry showed positive modifications in the both groups which were not, however, statistically significant (Table 5).
No systemic adverse reactions were observed.
Discussion
The vasomotor symptoms and dyspareunia due to urogenital atrophy are the main reasons for which a menopausal woman requires replacement therapy.
A meta-analysis on 77 studies [2] has shown estrogens (administered orally or vaginally, and in all dosage regimens) to be efficacious in the treatment of urogenital atrophy. In particular, the local low-dose estrogen therapy (using both estradiol and estriol) is as effective as systemic estrogen therapy in the treatment of urogenital atrophy in postmenopausal women.
The effectiveness of estriol on the vaginal epithelium and urethra is well documented [13, 16].
We have already demonstrated that intravaginal estriol therapy was effective in the treatment of symptoms and signs of urogenital aging [11].
Physical therapies involving pelvic floor muscle training with or without other treatments such as vaginal cones, biofeedback, and electrical stimulation are the standard for conservative treatment and prevention of SUI [24]. Castro et al. [25] in a randomized, single-blinded, controlled trial, compared the effectiveness of pelvic floor exercises, electrical stimulation, vaginal cones, and no active treatment in women with urodynamic stress urinary incontinence and demonstrated that these modalities are equally effective treatments and are far superior to no treatment in women with urodynamic SUI [25].
Pelvic floor rehabilitation is prescribed as the first-line treatment for women with SUI, particularly in cases with poor-quality perineal testing results or inverted perineal command [26].
Ishiko et al. [31] investigated the effects of the combination of pelvic floor exercise and estriol on postmenopausal SUI by a randomized study (pelvic rehabilitation and estriol vs. only rehabilitation): the authors demonstrated the efficacy of the combination therapy.
Recurrent urinary tract infections represent a serious complaint for many postmenopausal women. In the menopause period, the reduction of lactobacillus colonization, vaginal pH reduction and the atrophy of the vaginal mucosa are involved in the higher frequency of urinary tract infections. The use of intravaginal estriol for 6 months determined a significant reduction in the number of cases of bacteriuria and a decrease of vaginal pH.
The effectiveness of local estriol administration for the treatment of urogenital symptoms has been well documented by several authors [7–11].
Schar and co-workers [32] suggest estriol local therapy as an effective mode of primary treatment in postmenopausal women with urinary incontinence.
In a multicenter study on 251 postmenopausal women, Lose et al. [33] showed that the vaginal administration of low-dose estradiol and estriol are equally efficacious in alleviating lower urinary tract symptoms which appear after the menopause.
Iosif and colleagues [14] in a longitudinal study reported that 75% of the women referred significant subjective improvement of stress incontinence and a significant increase in pressure transmission ratio to the proximal urethra were noted after vaginal medication with estriol.
We have already demonstrated that the patients treated by vaginal estriol referred, on clinical examination, an improvement of their incontinence after local estrogenic therapy. Statistically significant increases were noted in urodynamic parameters such as MUP, MUCP, and PTR in comparison to the control subjects [11].
According to several authors, estrogen therapy relieves the symptoms of stress incontinence by causing proliferation and growth of the urethral mucosa and blood vessel engorgement, which in turn constitute the ‘urethral softness factor’ [14].
According to Bathia [34] the significant increase of abdominal pressure transmission to the proximal third of the urethra is to be considered a positive clinical response. This crucial effect is probably due to extraurethral factors such as improved functioning of the pelvic floor muscles [35]. In summary, increased tissue tension and urethral pressure, along with improved pressure transmission to the proximal urethra play an important role in the alleviation of genuine stress incontinence.
With regard to the risks of local estrogen therapy, estriol is considered to be free from potential adverse reactions of systemic estrogenic therapy [14]. Several authors have definitively demonstrated that intravaginal estriol does not stimulate the proliferation of endometrium [13]. Furthermore, local treatment of vaginal atrophy is not associated with possible risks of systemic HRT such as breast cancer [36].
Iosif et al. [14] recovered endometrial biopsies from 48 patients after 8–10 years of therapy with estriol suppositories and 7 showed weak proliferative changes, thus demonstrating that the risk of adverse reactions to estriol is insignificant.
In our series, the compliance of the tested patients was high and almost all completed the study. We did not observe adverse drug systemic effects, and collateral effects were limited to localized pruritus or burning.
In conclusion, combination therapy with estriol and rehabilitation (muscle exercise and electrostimulation) was highly efficacious in reducing the urogenital atrophy and frequency of urinary tract infections, as well as the symptoms and signs of stress urinary incontinence; furthermore, this treatment was seen to be safe and well-tolerated by the patients.
Indeed, we think that combination therapy should be considered the first-line treatment for SUI in post menopausal women.
References
Greendale GA, Judd HL (1993) The menopause: health implications and clinical management. J Am Geriatr Soc 41:426–436
Cardozo L, Bachmann G, McClish D, Fonda D, Birgenson L (1998) Meta-analysis of estrogen therapy in the management of urogenital atrophy in postmenopausal women: second report of the hormones and urogenital therapy committee. Obstet Gynecol 92:722–727
Barlow DH, Samsioe G, van Geelen JH (1997) A study of European women’s experience of the problems of urogenital ageing and its management. Maturitas 27:239–247
Samsioe G (1998) Urogenital aging. A hidden problem. Am J Obstet Gynecol 178:S245–S249
Iosif CS, Batra S, Ek A, Astedt B (1981) Estrogen receptors in the human female lower urinary tract. Am J Obstet Gynecol 141:817–820
Smith P (1993) Estrogens and the urogenital tract. Acta Obstet Gynecol Scand 157(suppl):1–25
Samsioe G, Jansson I, Mellstrom D, Svanborg A (1985) Occurrence, nature and treatment of urinary incontinence in a 70-year-old female population. Maturitas 7:335–342
Nilsson K, Heimer G (1992) Low-dose oestradiol in the treatment of urogenital oestrogen deficiency: a pharmacokinetic and pharmacodynamic study. Maturitas 15:121–127
Smith P, Heimer G, Lindskog M, Ulmsten U (1993) Oestradiol-releasing vaginal ring for treatment of postmenopausal urogenital atrophy. Maturitas 16:145–154
Fantl JA, Cardozo L, McClish DK (1994) Estrogen therapy in the management of urinary incontinence in postmenopausal women: a meta-analysis. First report of the hormones and urogenital therapy committee. Obstet Gynecol 83:12–18
Dessole S, Rubattu G, Ambrosini G, Gallo O, Capobianco G, Cherchi PL, Marci R, Cosmi E (2004) Efficacy of low-dose intravaginal estriol on urogenital aging in postmenopausal women. Menopause 11:49–56
Esposito G (1991) Estriol: a weak estrogen or a different hormone? Gynecol Endocrinol 5:131–153
Heimer GM, Englund DE (1992) Effects of vaginally-administered oestriol on post-menopausal urogenital disorders: a cytohormonal study. Maturitas 14:171–179
Iosif CS (1992) Effects of protracted administration of estriol on the lower genito urinary tract in postmenopausal women. Arch Gynecol Obstet 251:115–120
Foidart JM, Vervliet J, Buytaert P (1991) Efficacy of sustained-release vaginal oestriol in alleviating urogenital and systemic climacteric complaints. Maturitas 13:99–107
Van der Linden MC, Gerretsen G, Brandhorst MS, Ooms EC, Kremer CM, Doesburg WH (1993) The effect of estriol on the cytology of urethra and vagina in postmenopausal women with genito-urinary symptoms. Eur J Obstet Gynecol Reprod Biol 51:29–33
Henriksson L, Stjernquist M, Boquist L, Alander U, Selinus I (1994) A comparative multicenter study of the effects of continuous low-dose estradiol released from a new vaginal ring versus estriol vaginal pessaries in postmenopausal women with symptoms and signs of urogenital atrophy. Am J Obstet Gynecol 171:624–632
Bottiglione F, Volpe A, Esposito G, Aloysio DD (1995) Transvaginal estriol administration in postmenopausal women: a double blind comparative study of two different doses. Maturitas 22:227–232
Barentsen R, van de Weijer PH, Schram JH (1997) Continuous low dose estradiol released from a vaginal ring versus estriol vaginal cream for urogenital atrophy. Eur J Obstet Gynecol Reprod Biol 71:73–80
Dugal R, Hesla K, Sordal T, Aase KH, Lilleeidet O, Wickstrom E (2000) Comparison of usefulness of estradiol vaginal tablets and estriol vagitories for treatment of vaginal atrophy. Acta Obstet Gynecol Scand 79:293–297
Kanne B, Jenny J (1991) Local administration of low-dose estriol and vital Lactobacillus acidophilus in postmenopause. Gynakol Rundsch 31:7–13
Raz R, Stamm WE (1993) A controlled trial of intravaginal estriol in postmenopausal women with recurrent urinary tract infections. N Engl J Med 329:753–756
Dmochowski RR, Miklos JR, Norton PA, Zinner NR, Yalcin I, Bump RC (2004) Duloxetine urinary incontinence study group. Duloxetine versus placebo for the treatment of Noth American women with stress urinary incontinence. J Urol 170:1259–1263
Berghmans LCM, Hendrikis HJM, Bo K, Hay-Smith EJ, de Bies RA, van Waalwijk, van Doorn ESC (1998) Conservative treatment if stress urinary incontinence in women: a systematic review of randomized clinical trials. Br J Urol 82:181–189
Castro RA, Arruda RM, Zanetti MRD, Santos PD, Sartori MGF, Girao MJBC (2008) Single-blind, randomized, controlled trial of pelvic floor muscle training, electrical stimulation, vaginal cones, and no active treatment in the management of stress urinary incontinence. Clinics 63:465–472
Leriche B, Conquy S (2010) Guidelines for rehabilitation management of non-neurological urinary incontinence in women. Prog Urol 20:S104–S108
Archer DF (2010) Efficacy and tolerability of local estrogen therapy for urogenital atrophy. Menopause 17:194–203
Shumaker SA, Woman JF, Uebersax JS, McClish D, Fantl JA (1994) Health-related quality-of-life measures for women with urinary incontinence—the incontinence impact questionnaire and the urogenital distress inventory. Qual Life Res 3:291–306
Mishell DR (1987) Menopause: physiology and pharmacology. Yearbook Medical Publisher, Chicago
Wied GL, Bibbo M (1975) Evaluation of endocrinologic condition by exfoliative cytology. In: Gold JJ (ed) Textbook of gynaecologic endocrinology. Harper and Row, New York, pp 117–155
Ishiko O, Hirai K, Sumi T, Tatsuta I, Ogita S (2001) Hormone replacement therapy plus pelvic floor muscle exercise for postmenopausal stress incontinence. A randomized, controlled trial. J Reprod Med 46:213–220
Schar G, Kochli OR, Fritz M, Heller U (1995) Effect of vaginal estrogen therapy on urinary incontinence in postmenopause. Zentralbl Gynakol 117:77–80
Lose G, Englev E (2000) Oestradiol-releasing vaginal ring versus oestriol vaginal pessaries in the treatment of bothersome lower urinary tract symptoms. BJOG 107:1029–1034
Bhatia NN, Bergman A, Karram MM (1989) Effects of estrogen on urethral function in women with urinary incontinence. Am J Obstet Gynecol 160:176–181
Hilton P, Stanton SL (1983) The use of intravaginal estrogen cream in genuine stress incontinence. Br J Obstet Gynaecol 90:940–944
Sturdee DW, Panay N, International Menopause Society Writing Group (2010) Recommendations for the management of postmenopausal vaginal atrophy. Climacteric 13:509–522
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Capobianco, G., Donolo, E., Borghero, G. et al. Effects of intravaginal estriol and pelvic floor rehabilitation on urogenital aging in postmenopausal women. Arch Gynecol Obstet 285, 397–403 (2012). https://doi.org/10.1007/s00404-011-1955-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-011-1955-1