Abstract
Objective
The purpose of this study was to determine and compare the concentration of reactive oxygen species (ROS) and total antioxident (TAS) in seminal plasma of IVF (in vitro fertilization) and ICSI patients, to establish their effect on sperm quality (count, vitality, HOS, morphology, maturity, DNA strand breaks) and assess the fertilization potential of spermatozoa and IVF/ICSI outcome.
Method
IVF/ICSI patients (n = 48) 26 IVF and 22 ICSI were included in this study. A spermiogram was generated from each patient one-hour post ejaculation and smears were made from each semen sample to evaluate the morphology, sperm maturity (Chromomycin CMA3) and DNA strand breaks (Terminal deoxyribonucleotidyl transferase-mediated dUTP nick-end labelling, TUNEL-assay).
Results
In both groups a negative correlation was found between ROS concentration in seminal plasma and sperm vitality (r = −0.111; P = 0.453); membrane integrity and morphology (−0.141; P = 0.340) and fertilization rate (r = −0.0290; P = 0.045). However, TAS in seminal plasma correlated positive with fertilization rate (r = 0.081; P = 0.584). In addition, an inverse correlation was found between sperm DNA strand breaks (TUNEL-test) and spermatozoa global and progressive motility, vitality, and membrane integrity. Furthermore, the mean percentage of normal condensed spermatozoa (CMA3) was significantly higher (P = 0.0001) in patients undergoing IVF compared to ICSI. Spermatozoa of male ICSI patients were more susceptible to acid denaturation (acridine orange staining) compared to spermatozoa of male IVF patients (P = 0.041). However, ROS concentration was higher in IVF patients compared to ICSI patients (94.73 ± 102.84 vs. 54.78 ± 39.83 μmol/l, whilst TAS levels (1.43 ± 0.28 vs. 1.53 ± 0.22) and fertilization rate (67. 26 vs. 67.26) were similar in both groups.
Conclusion
ROS concentration and other sperm parameters were higher in IVF compared to ICSI patients. TAS concentration was comparable between the two groups. However, the fertilization rate was smilar in IVF and ICSI patients. Therefore, ROS concentration in seminal plasma affects the quality of spermatozoa but does not affect the fertilization rate in IVF/ICSI cycles.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reactive oxygen species (ROS) are free radicals that have a significant role in many of the sperm physiological processes such as capacitation, hyperactivation, and sperm–oocyte fusion [5, 6, 17]. In the presence of polyunsaturated fatty acids such as sperm plasma membrane, ROS triggers a chain of chemical reactions called lipid peroxidation [41, 81]. Furthermore, it has been shown that ROS can damage DNA by causing deletions, mutations, and other lethal genetic effects [55, 75]. Moustafa et al. [55] demonstrated that infertile patients with high ROS levels in their seminal plasma had a higher percentage of apoptosis than normal healthy donors.
There are two main sources of ROS in semen: leukocytes and immature spermatozoa [25]. Gomez et al. [28] demonstrated that levels of ROS produced by spermatozoa were negatively correlated with the quality of sperm in the original semen. Poor sperm quality is linked to increased ROS generation as a consequence of the presence of excess residual cytoplasma. Cytoplasmic droplets after spermiation are thought to be immature and functionally defective [8] and are capable of producing increased amounts of ROS [65]. A strong positive correlation exists between immature spermatozoa and ROS production, which in turn negatively affects the sperm quality [26, 59]. However, the cellular antioxidant mechanism present in almost all tissues and their secretions are likely to quench these ROS and protect against oxidative damage [38]. In addition, many studies have demonstrated that infertile men have an impaired seminal plasma non-enzymatic antioxidant capacity than fertile men, suggesting an association between decreased total antioxidant capacity and male infertility [47, 64]. Pasqualotto et al. [57] reported that levels of antioxidants in seminal plasma from infertile men were significantly lower than levels in fertile controls.
Excessive ROS production that exceeds critical levels can overwhelm all the antioxidant defence systems of spermatozoa and seminal plasma causing oxidative stress [16, 65]. Oxidative stress is a condition in which the elevated levels of ROS damage cells, tissues and organs [53, 62, 63] and is thought to be a mediator of sperm dysfunction and male infertility [2].
The semen quality score and ROS levels in semen samples appear to be strongly associated with male factor infertility, since both these parameters are more sensitive than individual sperm parameters in identifying male factor infertility [56]. A recent meta-analysis by Agarwal et al. [1] found that ROS levels correlated significantly with in vitro fertilization (IVF) rate (estimated overall correlation −0.374, 95% CI, −0.520 to −0.205).
Therefore, the purpose of this study was to determine and compare ROS and TAS concentration in seminal plasma of IVF and ICSI cycles, their effect on sperm quality and consequently the fertilization potential of spermatozoa in both IVF and ICSI cycles.
Materials and methods
Materials
Fourty eight male partners of females undergoing controlled ovarian hyperstimulation for IVF (n = 26) or ICSI (n = 22) were included in this study. Semen was collected by masturbation after 3 days of sexual abstinence.
Methods
Basic sperm parameters evaluation
In all patients, a standard semen analysis was performed according to WHO guidelines [79]. Morphology was assessed according to strict criteria described by Kruger et al. [44]. Semen samples were loaded onto a 45 over 90% discontinuous PureSperm® density gradient (Nidacon International AB, Sweden) and centrifugated at 500×g for 20 min at room temperature. The supernatant (seminal plasma) was immediately separated, and examined before storage to rule out the presence of spermatozoa in the supernatant. The seminal plasma was aliquoted into storage ampoules and stored at −80°C until the assay was performed (within 3 months). Repeated assays on fresh and frozen seminal plasma samples from the same patient showed that samples stored at −80°C did not significantly influence ROS, TAS concentrations in the seminal plasma.
The pellet containing the normal spermatozoa was washed, layered with 1 ml of culture media and incubated at 37°C. The motile sperm was used for injection (ICSI) or insemination (IVF) of oocytes.
Smears were prepared pre and post semen processing for morphological examination and chromatin condensation analysis, visible after staining (Chromomycin CMA3 DNA single strand breaks (acridine orange) staining and by terminal deoxynucleotidyl transferase (TDT) mediated d UTP nick end labelling (Terminal deoxyribonucleotidyl transferase-mediated dUTP nick-end labelling, TUNEL-test).
Eosin test (viability assessment)
The eosin test was performed according to the method described by Eliasson and Treich [20]. Briefly, one drop of semen was mixed on a slide with 1 drop of the 0.5% aqueous yellowish eosin solution and one drop of Nigrosin (10% in distilled water) and covered with a cover slip. After 1–2 min, the dead spermatozoa stained red, whilst live sperm remained unstained. Nigrosin was used as a counter-stain to facilitate visualization of the unstained live cells. One hundred sperm from each semen sample were evaluated per slide.
Assessment of membrane integrity of spermatozoa (hypoosmotic swelling test; HOS-test)
Hypoosmotic swelling test was carried out according to the method described by Jeyendran et al. [37]. One hundred microliters of sperm suspension added to 1 ml of hypo-osmotic solution (equal parts of 150 mOsmol fructose and 150 mOsmol sodium citrate solutions), followed by 60 min incubation at 37°C. After incubation, a minimum of 200 spermatozoa were examined per slide under the light microscope and the percentage of spermatozoa that showed typical tail abnormalities (curly tail) indicative of swelling were calculated.
Assessment of sperm morphology
Morphology was evaluated according to strict criteria described by Kruger et al. [44], and taking into consideration the specific effects, e.g., size alteration of a specific staining method. Briefly, a total of 5 μl of semen were pipetted onto a slide to make thin smears. The slide was air-dried for 3 min. then fixed for 15 s in methyl alcohol. It was stained with Papanicolaou staining. Bright field illumination with a magnification of 100× under an oil immersion objective was used for the evaluation. Two hundred spermatozoa were evaluated in each sample. At least ten high power fields from different areas of the slide were examined. The following criteria for normal spermatozoa were used: (1) smooth, oval head, 5–6 mm in length and 2.5–3.5 mm in diameter; (2) well defined acrosome, 40–70% of the sperm head; (3) mid-piece without defect, slender, axially attached with <1 μm; (4) tailpiece uniform, free from kinks, uncoiled, width thinner than mid-piece, length-45 μm; and (5) cytoplasmic droplets (remnants), which compromise less than half the head area, are acceptable but must be retained in the mid-piece region only. Multiple defects in individual cells were classified as amorphous unless there were only multiple tail defects. The semen samples were either classified as normal (≥14% morphologically normal spermatozoa) or abnormal (<14% morphologically normal spermatozoa).
Assessment of chromatin condensation (chromomycin CMA3) of spermatozoa
CMA3 was performed as previously described by Bianchi et al. [12]. Briefly, a semen aliquot was washed in Dulbecco’s Ca–Mg free phosphate buffer saline (PBS) and centrifuged at 300×g for 10 min. The spermatozoa were washed, fixed in methanol/glacial acetic acid (3:1) at 4°C for 5 min and then spread on clean slides. Each slide was treated for 20 min with 100 μl of CMA3 solution at 25°C in the dark (Sigma St Louis, MO, USA).
Slides were then washed and rinsed in PBS buffer and mounted with buffered glycerol. Fluorochrome was examined using a Zeiss photomicroscope III using a combination of excite dichroic barrier filters of BP 436/10:FT 580:LP 470. A total of 200 spermatozoa were evaluated on each slide. Evaluation of CMA3 staining is done by distinguishing spermatozoa that stain bright yellow (CMA3 positive) from those staining a dull yellow (CMA3 negative) [31].
Assessment of chromatin structure integrity of spermatozoa, single stand breaks (acridine orange-staining)
Ten microliters of each semen sample was spread onto a pre-cleaned slide that was allowed to dry at room temperature. Slides were then fixed overnight in freshly prepared Carnoys solution (three parts methanol and one part glacial acetic acid) and allowed to air dry for a few minutes before being stained with AO (CI 46005; sigma Chemical Company, St Louis, MO, USA).
Slides were stained in AO solution for 5 min at ambient temperature in the dark in an aluminium foil covered 500 ml glass container, gently rinsed with deionised water, and mounted with phenylenediamine mounting medium. The AO slides were kept in the dark and analysed by means of a fluorescence microscope (Leitz, Oberkochen, Germany) equipped with a 490-nm excitation filter and 530-nm barrier. At least 200 spermatozoa were assessed per slide. Spermatozoa with normal DNA content revealed a distinct green fluorescent colour, whereas sperm heads displaying a fluorescent spectrum varying from yellow–green–red were considered as being denatured (Abnormal DNA; 74].
Assessment of sperm DNA stand breaks (TUNEL-assay)
DNA fragmentation was assessed using the TUNEL assay. A detection kit (Apoptosis Detection System Fluorescence; Promega, Mannheim, Germany) was used. The procedure was done according to the manufacturer’s instructions.
Briefly Ejaculated sperm samples were separated from seminal plasma by slow speed centrifugation (250×g; 10 min). The supernatant was discarded, and the remaining pellet was washed in phosphate-buffered saline (PBS), pH 7.4. A droplet of this sperm suspension was smeared onto pre-treated slides (Superfrost; Menzel, Braunschwig, Germany), air dried and fixed by immersion in freshly prepared 4% methanol-free formaldehyde in phosphate-buffered pH 7.4 saline at 4°C for 25 min. The labelling reaction was carried out for 1 h in the dark at 37°C. The slides were then washed in fresh PBS for 25 min at 4°C. The slides were then washed further in fresh PBS for 5 min at room temperature, treated with 2% triton X-100 in PBS for 5 min, and rinsed several time in PBS for another 5 min at room temperature.
After the final rinse, excess water was drained off using a drop of anti fade solution and the sample were covered with a cover slip. Two hundred spermatozoa from various fields of view were immediately analysed with an epifluorescence microscope (Zeiss, Oberkochen, Germany) at 1,000× magnification. The percentage of green-fluorescing sperm (TUNEL-positive) was determined. Negative controls without TdT enzyme were prepared for each batch of analyzed slides.
Measurement of ROS concentrations in seminal plasma
The concentration of ROS was measured by a Colorimetric assay for the quantitative determination of peroxides in EDTA–plasma, serum and other biological fluids (Oxy Stat; Cat. No. BI-5007 Biomedica Medicine product GmbH & Co KG, Wien Austria).
Assay characteristics The Biomedica Oxy Stat assay measures the total concentration of peroxides, which is formed in the propagation-phase of the low-density lipoprotein oxidation process using peroxidase/TMB, 12 × 8 well microtiter plate format. Ninety-six tests per micro titer plate. Reference values EDTA plasma <400 μmoll/l, serum <350 μmol/l. Measuring range 7–6,000 μmol/l; detection limited 7 μmol/l. Sample volume 10 μl/test. Assay time 30 min.
Measurement of TAS concentrations in seminal plasma
Semen antioxidant capacity was determined by a method described by [52], and Rice-Evance and Miller [58], developed for the evaluation of antioxidant capacity in blood plasma. This is a Colorimetric assay using ABTS® (2, 2′-Azino-di-[3ethylbenzthiazoline sulphonate], and a commercially available kit, TAS (Randox Laboratory, Ltd Krefeld, Germany). Range 1.30–1.77 mmol/l plasma [52].
IVF/ICSI procedure
The female partners underwent controlled ovarian hyperstimulation, as described earlier by Hammadeh et al. [30] by pituitary desensitization using gonadotrophin-releasing hormone analogue (Gn-RHa) in the mid luteal phase. Thereafter, stimulation with follicle stimulating hormone (FSH Gonal-F, Serono, Germany) was performed. When at least three follicles were >18 mm in diameter and the serum oestradiol concentration > 800 pg/mL, ovulation was induced by administration of 10,000 IU human chorionic gonadotrophin (hCG). Transvaginal follicle aspiration was carried out 36 h post hCG. Retrieved oocytes were cultured in Vitro Life culture media (Sweden) at 37 h, in 5% CO2. The female patients underwent either IVF (n = 26) or ICSI (n = 22) treatment. Patients that achieved a pregnancy were aged 32.5 ± 4.0 years and patients that were unsuccessful were aged 34.0 ± 3.0 years.
Four hundred and eight oocytes were retrieved (278 from IVF and 200 from ICSI patients). Fertilization was recorded after 18–24 h post pronuclei assessment. Fifteen ongoing pregnancies were achieved (31.3%). The study protocol was approved by the institutional review board, University of Saarlandes, Germany, and written consent was obtained from all patients participating in this study.
Statistical analysis
Statictical analysis was performed using SPSS 11 for Windows Software Package (SPSS Inc., Chicago, IL, USA).
The relationship between ROS, TAS and DNA integrity, motility and morphology and their effect on fertilization rate after IVF and ICSI treatment was analysed. The Mann–Whitney (U test) was used for non-paired data. The results were presented by the mean ± SD values. Correlations were analysed by means of chi-square test or exact Fisher-test. Variants analysis was performed with the Kruskal–Wallis test and Levene-test. Differences were considered statistically significant at P < 0.05.
Results
The mean concentration of spermatozoa (73.9 ± 34.9 mill/ml), motility (42.9 ± 18.6%) and vitality (48.9 ± 23.8%) of IVF patients were significantly higher than in ICSI patients (45.9 ± 41.4 mill/ml; P = 0.013), motility 19.6 ± 11.5%; P = 0.0001) and (30.3 ± 25.1%; P = 0.012) (Table 1). In addition, the mean percentage of normal condensed spermatozoa (CMA3) was significantly higher (P = 0.0001) in patients undergoing IVF (79.88 ± 11.62%) in comparison to ICSI patients (60.6 ± 21.6%). However, spermatozoa of male partners of patients undergoing ICSI therapy were more susceptible tor acid denaturation (acridine orange staining) in comparison to spermatozozoa of male partners undergoing IVF treatment (98.9 ± 2.7 vs. 94.4 ± 7.5; P = 0.041) (Table 1).
ROS was higher in IVF compared to ICSI patients (94.73 ± 102.84 vs. 54.78 ± 39.83 μmol/l, P = 0.093). Whereas, TAS concentration was similar in both groups (1.43.0 ± 0.28 vs. 1.53 ± 0.22 mmol, P = 0.153). However, no significant difference between both groups could be found either for ROS or TAS concentration (Table 1). An inverse correlation was found between ROS concentration in seminal plasma and sperm vitality (r = −0.111; P = 0.453), membrane integrity (r = −0.042; P = 0.778) and morphology (r = −0.141; P = 0.340) as well as between TAS concentration and global (r = −0.313; P = 0.030), progressive motility (r = −0.409; P = 0.004) and morphology after semen processing (r = −0.321; P = 0.026) (Table 2).
However, in both groups a positive correlation was found between chromatin condensation (CMA3) of spermatozoa and sperm concentration (r = 0.437; P = 0.002), global and progressive motility (r = 0.299; P = 0.039), vitality (r = 0.336; P = 0.019), and morphology after semen processing (r = 0.319; P = 0.027). Nevertheless, an inverse correlation was shown between spermatozoa DNA strand breaks (TUNEL) and global motility (r = −0.078; P = 0.600) progressive motility (r = −0.167; P = 0.258), vitality (r = −0.108; P = 0.467) membrane integrity (r = −0.237; P = 0.105) and motility after semen processing (r = −0.111; P = 0.454) (Table 3).
In the IVF group, membrane integrity (HOS-test) correlated negatively with acridine orange test (DNA-strand break). CMA3 Chromatin integrity correlated positively with all investigated sperm parameters. Wherease, DNA strand breaks (TUNEL) correlated negatively with motility before and after sperm processing, vitality (Eosin test) and membrane integrity. In the ICSI group, DNA strand breaks (TUNEL) showed a negative correlation with volume, motility, membrane integrity, density after semen processing and morphology (Table 3).
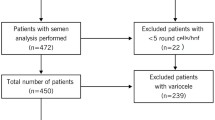
Embryo transfer was performed in 48 patients who were included in this study, 15 patients achieved a pregnancy and 33 patients were unsuccessful (31.25% pregnancy rate) (Fig. 1).
However, there was no significant difference between the mean number of retrieved, fertilized oocytes, fertilization rate between IVF and ICSI groups (10.7 ± 7.2, 5.5 ± 6.6 and 67.3 vs. 9.1 ± 4.1, 5.4 ± 3.6 and 67.9%, respectively). In the IVF group, eleven pregnancies were achieved (42.3%) with a 47.8% implantation rate per embryo transferred. In the ICSI group four pregnancies were achieved (18.2%) with a 40.7% implantation rate per embryo transferred. TAS concentration, morphologically normal spermatozoa and DNA integrity (non-fragmented DNA) as assessed by (Tunel) correlated positively with fertilization rate in both groups (Table 4; Figs. 2, 3, 4).
Discussion
Many studies have reported a connection between oxidative stress and DNA damage [15, 18, 35, 77]. The generation of high level of oxygen radicals can mediate the occurrence of high frequencies of single and double-strand DNA breaks commonly observed in the spermatozoa of infertile men [3, 7, 42]. A significant positive correlation between ROS and DNA fragmentation (r = 0.40; P = 0.02) was reported [10]. Saleh et al. [62] demonstrated a positive correlation between DFI levels and the concentration of ROS in the seminal plasma.
However, seminal plasma provides sperm cells with crucial protection against oxidative attack by different forms of ROS [9]. Lewis et al. [48] reported reduced ascorbate levels in seminal plasma of asthenozoospermic individuals and increased ROS activity. Eskenazi et al. [21] reported that high antioxidant intake was associated with better semen quality, especially motility. In addition, several reports have suggested that increased generation of ROS and/or variation in the levels of antioxidants defence are involved in the occurrence of oligospermia, sperm motility defects and/or abnormal sperm morphology [14, 40, 72].
In the present study, sperm concentration (73.85 ± 34.92 vs. 45.47 ± 41.44 mill/ml, P < 0.013), motility (42.88 ± 18.56 vs. 19.55 ± 11.54%; P < 0.0012), vitality (48.85 ± 23.85 vs. 30.27 ± 25.09%, P = 0.012), DNA integrity (98.04 ± 2.74 vs. 94.41 ± 7.51%, P < 0.041), chromatin condensation (CMA3) (79.88 ± 11.62 vs.60.55 ± 21.64%, P < 0.0001) were significantly better in IVF patients compared to ICSI patients (Table 1). However, ROS concentration was higher in IVF than in ICSI groups (94.73 ± 102.84 vs. 54.78 ± 39.83 μmol/l, P = 0.093). Though these difference was not statistically significant. Whereas, TAS concentration was similar in both groups (1.43 ± 0.28 vs. 153 ± 0.22 mmol/l, P = 0.153) (Table 1).
In addition, ROS in seminal plasma of IVF patients was negatively correlated with semen volume, global motility vitality, membrane integrity, density after semen processing, chromatin condensation, DNA double stand breaks and TAS concentration. However, in ICSI patients, a negative correlation was observed between ROS level and sperm concentration, progressive motility, vitality, membrane integrity, sperm density and motility after semen processing, chromatin condensation and DNA single stand breaks (Table 2). Kessopoulou et al. [39], and Henkel and Schill [32] previously reported a correlation between sperm concentration and increased production of ROS in infertile patients. Zorn et al. [82] found that ROS concentration in seminal fluid was negatively correlated with progressive motility, normal morphology and positively correlated with abnormal sperm head morphology. Iwasaki and Gagnon [36] and Mazzilli et al. [50] also reported a positive correlation between superoxide anion levels and sperm morphology abnormalities. According to the investigation of Aziz et al. [8] ROS positively correlates with the sperm deformity index, calculated by dividing the total number of deformities observed by the number of sperm evaluated [8].
Besides, many studies indicated that DNA damage is associated with abnormalities in conventional semen parameters [11, 13, 51, 61, 76]. Irvine et al. [35] have demonstrated significant negative correlations between semen quality and sperm DNA integrity. They have found an inverse correlation between DNA and sperm concentration (−0.54), motility (−0.38) and morphology (−0.38). Saleh et al. [62] have also shown that DNA damage exhibits a significant inverse correlation with motility (−0.47).
In the present study, DNA integrity was analysed by chromomycin CMA3, acridine orange stain and tunel assay, in order to gain complete insight into fertilization and pregnancy failure due to DNA damage. In both groups, a significantly positive correlation was found between the condensed nucleus chromatin and the sperm motility (r = 0.457; P = 0.001), morphology (r = 0.319; P = 0.027) and vitality (0.336; P = 0.019). DNA strand breaks (Tunel) correlates negatively with the motility (r = −0.078; P = 0.006) and vitality (r = −0.108; P = 0.467), but not with morphology (r = 0.127; P = 0.391) and density (r = 0.020; P = 0.891) (Table 3). These findings in the present study, confirm several previous studies [27, 67, 80] who found an association between DNA damage and sperm motility. The correlation between DNA strand breaks and sperm concentration and total sperm count is consistent with the previous findings [19, 24, 45, 68].
In addition, negative correlations were found between single strand breaks and global motility, vitality and membrane integrity, progressive motility, vitality, membrane integrity and motility after semen processing in IVF group. In spermatozoa of ICSI patients, similar correlations were observed as well as a negative correlation between DNA strand breaks and morphology (Table 3).
In contrast, Chen et al. [13] studied 59 men undergoing IVF treatment, and Donnelly et al. [19] studied 17 fertile men and 40 infertile men undergoing IVF or ICSI. Both studies reported no statistically significant correlation between sperm DNA damage and sperm morphology scored according to strict criteria. Morris et al. [54] reported a positive association between DNA damage and the percentage of sperm with abnormal morphology and a negative association between DNA damage and sperm concentration.
Said et al. [60] suggested that the sperm deformity index (SDI) may be a useful tool to detect the prevalence of sperm DNA damage and to identify potential infertile men. Infertile patients with a semen sample containing a high proportion of morphological abnormalities, specifically cytoplasma droplets, may be more susceptible to develop ROS-mediated sperm DNA damage [60]. Therefore, excessive ROS production is one of the best predictor of low fertilization in conventional IVF [43, 70, 71].
On the otherhand, spermatozoa with DNA fragmentation can fertilise oocytes, but the pregnancy process may be disturbed. This explains the positive association of the DNA strand breaks (Tunel) with fertilization rate (r = 0.108; P = 0.466) and the negative correlation with the number of pregnancies (r = −0.045; P = 0.763) (Table 4). In accordance with these results, Henkel and Schill [33] did not find an association of the percentage of Tunel positive spermatozoa in the ejaculate with fertilization (r = −0.087; P = 0.269), but with pregnancy (r = 0.228; P = 0.006). A further inverse correlation was shown between single strand breaks (acridine orange staining) and fertilization rate after ICSI (r = −0.144; P = 0.522) (Table 4). The mean percentage of morphologically normal spermatozoa correlated positively with fertilization in IVF (r = 0.203; P = 0.319) and ICSI (r = 0.152; P = 0.500) (Table 4; Fig. 3). In accordance with these results, it has been shown by several working groups that DNA damage in ejaculated spermatozoa as determined by TUNEL assay or other techniques such as the comet assay is negatively correlated with fertilization and pregnancy in IVF [34, 54, 73] and ICSI [49] program. It is well documented that there is a negative correlation between defective sperm chromatin structure (DNA-break) and fertility, in vivo [69] and in IVF cycles [22, 23, 29, 45, 46, 78]. Therefore, correct chromatin packaging around the protamine core seems to be necessary for optimal expression of the male gamete fertility potential. However, this condition does not seem mandatory for successful fertilization as demonstrated by ICSI, where normal fertilization and pregnancy rates can be achieved with cells that have not completed spermiogenesis, such as epididymal and testicular spermatozoa [29, 66]. In the present study, ROS are negatively associated with fertilization rate in conventional IVF and ICSI groups (r = −0.267; P = 0.187 and r = −0.241; P = 0.280, respectively). Whereas, a positive correlation was shown between TAS concentration and fertilization rate in both groups (r = 0.081; P = 0.584) (Table 4; Fig. 2). These results could indicate the influence of ROS on the plasma membrane and as a consequence the membrane fusion in IVF or sperm membrane swelling and chromatin decondensation after intracytoplasmic sperm injection. Besides, high levels of semen ROS are associated with low pregnancy rates in vivo [4]. Besides, the results of the present investigation also revealed that the mean number of retrieved oocytes (10.69 ± 7.24 vs. 9.09 ± 4.14 P = 0.346), number of fertilized oocytes (5.46 ± 6.59 vs. 5.36 ± 3.62 P = 0. 951), fertilization rate (67.26 vs. 67.87%) and number of transferred embryos (1.73 ± 1.00 vs. 2.18 ± 0.79, P = 0.097) were similar in both IVF and ICSI groups. However, 15 patients achieved a pregnancy whilst 33 patients were unsuccessful (31.25% pregnancy rate) (Fig. 1). In IVF group (n = 26) 11 pregnancies were achieved (42.3%) with a 47.8% implantation rate per embryo transferred. In ICSI patients (n = 22) four pregnancies were achieved (18.2%) with a 40.7% implantation rate per embryo transferred (Fig. 5) as previously thoroughly described [31].
Relationship between TAS concentration in seminal fluid and pregnancy rate of patients undergoing IVF/ICSI treatment. Higher TAS concentration in seminal plasma indicated increase in pregnancy rate (value represents median and interquartile (25, 75% percentile). No significant difference was shown between the groups
In conclusion ROS and TAS concentration in seminal plasma did not differ significantly between the patients undergoing IVF or ICSI therapy. A negative correlation was shown between ROS concentration in seminal plasma and sperm vitality membrane integrity sperm density Chromatin condensation, DNA single stand breaks in both IVF and ICSI groups. TAS level in seminal plasma was positively correlated to the fertilization rate of patients undergoing IVF/ICSI therapy. However, the potential role of no-enzymatic antioxidants in treatment of male infertility is still questionable.
References
Agarwal A, Prabakaran SA (2005) Mechanism, measurement, and prevention of oxidative stress in male reproductive physiology. Ind J Exp Biol 43:963–974
Agarwal A, Saleh R (2002) Role of oxidants in male infertility: rationale, significance, and treatment. Urol Clin North Am 29:817–827
Aitken RJ, Krausz C (2001) Oxidative stress, DNA damage and the Y chromosome. Reproduction 122:497–506
Aitken RJ, Irvine DS, Wu FC (1991) Prospective analysis of sperm–oocyte fusion and reactive oxygen species generation as criteria for the diagnosis of infertility. Am J Obstet Gynecol 164:542–551
Aitken RJ, Koopman P, Lewis SE (2004) Seeds of concern. Nature 432:48–52
Allmanaeni SS, Naughton CK, Sharma RK et al (2004) Increased seminal reactive oxygen species levels in patients with varicoceles correlate with varicocele grade but not with testis size. Fertil Steril 82:1684–1686
Azam S, Hadi N, Khan NU, Had SM (2003) Antioxidants and prooxidant properties of caffeine, theobromine and xanthine Med Sci Monit 9(9):BB325–BB330
Aziz N, Saleh RA, Sharma RK, Lewis-Jones I, Esfanndiari N, Thomas AJ Jr et al (2004) Novel association between sperm reactive oxygen species production, sperm morphological defects, and the sperm deformity index. Fertil Steril 81:349–354
Balercia G, Armeni T, Mantero F, Principato G, Regoli F (2003) Total oxyradical scavenging capacity toward different reactive oxygen species in seminal plasma and sperm cells. Clin Chem Lab Med 41(1):13–19
Barroso G, Morshedi M, Oehninger S (2000) Analysis of DNA fragmentation, plasma membrane translocation of phosphatidylserine aqnd oxidative stress in human spermatozoa. Hum Reprod 15:1338–1344
Benchaib M, Braun V, Lornage J et al (2003) Sperm DNA fragmentation decrease the pregnancy rate in an assisted reproduction technique. Hum Reprod 18:1023–1028
Bianchi PG, Manicardi GC, Bizzaro D, Bianchi U, Sakkas D (1993) Effect of deoxyribonucleic acid protamination on Fluorochrome staining and in situ nick-translation of murine and human spermatozoa. Biol Reprod 49:1083–1088
Chen SS, Chang LS, Wei YH (2001) Oxidative damage to proteins and decrease of antioxidants capacity in patients with varicocele. Free Rad Biol Med 30:1320–1334
Comhaire FH, Christophe AB., Zalata AA, Dhooge WS, Mahmoud AM, Depuydt CE (2000) The effect of combined conventional treatment, oral antioxidant and essential fatty acids on sperm biology in subfertile men. Prostaglandins Leukot Essent Fatty Acids 63:159–165
Cummins JM, Jequier AM, Kan R (1994) Molecular biology of human male infertility: links with againg, mitochondrial genetics and oxidative stress? Mol Reprod Dev 37:345–362
De Lamirande E, Leclerc P, Gagnon C (1997) Capacitation as a regulatory event that primes spermatozoa for the acrosome reaction and fertilization. Mol Hum Reprod 3:175–193
De Laminardi E, Tsai C, Harakat A, Gagnon C (1998) Involvement of reactive oxygen species in human sperm acrosome reactionn induced by A 23187, lysophosphatidylcholine, and biological fluid ultrafiltrates. J Androl 19(5):585–594
Donnelly ET, McClure N, Lewis SEM (1999) The effect of ascorbat (alpha tocopherol)- supplementation in vitro on DNA integrity and hydrogen peroxide-induced DNA damage in human spermatozoa. Mutagenesis 14:505–512
Donnelly ET, Steele EK, McClure N, Lewis SE (2001) Assessment of DNA integrity and morphology of ejaculated spermatozoa from fertile and infertile men before and after cryopreservation. Hum Reprod 16(6):1191–1199
Eliasson R, Treich L (1971) Supervital staining of human spermatozoa. Fertil Steril 22:134–137
Eskenazi B, Kidd SA, Arks AR, Sloter E, Block G, Wyrobeck AJ (2005) Antioxidants intake is associated with sperm quality in healthy men. Hum Reprod 20(4):1006–1012
Evenson DP, Wixon R (2006) Meta analysis of sperm DNA fragmentation using the sperm chromatin structure assay. Reprod BioMed Online 12:466–472
Evenson DP, Jost LK (2000) Sperm chromatin structure assay is useful for fertility assessment. Method Cell Sci 22(2–3):169–189
Evenson DP, Jost LK., Baer RK, Turner TW, Schrader SM (1991) Individuality of DNA denaturation patterns in human sperm as measured by the sperm chromatin structure assay. Reprod Toxicol 5(2):115–125
Garrido N, Messeguer M, Simon C, Pellicer A, Remoti J (2004) Pro-oxidative and anti-oxidative imbalance in human semen and its relation with male fertility. Asian J Androl 6(1):59–65
Gil-Guzman E, Ollero M, Lopez MC, Sharma RK, Alvarez JG, Thomas AJ, Agarwal A (2001) Differential production of reactive oxygen species by subsets of human spermatozoa at the different stages of maturation. Hum Reprod 16:1922–1930
Giwercman A, Richthoff J, Hjollund H, Bonde JP, Jepson K, Frohm B, Spano M (2003) Correlation between sperm motility and sperm chromation structure assay parameters. Fertil Steril (9):1404–1412
Gomez E, Irvine DS, Aitken RJ (1998) Evaluation of a spectrophotometeric assay for the measurement of malondialdehyde and 4-hydroxyalkenals in human spermatozoa: relationships with semen quality and sperm function. Int J Androl 21:81–94
Hammadeh ME, Stieber M, Haidl G, Schmidt W (1998) Association between sperm chromatin condensation, morphology based on strict criteria, and fertilization, cleavage and pregnancy rates in an IVF program. Andrologia 30:29–35
Hammadeh ME, Ertan K, Baltes S, Breamert B, George P, Rosenbaum P, Schmidt W (2002) Association between interleukin concentration in follicular fluid and intracytoplasmic sperm injection outcome. Am J Reprod Immunol 45:161–167
Hammadeh ME, Radwan M, Al-Hasani S, Micu R, Rosenbaum P, Lorenz M, Schmidt W (2006) Comparison of reactive oxygen species concentration in seminal plasma and semen parameters in partners of pregnant and non-pregnant patients after IVF/ICSI. Reprod BioMed Online 13(5):696–706
Henkel R, Schill WB (1998) Sperm separation in patients with urogenital infectins. Andrologia 30(suppl 1):91–97
Henkel R, Schill WB (2003) Sperm preparation for ART. Reprod Biol & Endocrinol 1:108–123
Host E, lindenberg S, Smith-Jensen S (2000) DNA strand breaks in human spermatozoa: correlation with fertilization in vitro in oligozoospermic men and in men with unexpected infertility. Acta Obstet Gynecol Scan 79:189–193
Irvine DS, Twigg JP, Gordon EL (2000) DNA integrity in human spermatozoa: relationship with semen quality. J Androl 21:33–44
Iwasaki A, Gagnon C (1992) Formation of reactive oxygen species in spermatozoa of infertile patients. Fertil Steril 57:409–417
Jeyendran R, Van der Ven H, Perez-Pelaez M et al (1984) Development of an assay to assess the functional integrity of human sperm membranes and its relationship to other semen characteristics. J Reprod Fertil 70:219–228
Jones R, Mann T, Sherins RJ (1979) Peroxidase breakdown of phospholipids in human spermatozoa: spermicidal effect effects of fatty acid peroxidase and protective action of seminal plasma. Fertil Steril 31:531–537
Kessopoulou E, Tomlinson MJ, Barratt CL, Bolton AE, Cooke ID (1992) Origen of reactive oxygen species in human semen–spermatozoa or leukocytes. J Reprod Fertil 94:463–470
Kim JG, Parthasarathy S (1998) Oxidation and the spermatozoa. Semin Reprod Endocrinol 16:235–239
Kobayashi H, Gil-Guzman E, Mahran AM, Rakesh S, Nelson DR, Thomas AJ Jr, Agarwal A (2001) Quality control of reactive oxygen species measurement by luminal-dependent chemiluminscence assay. J Androl 22(4):568–574
Kodama H, Yamaguchi R, Fukuda J, Kasai H, Tanaka T (1997) Increased oxidative deoxyribonucleic acid daage in the spermatozoa of infertile male patients. Fertil Steril 68(3):519–524
Krausz C, Mills C, Rogers S, Tan SL, Aitken RJ (1994) Stimulation of oxidant generation by human sperm suspensions using phorbol esters and formyl peptides: relationship with motility and fertilization in vitro. Fertil Steril 62:599–605
Krüger TF, Acosta AA, Simmons KF., Swanson RJ, Matta JF, Veek LL et al (1987) New method of evaluating sperm morphology with predictive value for human in vitro fertilization. Urology 30:248–251
Larson KL, DeJonge C, Barnes A, Jost L, Evenson DP (2000) Relationship between assisted reproduction techniques (ART) outcome and status of chromatin integrity as measured by the sperm chromatin structure assay (SCSA). Hum Reprod 15:1717–1722
Larson-Cook KL, Brannian JD, Hansen KA, Kasperson KM, Aamold ET, Evenson DP (2003) Relationship between the outcome of assisted reproductive techniques and sperm DNA fragmentation as measured by the sperm chromatin structure assay. Fertil Steril 80:895–902
Lewis SEM, Boyle PM, McKinney KA et al (1995) Total antioxidant capacity of seminal plasma is different in fertile and infertile men. Fertil Steril 64:868–870
Lewis SEM, Sterling ES, Young IS, Thompson W (1997) Comparison of individual antioxidants of sperm and seminal plasma in fertile and infertile men. Fertil Steril 67:142–147
Lopes S, Jurisicova A, Sun JG, Casper RF (1998) Reactive oxygen species: potential cause for DNA fragmentation in human spermatozoa. Hum Reprod 13:896–900
Mazzilli F, Rossi T, Marchesini M et al (1994) Superoxid anion in human semen related to seminal parameters and clinical aspects. Fertil Steril 62:862–868
McVicar, McClure N, Williamson K, Dalzell LH, Lewis SE (2004) Incidence of FAS positively and deoxyribonucleic acid double-stranded breaks in human ejaculated sperm. Fertil Steril Suppl 1:767–774
Miller NJ, Rice-Evans C, Davies MJ, Gopinathan V, Milner A (1993) A novel method for measuring antioxidants status in premature neonate. Clin Sci 84:407–412
Moller P, Wallin H, Knudsen LE (1996) Oxidative stress associated with exercise, psychological stress and life-style factors. Chem Biol Interact. 27:102 (1) 17–36
Morris ID, Iloti S, Dxon L, Brison DR (2002) The spectrum of DNA damage in human sperm assessed by single cell gel electrophoresis (Comet assay) its relationship fetilizationembryo development. Hum Reprod 17(4):990–998
Moustafa MH, Sharma PK, Thomton J, Mascha E, Abdel-Hafez MA, Thomas AJ Jr et al (2004) Relationship between ROS production, apoptosis and DNA denaturation in spermatozoa from patients examined for infertility. Hum Reprod 19:129–138
Nadella M, Bianchet MA, Gabelli SB, Barrila J, Amzel LM (2005) Structure and activity of the axon guidance protein MICAL. Proceeding National Academy of Science USA 15:102 (46):16830–16835
Pasqualotto FF, Sharma RK, Nelson DR, Thomas AJ, Agarwal A (2000) Relationship between oxidative stress, semen characteristics, and clinical diagnosis in men undergoing infertility investigation. Fertil Steril 73:459–464
Rice-Evance C, Miller NJ (1994) Total antioxidant status in plasma and body fluids. Methods Euzymol 234:279–293
Said TM, Agarwal A, Sharma RK, Mascha F, Sikka SC, Thomas AJJ (2004) Human sperm superoxid anion generation and correlation with semen quality in patients with mal infertility. Fertil Steril 82:871–877
Said TM, Aziz N, Sharma R, Lewis-Jones I, Thomas AJ, Agarwal A (2005) Novel association between sperm deformity index and oxidative stress-induced DNA damage in infertile male patients. Asian J Androl 7:121–126
Saleh RA, Agarwal A, Nelson DR, Nada EA, EL-Tonsy MH, Alvarez JG et al (2002) Increased sperm nuclear DNA damage in normozoospermic infertile men: a prospective study. Fertil Steril 78:313–318
Saleh RA, Agarwal A, Sharma RK et al (2003) Evaluation of nuclear DNA damage in spermatozoa from infertile men with varicocele. Fertil Steril 80:1431–1436
Sharma RK, Agarwal A (1996) Role of reactive oxygen species in male infertility. J Urol 48:835–850
Sharma RK, Pasqualotto FF, Nelson DR, Thomas AJ Jr, Agarwal A (1999) The reactive oxygen species-total antioxidant capacity score in a new measure of oxidative stress to predict male infertility. Hum Reprod 14:2801–2807
Sikka SC (2001) Relative impact of oxidative stress on male reproductive function. Curr Med Chem (Rev) 8(7):851–862
Silber SJ, Van Steirteghem AC, Liu J, Nagy Z, Tournaye H, Devroey P (1995) High fertilization and pregnancy rate after intracytoplasmic sperm injection with spermatozoa obtained from testicle biopsy. Hum Reprod 10(1):148–152
Sills ES, Fryman JT, Perloe M, Michels KB, Tucker MJ (2004) Chromatin fluorescence characteristics and standard semen analysis parameters: correlations observed in andrology testing among 136 males referred for infertility evaluation. J Obstet Gynecol 24(1):74–77
Silver EW, Eskenazi B, Evenson D, Block G, Young S, Wyrobek AJ (2005) Effect of antioxidants intake on sperm chromatin stability in healthy non-smoking men. J Androl 26(4):550–556
Spano M, Bonde JP, Hjollund HI, Kolstad HA, Cordelli E, Leter G (2000) The Danish first pregnancy planner study team. Sperm chromatin damage impairs human fertility. Fertil Steril 73:43–50
Sukcharoen N, Keith J, Irvine DS, Aitken RJ (1996) Prediction of the in-vitro fertilization (IVF) potential of human spermatozoa using sperm function test: the effect of the delay between testing and IVF. Hum Reprod 11:1030–1034
Sukcharoen N, Keith J, Irvine DS, Aitken RJ (1995) Predicting the fertilization potential of human sperm suspension in vitro; importance of sperm morphology and leukocyte contamination. Fertil Steril 63:1293–1300
Suleiman SA, Ali ME, Zaki ZM et al (1996) Lipid peroxidation and human sperm motility: protective role of vitamin E. J Andol 17:530–537
Sun J G, Jurisicova A, Camper RF (1997) Detection of deoxyribonucleic acid fragmentation in human sperm: correlation with fertilization in vitro. Biol Reprod 56:602–607
Tejada RI, Mitchel JC, Mark JJ, Friedman S (1984) A test for the practical evaluation of male fertility by Acridine orange fluorescence. Fertil Steril 42:87–91
Tominga H, Kodama S, Matsuda N et al (2004) Involvement of reactive oxygen speciec (ROS) in the induction of genetic instability by radiation. J Rad Res (Tokyo) 45:181–188
Tomlinson MJU, Moffat O, Manicardi GC et al (2001) Interrelationships between seminal parameters and sperm nuclear DNA damage before and after density gradient centrifugation: implications for assisted conception. Hum Reprod 16:2160–2165
Twigg JP, Fulton N, Gomez E, Irvine DS, Aitken RJ (1998) Analysis of the impact of intracellular reactive oxygen species generation on the structural and functional integrity of human spermatozoa: lipid peroxidation, DNA fragmentation and effectiveness of antioxidants. Hum Reprod 13(6):1429–1436
Virro MR, Larson-Cook KL, Evenson D (2004) Sperm chromatin structure assay (SCSA) parameters are related to fertilization, blastocyst development, and ongoing pregnancy in in vitro fertilization and intracytoplasmic sperm injection cycles. Fertil Steril 81:1289–1295
World Health Organization (1999) WHO laboratory manual for the examination of human sperm and semen–cervical mucus interaction, 4th edn. Cambridge University Press, Cambridge
Wyrobek AJ, Evenson D, Arnheim N, Jabs EW, Young S, Pearson FS, Glaser RL, Thiemann I, Eshkenazi B (2006) Advacing male age increase the frequencies of sperm with DNA fragmentation and certain gene mutations, but not aneuploidy or diploidyes. Proc Natl Acad Sci USA 103(25):9601–9606
Zalata AA, Ahmed AH, Alamaneni SS, Comhaire FH, Agarwal A (2004) Relationship between acrosin activity of human spermatozoa and oxidative stress. Asia J Androl 6(4):313–318
Zorn B, Vidamer G, Meden-Vrtovec H (2003) Seminal reactive oxygen species as predictors of fertilization, embryo quality and pregnancy rates after conventional in vitro fertilization and intracytoplasmic sperm injection. Int J Androl 26:279–285
Acknowledgments
The authors like to thank Dr. Maria Vourliotis for her careful revision of this manuscript regardiong the English language.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hammadeh, M.E., Al Hasani, S., Rosenbaum, P. et al. Reactive oxygen species, total antioxidant concentration of seminal plasma and their effect on sperm parameters and outcome of IVF/ICSI patients. Arch Gynecol Obstet 277, 515–526 (2008). https://doi.org/10.1007/s00404-007-0507-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-007-0507-1