Abstract
Introduction
The purpose of this study was to evaluate the results of a contoured focal articular femoral condyle resurfacing prosthetic in the treatment of full-thickness cartilage and osteochondral defects at the medial femoral condyle of the knee beyond 5 years.
Methods
In a multicenter case series, preoperative and follow-up scores of the Knee Injury and Osteoarthritis Outcome Score (KOOS), SF-36 and Tegner activity scale were evaluated. Standard radiographs were performed to evaluate the progression of osteoarthritis. Patients were also asked to report their satisfaction.
Results
A total of 21 patients were included in this study. The average follow-up was 5.3 years. The average age at the time of resurfacing was 54 years. Average KOOS scores significantly (P ≤ 0.005) improved for pain (51.1 to 77.6), symptoms (57.9 to 79.5), activities of daily living (ADL) (58.8 to 82.4), sports (26.3 to 57.8) and quality of life (QOL) (34.4 to 55.0). The Tegner activity level improved significantly (P ≤ 0.02) from 2.9 to 4. The physical health value of the SF-36 increased by 15.2 to 46.9 compared to the preoperative value. The mental health value almost (51.2) remained unchanged. As many as 16/21 of the patients in this series were satisfied with their outcome and would have the operation again. Radiographic results demonstrated solid fixation, preservation of joint space and no change in the osteoarthritic stage.
Conclusions
The device appears to be an effective reconstructive treatment option for large full-thickness cartilage and osteochondral lesions of the knee in middle-aged patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent years, there has been a steady rise in the volume of revision total knee arthroplasty (TKA) procedures [1] and the number of primary TKA procedures performed in the next two decades is expected to rise exponentially [2]. It is therefore critical to prolong focal defect management to preserve healthy articular surfaces, soft tissues and bone stock, and to delay the onset of conventional arthroplasty.
Full-thickness articular cartilage defects are commonly found in symptomatic knees [3, 4], the majority of which are observed in patients over the age of 40 years [5, 6]. Left untreated, these defects may result in progressive articular degeneration and predispose to the development of osteoarthritis [7–9]. Single or repeat repair procedures such as microfracture, OATS, allograft or cell-based technologies can provide relief from focal defects [10–15], but appear to be increasingly ineffective with increasing age [13, 16–18]. However, if these defects remain symptomatic, this patient population reflects a serious problem for the orthopaedic surgeon, as conventional joint arthroplasty does not provide joint preservation in middle-aged patients with isolated defects.
Focal defect management has been extended with the development of a reconstructive treatment option using a metallic inlay resurfacing prosthesis (HemiCAP®, Arthrosurface Inc., Franklin, MA, USA). The procedure allows the surgeon to precisely match the implant to the contour of the patient’s articular cartilage, thus filling the defect and restoring a smooth and continuous joint surface (Fig. 1). Healthy cartilage, bone and soft tissue structures are therefore preserved for conventional joint arthroplasty if it becomes necessary at a later stage.
The purpose of this multicenter study was to evaluate the durability of the focal resurfacing procedure in middle-aged patients with a minimum of 5 years follow-up. Outcomes were evaluated with validated outcome questionnaires, standard radiographs and comparison to normative, age-matched reference data.
Materials and methods
Study design
This is a level 4, multicenter (5 centers), case series. Patients, who had received the resurfacing device with a minimum of 5 years follow-up, were screened for eligibility according to the inclusion and exclusion criteria (Table 1) using pre- and intraoperative patient records. To achieve a homogenous middle-aged cohort, patients 65 years and older were excluded. Patients with kissing lesions and advanced grade cartilage degeneration in other compartments were excluded as well to support conclusions toward the potential benefit of the procedure. Institutional review board approval was obtained prior to screening. All patients gave informed consent before inclusion into the study.
Resurfacing prosthetic
The HemiCAP® contoured articular prosthetic (Fig. 1) for focal femoral condyle resurfacing consists of two components, a fixation and an articular component. The two are joined by a morse taper connection to provide a stable and immobile fixation of the implant and stress bearing contact at the bone/prosthetic interface. The fixation component is a cannulated cancellous screw with a tapering distal tip (titanium alloy). The articular dome-shaped component comes in 15- and 20-mm diameters, which are available in various off-set sizes corresponding to the radius of curvatures at the implant site (cobalt–chromium–molybdenum alloy with titanium plasma spray coverage on the underside).
Rehabilitation
Standard rehabilitation protocol included weight bearing as tolerated using crutches for 1–2 weeks with early functional rehabilitation and no restriction to range of motion.
Outcome measures
Demographic data and validated questionnaires were gathered at the follow-up visit. The Knee Injury and Osteoarthritis Outcome Score (KOOS) score [19] includes five domains: pain, symptoms, activities of daily living (ADL), sports and recreation ability, and knee-related quality of life (QoL) and has been used and validated for the treatment of focal cartilage defects [20, 21] (100 = best possible score, 0 = worst). Overall physical health assessment was obtained via the SF-36 Health Status Survey [22]. The SF-36 is a self-administered, patient-based scoring system assessing patient global health with 36 questions leading to a physical and mental health component score. The Tegner activity scale [23] is a numerical scale measuring the level of competitiveness of an individual sport’s participation. Each activity is measured from 0 to 10 where an elite professional athlete would compete in a sport at a level of 10, high-level recreational athletes would be at 6 and persons with knee problems or a disability would score at a level of 0. These scores were compared with their preoperative scores gathered at the latest follow-up to evaluate the effects of device implantation. Satisfaction was evaluated by a questionnaire that rated the general satisfaction with the treatment outcomes (1 = excellent, 2 = very good, 3 = good, 4 = fair, 5 = poor), if they would undergo the procedure again and if they would recommend this procedure to friends and family. Furthermore, results were compared to normative patient population data for KOOS, SF-36 and Tegner.
Radiographic assessment was evaluated comparing the preoperative radiographs to those obtained at the 5-year follow-up visit (AP and lateral standing X-rays). The radiographs were graded according to the Kellgren–Lawrence classification [24] and Osteoarthritis Research Society International (OARSI) Grading System [25, 26] to compare the stage of osteoarthritis with the preoperative status. Prosthetic connectivity, subsidence, migration, radiolucency, implant loosening and subchondral cyst formation were evaluated.
Secondary procedures following the index surgery were assessed according to the patient records and confirmed at the follow-up visit. Treatment failure was defined as removal of the prosthetic device and replacement by arthroplasty (uni-compartmental or total). All evaluations were carried out by the first and second author. Discrepancies in interpretation were settled by consensus.
Statistical analysis
Statistical analysis was performed using the software package SPSS 18.0 (SPSS Inc., Chicago, IL, USA). Mean and standard deviation values were recorded. To determine differences between the data of preoperative and follow-up scores, the Wilcoxon nonparametric test (significance, P ≤ 0.05) was used.
Results
Patient characteristics
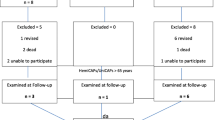
Forty patients were implanted with the resurfacing device; 21 patients were included in this study (Table 1; Fig. 2). The average age at implantation was 54 years (range 38–63). There were 11 males and 10 females. The average follow-up was 5.3 years (64 months; range 60–71 months). Six patients were not available for radiographic examination at follow-up; however, they were contacted by telephone and returned their questionnaires by mail. One patient underwent conversion to unicondylar knee replacement at 2 years postoperatively leaving 20 patients available for a 5-year analysis. Patient demographics are summarized in Table 2. Defects in four patients were resurfaced with a 15-mm surface implant and in 17 patients with a 20-mm implant. Surgical history and joint status are summarized in Table 3.
Score outcome measures
The results are summarized in Table 4. The average 5-year postoperative KOOS domains scores were 77.6 (pain), 79.5 (symptoms), 82.4 (ADL), 57.8 (sports/recreation), and 55.0 (QoL). The physical component score of the SF-36 was 46.9, the mental component score 51.2. The average Tegner Activity Scale was 4.0, meaning patients could engage in moderately heavy labor and recreational sports such as cycling. General satisfaction with the treatment outcomes was rated as 2.9 (SD 1.4, range 1–5). The result was rated excellent in 4/21 of the patients, very good in 6/21, good in 3/20, fair in 5/21 and in 3/21 as poor. Of the 21 patients, 16 (76%) patients indicated they would undergo the procedure again and would recommend it to friends and family.
Radiographic assessment
Fourteen patients were evaluated radiographically at a minimum of 5 years follow-up. No device disassembly, subsidence/migration, peri-prosthetic radiolucency, implant loosening or subchondral cyst formation was found. The average Kellgren–Lawrence score slightly increased from 2.2 to 2.5 over the course of the follow-up period. No clinically significant joint space narrowing was noted according to the OARSI Grading System. The average OARSI grade for osteophyte formation (tibial plateau and femoral condyles) remained unchanged when compared with baseline (Table 5).
Subsequent surgical procedures
One patient underwent arthroscopic debridement 4.9 years following the index procedure due to pain. Another patient underwent three further operations of the affected knee including arthroscopic debridement at 5 months due to persistent pain in the medial compartment, high tibial osteotomy for the correction of varus malalignment at 2 years and hardware removal/further debridement at 3 years. Satisfaction outcome for these two patients was rated fair and poor. One female patient underwent conversion from focal resurfacing to uni-compartmental knee arthroplasty at 2 years after the index procedure due to ongoing pain. This patient was regarded as a failure and was not included in the 5-year score analysis, but included in the general satisfaction analysis and rated as poor.
Comparison to normative data
When comparing the results of our patients to normative data for KOOS domains [27] and age 55–74 years, slightly better scores were found for our female patients for all domains except the Sports and Recreation domain, which was similar, and the QOL domain which was slightly worse (Fig. 3). When comparing the scores to those of normal scores for males, our patients were inferior across all domains (Fig. 4). Comparison to SF-36 normal scores [28] showed slightly inferior scores for the physical component (5 yr. = 46.9; normal = 52.1) and similar scores for the mental component (5 yr. = 51.2; normal = 52). When compared with a population of community volunteers 46–60 years of age [29], who considered their knee function normal and without any history of knee surgery or recent injury, the average Tegner activity level of our patients were again slightly inferior to the normal score published for this population (5 yr. = 4.0; normal = 4.6).
KOOS scores compared to normal female scores [27]. Population-based reference data for KOOS (females): 35–54 years; n = 80. 55–74 years; n = 85. Current study mean age at follow-up (female): 59 years (range 43–68); n = 9
KOOS scores compared to normal male scores [27]. Population-based reference data for KOOS (males): 35–54 years; n = 78. 55–74 years; n = 88. Current study mean age at follow-up (male): 58 years (range 46–68), n = 11
Discussion
Contoured metallic resurfacing is a novel reconstructive treatment option intended to extend the range of focal procedures in middle-aged patients who frequently find themselves between biological solutions and conventional arthroplasty. Unicondylar knee replacement has been shown to be a successful treatment option when larger surface reconstruction is warranted [30]. However, in middle-aged patients with isolated condylar defects, significant loss of healthy soft tissues and bone stock should be avoided.
The use of a metal implant, however, is of concern resulting in damage to the opposing articulating structures due to rigid fixation of the resurfacing prosthetic in the underlying bone. Basic science studies have previously investigated the validity of the resurfacing device: Kirker-Head et al. [31] assessed the functional and biological response in a goat model. One year after implantation, gross necropsy and histological data implied the biocompatibility and functionality of the implant, since no ongoing degenerative joint disease was apparent. Some chondral damage in the operated joint was found in several goats in opposing locations on the proximal tibia, including some focal meniscal injury. Similar results, however less frequently, were encountered in the un-operated controls [31]. This outcome was substantially better in comparison to other reported experimental animal studies in goats with untreated full-thickness defects [8, 32]. In vitro studies using fresh frozen human cadaver specimens to evaluate the effects of the prosthetic device on tibiofemoral contact pressure suggested biomechanical safety with no significant increase of contact pressures compared to untreated healthy knees, even under continuous dynamic loading [33, 34].
This clinical series represents the first report on patients with a minimum of 5 years of follow-up after focal prosthetic resurfacing of localized full-thickness cartilage and osteochondral condylar defects. All patients in our series had previously failed conservative treatment and the vast majority underwent single or repeat biological procedures prior to focal prosthetic resurfacing. The study looked specifically at middle-aged patients without kissing lesions and advanced grade cartilage degeneration in other compartments that would benefit the most from a limited resurfacing treatment. However, 42.5% of the implanted patients were operated outside the target population resulting in their exclusion from the study. Further weaknesses of the study were the small number of patients, the lack of prospective baseline data and that only 2/3 of the included patients were available for a radiographic follow-up.
Studies have shown that biological treatment measures are less suitable for middle-aged or active elderly patients resulting in inferior outcomes in older patients [13, 16–18]. Reports on microfracture found a strong and significant correlation between pain relief, functional results and age: better outcomes for patients under 35 years [16], less improvement for patients between 35 and 45 years compared to younger patients, [13] and significant increase in pain and decrease in function between 18 and 36 months after microfracture for patients over 40 years, while remaining relatively constant for those 40 and under at the same time points [17]. In a randomized trial comparing autologous chondrocyte implantation (ACI) to microfracture, Knutsen et al. [18] found that younger patients (less than 30 years old) had a significantly better clinical outcome at 5 years follow-up than older patients, regardless of their treatment group.
Recently, ACI as a primary procedure was reported not to result in inferior outcomes for patients aged older than 40 years [35] or 45 years [36] when compared to younger patients. ACI as a secondary procedure, in particular following microfracture, has shown a lower success rate [37]. High costs, high reoperation rates, complex rehabilitation and prolonged delay before return to sports [36, 38, 39] represent further limitations to the use of this technique.
Patients in our study rated their outcomes as: 62% excellent, very good or good; 24% fair; and 14% poor. As many as 76% would choose to undergo the procedure again. Slightly better rates were found in a study reporting results after ACI in patients over the age of 45 years. Patients rated their outcome at a mean follow-up of 4.7 years as: 72% good or excellent, 15% fair and 13% poor with 81% choosing to undergo the procedure again [36]. However, the failure rate requiring open repeat surgery was 14%, and even 43% of patients required second-look arthroscopy within the first year after ACI [36]. In a prospective study of ACI in patients with failed prior treatment (mean: 1.9 procedures), the subsequent surgical procedure rate observed was 49% overall and 40% related to ACI [39]. Results of the KOOS subscales at an average of 3.8 years follow-up were in all categories inferior compared to our results. Self-rating of the overall knee condition was 77% good or excellent [39]. In our study, one patient (1/21) was converted to a uni-compartmental knee arthroplasty due to ongoing pain and functional limitations. This failure rate is relatively low when compared with other treatments for focal defects [10, 18, 36, 39, 40].
In a study evaluating population-based reference data for the KOOS score, pain, physical function and knee-related quality of life varied with age and gender with a trend to lower normative levels with increasing age [27]. Although the average age of our patients at surgery was 54 years (range 38–63) and a higher number of women were treated compared to studies on ACI [36, 39], we showed superior results in the majority of KOOS subscales when compared to these reports.
Radiographic signs of osteoarthritis were common in our study. Knutsen et al. [18] reported that 34% of the patients had radiographic evidence of early osteoarthritis at 5 years after ACI or microfracture treatment. Our findings revealed a slight increase of osteoarthritis, but no progression in the grade of the Kellgren–Lawrence classification and OARSI Grading System. In an analytic literature synthesis on Kellgren–Lawrence progression in knee osteoarthritis, the annual progression by at least one grade ranged from 1.0 to 19.6% with an overall mean risk of progression of 5.6% at a mean follow-up of 5.3 years [41]. Thus, the osteoarthritic changes among our patients appear not to exceed the overall mean risk of progression.
In summary, by using validated outcome measures and standard radiographs, we have demonstrated clinically meaningful improvements in the majority of patients and radiological safety after focal prosthetic resurfacing for the treatment of full-thickness cartilage and osteochondral defects at the medial femoral condyle at a minimum follow-up of 5 years. Nevertheless, the results need to be confirmed in larger patient cohorts.
References
Bozic KJ, Kurtz SM, Lau E, Ong K, Chiu V, Vail TP, Rubash HE, Berry DJ (2010) The epidemiology of revision total knee arthroplasty in the United States. Clin Orthop Relat Res 468:45–51
Kurtz S, Ong K, Lau E, Mowat F, Halpern M (2007) Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am 89:780–785
vies-Tuck ML, Wluka AE, Wang Y, Teichtahl AJ, Jones G, Ding C, Cicuttini FM (2008) The natural history of cartilage defects in people with knee osteoarthritis. Osteoarthritis Cartilage 16:337–342
Wluka AE, Ding C, Jones G, Cicuttini FM (2005) The clinical correlates of articular cartilage defects in symptomatic knee osteoarthritis: a prospective study. Rheumatology (Oxford) 44:1311–1316
Curl WW, Krome J, Gordon ES, Rushing J, Smith BP, Poehling GG (1997) Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy 13:456–460
Hjelle K, Solheim E, Strand T, Muri R, Brittberg M (2002) Articular cartilage defects in 1,000 knee arthroscopies. Arthroscopy 18:730–734
Cicuttini F, Ding C, Wluka A, Davis S, Ebeling PR, Jones G (2005) Association of cartilage defects with loss of knee cartilage in healthy, middle-age adults: a prospective study. Arthritis Rheum 52:2033–2039
Jackson DW, Lalor PA, Aberman HM, Simon TM (2001) Spontaneous repair of full-thickness defects of articular cartilage in a goat model. A preliminary study. J Bone Joint Surg Am 83-A:53–64
Wang Y, Ding C, Wluka AE, Davis S, Ebeling PR, Jones G, Cicuttini FM (2006) Factors affecting progression of knee cartilage defects in normal subjects over 2 years. Rheumatology (Oxford) 45:79–84
Browne JE, Anderson AF, Arciero R, Mandelbaum B, Moseley JB, Jr., Micheli LJ, Fu F, Erggelet C (2005) Clinical outcome of autologous chondrocyte implantation at 5 years in US subjects. Clin Orthop Relat Res 237–245
Hangody L, Fules P (2003) Autologous osteochondral mosaicplasty for the treatment of full-thickness defects of weight-bearing joints: ten years of experimental and clinical experience. J Bone Joint Surg Am 85-A(Suppl 2):25–32
Peterson L, Vasiliadis HS, Brittberg M, Lindahl A (2010) Autologous chondrocyte implantation: a long-term follow-up. Am J Sports Med 38:1117–1124
Steadman JR, Briggs KK, Rodrigo JJ, Kocher MS, Gill TJ, Rodkey WG (2003) Outcomes of microfracture for traumatic chondral defects of the knee: average 11-year follow-up. Arthroscopy 19:477–484
Karataglis D, Green MA, Learmonth DJ (2006) Autologous osteochondral transplantation for the treatment of chondral defects of the knee. Knee 13:32–35
McCulloch PC, Kang RW, Sobhy MH, Hayden JK, Cole BJ (2007) Prospective evaluation of prolonged fresh osteochondral allograft transplantation of the femoral condyle: minimum 2-year follow-up. Am J Sports Med 35:411–420
Asik M, Ciftci F, Sen C, Erdil M, Atalar A (2008) The microfracture technique for the treatment of full-thickness articular cartilage lesions of the knee: midterm results. Arthroscopy 24:1214–1220
Kreuz PC, Erggelet C, Steinwachs MR, Krause SJ, Lahm A, Niemeyer P, Ghanem N, Uhl M, Sudkamp N (2006) Is microfracture of chondral defects in the knee associated with different results in patients aged 40 years or younger? Arthroscopy 22:1180–1186
Knutsen G, Drogset JO, Engebretsen L, Grontvedt T, Isaksen V, Ludvigsen TC, Roberts S, Solheim E, Strand T, Johansen O (2007) A randomized trial comparing autologous chondrocyte implantation with microfracture. Findings at five years. J Bone Joint Surg Am 89:2105–2112
Roos EM, Roos HP, Lohmander LS, Ekdahl C, Beynnon BD (1998) Knee Injury and Osteoarthritis Outcome Score (KOOS)—development of a self-administered outcome measure. J Orthop Sports Phys Ther 28:88–96
Bekkers JE, de Windt TS, Raijmakers NJ, Dhert WJ, Saris DB (2009) Validation of the Knee Injury and Osteoarthritis Outcome Score (KOOS) for the treatment of focal cartilage lesions. Osteoarthritis Cartilage 17:1434–1439
Hambly K, Griva K (2008) IKDC or KOOS? Which measures symptoms and disabilities most important to postoperative articular cartilage repair patients? Am J Sports Med 36:1695–1704
Ware JE Jr, Sherbourne CD (1992) The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 30:473–483
Tegner Y, Lysholm J (1985) Rating systems in the evaluation of knee ligament injuries. Clin Orthop Relat Res:43–49
Kellgren JH, Lawrence JS (1957) Radiological assessment of osteo-arthrosis. Ann Rheum Dis 16:494–502
Altman RD, Gold GE (2007) Atlas of individual radiographic features in osteoarthritis, revised. Osteoarthritis Cartilage 15 (Suppl A):A1–A56
Guermazi A, Hunter DJ, Roemer FW (2009) Plain radiography and magnetic resonance imaging diagnostics in osteoarthritis: validated staging and scoring. J Bone Joint Surg Am 91(Suppl 1):54–62
Paradowski PT, Bergman S, Sunden-Lundius A, Lohmander LS, Roos EM (2006) Knee complaints vary with age and gender in the adult population. Population-based reference data for the Knee injury and Osteoarthritis Outcome Score (KOOS). BMC Musculoskelet Disord 7:38
Hopman WM, Towheed T, Anastassiades T, Tenenhouse A, Poliquin S, Berger C, Joseph L, Brown JP, Murray TM, Adachi JD, Hanley DA, Papadimitropoulos E (2000) Canadian normative data for the SF-36 health survey. Canadian Multicentre Osteoporosis Study Research Group. CMAJ 163:265–271
Briggs KK, Steadman JR, Hay CJ, Hines SL (2009) Lysholm score and Tegner activity level in individuals with normal knees. Am J Sports Med 37:898–901
O’Rourke MR, Gardner JJ, Callaghan JJ, Liu SS, Goetz DD, Vittetoe DA, Sullivan PM, Johnston RC (2005) The John Insall Award: unicompartmental knee replacement: a minimum twenty-one-year followup, end-result study. Clin Orthop Relat Res 440:27–37
Kirker-Head CA, Van S, Ek SW, McCool JC (2006) Safety of, and biological and functional response to, a novel metallic implant for the management of focal full-thickness cartilage defects: preliminary assessment in an animal model out to 1 year. J Orthop Res 24:1095–1108
van Susante JL, Buma P, Schuman L, Homminga GN, van den Berg WB, Veth RP (1999) Resurfacing potential of heterologous chondrocytes suspended in fibrin glue in large full-thickness defects of femoral articular cartilage: an experimental study in the goat. Biomaterials 20:1167–1175
Becher C, Huber R, Thermann H, Paessler HH, Skrbensky G (2008) Effects of a contoured articular prosthetic device on tibiofemoral peak contact pressure: a biomechanical study. Knee Surg Sports Traumatol Arthrosc 16:56–63
Becher C, Huber R, Thermann H, Ezechieli L, Ostermeier S, Wellmann M, von Skrbensky G (2011) Effects of a surface matching articular resurfacing device on tibiofemoral contact pressure: results from continuous dynamic flexion–extension cycles. Arch Orthop Trauma Surg 131:413–419
Niemeyer P, Kostler W, Salzmann GM, Lenz P, Kreuz PC, Sudkamp NP (2010) Autologous chondrocyte implantation for treatment of focal cartilage defects in patients age 40 years and older: a matched-pair analysis with 2-year follow-up. Am J Sports Med
Rosenberger RE, Gomoll AH, Bryant T, Minas T (2008) Repair of large chondral defects of the knee with autologous chondrocyte implantation in patients 45 years or older. Am J Sports Med 36:2336–2344
Minas T, Gomoll AH, Rosenberger R, Royce RO, Bryant T (2009) Increased failure rate of autologous chondrocyte implantation after previous treatment with marrow stimulation techniques. Am J Sports Med 37:902–908
Gomoll AH, Farr J, Gillogly SD, Kercher J, Minas T (2010) Surgical management of articular cartilage defects of the knee. J Bone Joint Surg Am 92:2470–2490
Zaslav K, Cole B, Brewster R, DeBerardino T, Farr J, Fowler P, Nissen C (2009) A prospective study of autologous chondrocyte implantation in patients with failed prior treatment for articular cartilage defect of the knee: results of the Study of the Treatment of Articular Repair (STAR) clinical trial. Am J Sports Med 37:42–55
Wood JJ, Malek MA, Frassica FJ, Polder JA, Mohan AK, Bloom ET, Braun MM, Cote TR (2006) Autologous cultured chondrocytes: adverse events reported to the United States Food and Drug Administration. J Bone Joint Surg Am 88:503–507
Emrani PS, Katz JN, Kessler CL, Reichmann WM, Wright EA, McAlindon TE, Losina E (2008) Joint space narrowing and Kellgren–Lawrence progression in knee osteoarthritis: an analytic literature synthesis. Osteoarthritis Cartilage 16:873–882
Acknowledgments
The authors wish to thank 2med GmbH, Hamburg, Germany, for the help in communicating with the participating study centers.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Becher, C., Kalbe, C., Thermann, H. et al. Minimum 5-year results of focal articular prosthetic resurfacing for the treatment of full-thickness articular cartilage defects in the knee. Arch Orthop Trauma Surg 131, 1135–1143 (2011). https://doi.org/10.1007/s00402-011-1323-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-011-1323-4