Abstract
In the course of the chronic skin disease psoriasis, where a variety of treatment interventions is available, a strong growth of health economic studies comparing treatment costs and benefits can be noticed. The objective was to identify health economic evaluations of psoriasis treatments that have been published to date. Of particular interest were the mostly used analysis and outcome parameters, the compared treatments, and the question, if available health economic studies may be used to perform a meta-analysis of qualitative findings. A systematic literature search using PubMed Medline, Ovid Medline, and Cochrane Library was performed for articles, published and available until mid of January 2016. Among the key words were the terms “psoriasis” and “cost-effectiveness”. The search resulted in 318 articles without duplicates. Thereof 60 health economic analyses in psoriasis management were identified. Most of these are cost-effectiveness evaluations (45). The clinical parameter PASI (Psoriasis Area Severity Index) is the most often used cost-effectiveness outcome (33) followed by the Dermatology Life Quality Index (DLQI) (6). In case of cost-utility analyses, QALYs (quality-adjusted life-years) were mostly generated with the help of EuroQol five dimensions questionnaire (EQ-5D) (12), which was partly based on PASI and DLQI values. The majority of health economic studies is focusing on the direct medical and non-medical costs without consideration of productivity losses. Almost 70 % of 60 publications were conducted in Europe. Overall, most considered systemic treatments were the biological agents etanercept (36), adalimumab (27), and infliximab (26) followed by ustekinumab (17) and phototherapy (incl. UV-B, PUVA/psoralen combined with UV-A) (14). Comparisons including only topical treatments mostly focused on vitamin D treatment (14), corticosteroids (13), and coal tar products (6) followed by dithranol (5) and tazarotene (4). Given the setting, compared treatments, and study conditions, different results can be found for medical decision-making. Thereby, it can be noted that there are no standards on methods and outcomes measures available. This leads to a very limited comparability of health economic studies and presents no comfortable basis to examine a meta-analysis of health economic results. The presented systematic review shows the need for nationwide data and interpretation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Psoriasis vulgaris is a chronic, non-infectious inflammatory disease, and with a prevalence of 2–3 % one of the most frequent chronic skin diseases worldwide [9, 25, 58, 61].
Moreover, it is associated with numerous comorbidities such as cardiovascular and metabolic diseases as well as chronic immune diseases like rheumatism, chronic bowel disease, and Crohn’s disease. Thus, psoriasis is connected with a high disease burden, in particular marked reductions of patient’s quality of life.
Treatment of psoriasis consists of topical agents, UV treatment, traditional systemic treatments, and since 2003 also of biologics. Costs, especially for systemic treatments including biologics can be overwhelming to patients and the health care system [4].
Chronic diseases, which often require a lifelong treatment, cause significant follow-up costs and, therefore, have a high relevance concerning health economic aspects. While treatment costs as a monetary term are relatively easy to determine, different types to evaluate the benefit of a therapy exist. So far, the most often cited measurements to determine the benefit considering economic objectives are clinical outcomes like the Psoriasis Area and Severity Index (PASI) and cost outcomes, for example reduced follow-up costs [9].
In case of comparing the costs and benefits of treatment options, a cost-effectiveness analysis will be conducted. In cost-effectiveness analyses, outcomes are measured in program-specific units such as clinical parameters like the severity of disease or health outcomes like quality of life, cases cured, and days absent averted. When measuring quality of life in terms of a person’s health, factors to consider are for example pain level, mobility, and general mood. In case of combining outcome parameters of a treatment into a single aggregated summary outcome, the comparison with costs refers to a cost-utility analysis. There are different methods available to generate outcomes of a treatment in benefit units, for example the concept of quality-adjusted life-years (QALYs) [20]. A QALY gives an idea, how many extra months or years of life with a reasonable health-related quality a patient might gain as a result of treatment. Thereby, quality of life can range from 0 (worst possible health) to 1 (the best possible health). With the consideration of costs, cost–utility analyses present the cost of using a treatment to provide a year of the best quality of life available. Cost–utility analyses as kind of cost-effectiveness analyses using the QALYs to measure the effectiveness are expressed as cost per QALY [20, 34, 46].
Focusing on the costs of a cost-effectiveness/-utility analysis, costs are basically differentiated into direct and indirect ones. Direct costs include all resource consumptions that are associated with the medical treatment (e.g., costs of pharmaceuticals) as well as all resources that are used as a consequence of the treatment or disease, known as direct non-medical costs (e.g., for transportation, for household help, or for the educational system). Indirect costs generate all other costs caused indirectly through the treatment or the disease, including in particular disability days [25].
The variety of benefits and costs, economic analysis methods, and modeling techniques (e.g. decision tree, Markov model) indicate a wide range of different health economic evaluation studies. Many aspects have to be considered in planning a health economic study. Hence, there are several guidelines available [20, 37, 47, 64].
In the course of the chronic skin disease psoriasis, where a variety of interventions is available, a strong growth of health economic studies comparing treatment costs and benefits can be noticed. With the help of a systematic literature search, the researchers aimed to identify health economic evaluations of psoriasis treatments. Of particular interest were the following research questions:
-
1.
Which health economic studies on costs and benefits of psoriasis treatment have been published?
-
2.
Which analytical methods were used?
-
3.
Which outcomes were analyzed?
-
4.
Which models can be identified (decision tree, Markov model, or discrete event simulation)?
-
5.
Which treatments were investigated?
-
6.
What gaps exist in health economic studies of psoriasis care?
Materials and methods
Literature search
A literature search using Medline database (via PubMed and Ovid) and Cochrane Library was performed to identify the current situation of available health economic studies, evaluating treatment interventions for psoriasis patients and the respectively used benefit parameters and costs as well as modeling techniques. Search alerts were generated and reviewed for articles published until the 20 January 2016.
For example, the search algorithm in PubMed Medline was as follows:
-
(psoriasis) AND (“cost effectiveness” OR “cost effective” OR “health economic” OR “pharmacoeconomics” OR “pharmacoeconomic” OR “cost benefit” OR “cost utility”).
The literature search was limited to articles in English and German languages.
Study identification
Criteria for selecting the available research were: disease state is psoriasis, and the study is a health economic analysis, comparing costs as well as benefits of at least two interventions. Thus, analyses of one treatment only as well as cost-of-illness studies and cost-minimization analyses, in which no differences in the effectiveness of interventions are assumed, were excluded. In addition, only full texts, which were available for the authors, were considered. Pure abstracts based on a congress presentation were excluded. Finally, the researchers were only interested in original research articles. Thus, review papers were only included if synthesized data were generated.
Study inclusion was determined at two levels. At the first level, articles resulted from the literature search were screened on the basis of their title and abstract. At the second level, when fulfilling the inclusion criteria, full texts were reviewed to identify the applied method of the health economic study, the treatment options being compared, the kind of costs and outcome parameters as well as the modeling technique. Further characteristics of studies, which have been summarized, were the year of publication and the study setting.
Results
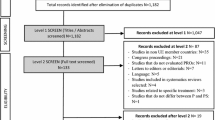
The search with PubMed Medline, Ovid Medline, and Cochrane Library combining “psoriasis” with terms of health economic analyses like “cost effectiveness” and “health economic” resulted in 318 articles without duplicates. Thereof 60 health economic analyses in psoriasis management were identified (see Tables 1 and 2). In total, 258 articles were excluded for not being an health economic study comparing costs and effectiveness of different treatment interventions or disease state other than psoriasis (Fig. 1). The 60 health economic analyses were from countries in Europe (32 including 13 times UK setting), North America (24), Asia (5), and from Brazil (1). A few studies were conducted in a multiple setting.
In addition to the extraction of publication year and country, included articles were compared with each other concerning their treatment comparisons, modeling method, and used costs and effectiveness parameters.
The analyses varied in type of treatment, see Figs. 2 and 3. The most considered systemic treatments were the biological agents etanercept (36), adalimumab (27), and infliximab (26) followed by ustekinumab (17) and phototherapy (incl. UV-B, PUVA/psoralen combined with UV-A) (14). Comparisons including topical treatments mostly focused on vitamin D treatment (14), corticosteroids (13), and coal tar products (6) followed by dithranol (5) and tazarotene (4). Some comparisons of systematic treatments with “basal” or “non-systemic” treatments contained no specific information about the topical treatment.
In addition, the timeline (Fig. 4) shows an overview of the number of topical, UV, and traditional systemic treatments as well as biologics analyzed in cost-effectiveness studies over the years. While the number of UV treatments, topical treatments, and traditional systemic treatments over the years ranges from 0 to 14, the number of biologics considered in health economic studies increases from 3 (2003) to 45 (2015).
A further comparison of included studies shows the distribution of comprised costs and effectiveness parameters in the respective cost-effectiveness and cost-utility analyses (Table 3). If there is a health economic study available, which includes a cost-effectiveness analysis as well as a cost-utility analysis, effectiveness parameters like PASI and DLQI (Dermatology Life Quality Index) were counted in the first row (cost-effectiveness), whereas utility parameters to measure QALYs are shown in the second row (cost-utility). Additionally in this case, modeling type and kind of costs were in a double reporting.
According to Table 3, most health economic analyses in psoriasis management are cost-effectiveness evaluations (45). The clinical parameter PASI is the most often used outcome to compare the costs with the effectiveness of a treatment (33 times in cost-effectiveness analyses). In most articles, the primary outcome was the proportion of patients achieving a 75 % reduction in the PASI score (so-called “PASI 75”). To include the patient-reported health-related quality of life, 5 cost-effectiveness studies used the DLQI. Further effectiveness parameters were disease-free days (DFD) (4), also reported as disease controlled days (DCD), and in 2 cost-effectiveness studies treatment free days (TFD). In the presence of cost-utility studies, the effectiveness is compared with costs in terms of QALYs. The latter were mostly measured by EuroQol five dimensions questionnaire (12), which was partly generated with the help of PASI or DLQI values. For example, a familiar research article to get QALYs with the help of DLQI is the publication of Currie and Conway [17].
Concerning the cost parameters, indirect costs are considered in 9 articles of 60 included hits. The majority of identified health economic studies is focusing on the direct medical and non-medical costs without consideration of productivity losses. As a result, data concerning productivity losses are rarely available in health economic analyses of psoriasis management.
Regarding the modeling type, Markov model is the most often applied kind of model. Discrete-event simulation is not yet applied in psoriasis management.
Discussion
Since it is already known that health economic studies are no new field in psoriasis management [9], the objective of the presented systematic review was to identify health economic analyses concerning psoriasis interventions that have been published to date. Until 20 January 2016, 60 health economic studies comparing costs and benefits were found with the help of the presented systematic literature research. Most of them were examined in Europe, particularly 13 in the UK. There, the National Institute for Health and Care Excellence (NICE) examines independently verified evidence on how well a drug works and whether it provides good value for money. This means whether it is cost-effective. With the use of QALYs as a standard and internationally recognized method to compare different treatments and measure their clinical effectiveness, NICE wants to ensure a fair evaluation of the effectiveness [14]. Therefore, it is not surprising that included evaluations from UK in the present systematic review are in most cases cost-effectiveness analyses using QALYs, known as cost-utility analyses.
It can be noted that since the approvals of the first biological agents by the Federal Drug Association (FDA) in 2003, biologics were directly considered in resulted articles (3). This number increased from 3 in 2003 to 45 in 2015. Biologics are known as considerably more expensive than traditional treatments [24, 55]. Due to their high benefit values for a specific group of psoriasis patients, they are now a permanent standard of treatment [45]. In some years, an update of this review will also yield in cost-effectiveness analyses considering additionally biosimilars. Biosimilars are biotechnologically processed drugs whose amino acid sequence is identical to the original biopharmaceutical [57].
This review of current health economic studies is limited by articles in English and German language with specific keywords. Thus, the search may have missed some relevant articles, published in languages other than English or German and not including selected keywords. Moreover, only full and available publications were considered. Abstracts based on a congress presentation or the like were excluded, given the fact that too little information is presented for this systematic review. Despite these limitations, this article gives an informative overview of health economic analyses, which have been conducted for the comparison of psoriasis treatments.
With regard to all 60 articles and the comparison in Table 3, it was noted that there are no standards on methods and outcome measures available. Even if the review shows that PASI was the most often used measurement in cost-effectiveness analyses of psoriasis interventions, no standardized measurement is used to compare patient-reported quality of life. Therefore, QALYs by EQ-5D or SF-36 (Short Form (36) Health Survey) and the DLQI were used mostly. The literature research yielded also health economic studies, in which no quality of life measurement was used (see Tables 1, 2). According to the increasing importance of the patient perspective, such health economic studies are less comparable with evaluations including patient-reported outcomes.
Concerning the use of costs it was noted that indirect costs are rarely considered in health economic studies of psoriasis treatment. The literature search showed that in only 9 of 60 articles indirect costs were included. However, cost-of-illness studies point out that psoriasis has a high impact on occupational disability [27, 63].
In view of available guidelines [20, 25, 36, 64], a variety of needed content to create a valid health economic evaluation exists. These mostly include details about the study design with objective, methodology, interventions, and target population, the perspective, the validity of data sources, the cost determination as well as the collection of outcome parameters, time horizon, discounting rates, modeling type, sensitivity analyses, and the discussion of outcomes including the presentation of limitations. While focusing on these items, it is possible to assess the quality of an economic analysis. While picking out some articles of the 60 included hits the researchers noted gaps on inter alia data conception, sensitivity analysis, critical discussion of limitations, and inclusion of patient-reported outcomes. Although several articles on topical and systemic agents are published, only a small number of well-conducted health economic studies exists.
In conclusion, the lack of standard on methods and outcome measures leads to a very limited comparability of health economic studies and presents no comfortable basis to examine a meta-analysis of health economic results. Given the setting, compared treatments, and study conditions, different results can be found for medical decision-making. In this context, it should be noted additionally that very heterogeneous requirements by authorities are given. Whereas the NICE requires health outcomes to be expressed in terms of QALYs, the Institute for Quality and Efficiency in Health Care (IQWiG) in Germany refers to the ethical and methodological problems being accompanied by the use of QALYs [36, 49]. The presented systematic review shows the need for nationwide data and interpretation.
References
Affleck AG, Bottomley JM, Auland M et al (2011) Cost effectiveness of the two-compound formulation calcipotriol and betamethasone dipropionate gel in the treatment of scalp psoriasis in Scotland. Curr Med Res Opin 27(1):269–284
Aggarwal K, Khandpur S, Khanna N et al (2013) Pandav CS Comparison of clinical and cost-effectiveness of psoralen + ultraviolet A versus psoralen + sunlight in the treatment of chronic plaque psoriasis in a developing economy. Int J Dermatol 52(4):478–485
Ahn CS, Gustafson CJ, Sandoval LF et al (2013) Cost effectiveness of biologic therapies for plaque psoriasis. Am J Clin Dermatol 14(4):315–326
Alora-Palli MB (2010) A cost-effectiveness comparison of liquor carbonis distillate solution and calcipotriol cream in the treatment of moderate chronic plaque psoriasis. Arch Dermatol 146:918
Anis AH, Bansback N, Sizto S et al (2011) Economic evaluation of biologic therapies for the treatment of moderate to severe psoriasis in the United States. J Dermatol Treat 22(2):65–74
Ashcroft DM, Li Wan Po A, Williams HC, Griffiths CE (2000) Cost-effectiveness analysis of topical calcipotriol versus short-contact dithranol. In the treatment of mild to moderate plaque psoriasis. Pharmacoeconomics 18(5):469–476
Augustin M, Peeter P, Radtke M, Moehling U, Lapp C (2007) Cost-effectiveness model of topical treatment of mild to moderate psoriasis vulgaris in Germany. A comparison of calcipotriol/betamethasone (Daivobet/Dovobet/Taclonex) once daily and a morning/evening non-fix combination of calcipotriol and betamethasone. Dermatology (Basel, Switzerland) 215(3):219–228
Augustin M, Radtke M, van Engen A et al (2009) Pharmacoeconomic model of topical treatment options of mild to moderate psoriasis vulgaris in Germany. J Dtsch Dermatol Ges 7(4):329–338
Augustin M, Radtke MA (2010) Gesundheitsökonomie und Versorgung der Psoriasis. UNI-MED, Bremen
Bergstrom KG, Arambula K, Kimball AB (2003) Medication formulation affects quality of life: a randomized single-blind study of clobetasol propionate foam 0.05 % compared with a combined program of clobetasol cream 0.05 % and solution 0.05 % for the treatment of psoriasis. Cutis 72(5):407–411
Bottomley JM, Auland ME, Morais J, Boyd G, Douglas WS (2007) Cost-effectiveness of the two-compound formulation calcipotriol and betamethasone dipropionate compared with commonly used topical treatments in the management of moderately severe plaque psoriasis in Scotland. Curr Med Res Opin 23(8):1887–1901
Chen S, Shaheen A, Garber A (1998) Cost-effectiveness and cost-benefit analysis of using methotrexate vs Goeckerman therapy for psoriasis. A pilot study. Arch Dermatol 134(12):1602–1608
Chern E, Yau D, Ho JC et al (2011) Positive effect of modified Goeckerman regimen on quality of life and psychosocial distress in moderate and severe psoriasis. Acta Derm Venereol 91(4):447–451
Chi C-C, Wang S-H (2014) Efficacy and cost-efficacy of biologic therapies for moderate to severe psoriasis: a meta-analysis and cost-efficacy analysis using the intention-to-treat principle. Biomed Res Int 2014:862851. doi:10.1155/2014/862851
Colombo G, Bruno M, Girolomoni G, Vena G (2012) Calcipotriol and betamethasone dipropionate in the treatment of mild-to-moderate psoriasis: a cost-effectiveness analysis of the ointment versus gel formulation. Clinicoecon Outcomes Res 4:261-268
Colombo GL, Di Matteo S, Peris K et al (2009) A cost-utility analysis of etanercept for the treatment of moderate-to-severe psoriasis in Italy. Clinicoecon Outcomes Res 1:53–59
Currie CJ, Conway P (2007) PSK11 evaluation of the association between EQ5D utility and dermatology life quality index (DLQI) score in patients with psoriasis. SKIN - Patient Reported Outcomes 10(6):A470–A471
de Portu S, Del Giglio M, Altomare G et al (2010) Cost-effectiveness analysis of TNF-alpha blockers for the treatment of chronic plaque psoriasis in the perspective of the Italian health-care system. Dermatol Ther 23(Suppl 1):S7–S13
Devaux S, Castela A, Archier E et al (2012) Topical vitamin D analogues alone or in association with topical steroids for psoriasis: a systematic review. J Eur Acad Dermatol Venereol: JEADV 26 Suppl 3:52–60
Drummond MF (2014) Methods for the economic evaluation of health care programmes, 3rd edn. Oxford University Press, New York
D’Souza LS, Payette MJ (2015) Estimated cost efficacy of systemic treatments that are approved by the US Food and Drug Administration for the treatment of moderate to severe psoriasis. J Am Acad Dermatol 72(4):589–598
Ellis CN, Reiter KL, Bandekar RR, Fendrick AM (2002) Cost-effectiveness comparison of therapy for psoriasis with a methotrexate-based regimen versus a rotation regimen of modified cyclosporine and methotrexate. J Am Acad Dermatol 46(2):242–250
Feldman SR, Garton R, Averett W, Balkrishnan R, Vallee J (2003) Strategy to manage the treatment of severe psoriasis: considerations of efficacy, safety and cost. Expert Opin Pharmacother 4(9):1525–1533
Feldman SR, Pearce DJ (2004) Management and costs of severe psoriasis: the role of new biologics. Expert Rev Pharmacoecon Outcomes Res 4(5):573–579
Ferrándiz C, García A, Blasco A, Lázaro P (2012) Cost-efficacy of adalimumab, etanercept, infliximab and ustekinumab for moderate-to-severe plaque psoriasis. J Eur Acad Dermatol Venereol 26:768–777
Freeman K, Marum M, Bottomley JM et al (2011) A psoriasis-specific model to support decision making in practice—UK experience. Curr Med Res Opin 27(1):205–223
Fowler JF, Duh MS, Rovba L et al (2008) The impact of psoriasis on health care costs and patient work loss. J Am Acad Dermartol 59(5):772–780
Greiner RA, Braathen LR (2009) Cost-effectiveness of biologics for moderate-to-severe psoriasis from the perspective of the Swiss healthcare system. Eur J Dermatol 19(5):494–499
Hakkaart-van Roijen L, Verboom P, Redekop WK, Touw KR, Rutten FF (2001) The cost effectiveness of tapered versus abrupt discontinuation of oral cyclosporin microemulsion for the treatment of psoriasis. Pharmacoeconomics 19(5 Pt 2):599–608
Hankin CS, Bhatia ND, Goldenberg G et al (2010) A comparison of the clinical effectiveness and cost-effectiveness of treatments for moderate to severe psoriasis. Drug Benefit Trends 22(1):17–27
Hankin CS, Feldman SR, Szczotka A et al (2005) A cost comparison of treatments of moderate to severe psoriasis. Drug Benefit Trends 17(5):200–214
Hartman M, Prins M, Swinkels OQJ et al (2002) Cost-effectiveness analysis of a psoriasis care instruction programme with dithranol compared with UVB phototherapy and inpatient dithranol treatment. Br J Dermatol 147(3):538–544
Heinen-Kammerer T, Daniel D, Stratmann L, Rychlik R, Boehncke WH (2007) Cost-effectiveness of psoriasis therapy with etanercept in Germany. Journal der Deutschen Dermatologischen Gesellschaft = J Ger Soc Dermatol: JDDG 5(9):762–768
Icks A, Chernyak N, Bestehorn K et al (2010) Methoden der gesundheitsökonomischen Evaluation in der Versorgungsforschung. Gesundheitswesen 72(12):917–933
Igarashi A, Kuwabara H, Fahrbach K, Schenkel B (2013) Cost-efficacy comparison of biological therapies for patients with moderate to severe psoriasis in Japan. J Dermatol Treat 24(5):351–355
Institute for Quality and Efficiency in Health Care (IQWiG) (2009) Working paper modelling version 1.0. Available via Dialog. https://www.iqwig.de/download/Working_Paper_Modelling.pdf. Accessed 27 Sept 2013
Institute for Quality and Efficiency in Health Care (IQWiG) (2009) General methods for the assessment of the relation of benefits to costs: version 1.0. IQWIG, Köln
Knight C, Mauskopf J, Ekelund M, Singh A, Yang S, Boggs R (2012) Cost-effectiveness of treatment with etanercept for psoriasis in Sweden. Eur J Health Econ 13(2):145–156
Koek MB, Sigurdsson V, van Weelden H et al (2010) Cost effectiveness of home ultraviolet B phototherapy for psoriasis: economic evaluation of a randomised controlled trial (PLUTO study). BMJ 340:1490
Liu Y, Wu EQ, Bensimon AG et al (2012) Cost per responder associated with biologic therapies for Crohn’s disease, psoriasis, and rheumatoid arthritis. Adv Ther 29(7):620–634
Lloyd A, Reeves P, Conway P, Reynolds A, Baxter G (2009) Economic evaluation of etanercept in the management of chronic plaque psoriasis. Br J Dermatol 160(2):380–386
Marchetti A, LaPensee K, An P (1998) A pharmacoeconomic analysis of topical therapies for patients with mild-to-moderate stable plaque psoriasis: a US study. Clin Ther 20(4):851–869
Marchetti A, Feldman SR, Kimball AB et al (2005) Treatments for mild-to-moderate recalcitrant plaque psoriasis: expected clinical and economic outcomes for first-line and second-line care. Dermatol Online J 11(1):1
Martin S, Feldman SR, Augustin M, Szapary P, Schenkel B (2011) Cost per responder analysis of ustekinumab and etanercept for moderate to severe plaque psoriasis. J Dermatol Treat 22(3):138–143
Nast A, Boehncke WH, Mrowietz U et al (2012) S3—guidelines on the treatment of psoriasis vulgaris (English version). Update. Journal der Deutschen Dermatologischen Gesellschaft = J Ger Soc Dermatol: JDDG 10 Suppl 2:S1–S95
National Institute for Health and Care Excellence (NICE) (2014) Measuring effectiveness and cost effectiveness: the QALY. Available via Dialog. https://www.nice.org.uk/proxy/?sourceurl=http://www.nice.org.uk/newsroom/features/measuringeffectivenessandcosteffectivenesstheqaly.jsp. Accessed 22 May 2014
National Institute for Health and Care Excellence (NICE) (2013) Guide to the methods of technology appraisal 2013. Available via Dialog. https://www.nice.org.uk/article/pmg9/resources/non-guidance-guide-to-the-methods-of-technology-appraisal-2013-pdf. Accessed 24 April 2014
Nelson AA, Pearce DJ, Fleischer AB, Balkrishnan R, Feldman SR (2006) New treatments for psoriasis: which biologic is best? [Review] [42 refs]. J Dermatol Treat 17(2):96–107
Nelson AA, Pearce DJ, Fleischer AB, Balkrishnan R, Feldman SR (2008) Cost-effectiveness of biologic treatments for psoriasis based on subjective and objective efficacy measures assessed over a 12-week treatment period. J Am Acad Dermatol 58(1):125–135
Pan F, Brazier NC, Shear NH, Jivraj F, Schenkel B, Brown R (2011) Cost utility analysis based on a head-to-head Phase 3 trial comparing ustekinumab and etanercept in patients with moderate-to-severe plaque psoriasis: a Canadian perspective. Value Health 4(5):652–656
Papp K, Poulin Y, Barber K et al (2012) Cost-effectiveness evaluation of clobetasol propionate shampoo (CPS) maintenance in patients with moderate scalp psoriasis: a Pan-European analysis. J Eur Acad Dermatol Venereol 26(11):1407–1414
Pearce DJ, Nelson AA, Fleischer AB, Balkrishnan R, Feldman SR (2006) The cost-effectiveness and cost of treatment failures associated with systemic psoriasis therapies. J Dermatol Treat 17(1):29–37
Peeters P, Ortonne JP, Sitbon R, Guignard E (2005) Cost-effectiveness of once-daily treatment with calcipotriol/betamethasone dipropionate followed by calcipotriol alone compared with tacalcitol in the treatment of Psoriasis vulgaris. Dermatology (Basel, Switzerland) 211(2):139–145
Polistena B, Calzavara-Pinton P, Altomare G et al (2015) The impact of biologic therapy in chronic plaque psoriasis from a societal perspective: an analysis based on Italian actual clinical practice. J Eur Acad Dermatol Venereol: JEADV 29(12):2411–2416
Puig L, López-Ferrer A, Vilarrasa E (2014) Incremental cost-effectiveness ratio analysis of biologic treatments for psoriasis at clinically significant evaluation time points. Actas dermo-sifiliográficas 105(10):951–953
Puig L, Lopez-Ferrer A, Vilarrasa E, Garcia I, Fernandez-Del Olmo R (2016) Model for assessing the efficiency of biologic drugs in the treatment of moderate to severe psoriasis for one year in clinical practice in Spain. Actas dermo-sifiliograficas 107(1):34–43
Radtke MA, Augustin M (2014) Biosimilars in psoriasis: what can we expect? Journal der Deutschen Dermatologischen Gesellschaft = J Ger Soc Dermatol: JDDG 12(4):306–312
Radtke MA, Augustin M (2008) Economic considerations in psoriasis management. Clin Dermatol 26:424–431
Riveros BS, Ziegelmann PK, Correr CJ (2014) Cost-effectiveness of biologic agents in the treatment of moderate-to-severe psoriasis. A Brazilian public health service perspective. Value Health Reg Issues 5:65–72
Ruano J, Isla-Tejera B, Jiménez-Puya R (2013) Long-term cost-effectiveness analysis of etanercept and adalimumab for plaque psoriasis not associated with arthritis. Dermatol Ther 3(2):131–142
Sawyer L, Samarasekera EJ, Wonderling D, Smith CH (2013) Topical therapies for the treatment of localized plaque psoriasis in primary care: a cost-effectiveness analysis. Br J Dermatol 168(5):1095–1105
Sawyer LM, Wonderling D, Jackson K, Murphy R, Samarasekera EJ, Smith CH (2015) Biological therapies for the treatment of severe psoriasis in patients with previous exposure to biological therapy: a cost-effectiveness analysis. Pharmacoeconomics 33(2):163–177
Schmitt-Rau K, Rosenbach T, Radtke M, Augustin M (2010) Cost-effectiveness of biological therapy in remission induction of moderate to severe plaque psoriasis. Dermatology 221:236–242
von der Schulenburg JG, Greiner W, Jost F et al (2008) German recommendations on health economic evaluation: third and updated version of the hanover consensus. Value Health 11(4):539–544
Shani J, Harari M, Hristakieva E, Seidl V, Bar-Giyora J (1999) Dead-Sea climatotherapy versus other modalities of treatment for psoriasis: comparative cost-effectiveness. Int J Dermatol 38(4):252–262
Sizto S, Bansback N, Feldman SR, Willian MK, Anis AH (2009) Economic evaluation of systemic therapies for moderate to severe psoriasis. Br J Dermatol 160(6):1264–1272
Spandonaro F, Ayala F, Berardesca E et al (2014) The cost effectiveness of biologic therapy for the treatment of chronic plaque psoriasis in real practice settings in Italy. BioDrugs 28(3):285–295
Staidle JP, Dabade TS, Feldman SR (2011) A pharmacoeconomic analysis of severe psoriasis therapy: a review of treatment choices and cost efficiency. Expert Opin Pharmacother 12(13):2041–2054
Stern RS (1988) The benefits, costs and risks of topical tar preparations in the treatment of psoriasis: considerations of cost effectiveness. Ann Acad Med Singap 17(4):473–476
Terranova L, Mattozzi C, Richetta AG et al (2014) Costs of therapy with biologics in the treatment of moderate to severe plaque psoriasis in the context of the Italian health-care system. G Ital Dermatol Venereol 149(1):131–143
Vañó-Galván S, Gárate MT, Fleta-Asín B (2012) Analysis of the cost effectiveness of home-based phototherapy with narrow-band UV-B radiation compared with biological drugs for the treatment of moderate to severe psoriasis. Actas Dermosifiliogr 103(2):127–137
Villacorta R, Hay JW, Messali A (2013) Cost effectiveness of moderate to severe psoriasis therapy with etanercept and ustekinumab in the United States. Pharmacoeconomics 31(9):823–839
Wang SH, Chi CC, Hu S (2014) Cost-efficacy of biologic therapies for moderate to severe psoriasis from the perspective of the Taiwanese healthcare system. Int J Dermatol 53(9):1151–1156
Woolacott N, Hawkins N, Mason A et al (2006) Etanercept and efalizumab for the treatment of psoriasis: a systematic review. [Review] [77 refs]. Health Technol Assess (Winch, Engl) 10(46):1–233
Related articles recently published in Archives of Dermatological Research (selected by the journal’s editorial staff)
Blome C, Gosau R, Radtke MA, Reich K, Rustenbach SJ, Spehr C, Thaci D, Augustin M (2016) Patient-relevant treatment goals in psoriasis. Arch Dermatol Res 308:69–78
Hawro T, Maurer M, Hawro M, Kaszuba A, Cierpialkowska L, Krolikowska M, Zalewska A (2014) In psoriasis, levels of hope and quality of life are linked. Arch Dermatol Res 306:661–666
Kuster D, Nast A, Gerdes S, Weberschock T, Wozel G, Gutknecht M, Schmitt J (2016) Cost-effectiveness of systemic treatments for moderate-to-severe psoriasis in the German health care setting. Arch Dermatol Res 308:249–261
Radtke MA, Reich K, Spehr C, Augustin M (2015) Treatment goals in psoriasis routine care. Arch Dermatol Res 307:445–449
Reich K, Mrowietz U, Radtke MA, Thaci D, Rustenbach SJ, Spehr C, Augustin M (2015) Drug safety of systemic treatments for psoriasis: results from The German Psoriasis Registry PsoBest. Arch Dermatol Res 307:875–883
Schmitt J, Kuster D (2015) Correlation between Dermatology Life Quality Index (DLQI) scores and Work Limitations Questionnaire (WLQ) allows the calculation of percent work productivity loss in patients with psoriasis. Arch Dermatol Res 307:451–453
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None.
Conflict of interest
Augustin M has served as consultant and/or paid speaker for and/or participated in clinical trials sponsored by companies that manufacture drugs used for the treatment of psoriasis including Abbott, Almirall, Amgen, Biogen, Celgene, Centocor, Janssen-Cilag, Leo, Medac, MSD (formerly Essex, Schering-Plough), Novartis, Pfizer (formerly Wyeth). Gutknecht M and Krensel M don’t have any conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Gutknecht, M., Krensel, M. & Augustin, M. Health economic analyses of psoriasis management: a systematic literature search. Arch Dermatol Res 308, 601–616 (2016). https://doi.org/10.1007/s00403-016-1673-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-016-1673-4