Abstract
Introduction
Treatment of displaced proximal humerus fractures remains challenging. The introduction of locking plates has renewed interest in treating these fractures with joint-preserving techniques rather than hemiarthroplasty, but high complication rates are still reported. Avascular necrosis is not solely dependent on the initial fracture pattern, but can also result from intraoperative and postoperative vascular insults.
Method
We describe a technique to minimize disruption of humeral head blood supply and maximize fracture fixation. A total of 34 patients with complex proximal humerus fractures were treated with a locking plate and endosteal implant through an anterolateral approach and followed for an average of 66 weeks to determine the rates of avascular necrosis.
Results
No patient suffered complete osteonecrosis (0%) and only one patient suffered partial necrosis (2.8%) of the humeral head. The length of the posteromedial hinge was not predictive of this complication.
Conclusion
Use of the anterolateral approach and endosteal augment of a lateral locking plate can minimize avascular necrosis following proximal humerus fracture.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fractures of the proximal humerus account for 4–5% of all fractures; 11% of these are three- and four-part injuries [4]. Despite this frequency, treatment remains challenging. With the advent of locked plating, there has been a recent trend toward plate osteosynthesis and away from hemiarthroplasty in treating these fractures. This choice often hinges upon the anticipated residual blood supply to the humeral head and the potential risk of avascular necrosis. There is, however, little evidence to guide this decision. The anterolateral branch of the anterior humeral circumflex artery has been considered the main nutrient artery of the proximal humerus [15]. However, disruption of this vessel occurs in 80% of all proximal humeral fractures [6] and clinical rates of osteonecrosis cannot be reconciled with this pattern of vascular anatomy. Recent studies have shown a dominant role for the posterior humeral circumflex artery and maintenance of humeral head perfusion even in displaced three- and four-part fractures [5, 18]. Reported rates of postoperative avascular necrosis are highly variable, however (3–37%), and preoperative prediction of this complication has been inaccurate [2, 17]. Intraoperative and postoperative factors may account for this variability rather than fracture pattern alone. Perfusion can be disrupted during dissection, manipulation of fracture pieces, osteosynthesis and thrombosis in the postoperative period [2]. The purpose of this study was to determine the rate of osteonecrosis after treatment of displaced proximal humerus fractures through a minimally invasive anterolateral approach using bicolumnar fixation with a lateral locking plate and an endosteal implant. We hypothesize that this technique will minimize surgical disruption of head perfusion and maximize stability to allow postoperative reperfusion when necessary, significantly reducing avascular necrosis.
Methods
After approval from the institutional review board, during the period of May 2006–March 2009, all patients with displaced fractures of the proximal humerus seen at a level 1 trauma center were treated with open reduction and internal fixation using an anterolateral approach, and fixation with a lateral locking plate and an endosteal augment. Any patient with a displaced fracture of the proximal humerus was eligible for inclusion except those with isolated fractures of a single tuberosity. Except for the exclusion of tuberosity fractures, fracture pattern was not used as a determinant of treatment. All patients underwent standard shoulder trauma radiographs and a CT scan upon presentation. Classification of fractures was made based on review of all preoperative imaging in both the Neer and AO systems and was confirmed intraoperatively. The length of the posterior–medial metaphyseal extension attached to the head as described by Hertel et al. was also measured from the CT scans [17]. Patients underwent a clinical evaluation and had X-rays of the shoulder at 2, 6, 12, 24 and 52 weeks after the operation. Radiographs were reviewed by three orthopedic surgeons including the senior author and assessed for evidence of avascular necrosis based on the criteria of Gerber et al. [14]. Necrosis was considered complete if the entire head had disappeared and partial if only a segment of the head had collapsed. Fisher’s exact test was used to determine the significance between rates of AVN in patients with and without an 8 mm posterior–medial hinge.
Operative technique
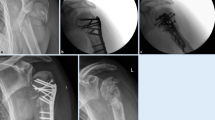
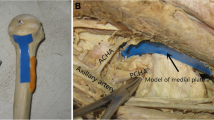
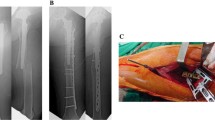
All patients were treated by the senior author. A more detailed description of this technique has been presented previously [9]. Patients are positioned in a sloppy lateral position, with fluoroscopy draped into the sterile field. The minimally invasive anterolateral approach was used in all cases [12]. In this approach, the anterior deltoid raphe is split and the anterior motor branch of the axillary nerve is identified and protected. Traction sutures through the rotator cuff or K-wires are used to control the humeral head and tuberosities. Reduction of the medial cortex is considered critical and cortical contact is achieved when possible. An endosteal implant is inserted through the lateral fracture lines. When there is comminution of the medial calcar region or cortical contact cannot be obtained, the implant is placed medially after being cut to the desired length. Push screws or k-wires are used to seat the implant along the medial column of the proximal neck in a position spanning the fracture and extending several centimeters into the humeral head (Fig. 1). In a valgus impacted fracture with an intact medial hinge, the endosteal implant is placed in a more lateral position to function as a buttress to the superior head (Fig. 2). A proximal humeral locking plate (Synthes, Paoli, PA) is then slid down the lateral aspect of the humerus under the axillary nerve and placed on the lateral vascular bare spot. This avoids any vessels penetrating the humeral head [11]. Coronal and sagittal alignment are obtained and confirmed with fluoroscopy. In 37 patients, a 6- to 8-cm allograft fibula was used as an endosteal implant. In four patients, a one-third tubular plate was used (Fig. 3). Locking screws are then placed so that they engage both the lateral plate and endosteal implant creating two columns of fixation. Additional screws are placed into the humeral head and shaft. Suction drains are used routinely. The fibula strut augment was chosen in most patients because it is technically easier to engage with the locking screws, has the potential for biologic incorporation and can be used as an indirect reduction tool.
Results
Forty patients met the inclusion criteria. Six patients were lost in follow-up. Three of these patients died of unrelated causes and two could not be located despite an exhaustive search. One patient suffered an infection and sought treatment elsewhere. The 34 remaining patients were used in this analysis. There were 26 females and 9 males. The average age was 66 years (range 44–83). Patients were followed with radiographs and clinical assessment for an average of 66 weeks (range 41–120). Using the Neer classification, there were 13 four-part fractures, 15 three-part fractures and 6 two-part fractures (Fig. 4). The distribution of the fractures in the AO classification is presented in Fig. 5.
All patients achieved radiographic union by 12 weeks. No patient had complete osteonecrosis (0%) and only one patient (2.8%) had evidence of partial osteonecrosis (Fig. 6). This patient suffered a four-part fracture and was the first to be treated using this technique. There was no posterior–medial hinge in this fracture. Despite collapse of a portion of the head, this patient has not required further surgery.
One patient suffered loss of reduction but went on to unite the fracture. This patient did not suffer osteonecrosis. Three patients had the lateral implant removed after union at their request.
Twenty patients had posterior–medial metaphyseal extensions that were less than 8 mm in length and 14 patients had no metaphyseal extension attached to the articular surface. The patient described above was the only one of these to suffer osteonecrosis. This was not statistically significant. (p = 0.59).
Discussion
In contrast to Neer’s original report which found 50% avascular necrosis in four-part fractures, more recent natural history studies have demonstrated infrequent AVN with nonoperative treatment [7, 28]. Using tetracycline labeling, Crosby et al. demonstrated that perfusion of the humeral head was preserved even when both tuberosities were displaced from the head [5]. With operative treatment, however, rates of AVN vary from 3 to 37% and efforts to predict ischemia based on fracture characteristics have been unsuccessful [2, 17]. In this investigation, we have confirmed that the 8-mm posterior–medial metaphyseal hinge is not a reliable predictor of AVN. Rather than being simply a product of the fracture pattern and thus the initial injury, avascular necrosis appears to be multifactorial with contributions from the injury, surgical trauma and stability of fixation.
Optimal treatment should address as many of these variables as possible. This study demonstrates that the comprehensive technique described provides a predictable method for avoiding avascular necrosis even when treating “at risk fracture” types. A single patient among this cohort of complex fractures suffered partial collapse of the humeral head. This patient was the first in the series to receive the endosteal implant and technical error is likely the cause of this failure rather then the technique itself. There are several aspects of this treatment protocol, which theoretically reduce AVN. The technique described seeks to achieve minimal soft tissue disruption while establishing rigid fixation of the fracture.
The anterolateral approach provides direct access to the lateral humeral bald spot obviating the need for circumferential dissection and potential vascular disruption either anteriorly or posteriorly [11]. Fragment manipulation and endosteal implant insertion can be done directly through the lateral fracture lines. The traditional deltopectoral approach requires significantly more dissection and retraction to access the lateral humerus increasing the potential for vascular insult. Hepp et al. compared the anterolateral deltoid split to the deltopectoral approach and demonstrated a trend toward increased AVN in the deltopectoral group [16].
Use of a minimally invasive approach likely reduces the risk of AVN, but the quality of fixation may also contribute to its reduction. This technique, in contrast to other minimally invasive techniques such as percutaneous pinning, allows for use of rigid fixation and we found that reduction was maintained in all but one patient. Although locked plating technology provides a substantial, biomechanical advantage over traditional compression plating, surprisingly high complication rates have been reported including varus displacement and screw cutout in 23–25% of cases [1, 3, 8, 13, 22, 23, 26, 27]. Loss of reduction has been shown to correlate with the absence of medial support [10, 20, 25]. The addition of the endosteal implant, particularly when used to re-establish medial support in the presence of calcar comminution, significantly increases the load to failure and stiffness of this construct. Use of a fibula strut is preferable to the one-third tubular plate, because the fibula is technically easier to engage with the locking screws, has the potential for biologic incorporation and can be used as an indirect reduction tool. Biomechanical testing has shown that addition of an intramedullary fibula increases the stiffness of the lateral proximal humerus plating by 3.84 times [21]. Maintenance of an adequate reduction may prevent postoperative thrombosis, and increased stability may allow for revascularization of fixed ischemic bone regardless of the initial fracture pattern [19]. Further study is needed to examine this phenomenon.
This study demonstrates that complex fracture patterns of the proximal humerus can be treated through joint-preserving methods with minimal risk of avascular necrosis. The minimally invasive approach diminishes further disruption of the humeral blood supply. Reduction, stabilization and augmentation of the calcar with the endosteal implant provide increased stability and may allow revascularization in areas ischemic from the initial fracture. Gerber et al. have shown that in the absence of this complication, good shoulder function can be obtained after such fractures [13]. Comparisons of hemiarthroplasty to ORIF have shown the most favorable outcomes in patients who have undergone plate fixation and avoided osteonecrosis [24]. Use of a minimally invasive approach with bicolumnar fixation can reliably avoid this complication even in fractures traditionally considered at high risk for AVN obviating the need for joint replacement.
References
Agudelo J, Schurmann M, Stahel P, Helwig P, Morgan SJ, Zechel W, Bahrs C, Parekh A, Ziran B, Williams A, Smith W (2007) Analysis of efficacy and failure in proximal humerus fractures treated with locking plates. J Orthop Trauma 21:676–681. doi:10.1097/BOT.0b013e31815bb09d
Bastian JD, Hertel R (2008) Initial post-fracture humeral head ischemia does not predict development of necrosis. J Should Elb Surg 17:2–8. doi:10.1016/j.jse.2007.03.026
Brunner F, Sommer C, Bahrs C, Heuwinkel R, Hafner C, Rillmann P, Kohut G, Ekelund A, Muller M, Audige L, Babst R (2009) Open reduction and internal fixation of proximal humerus fractures using a proximal humeral locked plate: a prospective multicenter analysis. J Orthop Trauma 23(3):163–172. doi:10.1097/BOT.0b013e3181920e5b
Court-Brown CM, Garg A, McQueen MM (2001) The epidemiology of proximal humeral fractures. Acta Orthop Scand 72:365–371. doi:10.1080/000164701753542023
Crosby LA, Finnan RP, Anderson CG, Gozdanovic J, Miller MW (2009) Tetracycline labeling as a measure of humeral head viability after 3- or 4-part proximal humerus fracture. J Should Elb Surg 18(6):851–858. doi:10.1016/j.jse.2008.12.016
Duparc F, Muller JM, Freger P (2001) Arterial blood supply of the proximal humeral epiphysis. Surg Radiol Anat 23(3):185–190
Edelson G, Safuri H, Salami J, Vigder F, Militianu D (2008) Natural history of complex fractures of the proximal humerus using a three-dimensional classification system. J Should Elb Surg 17:399–409. doi:10.1016/j.jse.2007.08.014
Fankhauser F, Boldin C, Schippinger G, Haunschmid C, Szyszkowitz R (2005) A new locking plate for unstable fractures of the proximal humerus. Clin Orthop Relat Res 430:176–181
Gardner MJ, Boraiah S, Helfet DL, Lorich DG (2008) Indirect medial reduction and strut support of proximal humerus fractures using an endosteal implant. J Orthop Trauma 22(3):195–200. doi:10.1097/BOT.0b013e31815b3922
Gardner MJ, Weil Y, Barker JU, Kelly BT, Helfet DL, Lorich DG (2007) The importance of medial support in locked plating of proximal humerus fractures. J Orthop Trauma 21(3):185–191. doi:10.1097/BOT.0b013e3180333094
Gardner MJ, Voos JE, Wanich T, Helfet DL, Lorich DG (2006) Vascular implications of minimally invasive plating of proximal humerus fractures. J Orthop Trauma 20:602–607. doi:10.1097/01.bot.0000246412.10176.14
Gardner MJ, Griffith MH, Dines JS, Briggs SM, Weiland AJ, Lorich DG (2005) The extended anterolateral acromial approach allows minimally invasive access to the proximal humerus. Clin Orthop Relat Res 434:123–129
Gerber C, Werner CM, Vienne P (2004) Internal fixation of complex fractures of the proximal humerus. J Bone Joint Surg Br 86:848–855
Gerber C, Hersche O, Berberat C (1998) The clinical relevance of posttraumatic avascular necrosis of the humeral head. J Should Elb Surg 7(6):586–590
Gerber C, Schneeberger AG, Vinh TS (1990) The arterial vascularization of the humeral head. An anatomical study. J Bone Jt Surg Am 72:1486–1494
Hepp P, Theopold J, Voigt C, Engel T, Josten C, Lill H (2008) The surgical approach for locking plate osteosynthesis of displaced proximal humeral fractures influences the functional outcome. J Should Elb Surg 17(1):21–28. doi:10.1016/j.jse.2007.03.029
Hertel R, Hempfing A, Stiehler M, Leunig M (2004) Predictors of humeral head ischemia after intracapsular fracture of the proximal humerus. J Should Elb Surg 13(4):427–433. doi:10.1016/S1058274604000795
Hettrich CM, Boraiah S, Dyke JP, Neviaser A, Helfet DL, Lorich DG (2010) Quantitative assessment of the vascularity of the proximal part of the humerus. J Bone Jt Surg Am 92(4):943–948. doi:10.2106/JBJS.H.01144
Lee CK, Hansen HR (1981) Post-traumatic avascular necrosis of the humeral head in displaced proximal humeral fractures. J Trauma 21(9):788–791
Lee CW, Shin SJ (2009) Prognostic factors for unstable proximal humeral fractures treated with locking-plate fixation. J Should Elb Surg 18:83–88. doi:10.1016/j.jse.2008.06.014
Mathison C, Chaudhary R, Beaupre L, Reynolds M, Adeeb S, Bouliane M (2010) Biomechanical analysis of proximal humeral fixation using locking plate fixation with an intramedullary fibular allograft. Clin Biomech (Bristol, Avon) 25:642–646. doi:10.1016/j.clinbiomech.2010.04.006
Owsley KC, Gorczyca JT (2008) Fracture displacement and screw cutout after open reduction and locked plate fixation of proximal humeral fractures. J Bone Jt Surg Am 90:233–240. doi:10.2106/JBJS.F.01351
Rose PS, Adams CR, Torchia ME, Jacofsky DJ, Haidukewych GG, Steinmann SP (2007) Locking plate fixation for proximal humeral fractures: initial results with a new implant. J Should Elb Surg 16(2):202–207. doi:10.1016/j.jse.2006.06.006
Solberg BD, Moon CN, Franco DP, Paiement GD (2009) Surgical treatment of three and four-part proximal humeral fractures. J Bone Jt Surg Am 91(7):1689–1697. doi:10.2106/JBJS.H.00133
Solberg BD, Moon CN, Franco DP, Paiement GD (2009) Locked plating of 3- and 4-part proximal humerus fractures in older patients: the effect of initial fracture pattern on outcome. J Orthop Trauma 23(2):113–119. doi:10.1097/BOT.0b013e31819344bf
Sudkamp N, Bayer J, Hepp P, Voigt C, Oestern H, Kaab M, Luo C, Plecko M, Wendt K, Kostler W, Konrad G (2009) Open reduction and internal fixation of proximal humeral fractures with use of the locking proximal humerus plate. Results of a prospective, multicenter, observational study. J Bone Jt Surg Am 91:1320–1328. doi:10.2106/JBJS.H.00006
Thanasas C, Kontakis G, Angoules A, Limb D, Giannoudis P (2009) Treatment of proximal humerus fractures with locking plates: a systematic review. J Should Elb Surg 18:612–621. doi:10.1016/j.jse.2009.06.004
Young TB, Wallace WA (1985) Conservative treatment of fractures and fracture-dislocations of the upper end of the humerus. J Bone Jt Surg Br 67:373–377
Conflict of interest
The authors declare that they have no conflicts of interest related to this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Neviaser, A.S., Hettrich, C.M., Dines, J.S. et al. Rate of avascular necrosis following proximal humerus fractures treated with a lateral locking plate and endosteal implant. Arch Orthop Trauma Surg 131, 1617–1622 (2011). https://doi.org/10.1007/s00402-011-1366-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-011-1366-6