Abstract
Introduction
Decompression surgery represents the standard operative treatment for lumbar spinal stenosis, but this procedure is often combined with fusion surgery. It is still discussed whether minimal-invasive decompression procedures are sufficient and if they compromise spinal stability as well. The aim of this study was to analyze the effects of different minimal-invasive decompression procedures on the range of motion (ROM) of the decompressed and adjacent segments under preload conditions.
Methods
Fourteen fresh frozen human cadaver lumbar spines (L2–L5) were tested in a spinal testing device with a moment of 7.5 N m in flexion/extension, lateral bending and rotation with and without a preload. The ROM of the decompressed segment L3/4 and the adjacent segments L2/L3 and L4/L5 was measured intact and after creating a gradual defect with resection of the interspinous ligament (ISL), bilateral undercutting decompression, detachment of the supraspinous ligament (SSL) and bilateral medial facetectomy.
Results
The resection of the ISL had no significant effect on the ROM of all segments. Undercutting decompression showed a significant increase in the ROM of all segments during flexion/extension and lateral bending. The detachment of the SSL caused a significant increase of ROM during flexion/extension in the instrumented and adjacent segments. After bilateral medial facetectomy, a decrease of ROM was observed in all directions of motion except flexion/extension with preload.
Conclusions
The results support minimal-invasive procedures for the preservation of spinal stability. Therefore, surgeons can determine which grade of decompression procedure can be performed in the individual patient without requiring additional fusion to maintain spinal stability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lumbar spinal stenosis (LSS) is the most common diagnosis for spine surgery in adults over 65 years, and its surgery rates increased more rapidly than all other major orthopedic procedures between 1980 and 2000 [1, 2]. Despite the increase in these operations, the indications for specific procedures to treat LSS are still lacking [3, 4]. Decompressing the spinal canal with a wide laminectomy is the standard operative treatment of LSS [5–8], but clinical studies have reported spinal instability after these procedures [9, 10]. These clinical findings were supported by biomechanical studies and finite element analyses that demonstrated that the surgical resection of the posterior anatomical structures, such as the facet joints, led to spinal instability [11–15]. These results caused an increasing number of surgeons to combine decompression with lumbar fusion surgery [16], despite a higher risk for complications, mortality and health-care costs, with a more invasive procedure to obtain a comparable clinical outcome [17–19]. Other studies observed increased mechanical stress at the segments adjacent to the fusion, which may cause adjacent segment degeneration (ASD) of the spine [20–22].
To reduce the complications associated with fusion surgery, several authors have evaluated less invasive procedures, such as laminotomy or undercutting decompression, which are intended to adequately decompress the spinal canal without compromising spinal stability [23–29]. Another alternative treatment for LSS is the use of motion-preserving implants, such as interspinous devices that allow resection of the posterior structures to a certain degree, because biomechanical and clinical studies demonstrated a stabilizing effect of these implants during extension motion [30, 31]. No consensus has been reached regarding whether less invasive procedures are sufficient for decompression and to what extent anatomical structures, such as the posterior ligament complex, must be preserved to maintain spinal stability [23, 24]. Several biomechanical cadaver studies evaluated the effect of graded facetectomies on lumbar spine range of motion (ROM) [14, 32, 33], but only a few studies have concentrated on less invasive decompression procedures [12, 34]. Furthermore, a literature search revealed no reports of biomechanical studies on the effect of less invasive decompression procedures on adjacent segments.
The aim of this biomechanical cadaver study was to analyze the effects of graded decompression procedures on the ROM of the instrumented and adjacent segments under preload conditions. Identifying the important anatomical structures, other than the facet joints, that interfere with spinal stability would help the surgeon determine whether certain decompression procedures must be combined with instrumented stabilization.
Materials and methods
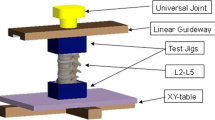
A total of 14 fresh human lumbar cadaver spines (L2–L5) were extracted at the time of the post-mortem autopsy and cryopreserved at −24 °C until the day of testing. There were eight male and six female lumbar spines with a mean age of 62 years. The surrounding soft tissue was dissected, but the intervertebral discs, the ligamentous structures and the capsules were kept intact. Visual inspection and screening via radiography of the specimens revealed no fractures, tumors, loss of disc height, osteophyte formation or scoliosis. The specimens were gradually thawed to room temperature on the day of testing and kept moist with saline solution throughout the testing procedure. The cranial and caudal vertebral bodies were embedded in polymethylmethacrylate (PMMA), and the lower end of the specimens was fixed to the base frame of a spinal testing device (Fig. 1) [35, 36]. A Cardan joint, which allowed free motion in all three dimensions, was connected to the upper end of the specimens. Three swivel joints were integrated into the Cardan joint, including angle sensors, force–torque sensors and drives for the loading. The sensors allowed a continuous assessment of the forces applied to the spine to record possible off-axis loads. The drives consisted of a harmonic drive gearing, an e-coupling and a step motor, which loaded the specimen by rotating the cardan joint at a constant angular velocity until the predetermined set point (7.5 N m in this study) was reached and the direction of motion was reversed. The weight of the cardan joint was balanced with a proportional–integral–derivative (PID)-controlled counterweight. The force–torque sensors continuously assessed the loading, and integrated angle sensors measured the corresponding angle data of the whole specimen. Additionally, the ultrasound-guided CMS 70 System® (Zebris Medical GmbH, Isny, Germany) provided a three-dimensional motion analysis of each spinal segment and was fixed with Kirschner wires to each vertebral body (Fig. 1). The data acquisition of the force–torque sensors, angle sensors and the CMS 70 System® was synchronized at 10 Hz and controlled by the LabVIEW® program (National Instruments Germany GmbH, München, Germany).
In our validated testing routine [35, 36], ±7.5 N m was applied during extension–flexion, lateral bending and axial rotation [37, 38], and the quantity of displacement (ROM) was measured [39]. The testing device also simulated a physiologic preload of 400 N during flexion–extension by inducing an optimized follower load path along the curvature of the spine using a cable and weights [40, 41] (Fig. 1).
After the specimen was fixed in the spinal testing device and the sensors were calibrated, the test routine started with a measurement of the intact spine in flexion–extension. This series of measurements was followed by testing the intact specimen during lateral bending, axial rotation and extension–flexion with a follower load of 400 N. The next series of measurements was performed with the same procedure after creating a gradual defect in the L3/L4 segment. The first defect was created by resecting the interspinous ligament (ISL). A bilateral posterior undercutting was performed using a flavectomy and partial resection of the cranial and caudal lamina. For the next defect, the supraspinous ligament (SSL) was detached from the spinous processes of L3 and L4. Finally, a bilateral medial facetectomy was performed.
The observed ROM values were displayed by graphs, and the resulting hysteresis loops were analyzed. In all three segments, the ROM of each decompression step was compared against each other and against the intact segment during all directions of motion.
Explorative testing for a normal distribution revealed that a non-parametric analysis should be performed. The statistical analysis included the Wilcoxon and Friedman tests, which were conducted with SPSS for Windows® Version v. 15.0 (SPSS Inc., Chicago, IL, USA), and a p value of <0.05 was considered significant.
Results
The resection of the ISL did not cause a significant increase in the median ROM during flexion–extension (the ROM increased from 6.4° to 6.8° in the L3/4 segment, from 8.2° to 8.4° in the cranial L2/3 segment and from 5.4° to 6.2° in the caudal L4/5 segment) (Fig. 2). After applying a follower load of 400 N, the ROM changed to 6° in the L3/4 segment, 6.6° in the L2/3 segment and 5.6° in the L4/5 segment, but these changes were not significant (Fig. 5).
The bilateral undercutting decompression significantly increased the median ROM of the L3/4 segment (10.5°), L2/3 segment (12.2°) and L4/5 segment (11.1°) during flexion–extension (Fig. 2). During lateral bending, the median ROM significantly increased from 7.2° to 7.7° in the L3/4 segment, from 6.2° to 8.1° in the L2/3 segment and from 8.6° to 10.7° in the L4/5 segment (Fig. 3). During the axial rotation, the median ROM did not significantly change in the L3/4 segment (3.7°–3.7°), the L2/3 segment (4.9°–5.1°) or the L4/5 segment (4.3°–4.6°) (Fig. 4). The follower load application during flexion–extension also significantly increased the ROM to 7.4° in the L3/4 segment, 10.5° in the L2/3 segment and 8.2° in the L4/5 segment (Fig. 5).
The detachment of the SSL caused a significant increase in the ROM to 11.6° in the L3/4 segment, 15.3° in the L2/3 segment and 11.6° in the L4/5 segment during flexion–extension (Fig. 2). During lateral bending, the ROM changed to 7.6° in the L3/4 segment and 10.9° in the L4/5 segment, which were both not significant, but a slightly significant increase to 8.8° in the L2/3 segment was observed (p < 0.042) (Fig. 3). The increased ROM during axial rotation to 4.2° in the L3/4 segment, 5.1° in the L2/3 segment and 6.7° in the L4/5 segment (Fig. 4) was also slightly significant (p < 0.042). Applying the follower load changed the ROM in the L3/4 segment to 7.9°, 11° in the L2/3 segment and 8.2° in the L4/5 segment during flexion–extension, which were not significant changes (Fig. 5).
The bilateral medial facetectomy significantly decreased the ROM during flexion–extension to 9.4° in the L3/4 segment, 12.6° in the L2/3 segment and 11.1° in the L4/5 segment (Fig. 2). During lateral bending, the ROM changes in the L3/4 segment (8.1°) and L4/5 segment (10.7°) were not significant, but the ROM significantly decreased to 7.8° in the L2/3 segment (Fig. 3). During the axial rotation, the ROM significantly decreased to 4° in the L3/4 segment and 4.9° in the L4/5 segment, but a significant change in ROM was not observed in the L2/3 segment (5.4°) (Fig. 4). The follower load had no significant influence on the ROM in the L3/4 segment (8°), the L2/3 segment (10.6°) or the L4/5 segment (8.5°) (Fig. 5).
The data showed that the resection of the ISL had no significant effect on the ROM of all segments; however, the undercutting decompression caused a significant increase in the ROM of all segments during the flexion–extension, with and without the follower load, and during lateral bending. Detaching the SSL caused a significant increase in the ROM of all segments during flexion–extension, but not after applying the follower load. A slightly significant increase was also observed during lateral bending in the L2/3 segment (+0.7°) and during the axial rotation of all segments (+0.02° in the L2/3 segment, +0.52° in the L3/4 segment, +1.95° in the L4/5 segment); however, increases of 0.5° lie within the possible measurement errors and should be interpreted critically.
Unexpectedly, the data showed a significant decrease in ROM after a bilateral medial facetectomy of all segments during the flexion–extension, in the L2/3 segment during lateral bending and in the L3/4 and L4/5 segments during axial rotation. This decrease was not observed upon applying the follower load.
Discussion
Biomechanical and clinical studies showed that the alteration of the facet joint integrity severely compromises spinal stability. Abumi et al. [32] evaluated the ROM of human cadaver spine segments after dividing the interspinous and SSLs and after a medial and total facetectomy. In contrast to the present study, dividing both ligaments had no influence on the ROM of the instrumented segment, but the authors found a slight increase in the ROM during flexion after a medial facetectomy and a significant increase in the ROM after a total facetectomy. The authors concluded that dividing the ISL and SSL and the medial facetectomy did not interfere with the spinal stability, but total facetectomy did. Fuchs et al. [33] performed a similar biomechanical study comparing the ROM of human cadaver lumbar spines after a graded facetectomy and implantation of an interspinous device (without resecting the ISL or SSL). The authors observed similar, but minor, ROM changes as Abumi et al. and concluded that the SSL played an important role in spinal stability. The stabilizing function of the SSL during flexion–extension was also observed in biomechanical porcine cadaver studies and finite element studies. Anasetti et al. [42] compared the ROM of human cadaver L4/5 segments with or without a resected SSL after implanting an interspinous device. After resecting the SSL, the authors found a higher ROM during flexion–extension, compared to the intact state. Tai et al. [43] compared the intervertebral disc displacement of human cadaver L4/5 segments during flexion–extension after two decompression procedures. In one group, a laminotomy with SSL preservation was performed, and a laminectomy with flavectomy and resection of the SSL was performed in the other group. During flexion, the intervertebral disc displacement was higher after resecting the SSL. This observation is supported by a finite element study conducted by Lee et al. [44], in which resecting the ligaments in the L2/3 segment caused an unstable flexion motion that might lead to spondylolisthesis. The experimental data of the present study also support the hypothesis that the SSL has an important role in preserving spinal stability. After resecting the ISL, a change in the ROM of the instrumented and adjacent segments was not observed during flexion–extension, but detaching the SSL caused a significant increase in ROM in the instrumented and adjacent segments during flexion–extension. Similarly, Chen et al. [45] observed a destabilization of the adjacent segments after decompression in their biomechanical study. The authors evaluated porcine lumbar spines after a partial and total laminectomy of L3/L4. The investigators removed the inferior portion of the L4 spinous process for the partial laminectomy and removed the entire spinous process L4 during complete laminectomy. During flexion, the intervertebral displacement on the adjacent discs was significantly larger after a complete laminectomy than after a partial laminectomy. The authors concluded that removing the anchoring point of the SSL leads to spinal instability at the adjacent segments with a higher risk of ASD development.
Clinical studies focused on radiological changes did not observe spinal instability after different undercutting decompression procedures [24, 46], but an actual biomechanical study showed a significant segmental instability. Schulte et al. [34] compared different functional spinal units (L1/2, L2/3, L3/4 and L4/5) with each other after bilateral undercutting decompression and implanting two dynamic implants into the individual segments. The decompression procedure consisted of a flavectomy, transection of the ISL and SSL and partial resection of the cranial and caudal lamina. After decompression, the authors found a significant increase in the ROM during flexion–extension (26 %) and minor increases in the ROM during lateral bending (6 %) and axial rotation (12 %). In the present study, similar results were observed by comparing the same decompressed (L3/4) and adjacent lumbar segments (L2/3, L4/5) with each other. Here, the undercutting decompression caused a significant increase in the ROM during the flexion–extension and lateral bending in the instrumented and adjacent segments.
In contrast to previous biomechanical studies, the hemifacetectomy in the present study decreased the ROM in the measured direction of motion. Sharma et al. [14] performed a finite element study and reported that only the orientation of the facet joints was responsible for resisting extension and anterior displacement. Therefore, the authors concluded, similar to Abumi et al. and Fuchs et al., that the selective removal of the facets would not compromise the segmental stability. However, actual biomechanical studies have shown that the capsule and surrounding ligaments provide segmental stability during flexion [47, 48]. Decompression with a partial facetectomy should cause substantial changes in segmental motion [49]. These changes were investigated by Natarajan et al. [50] using a finite element study. In this study, removing 75 % of one facet joint not only produced segmental instability, but also significant coupled motions. Another finite element simulation conducted by Lee et al. [44] also observed coupled motions and altered paths of loading by removing more than 75 % of the facets. This finding could explain the decrease in ROM observed after hemifacetectomy in our study, because the hemifacetectomy did not cause a decrease in ROM in all segments during the application of an optimized follower load. The use of a follower load records more realistic values in a biomechanical study because of the lack of actively stabilizing muscles in cadaver spines, which makes it difficult to transfer these results to in vivo conditions. However, calculating the center of rotation (COR) could help identify coupled motions or altered paths of loading in biomechanical studies and will be the purpose of future studies [51].
Nevertheless, the present biomechanical study identified important anatomical structures, including the SSL, for spinal stability. Resecting these structures may cause adjacent segment instability and subsequent ASD.
Before surgical interventions, the extent of decompression and additional instrumentation should be considered carefully for each patient, because the preservation of physiological motion and spinal stability could reduce the ASD and improve the long-term clinical outcome of patients. Still, further prospective randomized controlled trials are needed for evidence-based decompression procedures.
References
Taylor VM, Deyo RA, Cherkin DC et al (1994) Low back pain hospitalization: recent United States trends and regional variations. Spine 19(11):1207–1212
Deyo RA, Gray DT, Kreuter W et al (2005) United States trends in lumbar fusion surgery for degenerative conditions. Spine 30(12):1441–1445
Irwin ZN, Hilibrand A, Gustavel M et al (2005) Variation in surgical decision making for degenerative spinal disorders. Part I: lumbar spine. Spine 30(19):2208–2213
Katz JN, Lipson SJ, Lew RA et al (1997) Lumbar laminectomy alone or with instrumented or noninstrumented arthrodesis in degenerative lumbar spinal stenosis: patient selection, costs, and surgical outcomes. Spine 22(10):1123–1131
Airaksinen O, Herno A, Turunen V et al (1997) Surgical outcome of 438 patients treated surgically for lumbar spinal stenosis. Spine 22:2278–2282
Atlas SJ, Deyo RA, Keller RB et al (1996) The Maine Lumbar Spine Study, Part III. 1-year outcomes of surgical and nonsurgical management of lumbar spinal stenosis. Spine 21:1787–1795
Iguchi T, Kurihara A, Nakayama J et al (2000) Minimum 10-year outcome of decompressive laminectomy for degenerative lumbar spinal stenosis. Spine 25:1754–1759
Katz JN, Lipson SJ, Larson MG et al (1991) The outcome of decompressive laminectomy for degenerative lumbar stenosis. J Bone Joint Surg Am 73:809–816
Johnsson KE, Willner S, Johnsson K (1986) Postoperative instability after decompression for lumbar spinal stenosis. Spine 11:107–110
Nakai O, Ookawa A, Yamaura I (1991) Long-term roentgenographic and functional changes in patients who were treated with wide fenestration for central lumbar stenosis. J Bone Joint Surg Am 73:1184–1191
Detwiler PW, Spetzler CB, Taylor SB et al (2003) Biomechanical comparison of facet-sparing laminectomy and Christmas tree laminectomy. J Neurosurg 99(2 Suppl):214–220
Hamasaki T, Tanaka N, Kim J et al (2009) Biomechanical assessment of minimally invasive decompression for lumbar spinal canal stenosis: a cadaver study. J Spinal Disord Tech 22(7):486–491
Quint U, Wilke HJ, Loer F et al (1998) Functional sequelae of surgical decompression of the lumbar spine—a biomechanical study in vitro. Z Orthop 136(4):350–357
Sharma M, Langrana NA, Rodriguez J (1995) Role of ligaments and facets in lumbar spinal stability. Spine 20:887–900
Zander T, Rohlmann A, Klöckner C et al (2003) Influence of graded facetectomy and laminectomy on spinal biomechanics. Eur Spine J 12(4):427–434
Deyo RA, Nachemson A, Mirza SK (2004) Spinal-fusion surgery—the case for restraint. N Engl J Med 350:722–726
Ciol MA, Deyo RA, Howell E et al (1996) An assessment of surgery for spinal stenosis: time trends, geographic variations, complications, and reoperations. J Am Geriatr Soc 44(3):285–290
Fischgrund JS, Mackay M, Herkowitz HN et al (1997) Degenerative lumbar spondylolisthesis with spinal stenosis: a prospective, randomized study comparing decompressive laminectomy and arthrodesis with and without spinal instrumentation. Spine 22(24):2807–2812
Fritzell P, Hagg O, Nordwall A, Swedish Lumbar Spine Study Group (2003) Complications in lumbar fusion surgery for chronic low back pain: comparison of three surgical techniques used in a prospective randomized study. Eur Spine J 12(2):178–189
Ekman P, Möller H, Shalabi A, Yu YX, Hedlund R (2009) A prospective randomised study on the long-term effect of lumbar fusion on adjacent disc degeneration. Eur Spine J 18(8):1175–1186
Lee CS, Hwang CJ, Lee SW, Ahn YJ, Kim YT, Lee DH, Lee MY (2009) Risk factors for adjacent segment disease after lumbar fusion. Eur Spine J 18(11):1637–1643
Yang JY, Lee JK, Song HS (2008) The impact of adjacent segment degeneration on the clinical outcome after lumbar spinal fusion. Spine 33:503–507
Delank KS, Eysel P, Zöllner J et al (2002) Undercutting decompression versus laminectomy. Clinical and radiological results of a prospective controlled trial. Orthopade 31(11):1048–1056
Kleeman TJ, Hiscoe AC, Berg EE (2000) Patient outcomes after minimally destabilizing lumbar stenosis decompression: the “Port-Hole” technique. Spine 25:865–870
Thomé C, Zevgaridis D, Leheta O et al (2005) Outcome after less-invasive decompression of lumbar spinal stenosis: a randomized comparison of unilateral laminotomy, bilateral laminotomy, and laminectomy. J Neurosurg Spine 3(2):129–141
Tsai RY, Yang RS, Bray RS Jr (1998) Microscopic laminotomies for degenerative lumbar spinal stenosis. J Spinal Disord 11:389–394
Weiner BK, Walker M, Brower RS et al (1999) Microdecompression for lumbar spinal canal stenosis. Spine 24:2268–2272
Yamazaki K, Yoshida S, Ito T et al (2002) Postoperative outcome of lumbar spinal canal stenosis after fenestration: correlation with changes in intradural and extradural tube on magnetic resonance imaging. J Orthop Surg 10:136–143
Guiot BH, Khoo LT, Fessler RG (2002) A minimally invasive technique for decompression of the lumbar spine. Spine 27:432–438
Kong DS, Kim ES, Eoh W (2007) One-year outcome evaluation after interspinous implantation for degenerative spinal stenosis with segmental instability. J Korean Med Sci 22:330–335
Wilke HJ, Drumm J, Häussler K et al (2008) Biomechanical effect of different lumbar interspinous implants on flexibility and intradiscal pressure. Eur Spine J 17(8):1049–1056
Abumi K, Panjabi MM, Kramer KM et al (1990) Biomechanical evaluation of lumbar spinal stability after graded facetectomies. Spine 15(11):1142–1147
Fuchs PD, Lindsey DP, Hsu KY et al (2005) The use of an interspinous implant in conjunction with a graded facetectomy procedure. Spine 30(11):1266–12672
Schulte TL, Hurschler C, Haversath M et al (2008) The effect of dynamic, semi-rigid implants on the range of motion of lumbar motion segments after decompression. Eur Spine J 17:1057–1065
Hartmann F, Gercek E, Dietz SO et al (2011) Development of a multisegmental test body to calibrate and validate studies with spinal testing devices with follower load. Biomed Tech (Berl) 56(2):99–105
Hartmann F, Dietz SO, Hely H et al (2011) Biomechanical effect of different interspinous devices on lumbar spinal range of motion under preload conditions. Arch Orthop Trauma Surg 131(7):917–926
Goel VK, Panjabi MM, Patwardhan AG et al (2006) American Society for Testing and Materials. Test protocols for evaluation of spinal implants. J Bone Joint Surg Am 88(Suppl 2):103–109
Wilke HJ, Wenger K, Claes L (1998) Testing criteria for spinal implants: recommendations for the standardization of in vitro stability testing of spinal implants. Eur Spine J 7:148–154
Panjabi MM (1988) Biomechanical evaluation of spinal fixation devices: I. A conceptual framework. Spine 13(10):1129–1134
Dreischarf M, Zander T, Bergmann G et al (2010) A non-optimized follower load path may cause considerable intervertebral rotations. J Biomech 43(13):2625–2628
Patwardhan AG, Havey RM, Meade KP et al (1999) A follower load increases the load-carrying capacity of the lumbar spine in compression. Spine 24(10):1003–1009
Anasetti F, Galbusera F, Aziz HN et al (2010) Spine stability after implantation of an interspinous device: an in vitro and finite element biomechanical study. J Neurosurg Spine 13(5):568–575
Tai CL, Hsieh PH, Chen WP et al (2008) Biomechanical comparison of lumbar spine instability between laminectomy and bilateral laminotomy for spinal stenosis syndrome—an experimental study in porcine model. BMC Musculoskelet Disord 9:84
Lee KK, Teo EC, Qiu TX et al (2004) Effect of facetectomy on lumbar spinal stability under sagittal plane loadings. Spine 29(15):1624–1631
Chen LH, Lai PL, Tai CL, Niu C, Fu TS, Chen WJ (2006) The effect of interspinous ligament integrity on adjacent segment instability after lumbar instrumentation and laminectomy—an experimental study in porcine model. Biomed Mater Eng 16:261–267
Sanderson PL, Getty CJM (1996) Long-term results of partial undercutting facetectomy for lumbar lateral recess stenosis. Spine 1:1352–1356
Goel VK, Winterbottom JM, Weinstein JN et al (1987) Load sharing among spinal elements of a motion segment in extension and lateral bending. J Biomech Eng 109(4):291–297
Little JS, Khalsa PS (2005) Human lumbar spine creep during cyclic and static flexion: creep rate, biomechanics, and facet joint capsule strain. Ann Biomed Eng 33(3):391–401
Serhan HA, Varnavas G, Dooris AP et al (2007) Biomechanics of the posterior lumbar articulating elements. Neurosurg Focus 22(1):E1
Natarajan RN, Andersson GB, Patwardhan AG et al (1999) Study on effect of graded facetectomy on change in lumbar motion segment torsional flexibility using three-dimensional continuum contact representation for facet joints. J Biomech Eng 121(2):215–221
Schmidt H, Heuer F, Claes L et al (2008) The relation between the instantaneous center of rotation and facet joint forces—a finite element analysis. Clin Biomech (Bristol, Avon) 23(3):270–278
Acknowledgments
The authors would like to thank the Institute of Medical Biostatistics, Epidemiology and Informatics of the University Medical Center of the Johannes Gutenberg University Mainz for their help with the statistical analysis in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hartmann, F., Janssen, C., Böhm, S. et al. Biomechanical effect of graded minimal-invasive decompression procedures on lumbar spinal stability. Arch Orthop Trauma Surg 132, 1233–1239 (2012). https://doi.org/10.1007/s00402-012-1543-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-012-1543-2