Abstract
Introduction
Chondral and osteochondral lesions of the talus (OLTs) remain a challenging issue with numerous operative treatments proposed to date. The aim of this study was to evaluate 1-year follow-up data in the German Cartilage Registry (KnorpelRegister DGOU).
Methods
Among 401 patients in the database, 114 patients with a complete 1-year Foot and Ankle Outcome (FAOS) score for subscale Pain as the primary variable were included. A total of 12 different surgical treatments were performed. However, 8 techniques were carried out in negligible numbers of patients (n = 1–3), leaving 89 patients treated with the following techniques: arthroscopic antegrade bone marrow stimulation (group A; n = 32), autologous chondrocyte implantation with autologous cancellous bone grafting (group B; n = 9), matrix-augmented bone marrow stimulation (group C; n = 22), and matrix-augmented bone marrow stimulation with autologous cancellous bone grafting (group D; n = 26). Group differences and possible influencing variables such as age and sex were evaluated. Level of significance was set at p < 0.05 for all statistical tests.
Results
All four treatment groups showed significant improvement of the FAOS scores at 1 year postoperatively compared with their preoperative scores. No significant differences were found with respect to score changes among the groups. A positive correlation between FAOS subscale Pain improvement and defect size volume and negative correlations between increasing age and FAOS subscales Sports/Rec and QoL were found. Concomitant ankle stabilization led to greater improvement in FAOS subscales Symptoms and ADL than in patients with no stabilization. FAOS subscale Pain showed greater improvement in women than in men.
Conclusion
All analyzed treatment options were effective for treatment of OLTs. In particular, large defects appeared to benefit from treatment. In the presence of concomitant ankle instability, a stabilizing procedure appeared to have a positive impact on the outcome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chondral and osteochondral lesions of the talus (OLTs) are highly clinically relevant and lead to pain, swelling, and restricted range of motion of the ankle [21]. OLTs involve damage to the articular cartilage with the underlying subchondral bone [4]. A variety of surgical techniques are used for treatment of OLTs, including reparative and replacement techniques [15]. Among the treatments, bone marrow stimulation (BMS) such as microfracturing or drilling is commonly used [3]. Other common treatment options include matrix-augmented BMS (M-BMS) [5, 14], osteochondral autologous transplantation system (OATS) [10], and autologous chondrocyte implantation with or without a matrix (ACI/MACI) and autologous cancellous bone grafting [1, 9]. The selected surgical procedure is mainly based on defect size and localization of the lesion [15]. Furthermore, concomitant chronic ankle instability (CAI) appears to greatly influence patient quality of life [13]. Overall, there are few high-level studies and much missing evidence on the treatment and outcome of OLTs [17].
Registry data are considered more representative for evaluation of clinical results than data obtained in studies. The German Cartilage Registry (KnorpelRegister DGOU) is an observational, nationwide, longitudinal multicenter registry. Entries in the registry are welcomed for any kind of cartilage repair treatment for defects in the hip, knee, and ankle. The general aim is to determine real-life treatment patterns and clinical outcomes of chondral lesions.
The purpose of the present study was to analyze the outcomes of various procedures for treatment of OLTs with possible confounding variables that may influence the outcomes based on the registry data.
Materials and methods
The registry is conducted in accordance with the Declaration of Helsinki and registered at ger-manctr.de (DRKS00005617). Several different local ethics committees have welcomed the implementation of the KnorpelRegister DGOU in their jurisdiction (e.g. Ethics Commission of the Medical Center, University of Freiburg: EK-FR 105/13_130795). All participants provided written informed consent prior to inclusion in the registry.
Study design
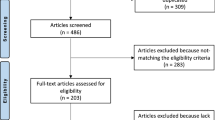
At the time of this data analysis, the KnorpelRegister DGOU included 401 patients in the ankle part of the database. Data collection was performed prospectively using a web-based Remote Data Entry System, which was developed by the Clinical Trials Unit (Freiburg, Germany) as an electronic data entry interface and data management system for clinical studies and other projects in clinical research. For this analysis, patients who underwent surgical treatment for an isolated OLT with a completed 1-year Foot and Ankle Outcome Score (FAOS) score for subscale Pain as the primary variable were included.
Treatment
A total of 114 patients met our inclusion criteria. Twelve different surgical treatments were performed: debridement, arthroscopic antegrade BMS, retrograde BMS, anterograde or retrograde drilling, ACI, ACI with autologous cancellous bone grafting, M-BMS, M-BMS with autologous cancellous bone grafting, cancellous bone grafting alone, OATS, osteochondral fragment refixation, and osteochondral fragment refixation with cancellous bone grafting. Eight techniques were carried out in negligible numbers of patients (n = 1–3) and were thus excluded. As a result, a total of 89 patients were included in the analysis, and divided into the following 4 treatment groups: arthroscopic antegrade BMS (group A; n = 32), ACI with autologous cancellous bone grafting (group B; n = 9), M-BMS (group C; n = 22), and M-BMS with autologous cancellous bone grafting (group D; n = 26). Only the OLTs were treated with these techniques.
Outcome measures
The clinical data analysis for the study was performed using the FAOS score [18]. The FAOS score is a 42-item questionnaire divided into five subscales: Pain, Symptoms, Activities of Daily Living (ADL), Sports and Recreation Ability (Sports/Rec), and Foot and Ankle-related Quality of Life (QoL). A translated and validated version is available in the German language [17].
For analysis of possible confounding variables that may influence the outcome, the following parameters were included: demographic characteristics such as age, sex, body mass index (BMI); and surgical data such as localization of defect, defect size (length × width), defect volume (length × width × depth), primary surgical repair technique for OLT, additional procedures, prior operative procedures at same ankle or OLT.
Statistical analysis
Analyses were conducted using SPSS software (version 24.0; SPSS Inc., Chicago, IL, USA). All outcome parameters were tested for deviation from a normal distribution. Because of the approximately normal distributions, mean and standard deviation (SD) were calculated for continuous variables (i.e. all outcome parameters). A univariate t test (ANOVA) with a post hoc Bonferroni correction was used to analyze the differences between treatment modalities. Possible influencing variables such as age and sex were examined by a bivariate t test and the tau-B correlation coefficient.
Results
Patient characteristics
The 89 patients comprised 42 males and 47 females with a mean age of 33.3 ± 13.2 years and a mean BMI of 26.5 ± 5.4 kg/m2. The mean defect size was 131.6 ± 83.76 mm2 and the mean defect volume was 813.7 ± 841.84 mm3. Thirty-nine (43.8%) patients underwent prior operative treatment of the same ankle, and 27 (31.4%) patients were treated at the same OLT. Simultaneous additional surgical procedures were performed in 21 (23.6%) patients. The characteristics of all patients are summarized in Table 1.
Outcome measures
All four treatment groups showed significant improvement of the FAOS scores at 1 year postoperatively compared with their preoperative scores (p < 0.05) (Fig. 1). Analysis by ANOVA with a post hoc Bonferroni correction revealed no significant differences with respect to score changes among the groups. However, group D had significantly better 1-year follow-up results than group C for FAOS subscale Symptoms (p = 0.041), while group B had significantly better 1-year follow-up results than group C for FAOS subscales Pain (p = 0.023), ADL (p = 0.020), and QoL (p = 0.007). No other significant differences were found among the groups (Fig. 1).
Results of the FAOS subscales pre- and postoperative (first column = preoperative; second column = 1 year postoperative) with respect to the different treatment options included (Group A: arthroscopic antegrade BMS; Group B: autologous chondrocyte implantation with autologous cancellous bone grafting; Group C: matrix-augmented BMS, Group D: matrix-augmented BMS with autologous cancellous bone grafting)
The bivariate t test revealed that patients with concomitant CAI who underwent a stabilizing procedure had significantly inferior preoperative scores in FAOS subscales Symptoms (p < 0.001) and ADL (p = 0.023) than patients without a concomitant stabilizing procedure. However, patients with an additional stabilizing procedure showed greater improvement in the 1-year follow-up results for FAOS subscales Symptoms (p = 0.046) and ADL (p = 0.043) (Table 2). Accordingly, FAOS subscale Pain showed a greater improvement in patients with a stabilizing procedure than in patients without a stabilizing procedure (11.7 ± 17.8 points vs. 20.5 ± 28.1 points), but the difference was not significant (p > 0.05) (Table 2). No significant differences were found for other confounding variables in terms of absolute scores or score changes, except for FAOS subscale Pain, which showed greater improvement in women compared with men (18.1 ± 22.3 points vs. 7.4 ± 15.7, p = 0.027). However, this difference was solely related to the inferior preoperative score in women, because the postoperative scores were similar (women: 75.5 ± 20.8; male: 75.9 ± 20.9).
Correlation analysis revealed that defect size volume was correlated with significantly greater improvement in FAOS subscales Pain, ADL, and Sports/Rec (all p < 0.05). Furthermore, improvements in FAOS subscales Sports/Rec and QoL had significant negative correlations with increasing age (both p < 0.05) (Table 3). No other significant differences were observed for other possible influencing parameters.
Discussion
This is the first study to present 1-year postoperative data from the German Cartilage Registry for OLT. One of the main advantages of this registry is the inclusion of a large number of patients with OLTs in a prospective approach.
All analyzed treatment options were effective for OLT treatment and no differences were found in terms of score improvements. Defect size volume was positively correlated with score improvement in FAOS subscales Pain, ADL, and Sports/Rec, which is encouraging for treatment of these challenging defects. In this context, M-BMS or ACI with autologous cancellous bone grafting appeared more effective than isolated M-BMS, indicating that bone grafting had benefits in filling the defect along with a matrix. This is consistent with recommendations from an expert- and evidence-based consensus statement for scaffold-based therapies [17]. The experts agreed that M-BMS and ACI can be considered for both primary and revision cases and recommended M-BMS in cases where bone grafting may be needed or one-step procedures are preferred. In single cohort studies, several authors reported good clinical results for M-BMS with autologous cancellous bone grafting [7, 8, 20, 22].
Presence of an OLT with concomitant CAI significantly worsens patient quality of life compared with presence of an OLT without CAI [13]. Although patients with concomitant CAI in our study had inferior FAOS scores preoperatively, their improvement in FAOS subscales Symptoms and ADL was significantly better and they achieved postoperative scores that were on the same level as patients without CAI. Thus, in presence of concomitant CAI, a stabilizing procedure should be added to the OLT treatment. Kim et al. analyzed 27 patients with medial compartment ankle osteoarthritis and concomitant CAI and reported a high satisfaction rate after ankle stabilization [12]. They concluded that a stabilizing procedure may play a crucial role in decreasing postoperative pain, considering that patients who underwent concomitant arthroscopic BMS had similar scores to patients who did not.
The influence of age on the outcomes of OLT treatments has been controversial [2, 6]. We found that improvement in FAOS subscales Sports/Rec and QoL was significantly negatively correlated with increasing age. However, whether the scores for these subscales represent less satisfaction in patients with increasing age remains questionable. Although female sex was described as a negative prognostic factor associated with higher risk for reintervention after ACI in the knee [11], female patients showed significantly greater improvement than male patients in FAOS subscale Pain according to our 1-year follow-up data after OLT treatment. However, because this was solely related to the difference in preoperative scores, a clear explanation for this finding is awaited.
This study has several limitations that need to be addressed. Treatment and post-treatment concepts can differ among the centers participating in the registry, and thus the analyzed data were rather heterogeneous. However, the limitations of cohort studies in single centers were negated. The analysis to date included the 1-year follow-up period, which is rather short. Evaluations of mid-term and long-term results will be needed. However, earlier prospective studies showed no significant improvement or worsening of the outcomes after the 1-year follow-up [2]. Lesion size or volume measurements are highly variable among examiners [16, 19], and different instruments or techniques for defect size measurements by surgeons may further bias the results. Thus, the analysis of defect sizes and postoperative outcomes should be interpreted with caution. Uniform measurement techniques need to be established to standardize defect size measurements for OLTs to allow validated correlations among outcomes, treatment strategies, and defect sizes.
Conclusions
All treatment options analyzed were effective for treatment of OLTs. In particular, large defects appeared to benefit from additional bone grafting. In the presence of CAI, a stabilizing procedure appeared to have a positive impact on the outcome. However, more data are necessary to determine possible differences with respect to influencing variables and group differences.
References
Baums MH, Heidrich G, Schultz W, Steckel H, Kahl E, Klinger HM (2006) Autologous chondrocyte transplantation for treating cartilage defects of the talus. J Bone Joint Surg Am 88(2):303–308
Becher C, Driessen A, Hess T, Longo UG, Maffulli N, Thermann H (2010) Microfracture for chondral defects of the talus: maintenance of early results at midterm follow-up. Knee Surg Sports Traumatol Arthrosc 18(5):656–663
Becher C, Thermann H (2005) Results of microfracture in the treatment of articular cartilage defects of the talus. Foot Ankle Int 26(8):583–589
Berndt AL, Harty M (1959) Transchondral fractures (osteochondritis dissecans) of the talus. J Bone Joint Surg Am 41-A:988–1020
Buda R, Castagnini F, Cavallo M, Ramponi L, Vannini F, Giannini S (2016) "One-step" bone marrow-derived cells transplantation and joint debridement for osteochondral lesions of the talus in ankle osteoarthritis: clinical and radiological outcomes at 36 months. Arch Orthop Trauma Surg 136(1):107–116
6Dambrosi R, Maccario C, Serra N, Ursino C, Usuelli FG (2017) Relationship between symptomatic osteochondral lesions of the talus and quality of life, body mass index, age, size and anatomic location. Foot Ankle Surg 10.1016/j.fas.2017.04.011
D’Ambrosi R, Maccario C, Ursino C, Serra N, Usuelli FG (2017) Combining microfractures, autologous bone graft, and autologous matrix-induced chondrogenesis for the treatment of juvenile osteochondral talar lesions. Foot Ankle Int 38(5):485–495
Galla M, Duensing I, Kahn TL, Barg A (2019) Open reconstruction with autologous spongiosa grafts and matrix-induced chondrogenesis for osteochondral lesions of the talus can be performed without medial malleolar osteotomy. Knee Surg Sports Traumatol Arthrosc 27(9):2789–2795
Giannini S, Buda R, Faldini C et al (2005) Surgical treatment of osteochondral lesions of the talus in young active patients. J Bone Joint Surg Am 87(Suppl 2):28–41
Hangody L, Kish G, Modis L et al (2001) Mosaicplasty for the treatment of osteochondritis dissecans of the talus: two to seven year results in 36 patients. Foot Ankle Int 22(7):552–558
Jungmann PM, Salzmann GM, Schmal H, Pestka JM, Sudkamp NP, Niemeyer P (2012) Autologous chondrocyte implantation for treatment of cartilage defects of the knee: what predicts the need for reintervention? Am J Sports Med 40(1):58–67
Kim SW, Jung HG, Lee JS (2020) Ligament stabilization improved clinical and radiographic outcomes for individuals with chronic ankle instability and medial ankle osteoarthritis. Knee Surg Sports Traumatol Arthrosc 28:3294–3300
Korner D, Ateschrang A, Schroter S et al (2020) Concomitant ankle instability has a negative impact on the quality of life in patients with osteochondral lesions of the talus: data from the German Cartilage Registry (KnorpelRegister DGOU). Knee Surg Sports Traumatol Arthrosc 28:3339–3346
Kubosch EJ, Erdle B, Izadpanah K et al (2016) Clinical outcome and T2 assessment following autologous matrix-induced chondrogenesis in osteochondral lesions of the talus. Int Orthop 40(1):65–71
Murawski CD, Kennedy JG (2013) Operative treatment of osteochondral lesions of the talus. J Bone Joint Surg Am 95(11):1045–1054
Oakley SP, Portek I, Szomor Z et al (2003) Accuracy and reliability of arthroscopic estimates of cartilage lesion size in a plastic knee simulation model. Arthroscopy 19(3):282–289
17Pinski JM, Boakye LA, Murawski CD, Hannon CP, Ross KA, Kennedy JG (2016) Low level of evidence and methodologic quality of clinical outcome studies on cartilage repair of the ankle. Arthroscopy, 32(1):214–222 e211
Roos EM, Brandsson S, Karlsson J (2001) Validation of the foot and ankle outcome score for ankle ligament reconstruction. Foot Ankle Int 22(10):788–794
Schafer D, Boss A, Hintermann B (2003) Accuracy of arthroscopic assessment of anterior ankle cartilage lesions. Foot Ankle Int 24(4):317–320
Valderrabano V, Miska M, Leumann A, Wiewiorski M (2013) Reconstruction of osteochondral lesions of the talus with autologous spongiosa grafts and autologous matrix-induced chondrogenesis. Am J Sports Med 41(3):519–527
van Dijk CN, Reilingh ML, Zengerink M, van Bergen CJ (2010) Osteochondral defects in the ankle: why painful? Knee Surg Sports Traumatol Arthrosc, 18(5):570-580
Walther M, Altenberger S, Kriegelstein S, Volkering C, Roser A (2014) Reconstruction of focal cartilage defects in the talus with miniarthrotomy and collagen matrix. Oper Orthop Traumatol 26(6):603–610
Acknowledgements
The German Cartilage Registry (Knorpel- Register DGOU) is initiated by the Working Group Clinical Tissue Regeneration and supported by the Deutsche Arthrose-Hilfe e.V. and the Stiftung Oskar-Helene-Heim.
Funding
For this study, data from the German Cartilage Registry (KnorpelRegister DGOU) were used, which is financially supported by the Deutsche Arthrose-Hilfe e.V. and the Stiftung Oskar-Helene-Heim.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The local ethic committees at all participating clinical centers gave their approval.
Informed consent
Informed consent was obtained from all individual participants included in the registry.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ettinger, S., Gottschalk, O., Kostretzis, L. et al. One-year follow-up data from the German Cartilage Registry (KnorpelRegister DGOU) in the treatment of chondral and osteochondral defects of the talus. Arch Orthop Trauma Surg 142, 205–210 (2022). https://doi.org/10.1007/s00402-020-03631-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-020-03631-z